Protein-polymer Self-assembly Conjugation Technologies
As a unique class of polypeptides with fully folded structures and globular shapes, proteins in most cases provide biological functions to protein-polymer conjugates. When hydrophobic polymers are attached to water-soluble proteins, the amphiphilic conjugates self-assemble in a manner similar to that of low molecular weight surfactants and synthetic block copolymers in aqueous solution. Therefore, these protein-based amphiphiles can also serve as building blocks for the construction of a wide range of solution nanostructures.
BOC Sciences provides the most complete protein-polymer conjugate solutions for the pharmaceutical and materials industries. Our chemistry team can tailor many parameters to achieve the desired protein-polymer conjugate combination, including the protein sequence and length, the chemical nature of the polymer, the length of the polymer, the solvent, the conjugation site and chemistry. We aim to change these properties to accommodate the engineering of new materials with desired behaviors and activities.
Introduction
Proteins are a promising class of pharmaceuticals due to their precise binding interactions and limited off-target effects. However, proteins frequently suffer from poor stability, rapid clearance, and immunogenicity, limiting their clinical application. The conjugation of a polymer to a protein has been shown to improve the pharmaceutical efficacy of the resulting bioconjugate by increasing protein stability and circulation time. A classical example is PEGylation, the attachment of polyethylene glycol (PEG) to a protein. Via steric repulsion, PEGylation can prevent proteolysis and shield antigenic groups, simultaneously increasing stability and lowering immunogenicity. Multiple PEG-protein bioconjugates have been approved to treat a variety of diseases including, chronic hepatitis C, acute lymphatic leukaemia, and rheumatoid arthritis. The strides made in the development of PEGylated drugs and their clinical success have led to an interest in creating the next-generation of protein-polymer bioconjugates with advanced functionality like biodegradability, targeted delivery, controlled drug release and the ability to cross biological barriers.
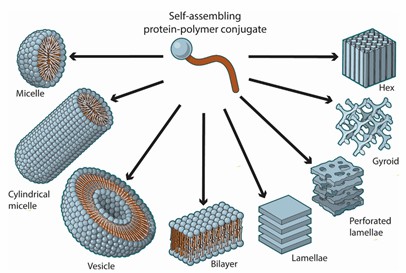 Fig. 1. Schematic representations of common self-assembled protein-polymer conjugates architectures (Advanced Drug Delivery Reviews. 2021, 174: 447-460).
Fig. 1. Schematic representations of common self-assembled protein-polymer conjugates architectures (Advanced Drug Delivery Reviews. 2021, 174: 447-460).
Mechanistically, self-assembly of protein-polymer bioconjugates relies on components designed to spontaneously order into a hierarchical structure with little external input. Noncovalent interactions between subunits such as hydrogen bonding, π-π stacking, ligand-binding interactions, metal-ligand interactions, electrostatic forces, dipole-dipole interactions, hydrophobic forces, and steric forces can be used to design self-assembling materials. That is, amphiphilicity can be engineered into these bioconjugates by conjugating a hydrophobic polymer to a hydrophilic protein. In concentrated and dilute solutions, the resulting amphiphile can self-assemble into higher order structures such as micelles, vesicles, bilayers, lamellae, perforated lamellae, and gyroids (Fig 1). In recent years, self-assembled nanoparticles based on protein-polymer conjugates have been intensively explored as carriers for delivery of anticancer drugs. Due to the presence of proteins, these self-assembled nanostructures possess the special advantage of built-in bioactivity.
Applications of Self-assembly Protein-Polymer Conjugates
Drug Delivery
Self-assembled protein-polymer conjugates have many distinct advantages over monomeric protein-polymer conjugates for drug delivery including drug encapsulation, temporal, and distribution control. For example, through encapsulation, self-assembled protein-polymer conjugates can improve the bioavailability of hydrophobic drugs, protect drugs from hydrolysis, enzymatic orchemical degradation, reduce toxicity, and lead to sustained release.
Bionanomaterials
As compared to monomeric protein-polymer conjugates, self-assembled bioconjugates demonstrate additional functionality, such as drug loading and stimuli-responsiveness. This additional functionality can be leveraged to develop the next generation of advanced biomaterials. These hybrid materials branch biological and synthetic research interfacing between many disciplines including materials science, polymer physics and structural biology.
Protein Therapeutics
The most commonly used polymer in hybrid biomaterials is PEG. PEGylation has proven to bean effective strategy to enhance the kinetic stabilization of proteins, to obtain catalytic activity at very high temperatures, and to improve the stability, pharmacokinetics, and biodistribution of therapeutic proteins. FDA-approved PEGylated proteins include Adagen, On-caspar, PEGASYS, and PEG-INTRON.
Our Bioconjugation Services for Self-assembly Protein-Polymer Conjugates
Synthetic Strategies for Self-assembly Protein-polymer Conjugates
Bioconjugation success relies on the availability of complementary reactive targets, termed reactive protein handles, on the surface of protein or biomacromolecule. Typically, these reactive protein handles are free amino acid side chains including amine, hydroxy, and thiol groups or functional groups present at the protein termini. As a professional polymerization service provider, BOC Sciences specializes in the generation protein-polymer conjugates via controlled radical polymerization (CRP), atom transfer radical polymerization (ATRP), reversible addition-fragmentation chain transfer (RAFT) polymerization and ring opening metathesis polymerization (ROMP) technologies to meet your research and production needs. Our ATRP and RAFT polymerization techniques allow for facile end-group modification of polymer chains, which can be functionalized to include reactive groups for bioorthogonal reactions. Our techniques for generating protein-polymer bioconjugates include:
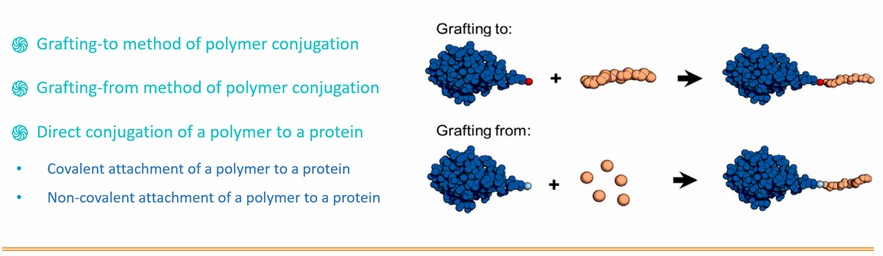
Designing Strategies for Self-assembly Protein-polymer Conjugates
One technique to achieve assembly of protein-polymer conjugates is to engineer amphiphilicity via modifying a hydrophilic protein with ahydrophobic polymer, generating a block copolymer like-molecule. Study shows that polymer blocks and protein blocks have different effects on protein-polymer self-assembly. BOC Sciences' protein-polymer conjugate self-assembly process platform provides protein and polymer screening and protein-polymer conjugate design services for customer research needs. For instance, protein cages such as ferritin and viral envelopes are common in nature, and our polymer bioconjugation technology supports the formation of nanostructures through the self-assembly of individual protein subunits. Additionally, we have developed a variety of block copolymer designs, including nonlinear, multiblock, and tapered block copolymers, especially diblock copolymers.
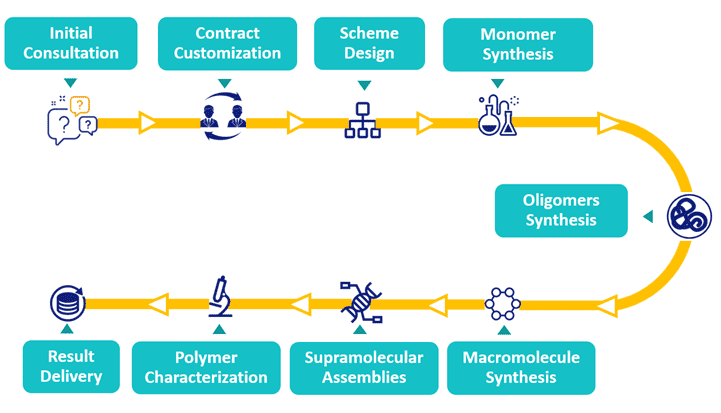
Our Polymer Bioconjugation Workflow
References
- Weil, T. et al. Polymer bioconjugates: modern design concepts toward precision hybrid materials. Progress in Polymer Science. 2020, 105: 101241.
- Stevens, C.A. et al. Self-assembly of protein-polymer conjugates for drug delivery. Advanced Drug Delivery Reviews. 2021, 174: 447-460.

 Fig. 1. Schematic representations of common self-assembled protein-polymer conjugates architectures (Advanced Drug Delivery Reviews. 2021, 174: 447-460).
Fig. 1. Schematic representations of common self-assembled protein-polymer conjugates architectures (Advanced Drug Delivery Reviews. 2021, 174: 447-460).




