- CAS: 91-97-4
- Molecular Weight: 264.28
- Molecular Formula: C16H12N2O2
- CAS: 7517-76-2
- Molecular Weight: 166.17700
- Molecular Formula: C8H10N2O2
- CAS: 67483-13-0
- Molecular Weight: 498.48
- Molecular Formula: C16H8N2Na2O6S4
- CAS: 5331-87-3
- Molecular Weight: 305.12
- Molecular Formula: C14H6Cl2N2O2
- CAS: 38661-72-2
- Molecular Weight: 194.23
- Molecular Formula: C10H14N2O2
- CAS: 3634-83-1
- Molecular Weight: 188.18
- Molecular Formula: C10H8N2O2
- CAS: 3173-72-6
- Molecular Weight: 210.19
- Molecular Formula: C12H6N2O2
- CAS: 28679-16-5
- Molecular Weight: 210.27
- Molecular Formula: C11H18N2O2
- CAS: 13879-35-1
- Molecular Weight: 252.35
- Molecular Formula: C14H24N2O2
- CAS: 123-61-5
- Molecular Weight: 160.13
- Molecular Formula: C8H4N2O2
- CAS: 104-49-4
- Molecular Weight: 160.13
- Molecular Formula: C8H4N2O2
- CAS: 10224-18-7
- Molecular Weight: 386.25
- Molecular Formula: C17H8 F6 N2 O2
- CAS: 10124-86-4
- Molecular Weight: 196.25
- Molecular Formula: C10H16N2O2
Introduction

Isocyanate monomers are a class of low molecular weight aromatic and aliphatic compounds containing isocyanate groups (-NCO). Due to the -NCO functional group, isocyanate derivatives are easy to react with active hydrogen compounds such as water, methanol, and amines, and have high reactivity. Presently, toluene diisocyanate (TDI) and diphenylmethane diisocyanate (MDI) are widely used, with the largest annual consumption.
Application
Mono-isocyanate monomers is an important intermediate in organic synthesis, which can be produced into a series of carbamate insecticides, fungicides, herbicides, and also utilized to enhance the waterproofing of plastics, fabrics, leather, etc. Difunctional isocyanate monomers can be used to synthesize high-performance polyurethane materials, including foam, rubber, elastic fibers, coatings, adhesives, synthetic leather, artificial wood, etc.[1]
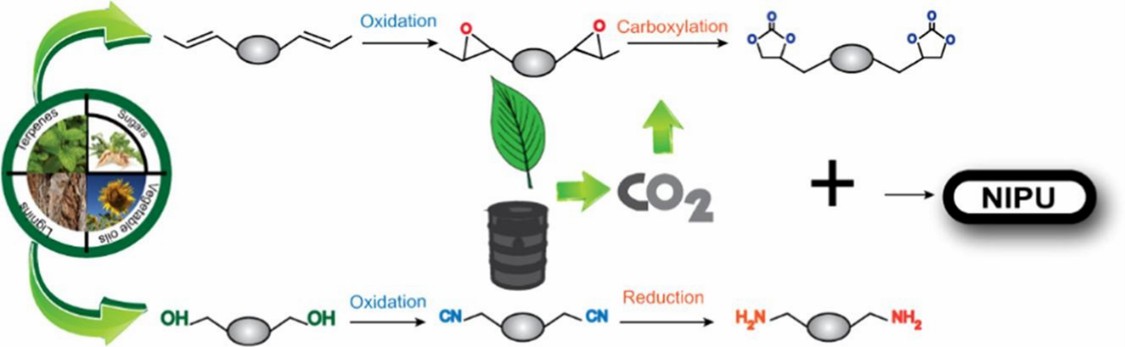 Fig. 1. Preparation of polyurethane by mono-isocyanate (European Polymer Journal. 2019, 118: 668-684).
Fig. 1. Preparation of polyurethane by mono-isocyanate (European Polymer Journal. 2019, 118: 668-684).
The most widely used isocyanate compound, toluene diisocyanate (TDI), is a colorless, irritating liquid.[2] TDI is mainly used in polyurethane foam, coatings, synthetic rubber, insulating paint, adhesives, etc. In addition, diphenylmethane diisocyanate (MDI) can also be used to produce rigid polyurethane foam, synthetic fibers, synthetic rubber, synthetic leather, adhesives, etc.
Diphenylmethane diisocyanate (MDI) is a polyurethane material synthesized from isocyanate, polyol and its additives. With its excellent performance and diverse varieties, MDI is unique among many synthetic materials and has become one of the fastest growing materials today. Its main products are widely used in polyurethane coatings, waterproof materials, sealing materials, ceramic materials, etc. In addition, polyurethane foams made from MDI can be used for heating (cooling), building materials, vehicles and ships. The finished product can be made into automobile baffle, synthetic leather, polyurethane elastic fiber, etc.
MDI and TDI are substitutes for each other, and both are raw materials for the production of polyurethane. The price of MDI is slightly lower than that of TDI, but the density of foaming production is higher than that of TDI system, and the polyurethane products formed by MDI have relatively good formability. If you are interested in our isocyanate monomers, please contact us immediately!
References
- Ghasemlou, M. et al. Bio-based routes to synthesize cyclic carbonates and polyamines precursors of non-isocyanate polyurethanes: A review. European Polymer Journal. 2019, 118: 668-684.
- Zhuang, J. et al. Bronchial epithelial pyroptosis promotes airway inflammation in a murine model of toluene diisocyanate-induced asthma. Biomedicine & Pharmacotherapy. 2020, 125: 109925.













![Benzene,1,1'-[2,2,2-trifluoro-1-(trifluoromethyl)ethylidene]bis[4-isocyanato- (9CI)](https://resource.bocsci.com/structure/10224-18-7.gif)


 Fig. 1. Preparation of polyurethane by mono-isocyanate (European Polymer Journal. 2019, 118: 668-684).
Fig. 1. Preparation of polyurethane by mono-isocyanate (European Polymer Journal. 2019, 118: 668-684).












