Polymer Micelle Platforms for Sustained and Targeted Drug Delivery
The formulation of advanced therapeutic agents frequently encounters profound physicochemical barriers, most notably extreme hydrophobicity and rapid systemic degradation. To overcome these inherent limitations, researchers are increasingly turning to self-assembling macromolecular architectures. Polymer micelles have emerged as highly sophisticated, thermodynamically stable nanocarriers engineered to revolutionize payload administration. Constructed from rationally designed amphiphilic block copolymers, these core-shell platforms provide a precisely tuned microenvironment for solubilizing lipophilic molecules while strictly shielding them from premature clearance. By modulating polymer topologies, molecular weights, and surface functionalities, formulators can dictate sustained release kinetics and facilitate highly specific active targeting mechanisms.
Resources
Introduction to Polymer Micelles in Drug Delivery
Polymer micelles have emerged as highly sophisticated nanoscale platforms designed to overcome the intrinsic limitations of administering poorly soluble therapeutic agents. These structures are thermodynamically stable, self-assembling colloidal particles characterized by a core-shell architecture. Driven by hydrophobic interactions in aqueous environments, amphiphilic polymers spontaneously aggregate above their critical micelle concentration. The resulting architecture features a hydrophobic core capable of encapsulating lipophilic molecules and a hydrophilic corona that provides steric stabilization and prolongs circulation time in biological fluids. By engineering the physicochemical properties of the constituent polymers, researchers can fine-tune these supramolecular assemblies to achieve sustained, controlled, and highly targeted delivery of various active pharmaceutical ingredients.
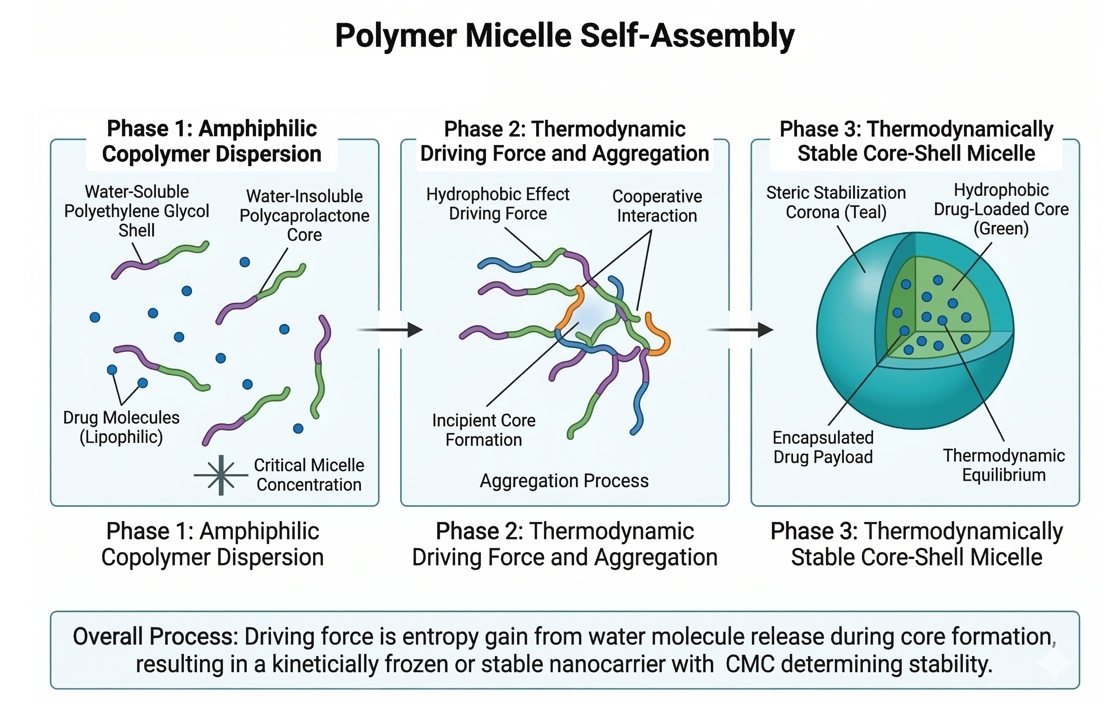 Fig. 1. Self-assembly process of amphiphilic copolymers into drug-loaded micelles (BOC Sciences Authorized).
Fig. 1. Self-assembly process of amphiphilic copolymers into drug-loaded micelles (BOC Sciences Authorized).
Challenges in Conventional Drug Delivery Systems
Traditional administration of active molecules frequently encounters significant biological and physicochemical hurdles. A primary obstacle is the poor aqueous solubility of many synthetic and biologically derived therapeutic agents, leading to suboptimal bioavailability and erratic pharmacokinetic profiles. Furthermore, conventional formulations often suffer from rapid clearance by the reticuloendothelial system, premature degradation by systemic enzymes, and a lack of tissue specificity. This indiscriminate distribution typically necessitates higher dosing to achieve an effect at the target site, which consequently increases the risk of systemic toxicity and off-target side effects.
Advantages of Polymer Micelles Over Other Carriers
Compared to liposomes, metallic nanoparticles, and solid lipid nanoparticles, polymer micelles offer unique structural and functional benefits. Their characteristically small size, typically ranging from 10 to 100 nanometers, allows for superior tissue penetration and reduced recognition by macrophages. The structural versatility of synthetic polymers enables precise control over the micellar size, surface charge, and drug-loading capacity. Additionally, polymeric micelles exhibit an exceptionally low critical micelle concentration, meaning they remain stable and intact even under severe dilution following systemic administration, preventing the premature burst release of their payload.
Design Considerations for Polymeric Micelle Formulations
Developing an optimal polymeric micelle requires a rigorous evaluation of thermodynamic and kinetic stability. Formulators must balance the hydrophilic-lipophilic balance to ensure proper self-assembly while maximizing drug encapsulation efficiency. The choice of the hydrophilic block, commonly polyethylene glycol, dictates the degree of steric hindrance and resistance to protein adsorption. Meanwhile, the hydrophobic block dictates the core environment, influencing drug compatibility, loading capacity, and the specific mechanism of release. Additional considerations include selecting polymers with appropriate molecular weights to ensure complete renal clearance and minimal bioaccumulation.
Functional Roles of Polymer Micelles in Targeted Delivery
Polymer micelles operate not merely as passive vehicles but as active participants in the pharmacokinetic lifecycle of a therapeutic agent. Through careful selection of monomeric building blocks and precise control over polymer architecture, these nanocarriers perform a variety of distinct, highly engineered functions. From mitigating solubility issues to enabling spatial and temporal control over payload release, the functional capabilities of polymeric micelles are driven by thermodynamic interactions and advanced surface chemistry.
- Solubilization of Hydrophobic Drugs: The hydrophobic core of a polymer micelle acts as a highly efficient microenvironment for the solubilization of lipophilic molecules. By partitioning into the core, therapeutic agents that are otherwise insoluble in aqueous media can be administered intravenously without the need for toxic cosolvents or aggressive surfactants. The high core capacity ensures that a significant payload can be delivered in a highly concentrated, yet stable, colloidal dispersion.
- Controlled and Sustained Drug Release: Polymer micelles excel at regulating the release kinetics of their encapsulated payloads. Release occurs through a combination of drug diffusion out of the polymeric core and the gradual degradation or dissociation of the micelle matrix. By adjusting the cross-linking density, core crystallinity, and polymer chain length, researchers can engineer formulations that provide a sustained release profile, maintaining localized concentrations over extended periods and minimizing the peak-and-trough fluctuations characteristic of free drug administration.
- Enhanced Stability and Protection from Degradation: Once encapsulated within the dense hydrophobic core, active pharmaceutical ingredients are physically shielded from enzymatic degradation, hydrolytic breakdown, and neutralizing binding proteins present in systemic circulation. The hydrophilic corona acts as a dense hydration layer, preventing opsonization and subsequent phagocytic clearance. This protective mechanism preserves the structural integrity and pharmacological activity of fragile molecules, including peptides and nucleic acids, during transit.
- Targeted Delivery and Improved Cellular Uptake: Polymer micelles exploit both passive and active targeting strategies. Their nanoscale dimensions allow them to passively accumulate in specific tissues exhibiting highly fenestrated vasculature and impaired lymphatic drainage, a phenomenon known as the enhanced permeability and retention effect. For active targeting, the terminal ends of the hydrophilic corona can be bioconjugated with specific ligands that recognize and bind to overexpressed receptors on target cell membranes, triggering receptor-mediated endocytosis and significantly increasing intracellular drug accumulation.
- Stimuli-Responsive Micelles: Advanced micellar platforms can be engineered to undergo structural destabilization in response to specific microenvironmental triggers. pH-responsive micelles utilize protonatable groups or acid-cleavable bonds to release their cargo in the acidic environments of endosomes or specific pathological sites. Temperature-responsive variants employ polymers with a lower critical solution temperature, causing them to undergo a phase transition and release drugs upon localized heating. Redox-responsive systems leverage disulfide bonds that rapidly cleave in the presence of high intracellular glutathione concentrations.
- Modular Modification: The synthetic nature of polymer chemistry allows for highly modular modifications. Researchers can functionalize the shell with imaging agents, contrast dyes, or multiple synergistic drugs to create theranostic platforms. Click chemistry and other highly efficient conjugation techniques enable the attachment of these functional moieties without disrupting the fundamental self-assembly thermodynamics of the underlying block copolymers.
Types of Polymer Micelles
The architectural and functional diversity of polymer micelles stems from the extensive library of available synthetic methodologies and monomer combinations. By precisely manipulating macromolecular topology, block ratios, and intermolecular forces, polymer chemists can engineer supramolecular assemblies tailored to highly specific physicochemical requirements. This structural versatility dictates the thermodynamic stability, payload retention, and ultimate biological fate of the delivery platform. Depending on the synthetic strategy, formulators can categorize these nanocarriers into several distinct classes, each offering unique advantages for encapsulating and shielding complex therapeutic molecules.
Amphiphilic Block Copolymer Micelles
Amphiphilic block copolymers represent the most rigorously studied and widely utilized class of micellar forming materials. These systems typically consist of linear AB di-block, ABA tri-block, or BAB tri-block topologies, where A represents a hydrophilic segment and B represents a hydrophobic segment. The self-assembly of these macromolecules is fundamentally driven by the hydrophobic effect. In an aqueous medium, the hydrophobic blocks aggregate to minimize unfavorable interactions with water, a thermodynamically spontaneous process governed by the standard free energy of micellization.
The structural integrity of the resulting core-shell architecture is heavily dependent on the Flory-Huggins interaction parameter between the hydrophobic block and the encapsulated payload. A highly compatible core restricts unimer exchange kinetics, leading to a kinetically frozen state that prevents premature drug leakage. The hydrophilic corona, most commonly constructed from polyethylene glycol, provides immense steric repulsion, dictated by the spatial volume of the hydrated polymer chains, effectively preventing inter-micellar aggregation and protein adsorption.
Graft Copolymer Micelles
Graft copolymers introduce complex branching into the macromolecular architecture, featuring a central polymer backbone covalently bonded to multiple side chains of contrasting solubility. This topological arrangement significantly alters the self-assembly dynamics compared to linear block copolymers. When the backbone is hydrophilic and the grafted chains are hydrophobic, intermolecular hydrophobic interactions drive the formation of multi-molecular micelles. The high localized density of the side chains often results in highly compact, rigid core structures that exhibit exceptional loading capacities for lipophilic compounds.
Perhaps the most significant advantage of graft architectures is their ability to form unimolecular micelles. By synthesizing a highly branched, star-like structure or a densely grafted brush polymer, a single macromolecule can undergo intramolecular folding to enclose its hydrophobic domains within a hydrophilic outer layer. Because self-assembly is confined to a single molecule, unimolecular micelles possess a critical micelle concentration effectively equal to zero. This grants them unparalleled thermodynamic stability, ensuring they remain structurally intact regardless of the severe dilution experienced following systemic administration.
Polyelectrolyte Copolymer Micelles
Polyelectrolyte copolymer micelles, frequently referred to as polyion complex micelles, assemble through a fundamentally different mechanism than their amphiphilic counterparts. Instead of hydrophobic interactions, these structures are driven by cooperative electrostatic complexation between a charged block copolymer and an oppositely charged polyion. When a diblock copolymer containing a neutral hydrophilic segment and a polycationic segment is mixed with a polyanionic therapeutic molecule, such as a nucleic acid or a charged peptide, the oppositely charged domains rapidly associate.
This complexation neutralizes the charges, creating a condensed, water-insoluble core, while the neutral hydrophilic blocks extend outward to form the stabilizing corona. The primary thermodynamic driving force for this assembly is the massive entropic gain resulting from the release of low-molecular-weight counterions into the bulk solution during complexation. Polyelectrolyte micelles are exceptionally sensitive to ionic strength and pH variations, making them highly suitable for stimuli-responsive delivery strategies where environmental changes trigger the disassembly of the polyion core and the subsequent release of the macro-molecular payload.
Non-Covalently Linked Micelles
Traditional block copolymers rely on robust, irreversible covalent bonds to connect their constituent domains. In contrast, non-covalently linked micelles utilize highly specific, dynamic supramolecular interactions to tether the hydrophilic and hydrophobic segments. These interactions include host-guest inclusion complexes (such as the interaction between cyclodextrin cavities and adamantane moieties), highly directional hydrogen bonding arrays, and transition metal-ligand coordination complexes.
Because these supramolecular linkages are dynamic and reversible, they imbue the micellar platform with sophisticated stimuli-responsive behaviors. By carefully engineering the association constant of the non-covalent bond, formulators can design micelles that remain stable under normal physiological conditions but undergo rapid structural dissociation upon exposure to specific environmental triggers. For example, a decrease in localized pH or the introduction of a competitive binding agent can sever the non-covalent linkage, separating the corona from the core and triggering an immediate, localized burst release of the encapsulated therapeutic agent.
Biodegradable vs Non-Biodegradable Micelle Systems
The selection between biodegradable and non-biodegradable structural materials dictates the long-term biological clearance and safety profile of the delivery vehicle. Biodegradable micelle systems are constructed from polymers containing chemically labile linkages within their backbone. Common examples include polyesters like poly-lactic acid and poly-caprolactone, or polyamino acids, which undergo predictable hydrolytic or enzymatic degradation. Over time, the massive macromolecular network breaks down into small, biologically benign metabolites that are easily metabolized or cleared by regular physiological pathways, virtually eliminating the risk of long-term tissue bioaccumulation.
Non-biodegradable systems, such as certain polyether-based formulations, utilize polymers with chemically inert backbones that resist physiological breakdown. While these materials often provide superior chemical stability and prolonged shelf-life, their formulation requires strict adherence to physiological clearance thresholds. Because the polymers cannot degrade, their molecular weight must be rigorously maintained below the renal filtration cutoff (typically around 40 to 50 kDa). Exceeding this threshold significantly increases the probability of macromolecular accumulation within the reticuloendothelial system, potentially leading to long-term toxicity.
Looking for Custom Polymer Micelles?
From amphiphilic block copolymers to stimuli-responsive systems, we offer tailored synthesis and formulation solutions for drug delivery research.
Formulation and Design Strategies of Polymer Micelles
The successful translation of a theoretical supramolecular concept into a viable, stable, and highly efficient delivery platform requires rigorous formulation optimization. Formulators must employ strategic methodologies to govern the self-assembly process, ensure maximal drug loading, maintain batch-to-batch consistency, and engineer robust stability under both storage and physiological conditions. The ultimate performance of the nanocarrier depends entirely on the meticulous orchestration of these fundamental design parameters.
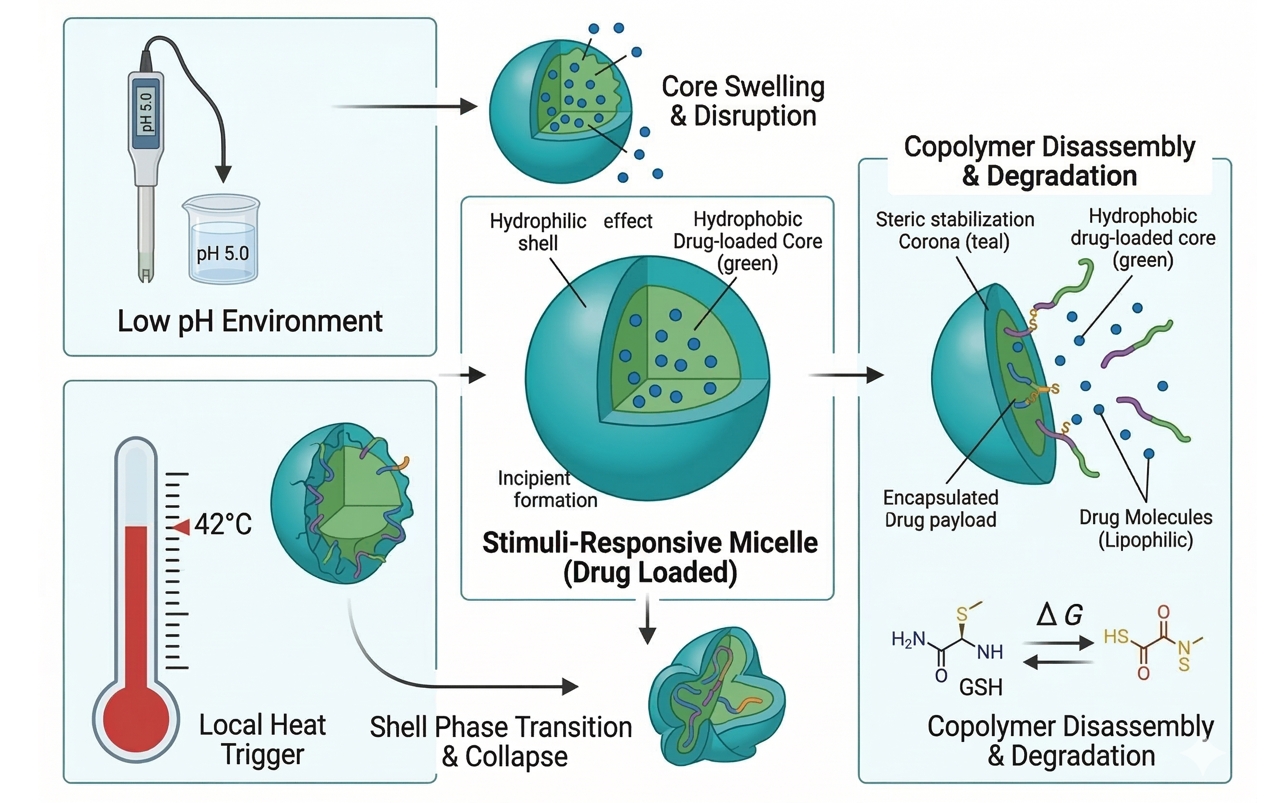 Fig. 2. Mechanisms of drug release from stimuli-responsive polymer micelles (BOC Sciences Authorized).
Fig. 2. Mechanisms of drug release from stimuli-responsive polymer micelles (BOC Sciences Authorized).
Drug–Micelle Compatibility and Loading Optimization
Maximizing the encapsulation efficiency and total drug loading capacity requires a profound understanding of the thermodynamic compatibility between the lipophilic payload and the micellar core-forming block. Formulators quantitatively evaluate this compatibility using the Flory-Huggins interaction parameter alongside Hansen Solubility Parameters. By minimizing the interaction parameter between the drug and the core polymer, researchers indicate a high thermodynamic affinity. This strong affinity suppresses the crystallization of the active ingredient and maximizes its partitioning into the hydrophobic core. This compatibility is fundamentally determined by evaluating and matching the cohesive energy densities and solubility parameters of the respective components.
To further optimize loading beyond simple hydrophobic partitioning, chemists often engineer specific intermolecular interactions within the core microenvironment. For instance, incorporating aromatic pendant groups into the hydrophobic polymer backbone can induce strong π-π stacking interactions with aromatic therapeutic molecules. Alternatively, formulators may employ a prodrug strategy, chemically conjugating the active pharmaceutical ingredient directly to the polymer backbone via hydrolytically labile linkers, thereby achieving near-perfect theoretical loading capacities and completely preventing premature drug leakage.
Controlling Release Kinetics and Duration
Engineering the precise temporal release of a payload is a critical objective in micelle formulation. The release profile is governed by the synergistic effects of molecular diffusion through the polymer matrix and the structural dissociation or degradation of the micelle itself. Researchers frequently model these profiles by mathematically evaluating the fractional drug release over time as a function of the polymer network kinetcs and fundamental diffusional mechanisms.
The physical state of the micelle core drastically influences the diffusion coefficient. A core formed by a polymer with a high glass transition temperature remains in a glassy, rigid state at physiological temperatures, severely restricting macromolecular chain mobility and resulting in highly sustained, diffusion-limited release over extended durations. Conversely, a polymer with a low glass transition temperature forms a rubbery core, facilitating rapid diffusion and faster release kinetics. To counteract the burst release phenomenon often observed upon systemic dilution, formulators frequently employ core-crosslinking strategies. By introducing stimuli-responsive disulfide bridges or applying localized ultraviolet irradiation to photoreactive core segments after self-assembly, the core becomes a covalently locked network, ensuring payload retention until the micelle encounters specific intracellular triggers.
Stability, Sterility, and Storage Considerations
Aqueous micellar dispersions exist in a state of dynamic equilibrium and are highly susceptible to physical instability phenomena, such as Ostwald ripening, macro-aggregation, and premature dissociation over extended periods. To ensure a commercially viable shelf-life, these colloidal systems are routinely subjected to lyophilization, transitioning the formulation into a stable, dry powder. This freeze-drying process subjects the delicate supramolecular architecture to severe structural stresses. To prevent irreversible micellar fusion during the freezing phase, formulators must strategically incorporate cryoprotectants and lyoprotectants, such as the disaccharides trehalose or sucrose. These specialized excipients form an amorphous glass matrix around the micelles and substitute for water molecules via extensive hydrogen bonding, perfectly preserving the hydration shell and ensuring rapid, uniform reconstitution upon the addition of aqueous media.
Furthermore, maintaining sterility is a paramount formulation hurdle. Due to the high thermodynamic energy involved, heat-based terminal sterilization methods, like autoclaving, typically induce polymer degradation or irreversible structural collapse. Similarly, gamma irradiation can trigger unintended polymer chain scission or excessive cross-linking. Therefore, strict aseptic processing combined with terminal membrane filtration through sterile filters remains the gold standard, leveraging the nanoscale dimensions of the micelles to achieve sterility without compromising physicochemical integrity.
In Vitro and In Vivo Evaluation of Micelle-Based Delivery Systems
Comprehensive physicochemical characterization and rigorous preclinical evaluation are mandatory prerequisites for validating any micellar platform. In vitro assessments rely on an array of advanced analytical techniques. Dynamic light scattering and electrophoretic light scattering determine the hydrodynamic radius, size distribution, and zeta potential, providing critical data on colloidal stability and surface charge. Morphological confirmation is typically achieved through cryogenic transmission electron microscopy or atomic force microscopy. Furthermore, gel permeation chromatography and high-performance liquid chromatography are utilized to quantify drug loading efficiency and map precise release kinetics across various simulated biological buffers.
Following rigorous in vitro validation, the formulations undergo comprehensive in vivo pharmacokinetic and biodistribution profiling using advanced animal models. These evaluations map the complex concentration-time curves to calculate critical parameters such as the elimination half-life, total body clearance, and area under the curve. By covalently attaching near-infrared fluorophores or incorporating radiolabeled isotopes into the polymer backbone, researchers can execute precise spatiotemporal tracking of the nanocarriers. This enables quantitative mapping of tissue-specific accumulation, verifying the efficiency of the enhanced permeability and retention effect or evaluating the success of active targeting ligands compared to standard, unencapsulated delivery methods.
Applications of Polymer Micelle Targeted Delivery
The transition from passive accumulation, which relies solely on the enhanced permeability and retention effect, to active targeting represents a monumental leap in precision delivery. Functionalizing the hydrophilic corona of polymer micelles with specific targeting ligands allows these supramolecular platforms to selectively interact with receptors overexpressed on specific cell membranes. This precise molecular recognition triggers receptor-mediated endocytosis, facilitating the direct intracellular translocation of the encapsulated payload. By strategically selecting the targeting moiety, formulators can dictate the biodistribution profile, minimize off-target accumulation, and significantly enhance the localized concentration of the active pharmaceutical ingredient at the highly specific cellular site without altering the thermodynamic stability of the micellar core.
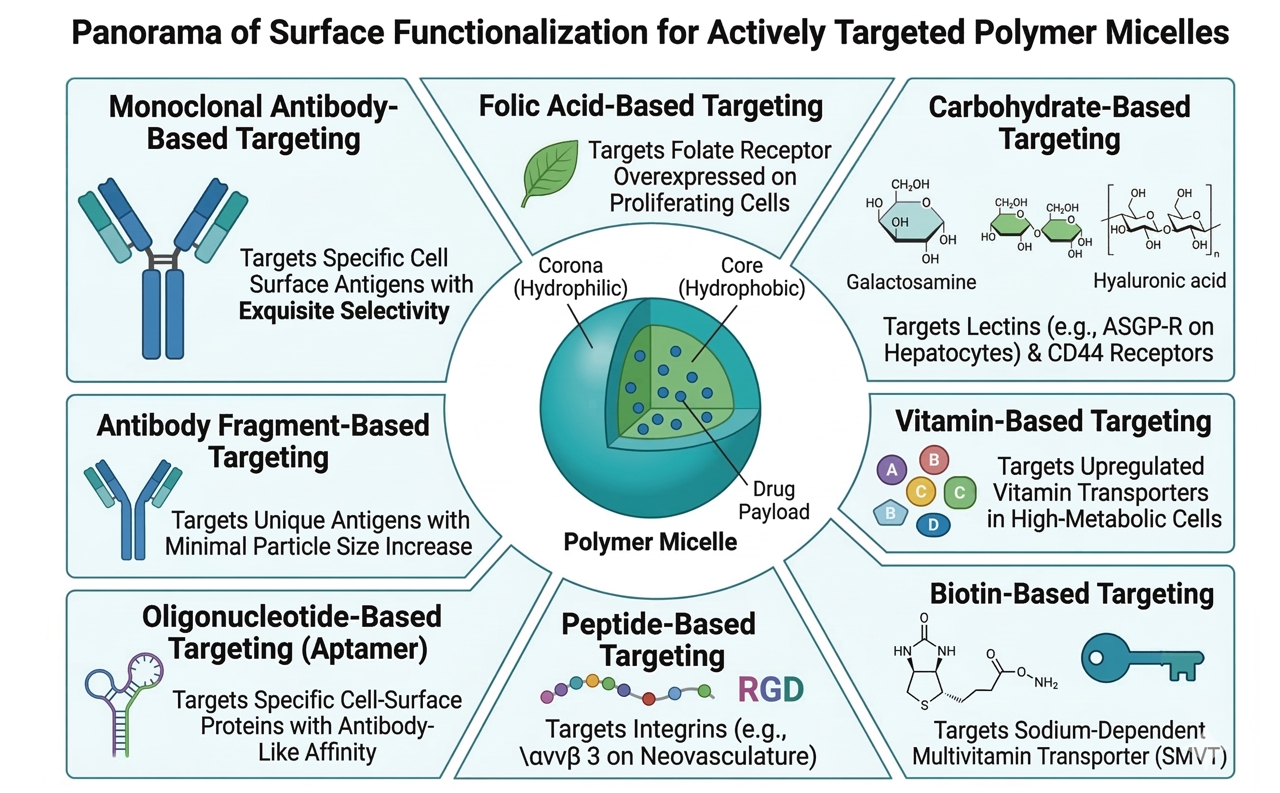 Fig. 3. Surface functionalization strategies for actively targeted polymer micelle platforms (BOC Sciences Authorized).
Fig. 3. Surface functionalization strategies for actively targeted polymer micelle platforms (BOC Sciences Authorized).
Carbohydrate-Based Targeting
Carbohydrate-functionalized micelles exploit the high binding affinity of specific cell-surface lectins and carbohydrate-recognition domains. Monosaccharides like galactose and N-acetylgalactosamine are extensively utilized to target the asialoglycoprotein receptor, a highly abundant receptor on hepatocytes, facilitating rapid and highly specific hepatic uptake. Polysaccharides, such as hyaluronic acid, serve a dual and highly efficient purpose: they can function as the primary hydrophilic block of the micelle while simultaneously acting as a highly specific ligand for CD44 and RHAMM receptors, which are frequently upregulated on rapidly proliferating cells. This multivalent presentation of carbohydrate moieties on the micellar surface significantly enhances the overall binding avidity compared to monovalent ligands, ensuring robust cellular adhesion.
Vitamin-Based Targeting
Cellular proliferation requires a continuous supply of essential vitamins, leading rapidly dividing cells to dramatically upregulate specific vitamin receptors on their surfaces. Conjugating vitamins to the terminal ends of the micellar corona allows the delivery platform to hijack these naturally occurring nutrient transport mechanisms. Because vitamins are endogenous molecules, they inherently exhibit extremely low immunogenicity and excellent biocompatibility profiles. Furthermore, their low molecular weight ensures that their chemical conjugation does not negatively impact the critical micelle concentration, the core-shell thermodynamics, or the overall hydrodynamic radius of the underlying polymer assembly.
Folic Acid-Based Targeting
Folic acid, widely known as Vitamin B9, is a premier targeting ligand due to its exceptionally high binding affinity for the folate receptor. This glycosylphosphatidylinositol-anchored protein is significantly overexpressed on the apical surfaces of numerous highly active cell types but remains highly restricted in healthy tissues. Folic acid conjugation typically involves the creation of a stable amide bond or an ester linkage between the gamma-carboxyl group of the folate molecule and the terminal functional groups of the hydrophilic polymer block, such as polyethylene glycol. This strategic chemical orientation ensures the critical pterin and p-aminobenzoic acid moieties remain entirely accessible for optimal receptor engagement and subsequent internalization.
Biotin-Based Targeting
Biotin, also identified as Vitamin B7, specifically targets the sodium-dependent multivitamin transporter. This specific transporter is broadly distributed and heavily overexpressed in various tissues requiring elevated metabolic rates. The primary advantage of utilizing biotin as a targeting ligand lies in its robust chemical stability and its ability to withstand diverse and aggressive formulation stresses without losing target binding affinity. When integrated into the micellar corona, biotinylated polymers facilitate rapid cellular internalization while maintaining a strictly stealth profile in systemic circulation, effectively bypassing physiological barriers that typically trap non-targeted colloidal carriers.
Peptide-Based Targeting
Peptide ligands offer a highly customizable and synthetically scalable approach to active cellular targeting. Short amino acid sequences can be rationally designed or identified via phage display technologies to bind with high specificity to target cell surface integrins or transmembrane receptors. A prominent example is the Arg-Gly-Asp sequence, which exhibits strong, specific binding to alphavbeta3 integrins heavily localized on developing neovasculature. Additionally, incorporating cell-penetrating peptides, such as transactivating transcriptional activator sequences or polyarginine tracts, dramatically enhances the membrane translocation efficiency of the micelle, facilitating direct cytoplasmic delivery of macromolecules that would otherwise remain trapped within endosomal compartments.
Monoclonal Antibody-Based Targeting
Intact monoclonal antibodies provide unparalleled, exquisite specificity for unique cell surface antigens. By covalently attaching full-length antibodies to the distal ends of the micellar corona, formulators equip the nanocarrier with the precise recognition capabilities inherent to the adaptive immune system. While this strategy yields exceptional binding affinity, integrating large glycoproteins can significantly alter the physicochemical properties of the micelle. The addition of intact antibodies increases the overall hydrodynamic volume, potentially hindering deep tissue penetration, and requires highly controlled bioconjugation strategies to prevent the unintended cross-linking of adjacent micelles and subsequent macroscopic aggregation.
Antibody Fragment-Based Targeting
To circumvent the size limitations and potential pharmacokinetic drawbacks associated with full-length monoclonal antibodies, researchers frequently employ engineered antibody fragments, including single-chain variable fragments and antigen-binding fragments. These truncated proteins retain the highly specific, high-affinity antigen-binding regions of the parent molecule but possess a mere fraction of the molecular weight. Conjugating these distinct fragments to the micelle surface minimizes the increase in particle size, perfectly preserving the superior tissue penetration characteristics of the nanoscale assembly. Furthermore, the absence of the crystallizable fragment region significantly reduces the likelihood of unintended recognition and premature clearance by phagocytic cells of the reticuloendothelial system.
Oligonucleotide-Based Targeting
Aptamers represent a highly sophisticated class of targeting ligands composed of single-stranded DNA or RNA oligonucleotides. Through complex intramolecular base-pairing, aptamers fold into distinct, highly stable three-dimensional conformations capable of binding specific target proteins with affinities and selectivities directly rivaling those of monoclonal antibodies. Aptamer-functionalized micelles offer several distinct formulation advantages: they possess excellent tissue penetration capabilities, demonstrate virtually no immunogenicity, and allow for exact, reproducible chemical synthesis without the biological variability associated with complex antibody production. Moreover, their secondary structures can be engineered to undergo rapid conformational changes in response to specific microenvironmental stimuli, providing an additional layer of targeted release control.
Services
Polymer Micelle for Targeted Drug Delivery Support Services
BOC Sciences leverages extensive expertise in functional monomer design, controlled polymerization technologies, nanocarrier development, and advanced analytical platforms to support research and development of polymer micelles for targeted drug delivery. Our services cover key stages including amphiphilic polymer design, functional modification, micelle formulation, drug loading optimization, and physicochemical characterization. With versatile polymer synthesis technologies and comprehensive nanomaterial analysis capabilities, BOC Sciences can help develop polymer micelle systems with enhanced stability, high drug-loading efficiency, and improved targeting performance, supporting projects from early research to advanced drug delivery development.

Functional Monomer Design and Synthesis
- Custom design and synthesis of hydrophilic and hydrophobic functional monomers for amphiphilic polymer construction.
- Structural optimization of PEG-based, lactide, and ε-caprolactone monomers to regulate hydrophilic–hydrophobic balance.
- Development of pH-responsive, redox-responsive, and enzyme-responsive monomers for stimuli-responsive delivery systems.
- Supply of azide, alkyne, and maleimide functional monomers for click chemistry modification.

Amphiphilic Polymer Design and Controlled Polymerization
- Custom design and synthesis of amphiphilic block copolymers for polymer micelle formation.
- Support for controlled polymerization techniques, including RAFT, ATRP, and ring-opening polymerization (ROP).
- Custom synthesis of PEG-PLA, PEG-PCL, and PEG-PLGA polymers for drug delivery applications.
- Development of gradient copolymers, multiblock copolymers, and functionalized polymers.

Targeting Ligand Conjugation and Surface Functionalization
- Targeting ligand modification services for actively targeted polymer micelle systems.
- Conjugation of folic acid, RGD peptides, antibody fragments, and aptamers.
- Support for click chemistry and bioorthogonal conjugation strategies.
- PEGylation and surface hydrophilization to improve circulation stability and biocompatibility.

Polymer Micelle Formulation and Characterization
- Polymer micelle preparation services, including self-assembly and dialysis methods.
- Support for hydrophobic drug encapsulation and co-loading systems.
- Characterization of particle size, morphology, and critical micelle concentration (CMC).
- Evaluation of drug release kinetics and formulation stability.
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What are polymer micelles used for in drug delivery?
Polymer micelles are primarily utilized as advanced nanoscale carriers to transport hydrophobic or highly sensitive therapeutic molecules. They encapsulate these molecules within a protective core, significantly enhancing their aqueous solubility, improving their pharmacokinetic profiles, and preventing premature degradation by systemic enzymes prior to reaching the desired biological target.
-
How do polymer micelles improve solubility and stability of drugs?
They improve solubility by utilizing an amphiphilic architecture where the lipophilic drug partitions into the hydrophobic core of the micelle, creating a stable colloidal dispersion in water. Stability is achieved through the dense hydrophilic corona, typically composed of polyethylene glycol, which acts as a steric shield against hydrolytic breakdown and inhibits the binding of neutralizing proteins in the bloodstream.
-
Can polymer micelles provide targeted and controlled release?
Yes. Passive targeting is achieved through their nanoscale size, allowing accumulation in fenestrated tissues. Active targeting is achieved by attaching specific biological ligands to the micelle surface. Controlled release is dictated by the structural properties of the polymer core, permitting the steady outward diffusion of the drug or enabling stimuli-responsive release triggered by precise environmental changes like pH or temperature.
-
How are polymer micelles customized for specific therapeutic applications?
Customization is achieved through modular polymer chemistry. Formulators can select specific hydrophobic monomers to match the chemical properties of the drug payload for optimal loading. The release rate can be tuned by adjusting the polymer's molecular weight and cross-linking density. Furthermore, the surface can be conjugated with bespoke targeting ligands, imaging agents, or stabilizing molecules to fit the precise physiological requirements of the intended target site.

 Fig. 1. Self-assembly process of amphiphilic copolymers into drug-loaded micelles (BOC Sciences Authorized).
Fig. 1. Self-assembly process of amphiphilic copolymers into drug-loaded micelles (BOC Sciences Authorized). Fig. 2. Mechanisms of drug release from stimuli-responsive polymer micelles (BOC Sciences Authorized).
Fig. 2. Mechanisms of drug release from stimuli-responsive polymer micelles (BOC Sciences Authorized). Fig. 3. Surface functionalization strategies for actively targeted polymer micelle platforms (BOC Sciences Authorized).
Fig. 3. Surface functionalization strategies for actively targeted polymer micelle platforms (BOC Sciences Authorized).