Polymer Micelles: A Complete Guide to Design, Assembly, and Applications
05-20-2026
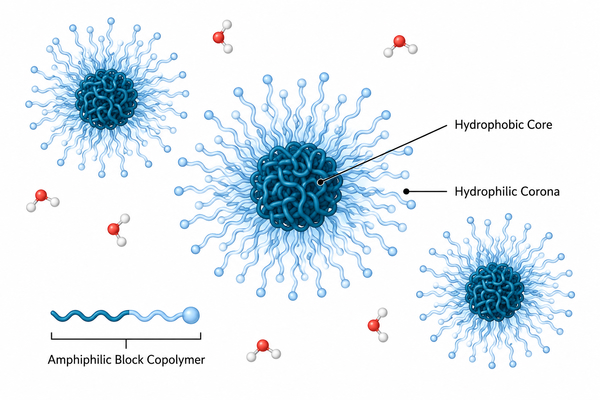
Polymer micelles are among the most important self-assembled nanostructures in modern drug delivery because they connect polymer architecture directly with formulation performance. Built from amphiphilic polymers, especially block copolymers, they organize in selective solvents into core-shell structures that can host poorly water-compatible molecules, protect sensitive cargo, and modulate release behavior through nanoscale compartmentalization. Their usefulness, however, does not come from nanosize alone. It comes from the fact that micelle formation, stability, loading, interfacial behavior, and route-specific performance all originate from molecular design choices that can be tuned deliberately. This page provides a pillar-level overview of polymer micelles, covering their structure, self-assembly, preparation, characterization, stability, and major application areas across drug delivery and related fields. The goal is not only to explain what polymer micelles are, but also to clarify how they should be designed, evaluated, and positioned relative to other carrier systems.
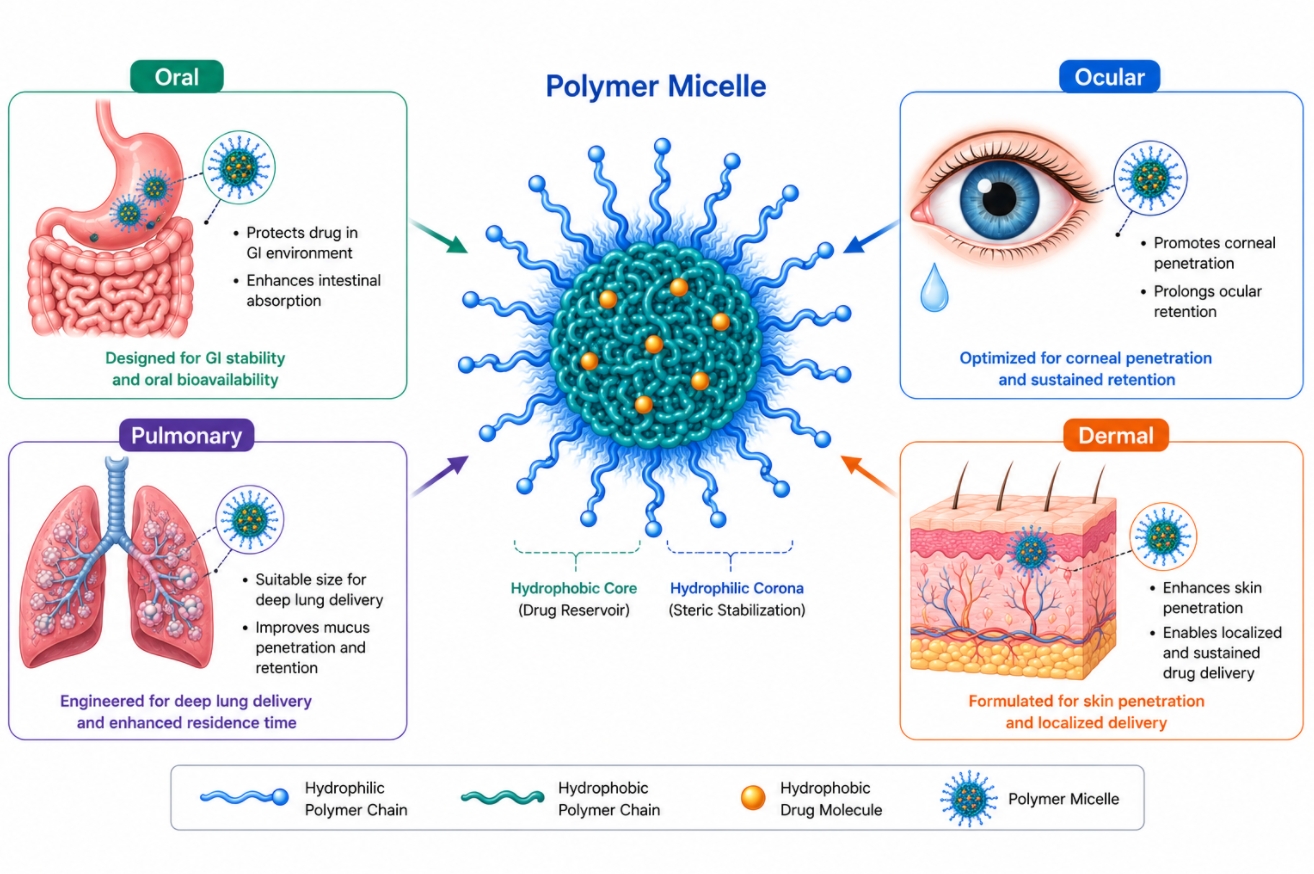
Comparing Polymer Micelles for Oral, Ocular, Pulmonary, and Dermal Delivery
05-20-2026
Polymer micelles are often described as versatile nanocarriers, but their real value becomes clearer when formulation is examined by delivery route rather than by platform alone. Oral, ocular, pulmonary, and dermal systems each expose polymer micelles to a different combination of fluid environment, clearance mechanism, barrier structure, and residence-time constraint. As a result, the same amphiphilic polymer that performs well in one route may behave poorly in another, even when the active compound and nominal particle size are unchanged. For this reason, route-specific micelle design should focus on the actual formulation problem being solved, such as improving dispersion of hydrophobic molecules, extending local residence, modulating interfacial interaction, or supporting more controlled local release. This page compares how polymer micelles function across four non-invasive delivery pathways and explains how barrier conditions, formulation variables, and evaluation priorities shift from one route to another. For additional background on polymer micelle formation and material use, Polymer Micelles: A Guide to Design, Assembly, and Applications introduces key concepts in polymer selection, assembly control, and functional performance.
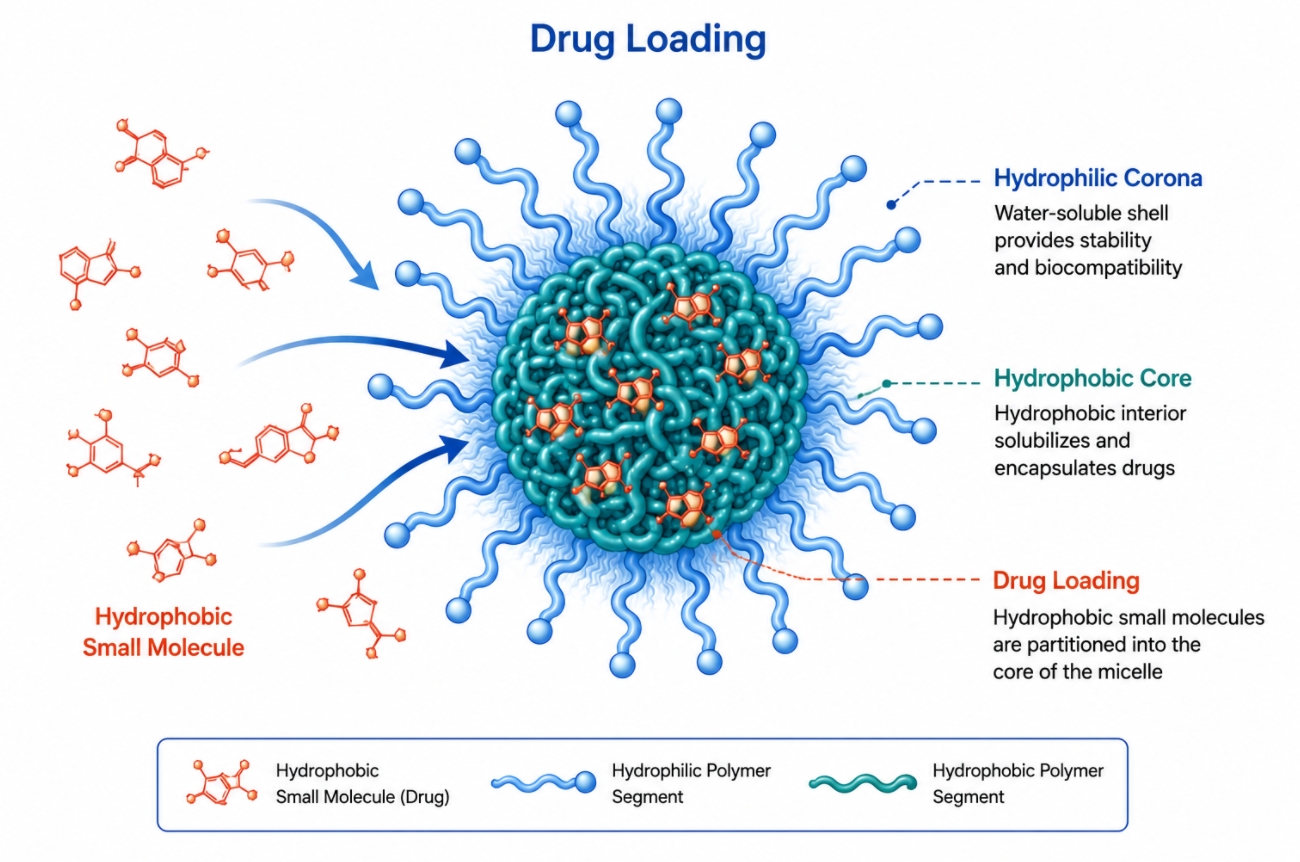
Polymer Micelles for Hydrophobic Small-Molecule Drugs: Formulation, Loading, and Release
05-20-2026
Polymer micelles are widely used to formulate hydrophobic small-molecule drugs because they offer more than a temporary increase in apparent solubility. Their value lies in creating a nanoscale core-shell environment where poorly water-compatible molecules can be partitioned, stabilized, and released in a more controlled way than they would be in simple cosolvent or surfactant systems. Yet successful formulation is never guaranteed by hydrophobicity alone. Drug structure, core chemistry, loading method, micelle stability, and release conditions all determine whether a micellar system becomes a practical formulation or merely a transient solubilization state. This page focuses on that formulation logic, explaining how polymer micelles accommodate hydrophobic small molecules, which types of compounds are most compatible, what controls leakage and controlled release, and how to evaluate whether a micelle formulation is truly robust.
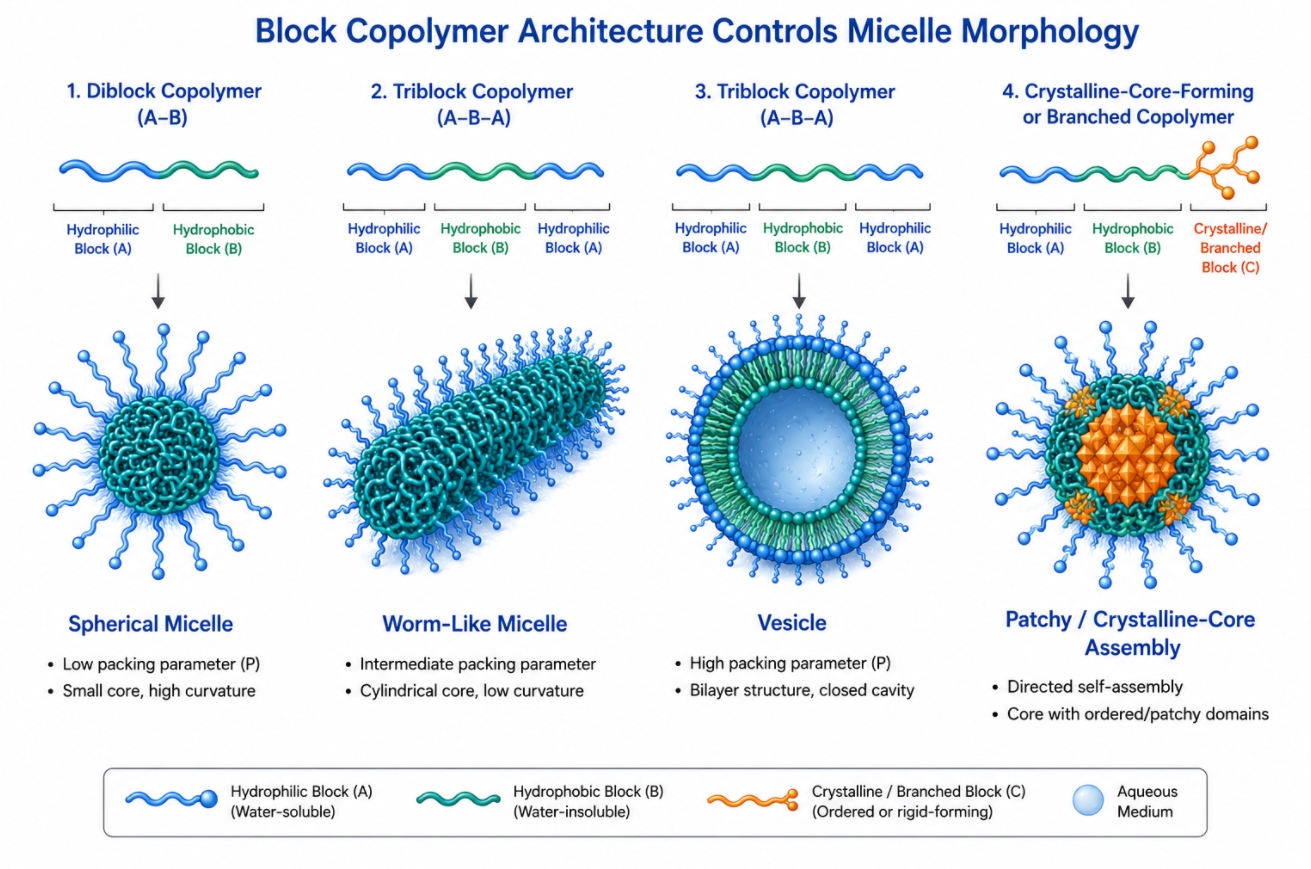
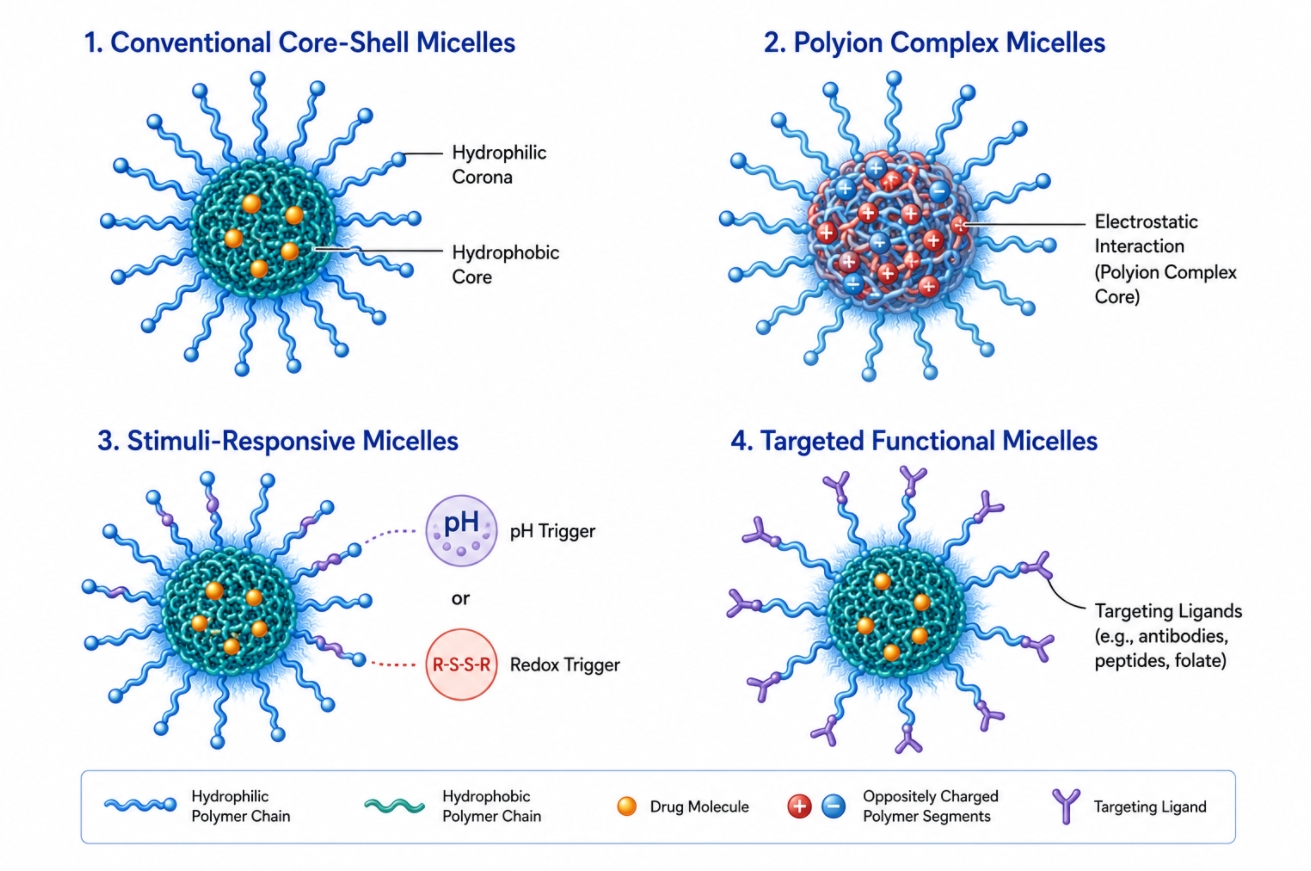
How Block Copolymer Architecture Controls Polymer Micelle Assembly?
05-20-2026
Block copolymer design sits at the center of polymer micelles development because the assembled structure is never independent from the molecular architecture that produces it. In selective solvents, amphiphilic block copolymers organize into nanoscale aggregates only when the balance between solvophilic and solvophobic segments, chain connectivity, topology, and intermolecular interactions favors assembly over molecular dispersion. This means polymer micelles are not generic carriers generated by any amphiphilic chain. Their size, morphology, stability, internal packing, and functional behavior are all consequences of design choices made at the block copolymer level. For researchers working with self-assembled nanostructures, the relevant question is therefore not only whether a polymer can form micelles, but how architecture controls what kind of micelles form, how persistent they remain, and how reliably they support a target formulation or materials task. Researchers developing amphiphilic polymer systems may find Polymer Micelles: A Guide to Design, Assembly, and Applications useful for understanding how molecular design influences micelle assembly, stability, and downstream applications.
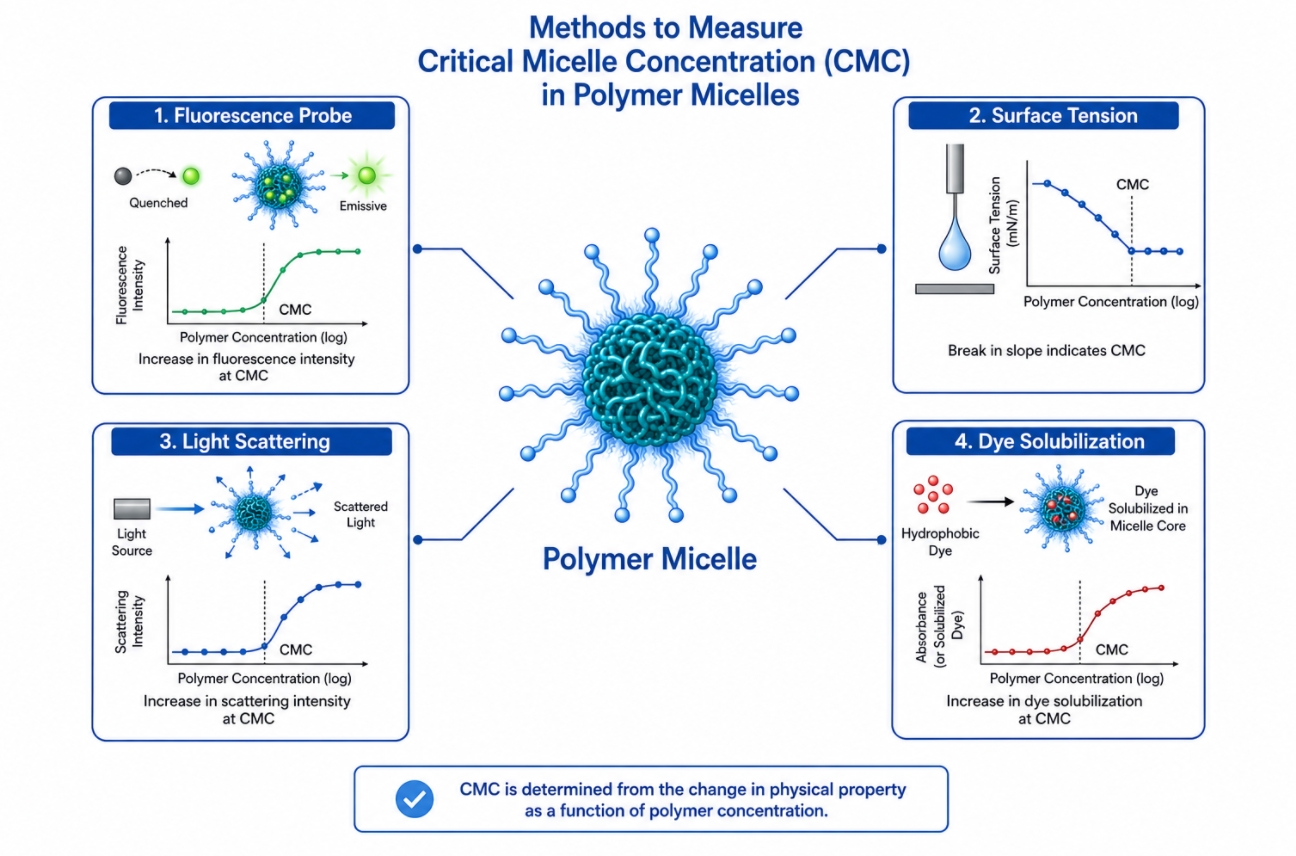
Critical Micelle Concentration in Polymer Micelles: Why It Matters for Self-Assembly and Stability
05-20-2026
Critical micelle concentration, or CMC, is one of the most frequently cited parameters in polymer micelles research, yet it is also one of the most frequently oversimplified. In amphiphilic polymer systems, CMC is not just a number describing when self-assembly begins. It also serves as an entry point for discussing how polymer structure affects micelle formation, how micelles respond to dilution, why some systems remain persistent while others dissociate, and how characterization data should be interpreted in formulation research. Because polymer micelles are widely used as self-assembled carriers and functional nanostructures, understanding CMC is essential for connecting molecular design with colloidal behavior. This page focuses on what CMC means in polymer micelles, how it is measured, why it matters for stability, and how it should be interpreted together with other structural and formulation parameters. For readers who want to better understand how amphiphilic polymers form nanoscale assemblies, Polymer Micelles: A Guide to Design, Assembly, and Applications explains key design principles, assembly behavior, and material applications.
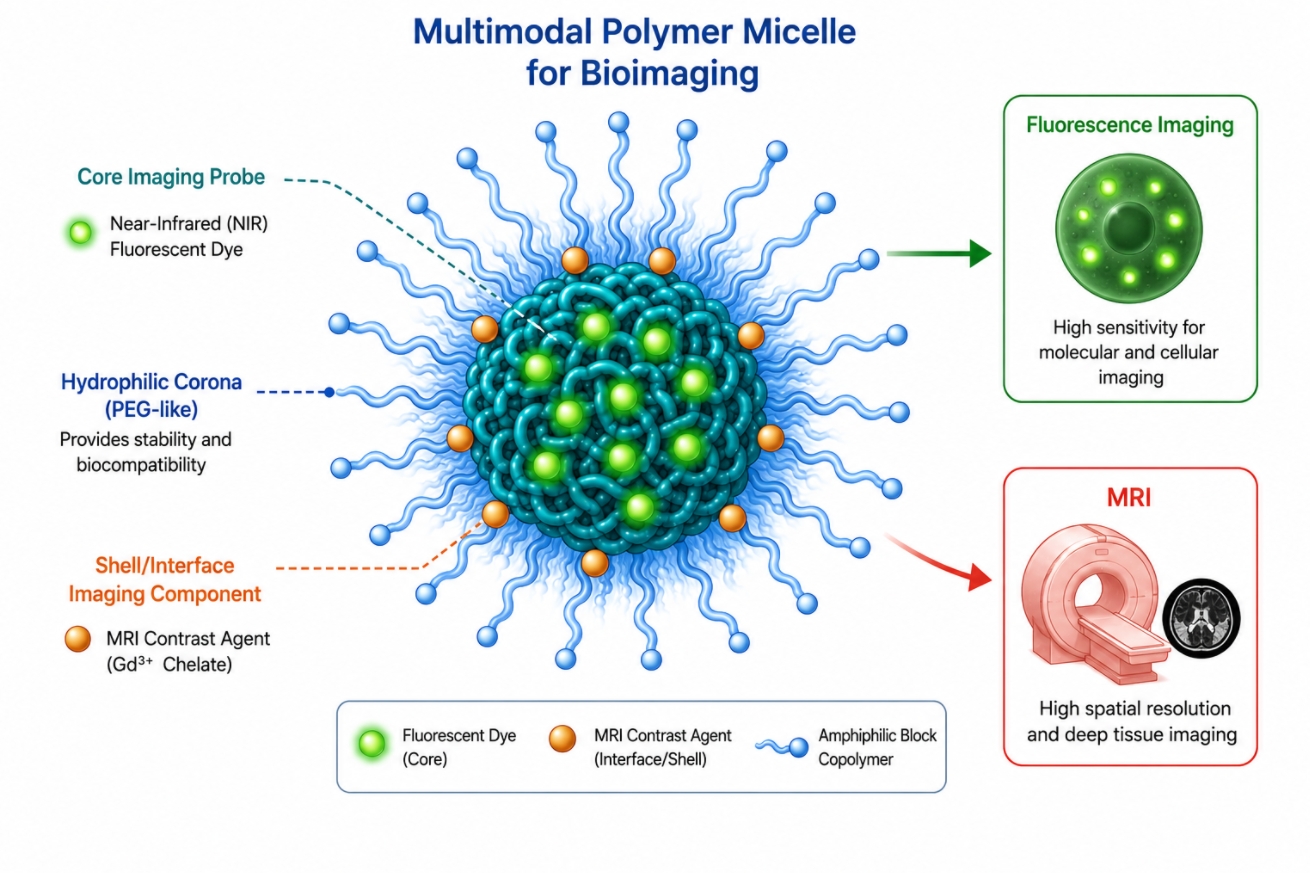
Polymer Micelles for Bioimaging: Design Strategies, Probe Loading, and Imaging Performance
05-20-2026
Polymer micelles have become increasingly valuable in bioimaging research because they do more than passively carry contrast agents or fluorescent probes. As self-assembled nanostructures built from amphiphilic polymers, they provide a controllable microenvironment for organizing imaging units, stabilizing hydrophobic or aggregation-prone probes, and modulating how signals are generated, retained, or activated in aqueous and biologically relevant conditions. Their core-shell architecture makes them especially useful when imaging performance depends not only on the probe itself, but also on probe location, colloidal stability, background suppression, and the ability to connect signal generation with structural change. For this reason, polymer micelles are now widely considered as imaging platforms rather than simple carriers, particularly in systems involving fluorescence, near-infrared imaging, magnetic resonance-related probes, responsive imaging, and multimodal designs. This page focuses on how polymer micelles support bioimaging through probe loading, signal design, responsive activation, and multimodal integration from a polymer materials perspective.
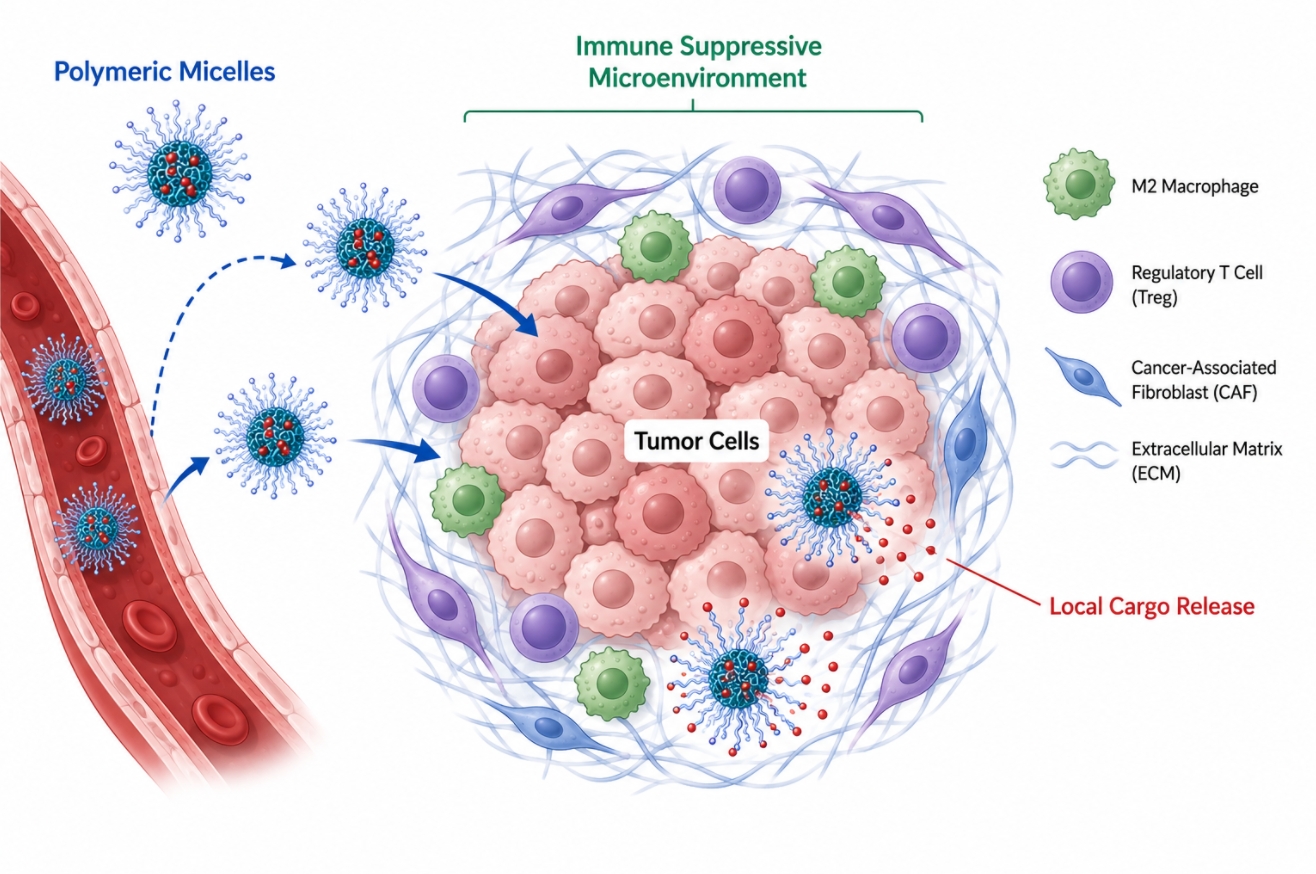
How Polymeric Micelles Support Cancer Immunotherapy?
05-20-2026
Polymeric micelles are increasingly discussed in cancer immunotherapy research because they offer more than simple nanoscale drug solubilization. Their value lies in the way amphiphilic block copolymers translate molecular design into controllable self-assembled structures that can influence local exposure, cargo protection, release timing, and interaction with the tumor microenvironment. In immunotherapy-oriented systems, the design objective is not only to transport an active component into tumor tissue, but also to reshape how immune-relevant signals are distributed, retained, and activated within a complex suppressive environment. That makes polymeric micelles especially relevant when researchers need to coordinate multiple formulation tasks at once, such as stabilizing hydrophobic immune modulators, co-delivering synergistic components, or introducing trigger-responsive release logic. This page focuses on those materials and formulation principles, explaining how polymeric micelles can support cancer immunotherapy research through tumor microenvironment modulation, immune cargo delivery, and combination design.
How to Design Stable Polymeric Micelles for Drug Delivery?
05-20-2026
Polymeric micelles are among the most versatile self-assembled nanostructures in advanced drug delivery research because they connect polymer design directly with formulation performance. Built from amphiphilic block copolymers, these carriers provide a hydrophobic interior for incorporating challenging payloads and a hydrophilic exterior for colloidal stabilization in aqueous environments. However, not every system that forms micelles is automatically useful for delivery. The practical value of polymeric micelles depends on how well their block architecture, critical micelle concentration (CMC), drug compatibility, preparation route, and characterization data work together to support a stable and interpretable formulation strategy. This page focuses on that design logic, helping researchers understand not only what polymeric micelles are, but also how to judge whether a given micelle system is structurally sound, formulation-relevant, and worth further optimization.
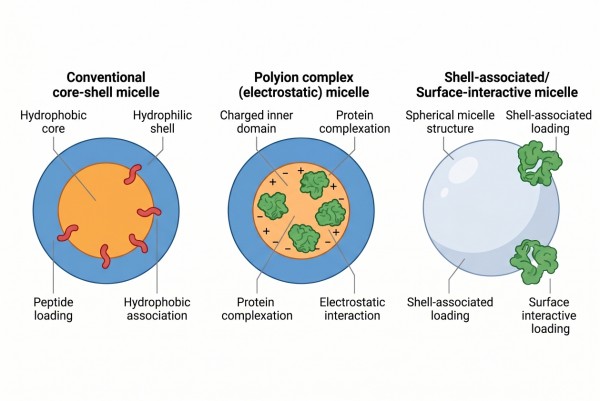
How Polymer Micelles Support Protein and Peptide Delivery?
04-23-2026
Protein and peptide delivery places unusual demands on polymer micelles because these cargos are far more structurally sensitive than many hydrophobic small molecules and often respond strongly to solvent exposure, interfacial stress, ionic conditions, and dehydration history. A useful micellar system must therefore do more than create a nanoscale carrier: it must preserve conformational integrity where possible, reduce premature leakage, maintain colloidal stability during processing, and support a release pathway that matches the physicochemical behavior of the biomacromolecule. In practice, successful design depends on aligning payload class, micelle architecture, loading mechanism, and preparation method instead of assuming that the same self-assembly logic used for conventional drug-loaded micelles will automatically translate to proteins and peptides.
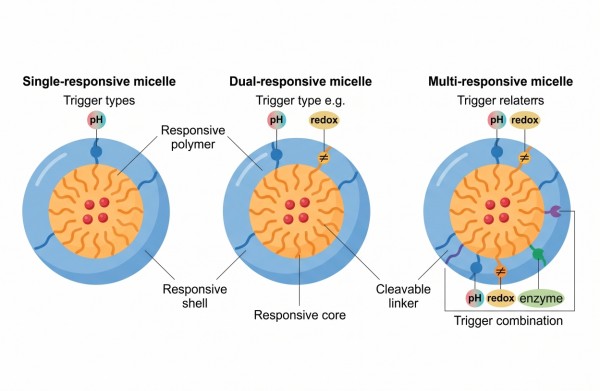
Stimuli-Responsive Polymer Micelles: Design Strategies, Trigger Mechanisms, and Controlled Release Applications
04-23-2026
Stimuli-responsive polymer micelles occupy a distinctive position in advanced polymer delivery because they combine the nanoscale advantages of self-assembled core-shell carriers with a second layer of control built into the polymer structure itself. Their defining feature is not simply that they can carry a payload, but that they can alter core integrity, corona hydration, interfacial behavior, or linker stability when exposed to a relevant trigger. In practical formulation terms, this means the same carrier can be designed to remain relatively stable during preparation and transport, yet become more permeable, destabilized, or disassembled when local conditions shift in a predictable way. The value of this design logic depends on whether the trigger is translated into a useful structural response rather than into uncontrolled instability. A responsive micelle must therefore be judged by more than the presence of a pH-sensitive group or a reducible linkage. The real question is whether the trigger changes the micelle at the right rate, in the right environment, and with enough structural contrast to improve release behavior without sacrificing formulation reproducibility. That requirement makes stimuli-responsive micelles a materials design problem, a colloid problem, and a process problem at the same time.
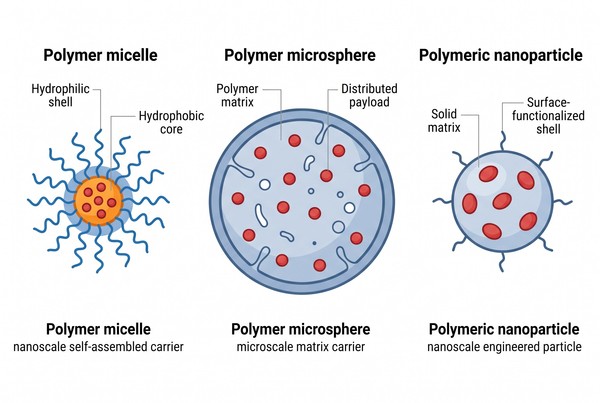
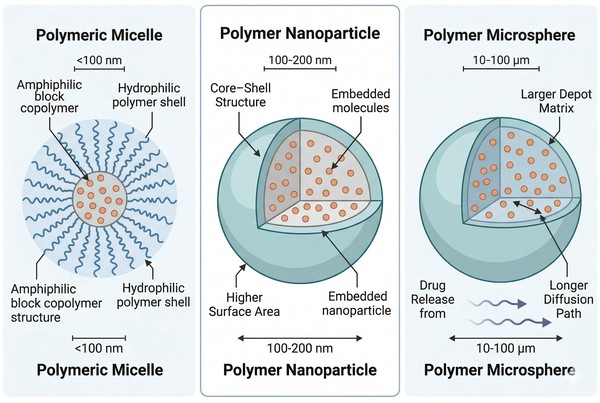
Polymer Micelles vs Polymer Microspheres vs Polymer Nanoparticles: How to Choose the Right Drug Delivery Platform
04-23-2026
Selecting the right polymer-based delivery platform is rarely a matter of choosing the most advanced material or the smallest particle size. In practice, the decision depends on how the carrier is built, how the payload is incorporated, how release is expected to occur, and how the formulation will be prepared, stored, and reproduced. Polymer micelles, polymer microspheres, and polymeric nanoparticles are often discussed together because all three can improve payload handling and controlled delivery, yet they operate through fundamentally different structural and process logics.
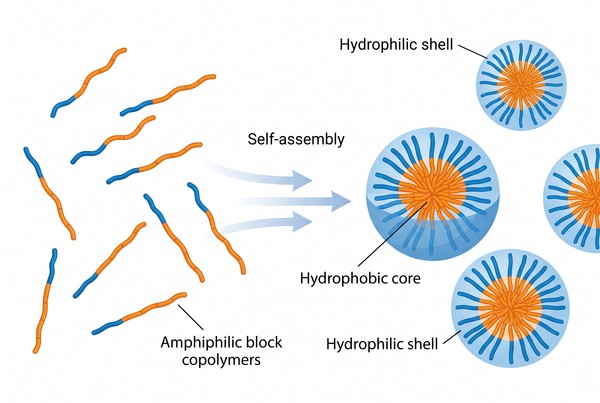
How Polymer Micelles Form and How to Prepare Them: Mechanisms, Methods, and Formulation Strategy
04-23-2026
Polymer micelles are self-assembled colloidal systems formed by amphiphilic macromolecules under selective solvent conditions. Their practical value lies in the fact that molecular design, solvent environment, and process history jointly determine whether the final dispersion becomes a useful nanoscale carrier or a poorly controlled aggregate population. For formulation work, it is therefore not enough to ask whether a polymer can form micelles in principle. What matters is how micellization proceeds, which route controls the transition from dissolved chains to core-shell assemblies, and how that route affects size, dispersity, loading behavior, and colloidal robustness in downstream use.
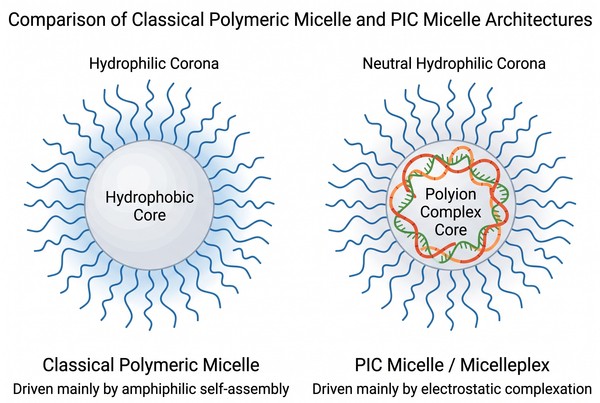
A Practical Guide to Polymer Micelles for Nucleic Acid Delivery
04-23-2026
Polymer micelles provide a versatile materials framework for nucleic acid delivery when the design goal extends beyond simple nanoparticle formation to controlled condensation, shielding, intracellular transport, and timely release of DNA, RNA, or oligonucleotide cargoes. Unlike conventional micellar systems developed for hydrophobic small molecules, nucleic acid-loaded polymer micelles are often built around electrostatic self-assembly between charged polymer segments and negatively charged nucleic acids, forming polyion complex micelles or micelleplexes with tunable core-shell organization. By controlling block composition, charge density, hydrophilic corona structure, degradability, and assembly conditions, these systems can be tailored to improve cargo protection, circulation stability, uptake behavior, endosomal escape, and intracellular decomplexation. As a result, polymer micelles have become an important design option for researchers developing non-viral nucleic acid delivery platforms with more adaptable structure-property relationships than many one-component polyplex systems.
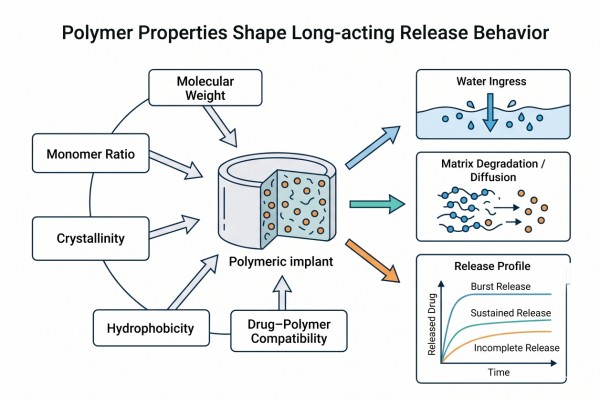
How to Choose the Right Polymer for Long-Acting Drug Delivery Formulations?
04-23-2026
Choosing the right polymer for long-acting drug delivery formulations requires more than selecting a familiar excipient or a commonly cited biodegradable material. In practice, polymer choice controls how the formulation absorbs water, how the drug is distributed and stabilized, how the matrix changes with time, and how reliably release can be tuned across a defined duration. The same active pharmaceutical ingredient (API) may behave very differently in a degrading polyester matrix, a hydrophilic hydrogel network, a membrane-controlled reservoir system, or an in situ forming depot. For this reason, effective polymer selection must connect API properties, delivery platform, polymer chemistry, processing route, and release objectives into one coherent design framework. This resource outlines that framework and shows how to evaluate polymer candidates for long-acting systems with a material-science-based approach.
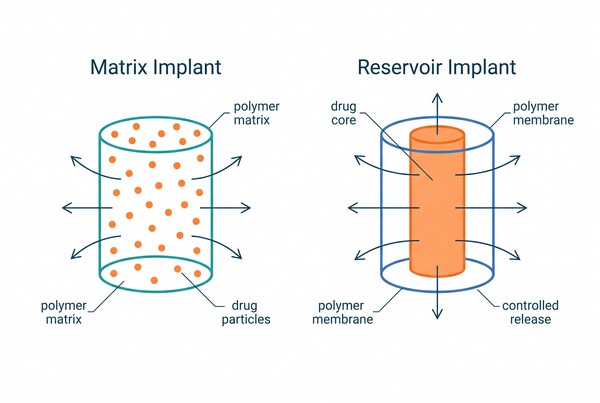
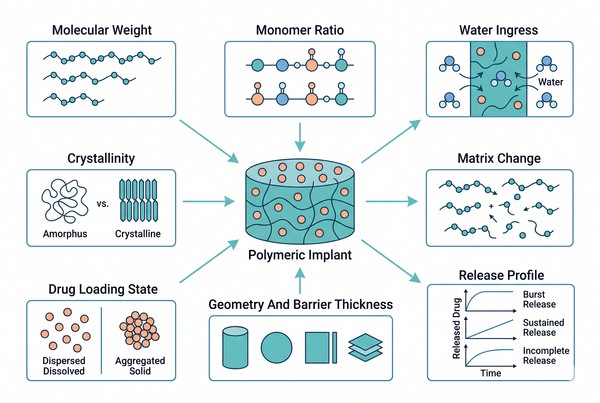
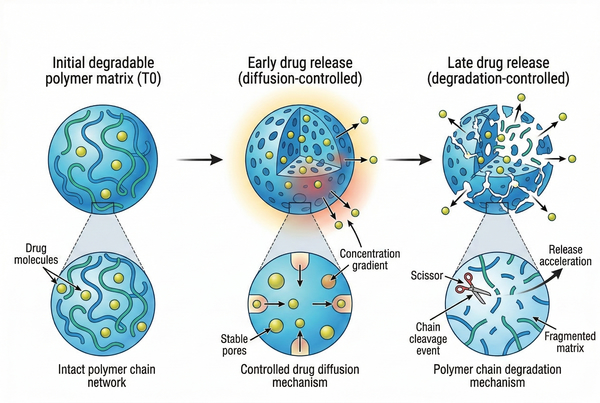
Polymeric Implants for Long-Acting Drug Delivery: Material Selection and Formulation Design
04-23-2026
Polymeric implants are an important platform for long-acting drug delivery because they combine material structure, drug–polymer compatibility, and controlled mass transport in a single dosage form. Unlike short-duration carriers that rely mainly on rapid dissolution or dispersion, implantable polymer systems are designed to maintain structural integrity and regulate drug release over extended periods through diffusion, polymer degradation, swelling, pore evolution, or a combination of these mechanisms. By adjusting polymer composition, molecular weight, crystallinity, geometry, wall thickness, and fabrication route, formulators can build implant systems that better match the stability, solubility, and release requirements of challenging active pharmaceutical ingredients (APIs). For research teams evaluating long-acting delivery strategies, polymeric implants offer a practical framework for balancing release duration, formulation robustness, and manufacturability.
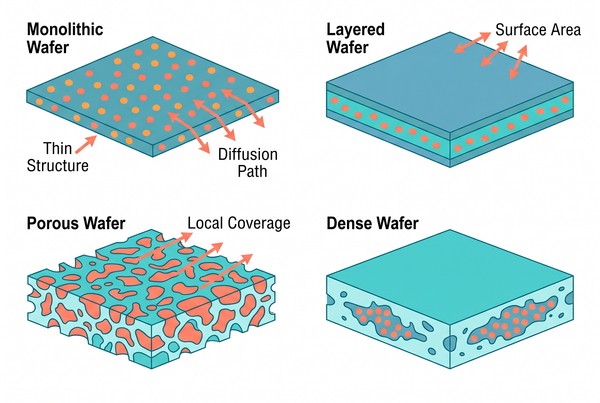
Implantable Polymeric Wafers for Long-Acting Drug Delivery: Structural Design and Release Strategy
04-23-2026
Polymer-based wafers represent a distinctive class of implantable drug delivery systems because their performance is strongly defined by geometry, local placement, and matrix-controlled release. Unlike particulate systems that disperse after administration or larger device-like implants that rely on bulk structure, polymeric wafers use thin, planar, or layered architectures to provide localized coverage and sustained drug exposure from a defined surface. This makes them especially relevant when the development goal is long-acting local delivery supported by controllable diffusion distance, manageable structural persistence, and tunable release behavior. In practical formulation work, the value of a wafer lies not only in what polymer it contains, but in how that polymer behaves inside a thin implantable structure over time.
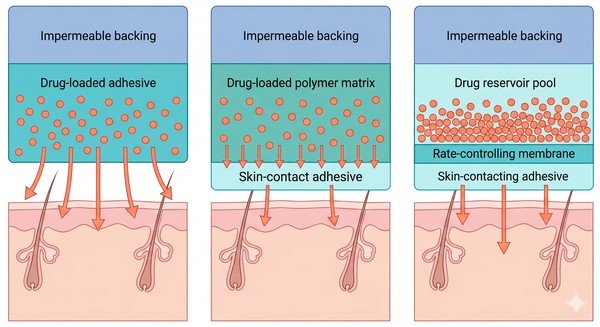
Polymeric Transdermal Patches for Long-Acting Drug Delivery: Matrix Design and Release Control
04-23-2026
Polymer-based transdermal patches remain one of the most practical long-acting drug delivery formats because they combine controlled release with noninvasive administration, scalable manufacturing, and flexible design options. Unlike simple topical films or rapidly depleted surface formulations, polymeric patch systems are engineered to regulate how a drug is stored, how it moves through the matrix, how it contacts the skin, and how long it can maintain a useful input profile. In long-acting delivery, that balance is especially important because successful patch performance depends on more than drug release alone. The formulation must also support stable skin contact, predictable transport across the stratum corneum, resistance to crystallization or phase separation, and sufficient physical stability during wear and storage.
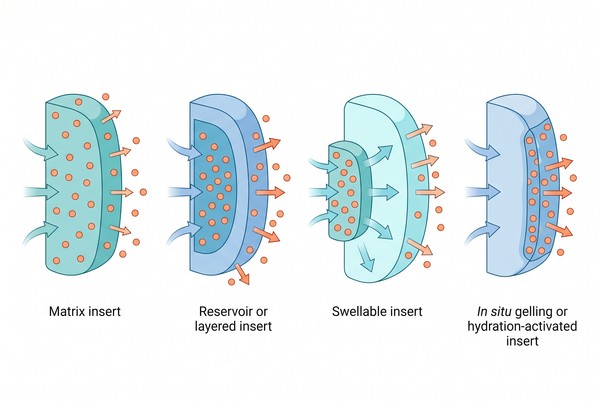
Polymeric Inserts for Long-Acting Drug Delivery: Material Selection and Site-Specific Design
04-23-2026
Polymer-based inserts are a distinct long-acting drug delivery platform because they are designed to remain at a defined local site long enough to improve residence, regulate the local microenvironment, and extend drug release beyond what conventional liquids, semisolids, or rapidly cleared local formulations can usually achieve. Unlike systems that depend mainly on immediate spreading or passive diffusion, polymeric inserts use matrix architecture, hydration, mucoadhesion, swelling, erosion, or structural persistence to hold the dosage form in place and modulate how drug leaves the system over time. This makes them especially valuable when the local site is affected by rapid clearance, fluid turnover, leakage, motion, or short contact time. In practical formulation work, the performance of an insert depends less on the label "insert" itself than on how well the material, geometry, and release logic match the site of administration and the intended long-acting delivery goal.
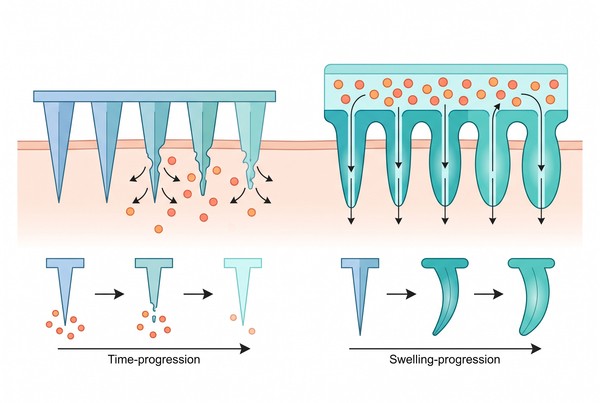
Polymeric Microneedles and Transdermal Systems for Long-Acting Drug Delivery
04-23-2026
Polymer-based microneedles and transdermal systems are emerging as versatile long-acting drug delivery platforms because they combine skin-access enhancement with tunable release control. By integrating needle geometry, matrix design, reservoir coupling, and material selection, these systems can support more prolonged input than conventional topical formulations while avoiding the complexity of larger implantable devices. Depending on the platform design, long-acting behavior may come from rapid skin barrier penetration followed by sustained release from the needle matrix, or from a microneedle-enabled pathway that allows a connected patch or reservoir to maintain controlled delivery over an extended period.
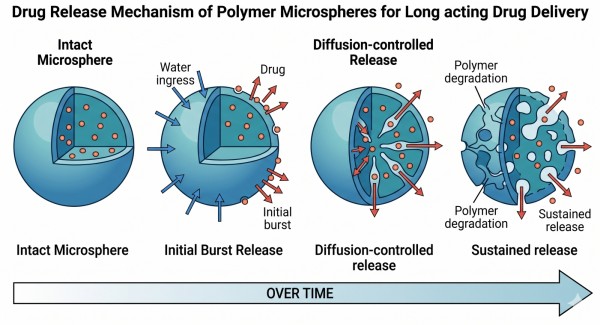
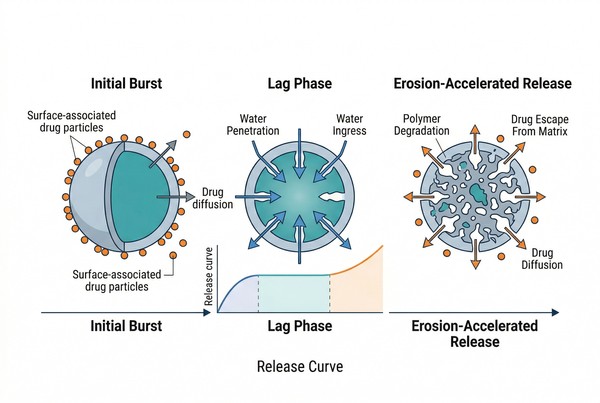
Polymer Microspheres for Long-Acting Drug Delivery: Materials, Release Control, and Formulation Strategy
04-23-2026
Polymer microspheres are one of the most established application forms in polymeric long-acting drug delivery systems, offering a practical way to convert rapid drug exposure into sustained and more controlled release profiles. By selecting the right polymer chemistry, particle structure, loading approach, and manufacturing process, formulators can tailor microsphere systems for small molecules, peptides, proteins, and other challenging payloads. Compared with immediate-release formulations, polymer microspheres can reduce concentration fluctuation, extend dosing intervals, improve formulation robustness, and support both local and systemic delivery goals through tunable degradation and diffusion behavior.
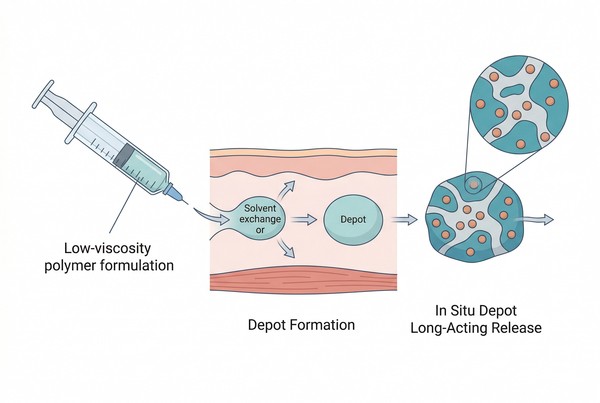
In Situ Forming Polymeric Depots for Long-Acting Drug Delivery: Formulation Design and Release Control
04-23-2026
Polymeric in situ forming depots are long-acting drug delivery systems designed to be administered as low-viscosity liquids, solutions, or suspensions that transform into a gelled or solidified reservoir after administration. This combination of injectability before use and depot persistence after administration makes the platform especially attractive when formulators need extended release without relying on preformed implants or large particulate suspensions. By selecting suitable polymer materials, trigger mechanisms, and formulation variables, these systems can be engineered to control depot formation, internal structure, drug distribution, and long-term release behavior. Their practical value lies in the ability to combine relatively simple administration with programmable release, adaptable depot architecture, and compatibility with both localized and systemic long-acting delivery goals.
Polymer Nanoparticles for Long-Acting Drug Delivery: Formulation Strategies, Release Logic, and Material Trade-Offs
04-23-2026
Polymer nanoparticles are highly adaptable carriers for long-acting drug delivery because they combine tunable polymer chemistry with controllable particle architecture, surface functionality, and payload association strategies. By adjusting polymer class, particle size, surface charge, internal morphology, and loading method, formulators can influence colloidal stability, payload retention, tissue distribution, and release behavior across a wide range of therapeutic formats. Unlike simple nanosuspensions, polymer nanoparticles can be engineered to provide not only transport and protection, but also matrix-mediated release control, making them especially useful when formulation goals include prolonged local retention, improved payload stability, or better balance between delivery efficiency and release duration.
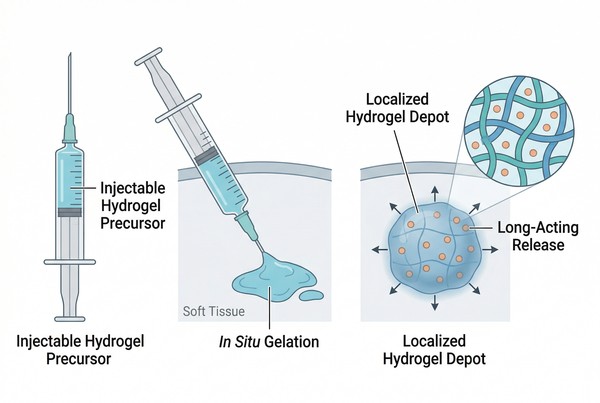
Injectable Polymer Hydrogels for Long-Acting Drug Delivery: Design Principles and Material Selection
04-23-2026
Injectable polymer hydrogels are versatile long-acting drug delivery platforms that can be administered in a flowable state and converted into localized depots after injection through physical association, ionic interaction, solvent exchange, or chemical crosslinking. Unlike rigid implants or discrete particulate systems, hydrogel depots provide a water-rich three-dimensional polymer network that can retain payloads, regulate transport, and adjust release behavior through mesh size, swelling, degradation, and drug–matrix interactions. Because these systems can be tuned for injectability, gelation rate, mechanical integrity, and residence time, they are widely explored for localized and depot-style delivery of small molecules, peptides, proteins, and selected nucleic acid-related payloads. For formulation design, injectable hydrogels are especially useful when the goal is to combine minimally invasive administration with controllable matrix-based long-acting release.
PLGA for Long-Acting Drug Delivery: Selection, Release Mechanisms, and Formulation Design
04-23-2026
Poly(lactic-co-glycolic acid) (PLGA) is one of the most practical and extensively used biodegradable polymers for long-acting drug delivery because it gives formulators a rare combination of tunable degradation, broad processing flexibility, and compatibility with multiple sustained-release platforms. By adjusting copolymer ratio, molecular weight, end-group chemistry, and matrix structure, PLGA systems can be designed to regulate water uptake, erosion, pore formation, and payload diffusion across weeks or months. This makes PLGA a central material choice for depot formulations, microspheres, nanoparticles, implants, and in situ forming systems intended to improve exposure control, reduce dosing frequency, and support more stable formulation performance for small molecules, peptides, proteins, and selected macromolecular payloads.
Polymer-Based Long-Acting Drug Delivery Systems: Application Forms, Material Selection, and Formulation Design
04-23-2026
Polymer-based long-acting drug delivery systems provide a versatile framework for extending drug availability by integrating material structure, dosage architecture, and mass transport control into one formulation strategy. Rather than relying on rapid dissolution, these systems use engineered polymer phases to regulate hydration, diffusion, degradation, swelling, retention, and payload stabilization across different release windows. Their value lies in the ability to adapt polymer chemistry and formulation geometry to the requirements of small molecules, peptides, proteins, charged payloads, and poorly soluble compounds. As a result, polymer-based platforms have become central to long-acting formulation design, especially when predictable release behavior and platform-specific tunability are required.
Biodegradable Polymers for Drug Delivery: Materials, Design, and Controlled Release Strategies
03-23-2026
As pharmaceutical innovation increasingly focuses on complex and poorly soluble active compounds, biodegradable polymers have emerged as essential materials for advanced drug delivery system (DDS) design. Rather than acting as passive carriers, these polymers actively regulate drug release, stability, and distribution by undergoing controlled degradation in physiological environments. Their ability to break down into non-toxic byproducts makes them particularly attractive for designing safe and tunable delivery platforms. By precisely engineering polymer composition, molecular weight, and architecture, researchers can tailor degradation kinetics and release profiles to match specific formulation requirements. Biodegradable polymers enable the development of delivery systems that improve solubility, protect sensitive molecules, and achieve sustained or stimuli-responsive release. As a result, they play a central role in modern formulation strategies for small molecules, peptides, proteins, and nucleic acids.
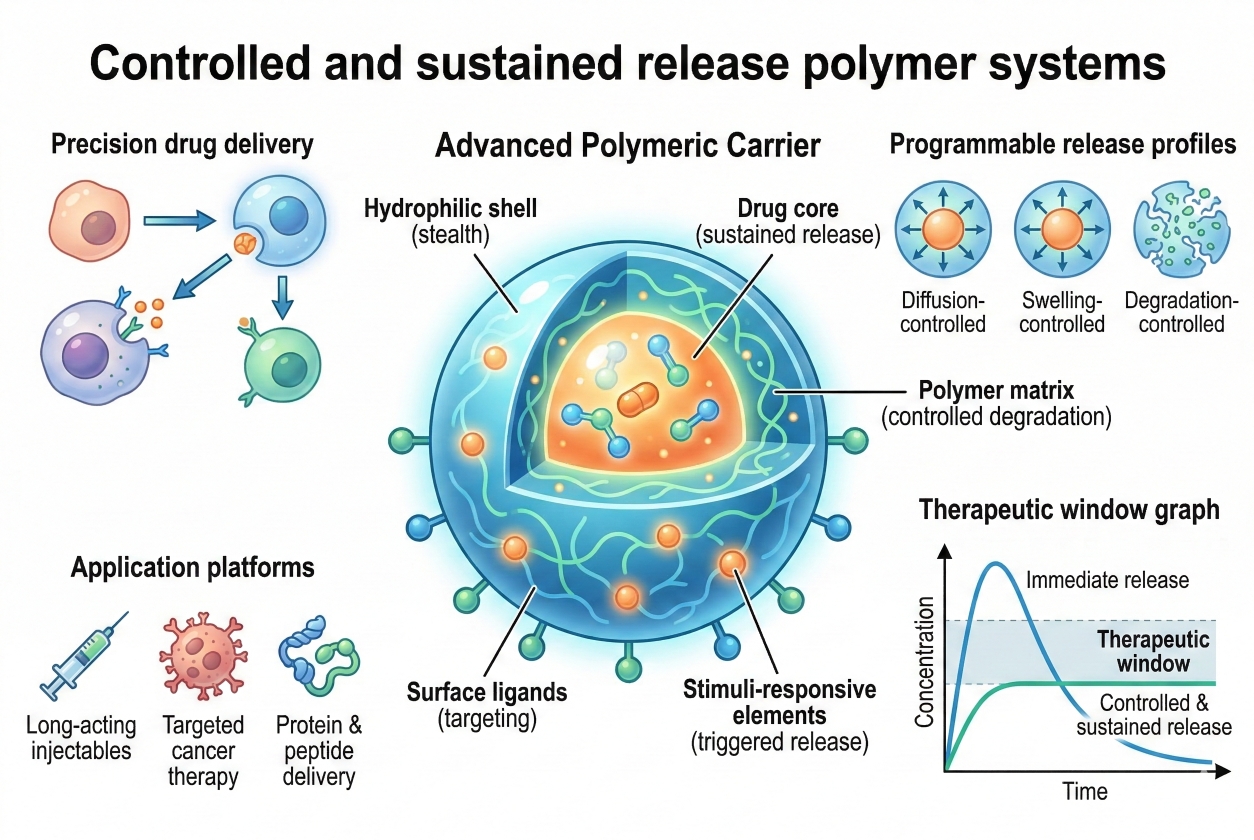
Controlled & Sustained Release Polymer Systems for Precision Drug Delivery
03-23-2026
Controlled and sustained release polymer systems have become essential technologies in modern pharmaceutical science, enabling more precise control over drug pharmacokinetics and therapeutic performance. By incorporating active pharmaceutical ingredients into polymer matrices, nanoparticles, hydrogels, or implantable devices, these systems can regulate the rate, duration, and location of drug release within the body. Compared with conventional formulations, polymer-based delivery platforms help maintain stable drug concentrations, reduce dosing frequency, and minimize systemic side effects. Biodegradable polymers, functionalized synthetic polymers, and natural polymers derived biomaterials can be carefully designed to achieve predictable degradation profiles, tunable drug diffusion, and responsive release behaviors under specific physiological conditions. These capabilities make polymer systems particularly valuable for the delivery of small molecules, peptides, proteins, and nucleic acid therapeutics.
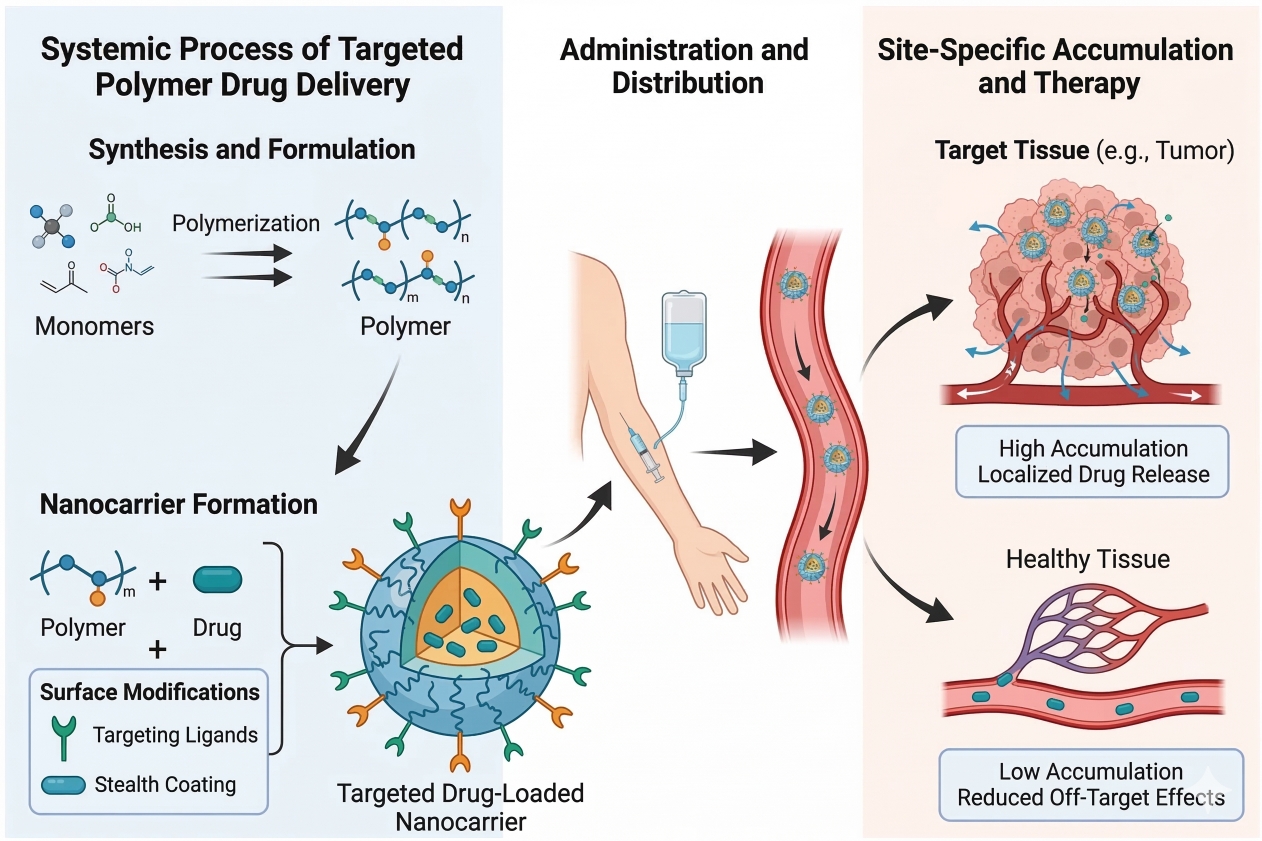
Targeted Polymer Drug Delivery Systems: Site-Specific Therapy and Controlled Release
03-23-2026
Targeted polymer drug delivery systems represent a transformative approach in modern therapeutics, enabling precise site-specific therapy and highly controlled drug release. By leveraging advanced polymer chemistry, functional design strategies, and smart material engineering, these systems can improve drug stability, enhance bioavailability, and minimize systemic side effects. From cancer treatment and gene delivery to inflammatory and chronic disease management, polymer-based delivery platforms are reshaping how active pharmaceutical ingredients are transported and released in the body. With increasing demand for precision medicine and optimized therapeutic performance, targeted polymer technologies are becoming a core focus in pharmaceutical research and development, driving innovation in drug formulation, nanomedicine, and next-generation controlled release systems.
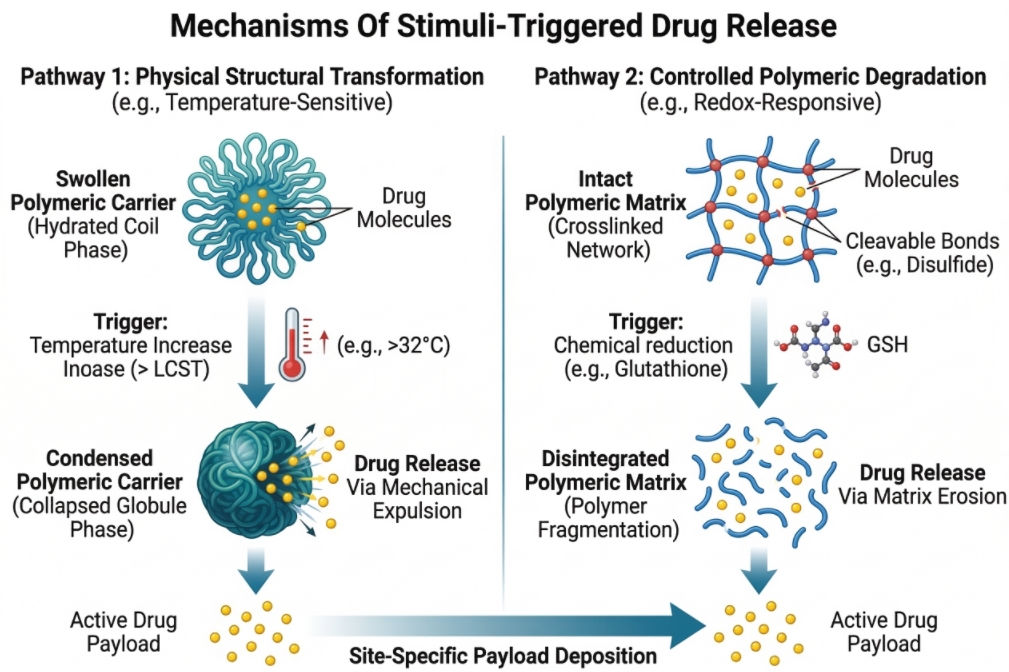
Stimuli-Responsive Polymer Drug Delivery Systems for Targeted Drug Release
03-23-2026
Targeted drug delivery has become a central focus in modern pharmaceutical and biomedical research, as conventional drug administration often suffers from limited therapeutic efficiency, systemic toxicity, and poor control over drug release profiles. To address these challenges, stimuli-responsive polymer drug delivery systems—also known as smart polymer systems—have emerged as an innovative strategy for achieving controlled and site-specific drug release. These advanced materials are designed to respond to specific environmental triggers, enabling therapeutic agents to be released only when and where they are needed. Stimuli-responsive polymers can undergo physical or chemical changes when exposed to particular internal stimuli (such as pH variations, enzymatic activity, or redox conditions) or external stimuli (including temperature, light, magnetic fields, ultrasound, or electric signals). These responsive behaviors allow polymer carriers to precisely regulate drug release kinetics, improve drug stability, and enhance the accumulation of therapeutic molecules at target tissues. As a result, smart polymer-based delivery systems have attracted significant interest in areas such as cancer therapy, gene delivery, inflammatory disease treatment, and regenerative medicine.
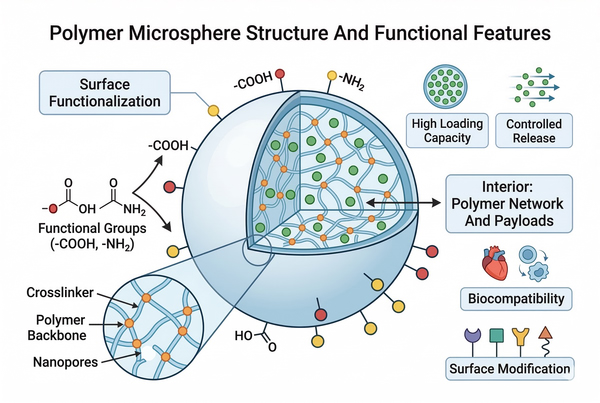
How Polymer Microspheres Work in Drug Delivery: Mechanisms, Design, and Applications
03-23-2026
Polymer microsphere drug delivery systems represent one of the most versatile and scientifically advanced platforms in controlled release technology. By engineering polymeric materials into microscale spherical carriers, researchers can precisely regulate drug encapsulation, release kinetics, stability, and targeting behavior. These systems are widely explored in pharmaceutical formulation development, biomaterials research, and advanced drug delivery design. Their performance depends on polymer chemistry, physicochemical interactions, structural morphology, and degradation behavior, all of which can be tailored through rational material selection and formulation strategies.
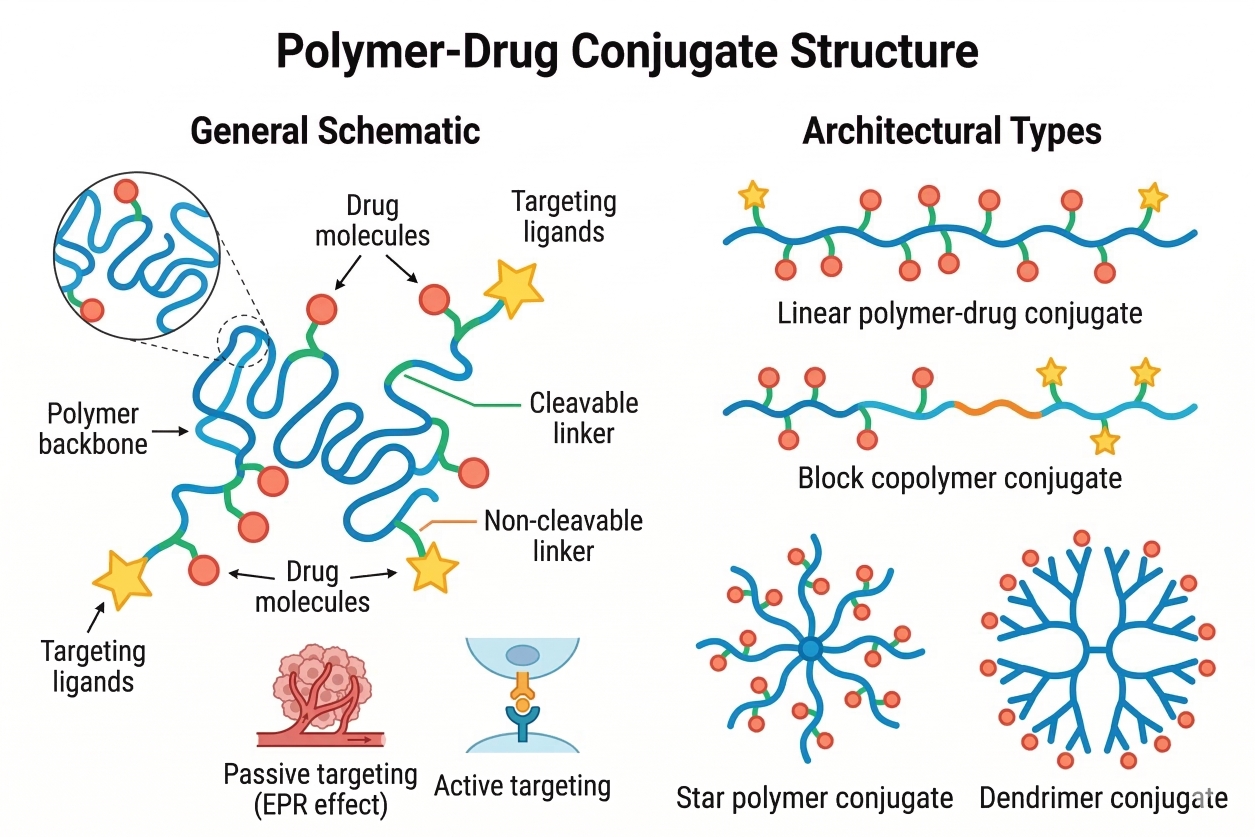
Polymer–Drug Conjugates for Drug Delivery: Design Strategies, Materials, and Applications
03-23-2026
Polymer–drug conjugates (PDCs) have emerged as an important strategy for improving the delivery of therapeutic molecules, particularly compounds with poor solubility or limited stability in aqueous environments. In these systems, drug molecules are covalently attached to polymer carriers, allowing the physicochemical properties of the polymer to enhance the overall performance of the drug formulation. The polymer component can improve dispersion in aqueous media, protect the drug from premature degradation, and influence release behavior through carefully designed linker chemistry. The effectiveness of polymer–drug conjugates largely depends on rational molecular design. Factors such as polymer type, molecular weight, linker structure, and conjugation strategy play critical roles in determining drug loading capacity, stability, and release kinetics. With advances in polymer synthesis and bioconjugation technologies, a wide variety of polymer backbones and functional materials can now be engineered to construct highly tailored conjugate systems for modern drug delivery research.
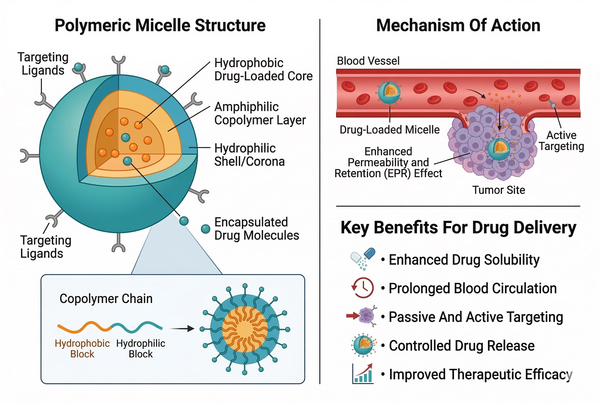
Conventional Micelles vs. Polymeric Micelles for Drug Delivery: Choosing the Right Carrier
03-23-2026
Micellar nanostructures have become important tools for improving the formulation of hydrophobic or poorly soluble therapeutic molecules. By exploiting the self-assembly of amphiphilic molecules in aqueous environments, micelles create nanoscale compartments capable of solubilizing compounds that otherwise exhibit limited bioavailability in water-based systems. Among the various micellar systems studied in pharmaceutical research, conventional surfactant micelles and polymeric micelles represent two widely investigated carrier platforms. Although both systems rely on amphiphilicity-driven self-assembly, their physicochemical characteristics, stability, and drug delivery performance differ substantially. Understanding these differences is essential when selecting an appropriate carrier for specific drug molecules and formulation objectives.
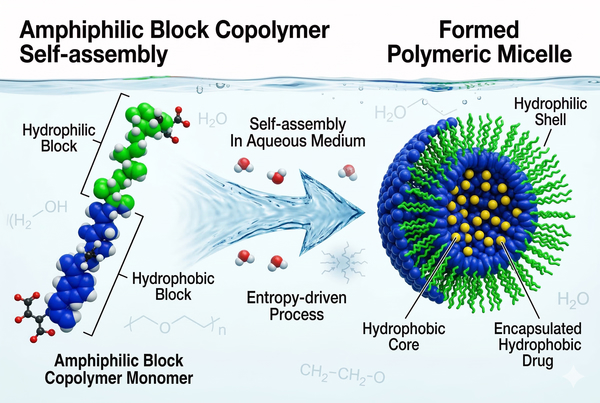
Polymeric Micelles for Poorly Soluble Drugs: Enhanced Solubility and Controlled Delivery
03-23-2026
High hydrophobicity severely restricts systemic absorption, resulting in erratic bioavailability and rendering many fundamentally promising molecules pharmacologically ineffective. Traditional formulation approaches, which often rely on harsh organic co-solvents or low-molecular-weight surfactants, frequently introduce severe dose-limiting toxicities and thermodynamic instability upon systemic administration. Polymeric micelles have emerged as a highly sophisticated, supramolecular solution to these pervasive delivery challenges. Through the spontaneous self-assembly of amphiphilic block copolymers, these advanced nanocarriers form a thermodynamically stable core-shell architecture. The inner lipophilic core serves as an optimized nanoreservoir to encapsulate and seamlessly solubilize hydrophobic drugs, while the protective hydrophilic corona prevents aggregation and significantly prolongs systemic circulation.
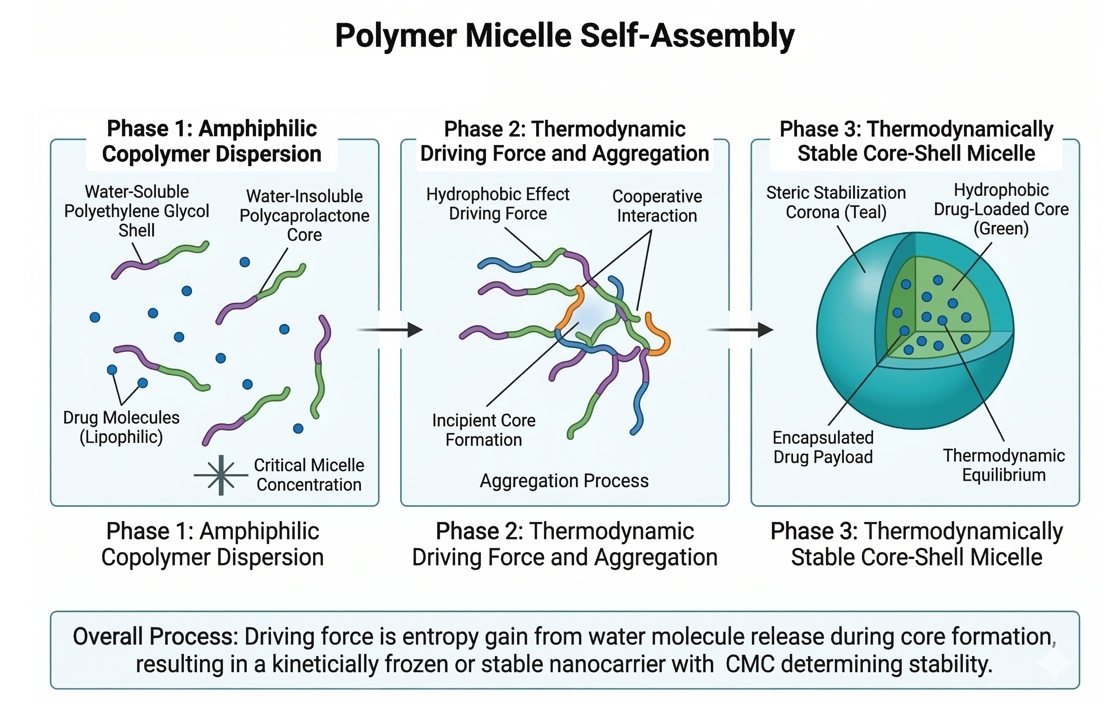
Polymer Micelle Platforms for Sustained and Targeted Drug Delivery
03-23-2026
The formulation of advanced therapeutic agents frequently encounters profound physicochemical barriers, most notably extreme hydrophobicity and rapid systemic degradation. To overcome these inherent limitations, researchers are increasingly turning to self-assembling macromolecular architectures. Polymer micelles have emerged as highly sophisticated, thermodynamically stable nanocarriers engineered to revolutionize payload administration. Constructed from rationally designed amphiphilic block copolymers, these core-shell platforms provide a precisely tuned microenvironment for solubilizing lipophilic molecules while strictly shielding them from premature clearance. By modulating polymer topologies, molecular weights, and surface functionalities, formulators can dictate sustained release kinetics and facilitate highly specific active targeting mechanisms.
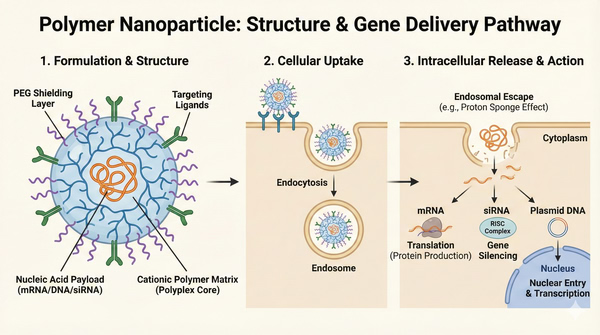
Polymer Nanoparticles for Gene Delivery: DNA, mRNA, siRNA and Oligo Therapeutics
02-25-2026
The development of robust and efficient delivery vectors remains a cornerstone of modern nucleic acid research. While viral vectors have historically dominated early biological studies, synthetic non-viral systems, particularly polymer nanoparticles, have emerged as highly versatile alternatives. These macromolecular systems offer unparalleled chemical flexibility, allowing researchers to engineer custom carriers tailored to the specific physicochemical properties of the genetic payload. By neutralizing the negative charge of nucleic acids and condensing them into nanometric structures, polymers facilitate the safe transit of genetic material across complex biological barriers.
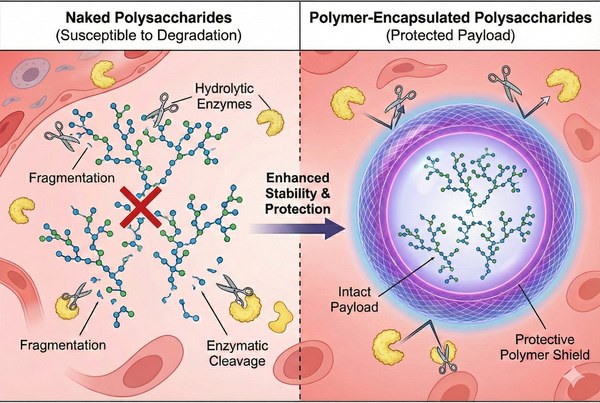
How Polymer Particles Improve Polysaccharide & Glycan Delivery Efficacy?
02-25-2026
The successful delivery of complex biomolecules remains a primary hurdle in modern formulation science. While polysaccharides and glycans possess immense potential for advanced therapeutics, their inherent physicochemical properties often limit their bioavailability and targetability. Polymer particles have emerged as a sophisticated solution, providing a customizable, highly engineered vehicle to encapsulate, protect, and deliver these complex carbohydrates. By leveraging advanced polymer chemistry, researchers can dramatically improve the pharmacokinetic profiles of glycans, ensuring they reach their intended biological targets with high efficiency.
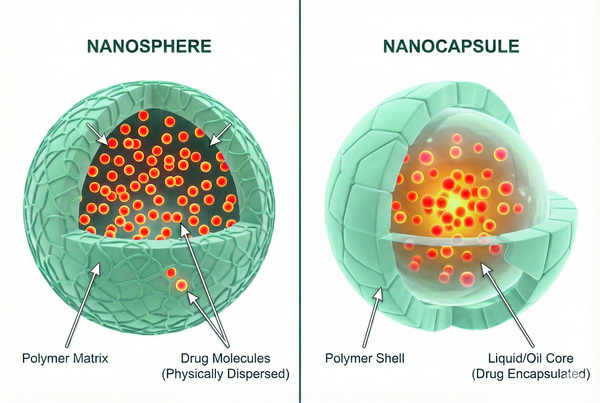
Polymer Nanoparticles for Drug Delivery: Formulation Strategies and Applications
02-25-2026
The pharmaceutical landscape is increasingly shifting away from conventional systemic administration toward sophisticated, localized, and smart delivery systems. Among the most versatile tools in this domain are polymer nanoparticles (PNPs). Unlike pre-formed solid implants or simple solutions, PNPs represent a class of colloidal drug delivery systems that offer precise control over pharmacokinetics and biodistribution. These systems are engineered to overcome historic challenges such as poor bioavailability, rapid renal clearance, and off-target toxicity, serving as dynamic reservoirs that modulate the therapeutic index of encapsulated payloads.
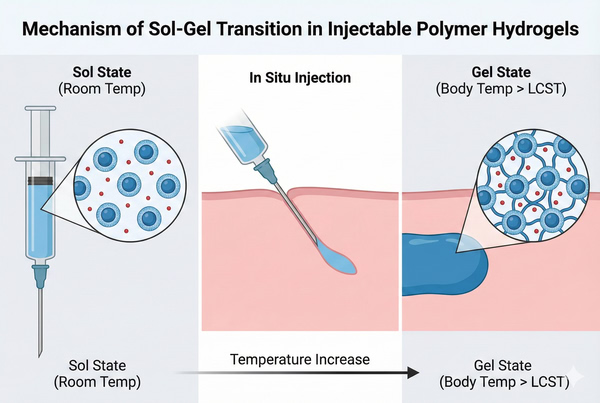
Injectable Polymer Hydrogels for Sustained Drug Delivery and Controlled Release
02-25-2026
The landscape of pharmaceutical formulation is undergoing a paradigm shift, moving away from systemic administration toward localized, smart delivery systems. Among the most promising advancements in this field are injectable polymer hydrogels. These stimuli-responsive biomaterials offer a unique solution to the historic challenges of bioavailability, patient compliance, and therapeutic precision. For researchers and formulators, understanding the physicochemical properties and rheological behaviors of these polymers is essential for designing next-generation drug delivery systems (DDS). This article explores the mechanisms, critical polymer classes, and design strategies behind injectable hydrogels.
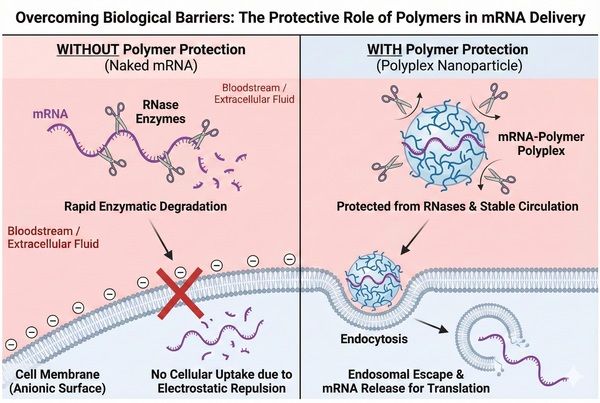
Overcoming mRNA Stability and Delivery Challenges with Polymers
02-25-2026
The clinical validation of mRNA technology has ushered in a new era of medicine, yet the transition from bench to bedside remains hindered by the inherent fragility of the mRNA molecule. While lipid nanoparticles (LNPs) have established the foundational success for current vaccines, the field is increasingly pivoting toward polymer-based delivery systems to address limitations in thermal stability, immunogenicity, and tissue specificity. Synthetic polymers offer a broad chemical design space, allowing for precise control over molecular architecture and functionalization.
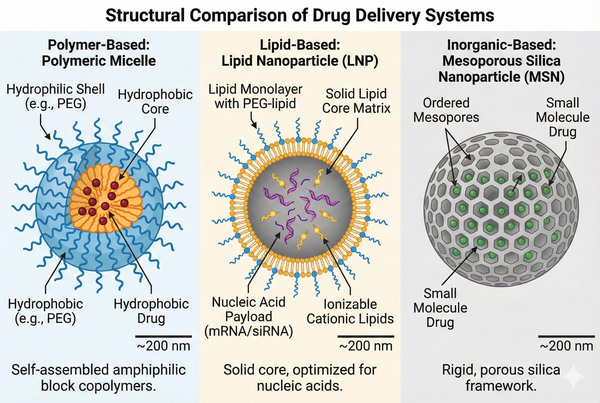
Choosing the Right Drug Delivery System: Polymer vs Lipid vs Inorganic Carriers
02-25-2026
As the industry moves from small, soluble molecules to complex biologics—including proteins, peptides, monoclonal antibodies, and nucleic acids—the limiting factor in clinical success is often no longer the potency of the active pharmaceutical ingredient (API), but its bioavailability. A drug delivery system (DDS) is defined not merely as a vehicle, but as a specialized interface between the drug and the biological environment. Its primary function is to modulate the pharmacokinetics (PK) and pharmacodynamics (PD) of the therapeutic agent. By controlling the rate, time, and place of release, a well-engineered DDS can transform a potent but toxic or unstable molecule into a viable clinical therapy. The modern approach to DDS design focuses on overcoming the body's natural defense mechanisms while ensuring the cargo reaches its intracellular or extracellular target in a bioactive state.
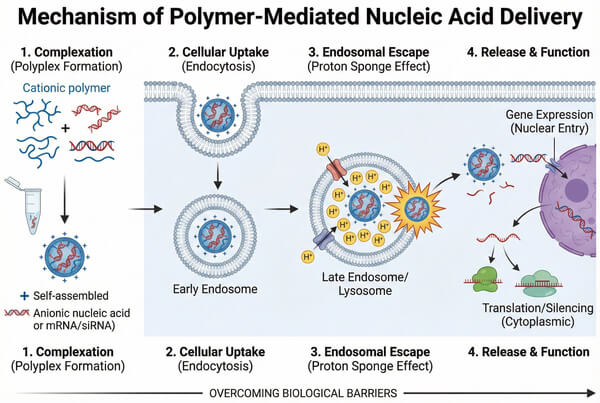
Polymers for Nucleic Acid Delivery: Strategies and Technologies
01-30-2026
With the breakthrough advances in gene therapy, mRNA vaccines, and CRISPR/Cas gene-editing technologies, nucleic acid therapeutics have emerged as a frontier of modern biomedicine. However, the clinical application of nucleic acids as therapeutic agents is primarily limited by inefficient in vivo delivery. Naked nucleic acid molecules are highly susceptible to enzymatic degradation by nucleases in biological fluids, and their strong negative charge and hydrophilic nature severely hinder cellular membrane penetration. As a result, the development of safe and efficient delivery carriers is critical to unlocking the full potential of gene-based therapies. Among various delivery platforms, polymers have become a central pillar of non-viral gene delivery systems due to their highly tunable chemical structures, favorable biocompatibility, and excellent nucleic acid loading capacity.
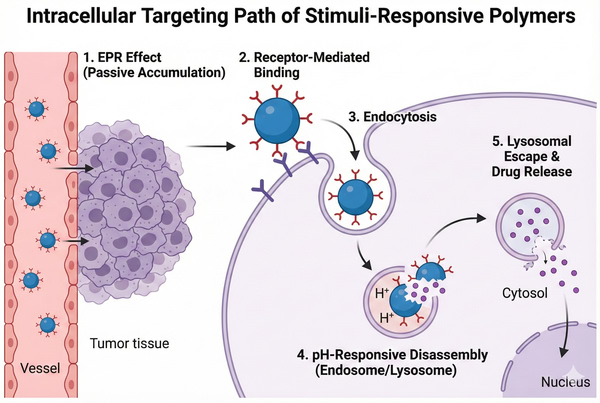
Polymers for Small Molecule Drug Delivery: Formulation Strategies and Design Principles
01-30-2026
In the field of modern drug development, despite the rapid rise of biologics, small molecule therapeutics continue to dominate the pharmaceutical market due to their well-defined chemical structures, mature synthetic processes, and ability to penetrate cell membranes to act on intracellular targets. However, with the advancement of high-throughput screening (HTS) and combinatorial chemistry, approximately 70%-90% of newly identified lead compounds fall into BCS Class II or IV, exhibiting low solubility or low permeability. Advances in polymer science offer revolutionary tools to address these pharmacological challenges. Functional polymers are no longer merely inert excipients; they have become key materials capable of precisely modulating drug release, improving pharmacokinetic (PK) profiles, and enabling targeted delivery.
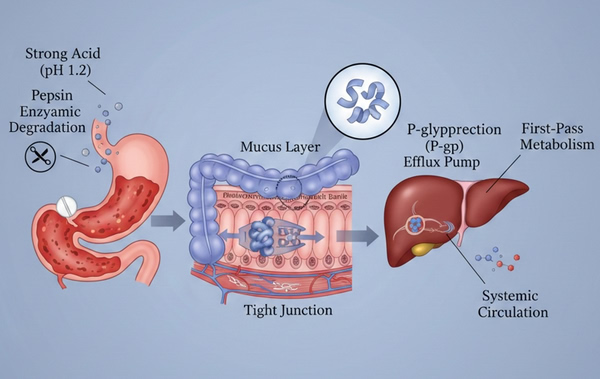
Overcoming Oral Drug Delivery Challenges with Polymer Carriers
01-30-2026
In the contemporary field of drug development, oral administration remains the preferred route of drug delivery due to its non-invasive nature, high patient compliance, and cost-effectiveness. However, with advances in modern medicinal chemistry, new molecular entities (NMEs) often exhibit poor solubility, low permeability, or instability within the gastrointestinal (GI) environment, posing severe challenges to conventional formulation technologies. Polymers, as versatile drug delivery platforms, provide powerful solutions to overcome these biological barriers through physicochemical modification and structural design. Customized polymer-based drug delivery strategies, biomimetic and biopolymer architectures, and drug-free macromolecular processing approaches have all been applied to address challenges in polymer drug delivery. Polymer drug delivery systems based on natural and synthetic polymers have been rapidly adopted across the pharmaceutical industry. Integrating perspectives from both artificial and biological disciplines will facilitate the development of innovative models for polymer-based therapeutic and nucleic acid delivery systems.
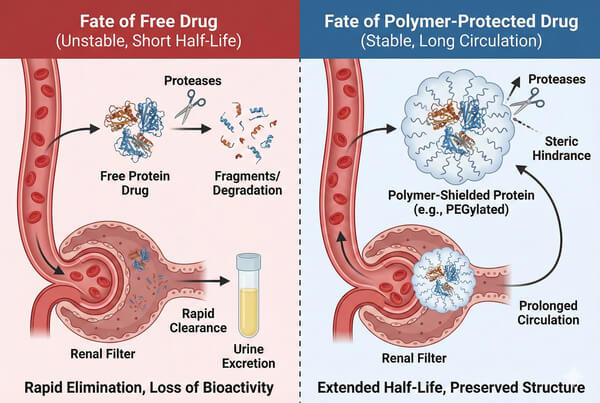
Polymer Carriers for Protein and Peptide Drug Delivery
01-30-2026
Proteins and peptide drugs, with their high specificity and low toxicity, have become central to modern biopharmaceutical research. However, their intrinsic physicochemical instability and limitations in in vivo pharmacokinetics (PK) hinder clinical translation. Functional polymer carriers, as an advanced drug delivery strategy, can significantly improve the in vivo performance of biomacromolecules through mechanisms such as steric hindrance, surface modification, and structural encapsulation. This article explores, from both biochemical and materials science perspectives, the mechanisms by which polymer carriers overcome physiological barriers, the types of carriers available, and key design considerations in drug development.
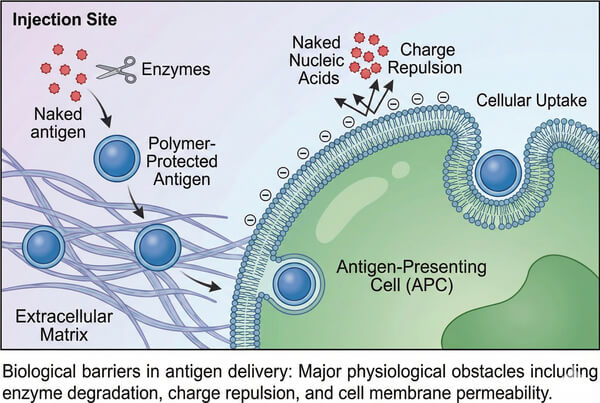
How Synthetic Polymers Enable Targeted Vaccine Delivery?
01-30-2026
The paradigm of vaccinology is rapidly shifting from traditional whole-pathogen formulations toward molecularly defined antigens, including recombinant subunits, synthetic peptides, and emerging nucleic acid (mRNA/DNA) technologies. While these next-generation antigens offer significant advantages in specificity and safety, their clinical efficacy is often limited by intrinsic physicochemical instability, short in vivo half-life, and poor membrane permeability. Exposed antigens are highly susceptible to nuclease or protease degradation in the complex physiological environment and are inefficiently taken up by antigen-presenting cells (APCs), resulting in weak immune responses. In this context, synthetic polymers, with their exceptional chemical versatility and controllable structures, have emerged as key tools to overcome these delivery bottlenecks. Compared with viral or lipid-based systems, synthetic polymers allow precise molecular engineering of molecular weight, topology, and surface functionalities, enabling the creation of intelligent delivery platforms that mimic pathogen features, protect fragile cargo, and achieve targeted lymph node delivery.
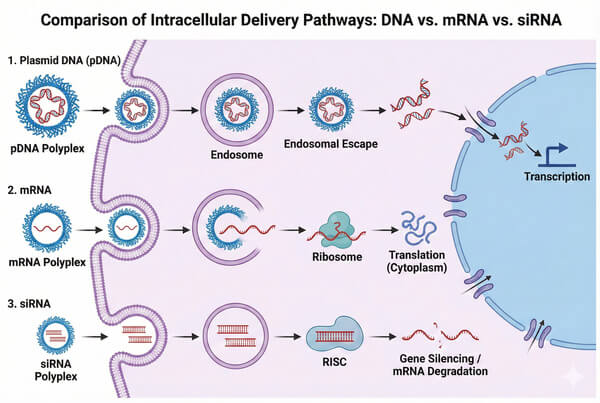
Polymer-Based Gene Delivery Platforms for DNA, RNA, and Oligonucleotide Therapeutics
01-27-2026
With the breakthrough advances in gene therapy and mRNA vaccine technologies, the safe and efficient delivery of nucleic acid therapeutics to target cells has become a central challenge in the biopharmaceutical field. Although viral vectors are widely used in clinical applications, issues such as immunogenicity, limited cargo capacity, and high manufacturing costs have driven the scientific community to explore non-viral alternatives. Among these, polymer-based gene delivery systems have emerged as a leading platform for next-generation nucleic acid delivery, owing to their structural design flexibility, low immunogenicity, and ability to accommodate high–molecular-weight genetic payloads.

