Polymer-Based Long-Acting Drug Delivery Systems: Application Forms, Material Selection, and Formulation Design
Polymer-based long-acting drug delivery systems provide a versatile framework for extending drug availability by integrating material structure, dosage architecture, and mass transport control into one formulation strategy. Rather than relying on rapid dissolution, these systems use engineered polymer phases to regulate hydration, diffusion, degradation, swelling, retention, and payload stabilization across different release windows. Their value lies in the ability to adapt polymer chemistry and formulation geometry to the requirements of small molecules, peptides, proteins, charged payloads, and poorly soluble compounds. As a result, polymer-based platforms have become central to long-acting formulation design, especially when predictable release behavior and platform-specific tunability are required.
Resources
What Are Polymer-Based Long-Acting Drug Delivery Systems?
Polymer-based long-acting drug delivery systems encompass a broad family of dosage platforms in which polymers act as matrices, membranes, gels, carrier shells, self-assembled structures, or structural supports that control drug behavior over time. The central formulation challenge is not simply extending release duration, but balancing loading, stability, transport, and structural evolution in a way that remains useful throughout the full release window. Different platforms solve this challenge differently. Some rely on polymer degradation, others on diffusion barriers, swelling networks, or dynamic carrier assemblies. Understanding that broader framework is essential before choosing a material family or narrowing a formulation strategy.
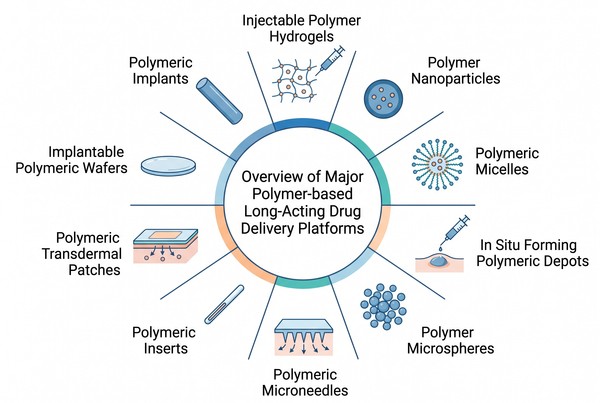 Fig. 1. Overview of major polymer-based long-acting delivery platforms (BOC Sciences Authorized).
Fig. 1. Overview of major polymer-based long-acting delivery platforms (BOC Sciences Authorized).
The polymer may function as a continuous matrix, a rate-controlling membrane, a hydrated network, a particulate carrier, or a self-assembled structure that alters payload transport. Unlike short-duration systems that depend mainly on prompt dissolution, long-acting polymer systems are designed to manipulate the time-dependent relationship between drug location, water access, polymer reorganization, and release kinetics. This makes polymer selection inseparable from overall formulation design rather than a simple excipient choice.
How Long-Acting Polymer Systems Differ from Immediate-Release Formulations?
Immediate-release formulations are generally built to maximize rapid drug availability, whereas long-acting polymer systems are designed to regulate when and how the payload becomes available. This changes the role of the material completely. In long-acting systems, the polymer must preserve internal organization, influence solvent ingress, and maintain a controlled transport environment over time. Even small changes in composition, molecular weight, or geometry can alter release behavior substantially. As a result, performance depends less on the initial dose alone and more on how the formulation evolves structurally during storage, administration, hydration, and prolonged release.
Why Polymer Architecture Matters in Sustained and Extended Drug Delivery?
Polymer architecture influences long-acting performance because chain composition, block arrangement, branching, and network structure all change how water enters, how the drug is distributed, and how the carrier transforms during release. A dense hydrophobic matrix behaves differently from an amphiphilic micellar assembly or a crosslinked hydrogel, even when the same API is used. Architecture also affects whether the system erodes, swells, stays intact as a barrier, or opens progressively through pore formation. In practical formulation work, this means architecture is not an abstract chemical feature; it is one of the main determinants of release mechanism and dosage-form feasibility.
Why Polymers Matter in Long-Acting Drug Delivery?
Polymers matter in long-acting drug delivery because they do more than hold the drug inside a dosage form. They define the physical environment in which the payload is distributed, the pathway through which water and dissolved drug move, and the structural logic that sustains release over time. In long-acting systems, the polymer is often the primary design variable that determines whether the formulation behaves as a depot, a stable membrane device, a swelling carrier, or a degrading matrix. This material-driven control is what separates advanced long-acting formulations from conventional excipient-based drug products with limited release-modulating capability.
What Makes Polymers Different from Conventional Excipients?
Conventional excipients often serve supportive roles such as bulking, wetting, stabilizing, or adjusting flow and manufacturability. Polymers in long-acting systems, however, frequently act as the dominant functional phase. They regulate hydration, control transport path length, provide mechanical persistence, modify local microenvironment, and determine whether the dosage form retains integrity or changes progressively during release. Their role is therefore structural and mechanistic rather than merely supportive. This distinction is especially important in long-acting design, where polymer behavior over time directly affects release profile quality, formulation reproducibility, and the ability to tune performance without redesigning the entire dosage concept.
Why Long-Acting Systems Depend on Material Design?
Long-acting systems depend on material design because release duration is rarely achieved by one variable alone. Instead, performance emerges from the interaction between polymer chemistry, internal microstructure, dosage geometry, and payload properties. A material that hydrates too quickly may cause burst release, while one that remains too impermeable may trap the drug and create incomplete release. Material design therefore determines not only whether release is prolonged, but whether it is controlled, reproducible, and compatible with the intended platform. For this reason, successful long-acting formulations are usually built around deliberate material design rather than platform labels alone.
How Polymers Support Different Long-Acting Delivery Objectives?
Different long-acting delivery objectives require different polymer functions. Some formulations prioritize extended release from a solid matrix, while others require localized retention, self-assembly for poorly soluble compounds, or mild hydrated environments for sensitive biomolecules. Polymers can be tuned to serve each of these objectives by altering degradation rate, hydrophilic balance, network density, surface behavior, and compatibility with the payload. A single polymer family may even support multiple platform types if its composition or formulation context changes. This flexibility is what makes polymers so valuable in long-acting design, but it also means that material selection must always be purpose-driven.
Why Material Tunability Matters More than Polymer Familiarity?
A familiar polymer is not always the right polymer. In long-acting delivery, the best candidates are usually those with enough tunability to support rational optimization rather than those most often cited in the literature. Tunability may come from molecular weight range, monomer ratio, end-group chemistry, hydrophilic modification, blend compatibility, or crosslinking options. These variables create formulation space that can be explored systematically when the initial profile is too fast, too slow, unstable, or incomplete. A polymer with flexible design space often provides more practical value than a popular material whose behavior cannot be adjusted without abandoning the platform entirely.
Main Long-Acting Delivery Platforms Built from Polymers
Polymer-based long-acting delivery is not a single dosage form but a collection of platform types that use polymers in structurally different ways. Some platforms rely on hydrated networks, others on solid matrices, particles, membranes, or in situ transformations that generate depots after administration. These distinctions matter because each platform creates different constraints for loading, processing, transport, and stability. A polymer that performs well in one architecture may behave poorly in another because geometry, surface area, phase continuity, and hydration mode change. Reviewing the main platform types first provides the most useful map for understanding downstream material selection and formulation strategy.
| Delivery Platform | Structural Logic | Main Release Basis | Typical Material Strategy | Best-Fit Development Goal |
|---|
| Polymer Hydrogels | Crosslinked hydrated network | Diffusion, swelling, network relaxation | Hydrophilic or stimuli-responsive polymers | Mild localized long-acting release |
| Polymer Nanoparticles | Nanoscale polymer matrix or shell carrier | Diffusion, degradation, particle retention | Biodegradable or functional polymers | Carrier-based sustained transport |
| Polymeric Micelles | Self-assembled amphiphilic nanostructure | Carrier stability and payload retention | Amphiphilic block copolymers | Poorly soluble drug delivery |
| In Situ Depots | Polymer phase formed after administration | Phase inversion, diffusion, degradation | Depot-forming biodegradable polymers | Minimally invasive depot formation |
| Polymer Microspheres | Micron-scale matrix particles | Diffusion plus matrix erosion | Polyester-rich biodegradable systems | Long-acting particulate depots |
| Microneedles/Transdermal Systems | Polymer-assisted skin delivery architecture | Dissolution, diffusion, skin-facing transport | Film-forming, dissolvable, or hybrid polymers | Nontraditional long-acting access |
| Inserts/Wafers/Implants | Structured solid devices | Matrix diffusion, barrier control, degradation | Persistent or degradable structural polymers | Geometry-defined extended release |
Injectable hydrogels are network-based systems that form or remain as water-rich polymer matrices capable of entrapping the API within a hydrated environment. Their utility in long-acting delivery comes from controllable mesh size, swelling, and localized retention rather than from dense barrier behavior. These systems are often attractive when the payload benefits from a mild aqueous environment and when diffusion must be moderated without exposing the drug to harsh thermal or solvent conditions. Polymer selection focuses on gelation pathway, crosslink density, and structural persistence, because these variables determine whether the hydrogel behaves as a stable depot or a rapidly relaxing matrix.
Polymer nanoparticles provide long-acting potential through carrier-based control at the nanoscale, where particle composition, surface chemistry, and internal morphology determine loading, circulation behavior, and release rate. Unlike bulk depots or implants, these systems must maintain colloidal integrity while also modulating payload release through matrix density, degradation, or shell permeability. They are especially useful when controlled transport, cellular interaction, or interface engineering is important. Material choice therefore extends beyond basic biodegradability and includes surface shielding, charge balance, and the ability to maintain a stable nanoscale architecture during storage, handling, and exposure to biological fluids.
Polymeric micelles are self-assembled nanostructures formed from amphiphilic polymers that organize into a hydrophobic core and hydrophilic corona in aqueous media. Their long-acting relevance is strongest for poorly soluble compounds that need both solubilization and improved retention in a carrier phase. Micellar performance depends on core compatibility, assembly stability, critical micelle behavior, and resistance to premature dilution-driven disassembly. Because the formulation is maintained by dynamic supramolecular organization rather than a fixed solid matrix, polymer selection focuses on block balance and thermodynamic stability as much as on conventional material descriptors such as degradation rate or mechanical strength.
In situ forming depots are systems that begin as injectable liquids, solutions, or suspensions and then transform into drug-containing depots after placement through solvent exchange, precipitation, or another phase transition. Their appeal lies in combining administration flexibility with depot-style sustained release, but their behavior is highly sensitive to polymer–solvent relationships and phase inversion kinetics. Early pore formation, burst release, and variable internal morphology are common design challenges. The most suitable polymers for these systems are those that can generate sufficiently cohesive depots while controlling water ingress, internal porosity, and later-stage degradation or diffusion without sacrificing injectability.
Polymer microspheres function as long-acting particulate depots in which the drug is entrapped within or distributed across micron-scale polymer particles. Their high surface area and process-derived internal morphology make them powerful but complex dosage forms. Release can arise from early surface-near diffusion, later polymer erosion, and progressive opening of internal porosity. As a result, microsphere performance depends strongly on polymer composition, emulsification conditions, solvent removal, and drug-state control. These systems are especially valuable when sustained release from an injectable particulate format is desired, but they require careful material tuning to reduce burst release and support consistent total release.
Polymeric microneedles create long-acting opportunities by combining minimally invasive skin access with polymer-controlled release or depot formation near the application site. Depending on the design, the polymer may dissolve quickly after insertion, persist as a structural scaffold, or deliver drug through hybrid architectures that incorporate sustained-release elements. Material selection therefore requires a balance between mechanical strength, fabrication precision, dissolution or persistence profile, and compatibility with the payload. Because microneedles operate at small dimensions and under skin-penetration constraints, polymers that are attractive in bulk implants may not perform well unless they also satisfy micromolding and structural reliability requirements.
Polymeric inserts are site-specific dosage systems designed to remain at a local administration site while releasing the drug over time through matrix diffusion, hydration, or structural evolution. Their long-acting value comes from controlled residence in a confined environment, which places strong emphasis on shape retention, local compatibility, and predictable hydration behavior. Polymer choice is influenced by site conditions, fluid exposure, and the need to avoid premature fragmentation or uncontrolled swelling. Compared with bulk depots, inserts often require more deliberate control of dimensions, surface properties, and local interaction because they function within defined anatomical microenvironments rather than broad tissue spaces.
Polymeric transdermal patches depend on polymer films, matrices, adhesives, or membrane layers that maintain drug distribution and skin contact while regulating sustained transport across the skin interface. Unlike injectable or implantable systems, patch performance is tied closely to film mechanics, tack, matrix diffusion behavior, and long-term compositional stability in a thin geometry. The most suitable polymers in these systems are not only those that modulate release, but those that also provide processable films and stable skin-facing performance. This makes transdermal selection more dependent on polymer mechanics and interfacial behavior than many bulk long-acting devices.
Implantable polymeric wafers are thin structured dosage forms that combine relatively high local drug density with geometry-defined release behavior. Their thinner dimensions create strong dependence on surface area, thickness uniformity, and polymer continuity, meaning that even small formulation inconsistencies can change release markedly. Material choice must therefore support thin-device processability, stable payload distribution, and controlled diffusion or degradation across a relatively flat structure. Wafers are especially useful when localized solid-state delivery is preferred, but they require polymers that maintain structural coherence without becoming so impermeable that useful release is delayed or incomplete.
Polymeric implants are among the most structurally defined long-acting systems because they rely on a preformed solid device with an intentional geometry, matrix design, or membrane barrier that regulates release over an extended period. They may function as degradable monoliths, reservoir systems, or hybrid architectures in which both diffusion and degradation contribute to performance. Material selection is therefore shaped by mechanical persistence, drug distribution, microstructural evolution, and the extent to which water ingress must be slowed or encouraged. Implants are highly tunable, but they require careful matching between device architecture and polymer transport behavior to avoid burst or residual drug trapping.
How API Properties Change Polymer and Platform Selection?
The same polymer or platform can perform very differently depending on the physicochemical profile of the API. For long-acting formulations, polymer selection should therefore start with the drug rather than with a preferred material family. Solubility, charge, molecular size, structural fragility, dose requirement, and compatibility with water, solvents, or thermal processing all shape which platforms remain realistic. A highly hydrophobic small molecule may favor matrix or micellar logic, while a protein or charged payload may require milder environments or functional interaction with the carrier. Starting from the API prevents material selection from becoming a list-based exercise disconnected from formulation reality.
Small Molecules and Matrix-Based Release Logic
Small molecules often offer the broadest platform flexibility, but their selection logic still depends heavily on solubility, partitioning, and dose density. Hydrophobic small molecules may fit well into polyester matrices, implant systems, microspheres, or amphiphilic carriers if the formulation maintains both loading stability and eventual release. More water-soluble compounds may require stronger barrier control to prevent rapid early escape. For many small molecules, the central question is whether the polymer environment supports a controlled concentration gradient without causing crystallization, phase migration, or excessive surface enrichment during processing and storage.
Peptides and Proteins in Sensitive Polymer Environments
Peptides and proteins introduce stronger constraints because they are often sensitive to water activity, interfacial stress, microenvironmental pH changes, and thermal or solvent exposure. Polymers for these payloads must therefore do more than slow release. They must preserve structural integrity and reduce conditions that promote unfolding, aggregation, adsorption, or degradation. Hydrogels, carefully designed microspheres, and selected biodegradable matrices may all be useful, but the success of each approach depends on how gently the polymer platform handles the biomolecule during fabrication, storage, and gradual release. Mildness and protective capacity become as important as release duration itself.
Charged Payloads and Functional Polymer Interactions
Charged payloads often require polymers that provide controlled electrostatic interaction rather than neutral encapsulation alone. Cationic polymers such as polyethyleneimine, linear or related functional materials can improve association with negatively charged macromolecules, but excessive interaction may reduce release efficiency or compromise stability. In these systems, polymer selection must balance binding strength, structural support, and the ability to maintain the payload in an active and releasable state. Hybrid systems are frequently valuable because one polymer can provide charge-mediated association while another supplies matrix integrity or long-acting transport control.
Poorly Soluble Compounds and Amphiphilic Carrier Selection
Poorly soluble compounds often need more than prolonged release; they need a polymer environment that improves loading state and prevents precipitation or overly strong entrapment. Amphiphilic carriers, hydrophobic matrices, and controlled-porosity systems can each be useful, depending on whether the main problem is dissolution, distribution, or late-stage release. In many cases, the best polymer is not the one that holds the drug most tightly, but the one that allows stable loading while still supporting transport once the formulation is hydrated. This is why micelles, nanoparticles, and certain matrix systems are especially relevant for poorly soluble payloads.
Dose, Stability, and Loading Constraints across Long-Acting Systems
Long-acting dosage forms must often carry meaningful dose levels in compact volumes or devices, so loading constraints become a central platform-selection variable. A polymer that supports an elegant release profile at low loading may become unreliable once the dose is increased and the drug begins to crystallize, migrate, or disrupt matrix continuity. Stability concerns also differ across APIs and platforms. Some compounds tolerate hydrophobic solid devices well, while others require highly controlled hydrated environments. Dose, physical stability, and formulation concentration therefore act as practical filters that narrow platform options before fine material optimization even begins.
Common Polymers in Long-Acting Delivery Platforms
Long-acting drug delivery platforms draw from several recurring polymer families, each contributing a distinct combination of transport behavior, structural role, and formulation flexibility. The importance of these materials lies less in their names alone and more in how they are used: some act as hydrophilic modifiers, others as barrier-forming device materials, degradable matrices, cationic interaction polymers, or functional components in hybrid systems. A useful overview of common polymer families helps connect platform categories with realistic material choices. It also clarifies why many successful long-acting systems are built from combinations of polymers rather than a single material performing every required function.
| Polymer Family | Primary Role | Key Strength | Main Limitation | Typical Platform Use |
|---|
| Polyethylene Glycol (PEG) | Hydrophilic modifier and transport regulator | Improves hydration and interfacial tuning | Can accelerate ingress if overused | Hydrogels, nanoparticles, blends, micelles |
| Ethylene-Vinyl Acetate (EVA) | Non-degradable membrane or barrier polymer | Stable diffusion control | Does not provide degradable matrix opening | Patches, inserts, reservoir systems |
| Polyvinyl Alcohol (PVOH) | Hydrophilic film-forming and matrix-modifying polymer | Useful hydration and film adjustment | Can increase water sensitivity | Hydrogels, patches, microneedles, blends |
| Polyurethanes (PUs) | Durable structural or barrier polymer | Tunable mechanics and permeability | Performance depends strongly on chemistry | Membranes, patches, persistent devices |
| Polyesters (PGA, PLA, PLGA, PCL) | Biodegradable matrices and depot-forming polymers | Broad long-acting formulation versatility | Can show burst, tail, or microenvironment complexity | Microspheres, depots, wafers, implants |
| Chitosan and HA | Functional natural polymers | Interactive hydrated-system utility | Often need structural partners | Hydrogels, inserts, microneedles, hybrids |
| Polymer Blends | Performance balancing through combined material roles | Greater design flexibility | Compatibility and reproducibility challenges | Hybrid long-acting systems |
Polyethylene Glycol (PEG)
Polyethylene glycol is widely used in long-acting delivery not usually as the only structural polymer, but as a hydrophilic modifier that changes water uptake, steric stabilization, and interfacial behavior. In nanoparticles, micelles, and hybrid matrices, PEG can reduce aggregation, improve dispersion, and adjust how water accesses the dosage system. In gel systems, PEG-derived materials may also contribute directly to network formation or mesh tuning. Its main value lies in controllable hydrophilicity and formulation flexibility. However, excessive PEG content can increase water ingress too aggressively and undermine long-duration control if the rest of the polymer architecture is not balanced appropriately.
Ethylene-Vinyl Acetate (EVA)
Ethylene-vinyl acetate is a classic example of a non-degradable polymer used for barrier or membrane control rather than degradable matrix opening. Its utility lies in stable permeability, film processability, and long-term structural consistency, which make it attractive in reservoir-style systems, patches, and inserts. EVA is especially useful when the formulation relies on controlled diffusion through a persistent polymer phase. The tradeoff is that it does not provide biodegradation-driven release adjustment, so performance depends heavily on membrane thickness, geometry, and internal drug distribution. In long-acting design, EVA is best understood as a barrier material rather than a general-purpose matrix polymer.
Polyvinyl Alcohol (PVOH)
Polyvinyl Alcohol is a hydrophilic, film-forming polymer that appears frequently in long-acting delivery as a matrix modifier, coating component, hydrogel contributor, or fabrication aid in structured polymer systems. Its strength lies in controllable hydration and mechanical film behavior, which can be useful in microneedles, patch-type designs, and hybrid matrices where water access must be adjusted rather than blocked entirely. Because it is highly water-responsive, PVOH can improve release pathways in otherwise restrictive systems. However, if it dominates the formulation or is poorly balanced with hydrophobic components, it may accelerate early release or reduce long-duration structural control.
Polyurethanes (PUs)
Polyurethanes are important long-acting materials because they provide durable mechanics and tunable permeability across a broad range of segmented structures. Depending on composition, they may function as flexible membrane materials, structural films, or device components in systems where persistent barrier behavior is required. Their usefulness is especially apparent in patches, inserts, and other device-based dosage forms that need long-term dimensional stability. At the same time, polyurethane performance is highly chemistry-dependent, which means the term itself is too broad to predict behavior without further structural detail. In formulation work, polyurethanes are most valuable when membrane mechanics and sustained permeability must be controlled together.
Polyesters
Biodegradable polyesters remain the most prominent material family in long-acting delivery because they can serve as structural matrices while also providing controlled degradation and evolving transport pathways. PLA and related materials favor slower hydration and stronger matrix retention, while glycolide-containing systems can support more dynamic opening and degradation. Among them, PLGA for long-acting drug delivery stands out because its monomer ratio, molecular weight, and end-group chemistry can be tuned to alter release behavior across multiple platforms. These materials are widely used in microspheres, depots, wafers, and implants, but their performance remains highly dependent on morphology and internal drug-state control.
Chitosan and Hyaluronic Acid (HA)
Chitosan and hyaluronic acid are natural or semi-natural functional polymers that play especially important roles in hydrated and site-specific long-acting systems. Chitosan contributes cationic interaction, mucoadhesion, and matrix-forming utility, while hyaluronic acid supports water-rich network behavior and local interface compatibility in soft delivery environments. These materials are particularly useful in hydrogels, inserts, microneedles, and hybrid depots where interaction with the local environment is part of the design logic. On their own, however, they do not always provide the long-term mechanical persistence needed for every platform, so they are frequently paired with additional structural polymers.
Polymer Blends and Hybrid Material Systems
Polymer blends and hybrid systems are often where long-acting design becomes most practical, because a single material rarely provides ideal loading, stability, mechanics, and release control simultaneously. Blending can combine hydrophobic matrix strength with hydrophilic transport tuning, or pair a structural polymer with a functional component that improves charge interaction, gelation, or local compatibility. The advantage is expanded design space and finer adjustment of release behavior. The challenge is that incompatibility between phases may create morphology drift, phase separation, or inconsistent performance. Successful blends therefore require careful attention to miscibility, process history, and long-term microstructural stability.
Need Help Choosing the Right Polymer Platform for Long-Acting Drug Delivery?
BOC Sciences supports polymer screening, platform selection, and release-oriented formulation design for long-acting delivery systems across hydrogels, depots, microspheres, implants, wafers, and transdermal formats.
How to Select Polymer Materials for Long-Acting Delivery Platforms?
Once platform type and API constraints are defined, polymer selection becomes a process of matching material behavior to the dominant release logic of the system. A useful material-selection workflow considers whether the dosage form needs degradability, barrier persistence, hydrophilic transport modulation, or interactive functionality such as charge-based association or gel formation. This section moves beyond material names and focuses on how polymer families are actually chosen in long-acting formulation work. The goal is not to identify a universally best polymer, but to understand which material strategy fits a given platform, payload, and release objective with the least structural compromise and the greatest tunability.
Biodegradable Polyesters in Long-Acting Formulation Design
Biodegradable polyesters are often the first materials considered for long-acting depots, microspheres, wafers, and implants because they offer both matrix structure and time-dependent opening through hydrolytic degradation. Their usefulness comes from the ability to tune chain composition, hydrophobicity, and erosion pace rather than from degradation alone. These polymers can support extended release when drug diffusion through the intact matrix is limited initially and becomes more permissive over time. However, they require careful design because degradation products, internal porosity, and drug distribution can all alter release unpredictably if composition and morphology are not tightly controlled from the start.
Hydrophilic and Amphiphilic Polymers for Transport Modulation
Hydrophilic and amphiphilic polymers become especially important when the main design challenge is controlling water access, local transport pathways, or self-assembly behavior rather than creating a rigid degrading matrix. These materials can increase hydration, stabilize dispersed drug states, or support carrier systems such as micelles and nanoparticles. They are useful for poor-solubility problems, water-rich depot environments, and hybrid systems where complete release would otherwise be difficult. Their role is often modulatory rather than purely structural, which means they work best when their contribution to hydration and interfacial behavior is tuned carefully against the need for sustained release and physical consistency.
Functional and Natural Polymers for Hybrid Delivery Systems
Functional and natural polymers are particularly valuable when long-acting performance depends on more than transport control alone. They may provide charge interaction, mucoadhesion, gelation, local compatibility, or soft-matrix behavior that strengthens a specialized platform even if they are not the sole structural polymer. In hybrid systems, these materials can be paired with more persistent matrices to create better balance between interaction and durability. Their selection is often driven by localized delivery goals or payload sensitivity rather than by conventional sustained-release logic alone. This makes them especially relevant in hydrogels, inserts, microneedles, and other site-specific or interface-dependent long-acting platforms.
Non-Biodegradable Polymers for Reservoir and Barrier-Controlled Systems
Non-biodegradable polymers are preferred when long-acting behavior depends on stable diffusion control instead of progressive matrix erosion. In reservoir systems, inserts, patches, or persistent device components, these materials provide reproducible barrier performance, film stability, and mechanical persistence across the entire release window. Their value lies in maintaining a predictable pathway for transport, often governed by permeability, thickness, and exposed area. Because they do not rely on degradation to enable release, their design demands greater attention to geometry and core-shell organization. When barrier consistency is more important than matrix clearance, non-biodegradable materials often become the more rational strategy.
Matching Polymer Families with Platform Architecture
A polymer family should always be chosen in relation to platform architecture rather than in isolation. Hydrogels need network-forming or swelling-manageable polymers, micelles need amphiphilic self-assembly, microspheres and depots often need tunable biodegradable matrices, and patches or reservoir systems may require persistent barrier materials. The same polymer can behave differently across these formats because geometry, phase continuity, and hydration mode change the governing mechanism. Matching material family with architecture therefore means understanding which polymer role is required first, whether matrix former, membrane, carrier shell, modifier, or hybrid component, and then selecting chemistry that supports that role with adequate processability.
Release Mechanisms in Polymer-Based Long-Acting Drug Delivery Systems
Long-acting performance is best understood through release mechanisms rather than through platform names alone. A dosage form may be called a depot, implant, or microsphere, but its actual behavior depends on whether release is governed primarily by diffusion, swelling, barrier permeability, degradation, or combinations of these processes over time. In many cases, different mechanisms dominate at different stages of the release profile. Early transport may be controlled by surface-near diffusion, while later behavior depends on erosion or pore connectivity. Understanding these transitions is essential because material selection, geometry, and processing all influence how and when one mechanism gives way to another.
Diffusion-Controlled Release
Diffusion-controlled release occurs when dissolved drug molecules migrate through a polymer phase, water-filled channels, or a rate-controlling membrane according to concentration gradients. This mechanism is common in reservoir devices, films, and some dense matrices where polymer degradation is slow relative to transport. The main formulation variables are polymer permeability, drug solubility within the matrix, path length, and hydration state. Diffusion control can provide smooth sustained release if the internal structure remains stable. However, it is sensitive to surface enrichment, pore connectivity, and any change in membrane integrity, all of which can distort the intended profile.
Degradation- and Erosion-Controlled Release
Degradation- and erosion-controlled release becomes important when the drug cannot leave efficiently until the polymer phase changes chemically or structurally. In biodegradable matrices, hydrolysis reduces chain length, weakens matrix continuity, and eventually opens transport pathways that were initially restrictive. This mechanism is central to many polyester-based depots, microspheres, wafers, and implants. It is especially useful when an initially dense structure is needed for loading stability or device integrity. The challenge is that degradation is rarely uniform, so changes in internal microenvironment, porosity, and local acidity can complicate the release profile if material composition is not optimized carefully.
Swelling-Controlled and Osmotically Assisted Release
Swelling-controlled release is driven by the ability of the polymer network to absorb water, expand, and alter internal mesh or channel dimensions over time. This mechanism is particularly relevant in hydrogels and other hydrophilic systems where structural relaxation directly affects payload transport. In some designs, osmotic gradients further accelerate water entry or drive internal pressure changes that support release. Swelling can be advantageous because it allows controlled opening without requiring full polymer degradation. At the same time, it must be balanced carefully, since excessive or poorly timed swelling may destabilize geometry, increase burst release, or shorten the effective duration of the formulation.
Matrix Opening, Pore Formation, and Water Ingress
Water ingress is one of the most influential hidden variables in long-acting systems because it determines when the internal matrix begins to change. As water enters the dosage form, it can dissolve near-surface drug, plasticize the polymer, initiate hydrolysis, and create channels or pores that accelerate later transport. In in situ depots and many degrading matrices, pore formation is central to the shift from slow initial release to faster internal depletion. Material composition, hydrophilic content, process history, and drug loading all influence how these pathways develop. Matrix opening is therefore a mechanistic bridge between material design and observed release behavior.
Burst Release, Tail Release, and Incomplete Release
Many long-acting formulations fail not because they release too little overall, but because they release in the wrong pattern. Burst release often reflects surface-associated drug, early porosity, or rapid water ingress. Tail release may appear when the device becomes too restrictive at later stages or when residual drug remains trapped in slowly opening regions. Incomplete release can arise from strong entrapment, poor channel connectivity, phase separation, or insufficient polymer transformation. These phenomena are not independent defects; they are often the visible consequences of mismatches between polymer chemistry, internal drug state, platform architecture, and processing-induced microstructure.
How Polymer Properties Shape Drug Release and Formulation Design?
Release mechanisms explain what happens, but polymer properties explain why it happens in a specific way. Within the same platform, changes in molecular weight, monomer composition, crystallinity, drug distribution, geometry, or processing history can produce very different release outcomes. This is why effective long-acting formulation design depends on property-level reasoning rather than material labels alone. A polymer family may appear suitable on paper, yet fail in practice if its morphology, hydration behavior, or drug compatibility generate the wrong internal microstructure. Understanding these property–performance relationships is essential for moving from broad platform selection to robust formulation design and rational optimization.
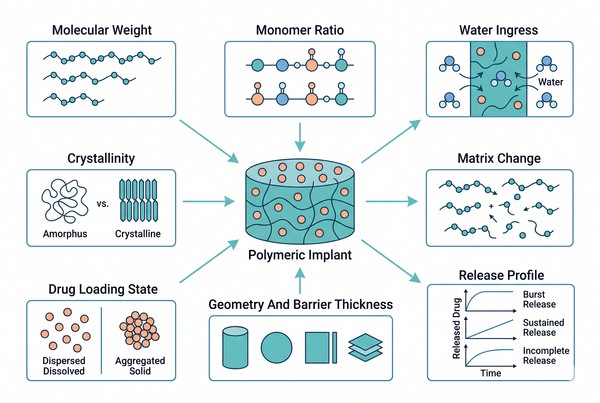 Fig. 2. How polymer properties shape release behavior and formulation design (BOC Sciences Authorized).
Fig. 2. How polymer properties shape release behavior and formulation design (BOC Sciences Authorized).
Molecular Weight, Monomer Ratio, and Crystallinity
Molecular weight influences chain mobility, entanglement, and the rate at which the matrix reorganizes or degrades. Monomer ratio alters hydrophobicity, hydrolysis tendency, and thermal behavior, especially in copolyesters. Crystallinity affects water penetration and transport by creating ordered regions that resist diffusion and structural change. Together, these three variables strongly influence whether a formulation releases too quickly, retains drug too long, or transitions between phases of release smoothly. In long-acting development, they are not merely descriptive polymer attributes; they are primary formulation levers that determine how the dosage form behaves once exposed to the release environment.
Drug Loading State and Internal Distribution
The internal state of the drug inside the polymer can range from molecular dispersion to amorphous domains, fine crystals, or large segregated particles, and each state produces different release behavior. Uniform internal distribution generally improves predictability, while surface enrichment and phase separation often increase burst risk. High drug loading may create useful concentration gradients, but it can also destabilize matrix continuity or induce crystallization during storage. For long-acting platforms, drug state should be treated as a critical formulation variable because release depends not only on how much drug is present, but on how that drug is physically arranged inside the polymer phase.
Geometry, Surface Area, and Barrier Thickness
Geometry provides a practical way to tune long-acting behavior because it changes exposed area, diffusion path length, and the ratio of surface-associated material to total payload. Thin devices, small particles, and porous structures generally release faster than larger or thicker systems built from the same polymer. In barrier-controlled designs, membrane thickness can be as important as material chemistry, since transport may depend more on path length than on degradation. Geometry also affects hydration gradients and the rate at which the internal structure transforms. This makes dimensional design one of the most direct and scalable tools in long-acting formulation optimization.
Solvent Systems, Thermal Processing, and Phase Transformation
Processing determines microstructure, and microstructure determines long-acting behavior. Solvent-based fabrication can create porosity, alter drug distribution, and leave residual domains that change early hydration. Thermal processing may improve uniformity for some APIs but damage others or alter crystallinity in ways that affect release. In situ forming depots add further complexity because phase transformation occurs during administration rather than before it. The correct polymer for a platform is therefore inseparable from the process used to fabricate that platform. A material that appears suitable chemically may still fail if its preferred processing pathway is incompatible with the payload or final morphology target.
Stability, Morphology, and Reproducibility in Formulation Development
Long-acting formulations must remain structurally reliable over time, which means polymer selection must account for storage stability, morphology retention, and reproducible manufacturing behavior. Changes in crystallinity, moisture content, phase distribution, or drug domain structure can alter the release profile before the formulation is ever used. Reproducibility is equally important because small process variations can produce different pore structures, surface states, or matrix densities. For this reason, polymer screening should include not only release testing, but also morphology and stability evaluation. A polymer is genuinely suitable only when it supports both the intended mechanism and consistent formulation behavior.
How to Match the Right Delivery Form with the Right Polymer Strategy?
Choosing the right long-acting system requires combining all the previous layers of reasoning into one final decision: which platform best fits the API and which polymer strategy best fits that platform. This is where many projects become inefficient, because teams compare platforms and polymers independently instead of linking them through mechanism and dosage architecture. A useful match should reflect the payload's sensitivity, the desired release logic, the practical loading requirement, and the level of structural persistence needed. In many cases, platform choice narrows polymer options more effectively than material screening alone, especially when geometry and processing constraints are strong.
When Hydrogels Are More Suitable than Solid Devices
Hydrogels are often more suitable than solid devices when the payload benefits from a water-rich environment and when localized retention must be achieved without relying on a rigid structure. They are especially useful for sensitive molecules that do not tolerate strong dehydration, harsh thermal processing, or highly hydrophobic matrices. Their network-based release logic also allows adjustment through crosslink density and swelling rather than through dense barrier design. However, hydrogels are not ideal when strong mechanical persistence or highly geometry-defined release is required. Their value is greatest when mildness and adaptable hydration matter more than device-like structural control.
When Microspheres or Depots Offer Better Long-Acting Control
Microspheres and in situ depots become stronger options when sustained release must be achieved from injectable dosage forms that create a localized polymer-rich phase after administration. Microspheres are advantageous when particle-based manufacturing and preformed morphology control are desirable, while depots are attractive when minimally invasive placement and post-administration phase formation are priorities. In both cases, biodegradable matrix polymers often dominate the strategy because controlled erosion and porosity development contribute to release. These systems are particularly valuable when a compact injectable long-acting format is needed, but they demand close attention to burst release, solvent effects, and internal drug-state control.
When Implants, Inserts, or Wafers Provide Stronger Structural Advantages
Implants, inserts, and wafers are often preferred when strong structural definition is needed and when geometry itself is a major release-control variable. These formats allow direct tuning through thickness, surface area, path length, and matrix continuity in ways that are harder to achieve in dispersed or self-assembled carriers. They are especially useful when long residence, stable shape, or localized delivery from a preformed solid structure is desired. The best polymer strategy in these systems depends on whether the design relies on a degrading matrix, a persistent diffusion barrier, or a hybrid structure that combines both mechanisms.
When Nanoparticles and Micelles Are Better for Solubility or Carrier Logic
Nanoparticles and micelles become the better route when the formulation problem is tied to carrier logic as much as to release duration. They are especially relevant for poorly soluble compounds, payloads that benefit from nanoscale interface control, and systems where matrix or device formats would be too restrictive or impractical. Polymeric micelles are generally favored when solubilization and amphiphilic stabilization are central, whereas nanoparticles are more appropriate when a defined polymer matrix or shell is needed at small scale. In both cases, release depends on carrier stability and payload retention rather than on bulk geometry alone.
When Transdermal and Microneedle Systems Become the Better Route
Transdermal patches and microneedle systems become better routes when long-acting delivery benefits from surface-based access rather than from implanted or injected depots. Their polymer strategies are strongly shaped by film mechanics, adhesive behavior, dissolution profile, and skin-interface transport rather than by bulk erosion or large depot formation. These platforms are valuable when dosing convenience, device-like application, or localized skin-mediated delivery is part of the design objective. However, they demand polymers that perform reliably in thin, mechanically constrained formats. This makes material selection more dependent on fabrication precision and interfacial control than in many injectable systems.
Services
Advanced Polymer Synthesis and Formulation Services
At BOC Sciences, we support polymer-based long-acting drug delivery development from the perspective of material logic, platform fit, and release-oriented formulation design. Our capabilities span polymer screening, custom synthesis, functional modification, and characterization strategies that help researchers connect polymer structure with long-acting performance across depots, hydrogels, microspheres, implants, wafers, and transdermal systems. We work to clarify how molecular weight, hydrophilic balance, charge characteristics, and processing conditions affect loading, transport, stability, and morphology. By combining material development with formulation insight, BOC Sciences helps teams build stronger polymer strategies for complex sustained and extended-release programs.

Custom Polymer & Copolymer Synthesis
- Tailored development of biodegradable, hydrophilic, and functional polymers for long-acting platforms.
- Control over composition, architecture, and molecular-weight-related design variables.
- Support for platform-fit material design across depots, particles, hydrogels, and devices.
- Polymer candidates aligned with release behavior and formulation feasibility goals.

Polymer Functionalization & Material Optimization
- Hydrophilic, cationic, and hybrid modification strategies for tuning polymer performance.
- Material optimization for carrier stability, matrix opening, and barrier control.
- Blend design support for combining structural and functional polymer roles.
- Refinement of polymer candidates around platform-specific long-acting requirements.

Long-Acting Formulation Development Support
- Material and platform screening for hydrogels, depots, microspheres, wafers, and implants.
- Assessment of drug loading, internal distribution, and release-mechanism alignment.
- Support for morphology-aware process selection and formulation refinement.
- Practical guidance on matching polymer strategy with dosage-form architecture.

Analytical Characterization & Technical Evaluation
- Characterization of molecular weight, crystallinity, thermal behavior, and drug state.
- Morphology and stability assessment for long-acting formulation consistency.
- Evaluation of release-related structural changes during formulation screening.
- Technical support for building more reproducible polymer-based delivery systems.
Do You Need A Consultation?
BOC Sciences supports polymer selection, platform comparison, and formulation design for long-acting drug delivery systems built around hydrogels, depots, microspheres, implants, and other advanced polymer-based dosage architectures.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What are polymer-based long-acting drug delivery systems?
Polymer-based long-acting drug delivery systems are formulations in which a polymer controls how a drug is retained, protected, and released over time. The polymer may function as a matrix, membrane, network, carrier shell, or self-assembled structure, depending on the dosage platform and intended release mechanism.
-
Why are polymers important in long-acting drug delivery?
Polymers are important because they directly shape hydration, diffusion, degradation, swelling, and structural persistence within a long-acting formulation. Unlike conventional excipients, they often define the release mechanism itself, making polymer selection central to both dosage architecture and long-term formulation performance.
-
What are the main polymer-based platforms for long-acting delivery?
Main polymer-based platforms include hydrogels, nanoparticles, micelles, in situ depots, microspheres, microneedles, inserts, transdermal patches, wafers, and implants. Each platform uses polymers differently, so the best choice depends on the API, release objective, processing route, and structural demands of the formulation.
-
How do API properties affect polymer selection?
API properties influence polymer selection through solubility, charge, molecular size, stability, and dose requirement. These factors determine whether the payload fits better in a degrading matrix, a hydrated network, an amphiphilic carrier, or a barrier-controlled device, and they also define acceptable processing conditions.
-
What polymers are most commonly used in long-acting delivery systems?
Common polymers include biodegradable polyesters such as PLA, PLGA, and PCL, hydrophilic modifiers such as PEG and PVOH, barrier materials such as EVA and selected polyurethanes, and natural functional polymers such as chitosan and hyaluronic acid used in hybrid or hydrated systems.
-
What release mechanisms are involved in polymer-based long-acting systems?
Key release mechanisms include diffusion-controlled transport, degradation- and erosion-driven release, swelling-mediated release, and transport caused by matrix opening or pore formation. In many long-acting formulations, different mechanisms dominate at different stages rather than one mechanism controlling the entire profile.
-
Why do burst release and incomplete release happen?
Burst release often results from surface-associated drug, early porosity, or rapid water ingress, while incomplete release may reflect strong entrapment, poor pore connectivity, or matrix regions that do not open sufficiently. Both effects usually indicate mismatch between polymer properties, processing history, and internal drug distribution.
-
How do I match the right platform with the right polymer strategy?
The best match comes from linking API constraints with the dominant release mechanism and structural needs of the platform. Hydrogels favor hydrated control, microspheres and depots often rely on degradable matrices, and implants or patches may require more geometry-defined or barrier-based polymer strategies.

 Fig. 1. Overview of major polymer-based long-acting delivery platforms (BOC Sciences Authorized).
Fig. 1. Overview of major polymer-based long-acting delivery platforms (BOC Sciences Authorized). Fig. 2. How polymer properties shape release behavior and formulation design (BOC Sciences Authorized).
Fig. 2. How polymer properties shape release behavior and formulation design (BOC Sciences Authorized).