Polymer Nanoparticles for Long-Acting Drug Delivery: Formulation Strategies, Release Logic, and Material Trade-Offs
Polymer nanoparticles are highly adaptable carriers for long-acting drug delivery because they combine tunable polymer chemistry with controllable particle architecture, surface functionality, and payload association strategies. By adjusting polymer class, particle size, surface charge, internal morphology, and loading method, formulators can influence colloidal stability, payload retention, tissue distribution, and release behavior across a wide range of therapeutic formats. Unlike simple nanosuspensions, polymer nanoparticles can be engineered to provide not only transport and protection, but also matrix-mediated release control, making them especially useful when formulation goals include prolonged local retention, improved payload stability, or better balance between delivery efficiency and release duration.
Resources
Polymer Nanoparticles for Long-Acting Drug Delivery
In long-acting formulation development, polymer nanoparticles sit at the intersection of carrier design, payload protection, and controlled release engineering. They are smaller and more interfacially active than depot microspheres, but more structurally programmable than many self-assembled low-molecular-weight systems. This makes them attractive when a project requires a carrier that can encapsulate or complex a drug, adjust transport through tissues or biological fluids, and still provide some degree of sustained or delayed release. At the same time, not every nanoparticle is truly a long-acting platform. Some nanoparticles improve solubility, circulation, or uptake without meaningfully slowing payload escape. A useful long-acting nanoparticle strategy therefore depends on more than size reduction. It depends on how the polymer matrix, the payload state, and the particle structure work together over time.
What Defines a Polymer Nanoparticle System?
A polymer nanoparticle system is a colloidal carrier in which the structural framework is built primarily from polymeric materials rather than from simple surfactants, inorganic solids, or lipid-only assemblies. Depending on the formulation route, the drug may be dissolved in a polymer matrix, confined inside a core-shell particle, adsorbed near the surface, or associated electrostatically with charged polymer domains. This flexibility allows polymer nanoparticles to serve as more than passive nanocarriers. Their composition and internal organization can actively shape release behavior, payload stability, and transport through complex formulation environments.
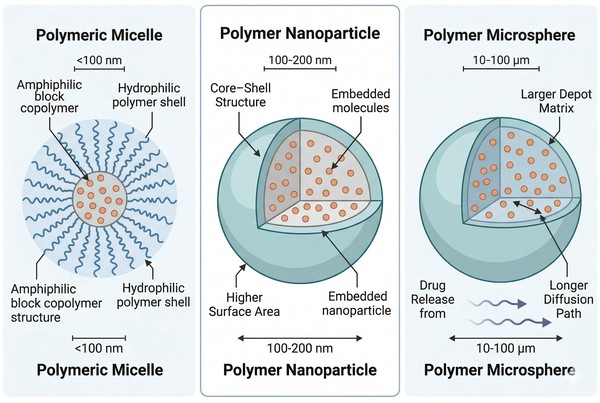 Fig. 1. Polymeric nanoparticles differ from micelles and microspheres in structure, scale, and release behavior (BOC Sciences Authorized).
Fig. 1. Polymeric nanoparticles differ from micelles and microspheres in structure, scale, and release behavior (BOC Sciences Authorized).
How Polymer Nanoparticles Differ from Microspheres and Micelles?
Polymer nanoparticles differ from microspheres mainly in scale, interfacial area, and transport behavior. polymeric microspheres are larger depot systems that often depend on bulk matrix degradation and longer diffusion paths, while nanoparticles offer higher surface area and more direct interaction with the surrounding medium. They also differ from polymeric micelles, which are usually self-assembled from amphiphilic polymers and can dissociate if the assembly is not sufficiently stable. Polymer nanoparticles are often more structurally persistent than micelles, but they can still release too quickly if the payload is weakly retained or concentrated near the surface.
Why Polymer Nanoparticles Are Used in Long-Acting Delivery?
Polymer nanoparticles are used in long-acting delivery when the formulation requires a combination of small carrier size, tunable release, and payload protection. They are especially attractive when a dense implant or large particulate depot is not ideal, or when the payload benefits from nanoscale encapsulation rather than bulk matrix embedding. In some systems, long-acting performance comes from slow polymer degradation; in others, it comes from strong matrix retention, local tissue residence, or reduced burst release compared with free drug. Their value is therefore highly context-dependent and tied directly to the intended delivery objective.
What Polymer Materials Are Used in Nanoparticle Formulations?
Material selection determines how a nanoparticle is formed, how it interacts with the payload, and how it behaves after formulation. Different polymer classes provide different balances of degradability, hydrophobicity, charge, mechanical integrity, colloidal stability, and release control. For this reason, polymer choice should be linked directly to the payload's physicochemical properties and the intended release mechanism, rather than treated as a secondary variable after particle fabrication has already been chosen.
Biodegradable Polyester Nanoparticles
Nanoparticles based on biodegradable polymers such as PLGA, PLA, and PCL are widely used because they can combine encapsulation with degradation-assisted release. These materials are particularly useful for hydrophobic payloads or projects that require matrix erosion to contribute to sustained release. Their hydrophobic cores often support better retention than highly water-rich systems, but the resulting particles may still show significant burst release if morphology is porous or the drug is concentrated near the surface. Within this class, differences in polymer composition, molecular weight, crystallinity, and end-group chemistry strongly affect release behavior and fabrication outcomes. A nanoparticle made from a faster-hydrating polyester may release more quickly than one made from a more hydrophobic or slower-relaxing matrix, even when particle size looks similar. For users comparing polyester nanocarriers more broadly, these systems often overlap conceptually with polymer nanoparticles for drug delivery and with sustained-release logic discussed in polyester-based depot systems.
Amphiphilic Copolymer Nanoparticles
Amphiphilic copolymers are useful when the formulation must balance hydrophobic drug loading with aqueous dispersibility and steric stabilization. Depending on composition and fabrication route, these polymers can form nanoparticles with core-shell organization, interfacial protection, and improved colloidal behavior. They are often selected when simple hydrophobic matrices are too aggregation-prone or when surface hydration is needed to improve particle handling and reduce nonspecific interactions. The trade-off is that amphiphilic systems can behave closer to stabilized assemblies than dense depot particles if the structure is not sufficiently locked or if the payload partitions easily into water. This means they may improve loading and dispersion without necessarily producing strong long-acting release unless the polymer composition, core density, and process conditions are chosen carefully. They are especially useful when the formulation goal includes both nanocarrier stability and controlled payload residence, rather than pure degradation-driven release.
Cationic and Functional Polymer Nanoparticles
Cationic and functional polymers are often used when the payload is charged, structurally sensitive, or dependent on electrostatic association rather than hydrophobic encapsulation. Polymers such as poly-γ-glutamic acid-related systems, polyamines, polycationic derivatives, and functional copolymers can form nanoparticles that associate with nucleic acids, peptides, or other charged molecules through ionic interactions. In these systems, the polymer does not just define the particle structure. It also defines how strongly the payload is retained, shielded, and later released. These materials are especially relevant when the project extends into polymer nanoparticles for gene delivery or related polymer-based gene delivery platforms. However, they require careful tuning because stronger charge can improve loading while simultaneously increasing aggregation risk, toxicity concerns in some contexts, or overly tight payload binding. For long-acting use, the key is to balance protection and controlled release rather than maximize electrostatic condensation alone.
Natural and Semi-Synthetic Polymer Nanoparticles
Materials from the broader family of natural polymers are often selected when aqueous processing, mild formulation conditions, or intrinsic biofunctionality are important. Examples such as chitosan and dextran derivatives can support nanoparticle formation through ionic gelation, polyelectrolyte association, or modified self-assembly routes. These materials are especially attractive for sensitive payloads because they can reduce exposure to harsh solvents and allow more adaptable surface chemistry. Their limitations usually involve broader material variability, less predictable matrix density, and more complex control of long-term release. In some cases, they are better suited to payload protection or mucoadhesion than to true extended release. For this reason, natural and semi-synthetic nanoparticles should be selected when their specific material advantages solve a real formulation problem, not simply because they are perceived as inherently gentle or general-purpose.
How Polymer Class Affects Loading and Release Behavior?
Polymer class affects not only whether a nanoparticle can be formed, but how the payload is distributed and how the release profile evolves. Hydrophobic polyesters usually support matrix encapsulation and degradation-assisted release. Amphiphilic copolymers often improve aqueous stability and solubilization but may release more quickly if the core is insufficiently retentive. Cationic polymers can strongly associate with charged payloads but may create incomplete release if binding is too strong. Natural polymers may offer mild processing but less rigid control over long-term structure. The best material class is therefore the one whose dominant interaction mode matches the formulation objective. If the project needs a hydrophobic matrix depot at nanoscale, a biodegradable polyester may be appropriate. If it needs protected association with a charged payload, a functional polymer may be better. If it needs colloidal stability with a self-organized core-shell structure, an amphiphilic system may be preferred. Rational selection begins by identifying which release and retention mechanism is actually needed, then choosing the polymer class that supports it with the least added complexity.
| Polymer Class | Typical Examples | Main Loading Logic | Main Release Logic | Key Advantage | Main Limitation |
|---|
| Biodegradable polyesters | PLGA, PLA, PCL | Matrix encapsulation of hydrophobic or dispersed payloads | Diffusion plus degradation-assisted release | Strong structural retention and tunable degradation | Burst release if drug is surface enriched |
| Amphiphilic copolymers | PEG-containing block or graft copolymers | Core loading with shell stabilization | Diffusion and matrix partitioning | Good colloidal stability and dispersibility | May release too fast if core retention is weak |
| Cationic and functional polymers | Polyamines, pyridinium polymers, polylysine-related systems | Electrostatic association or complexation | Desorption, disassembly, or environment-triggered release | Useful for charged and sensitive payloads | Aggregation and overbinding risk |
| Natural and semi-synthetic polymers | Chitosan, dextran derivatives | Ionic gelation or mild aqueous assembly | Diffusion with matrix relaxation | Mild processing and adaptable surface chemistry | Less rigid long-term release control |
Need a Polymer Nanoparticle Strategy for Long-Acting Delivery?
From polymer selection and nanoparticle synthesis to loading optimization and release tuning, we support long-acting nanocarrier development for small molecules, proteins, and nucleic acid-related payloads.
Which Structural Features Most Affect Nanoparticle Performance?
In polymer nanoparticle development, structural attributes are not just characterization outputs. They are direct controls on release, colloidal stability, payload exposure, and platform fit. Two formulations made from the same polymer can perform very differently if they differ in size distribution, surface properties, internal organization, or drug placement. That is why nanoparticle structure must be designed as part of the formulation strategy rather than treated as a passive consequence of processing.
Particle Size and Size Distribution
Particle size affects surface area, diffusion path length, interfacial exposure, and transport through the formulation environment. Smaller nanoparticles often disperse well and can improve interaction with tissues or cells, but they also expose more payload near or at the surface, which can accelerate release. Larger nanoparticles may improve retention within the matrix, but they can also broaden size-related variability if the fabrication process is not tightly controlled. Size distribution matters just as much as average size because broad distributions often create mixed release behavior across the same batch. For long-acting design, size should be selected based on the intended release logic rather than on a generic preference for smaller particles. A system optimized for circulation, local tissue residence, or slower release may require a different size range than one optimized for uptake or rapid dispersion. Good nanoparticle design therefore treats size as a functional variable connected to release and stability, not just a target number on a characterization report.
Surface Charge and Colloidal Stability
Surface charge is a major determinant of colloidal stability because it influences interparticle repulsion, interaction with proteins or salts in the surrounding medium, and the likelihood of aggregation during storage. Strong surface charge can help stabilize a suspension, but it can also alter biological interaction and accelerate payload loss if the drug is loosely held near the particle exterior. In functional or cationic systems, charge also influences how the payload associates with the carrier and how easily it may be displaced. From a long-acting perspective, the best surface charge is not necessarily the highest magnitude. It is the charge profile that maintains colloidal stability while preserving the desired payload state and release pattern. This often requires balancing stabilizer choice, polymer end groups, ionic environment, and surface composition instead of relying on one nominal zeta potential target.
Core-Shell Structure and Surface Modification
Many polymer nanoparticles are not uniform solid spheres. They may have dense cores, hydrated shells, layered interfaces, or modified outer surfaces designed to improve dispersion or alter interaction with the external medium. Core-shell structure becomes especially important when hydrophobic loading and aqueous stability must be balanced at the same time. A stable shell can reduce aggregation and protect the particle, while a more retentive core can slow payload escape. Surface modification further changes how the nanoparticle behaves by affecting steric stabilization, hydration, and interaction with surrounding components. However, modifications that improve colloidal stability do not always improve long-acting release. In some cases, they may even accelerate payload leakage by increasing matrix hydration or reducing internal packing density. This is why surface design should always be evaluated together with loading state and release profile, not as a separate optimization track.
Particle Morphology and Internal Drug Distribution
Internal morphology determines whether the nanoparticle is dense, porous, hollow, phase-separated, or compositionally heterogeneous. These structural differences strongly influence how water enters the matrix and how quickly the payload becomes exposed to release media. Internal drug distribution is equally important. Payload confined deep within a dense matrix usually releases more slowly than drug concentrated near the surface or trapped in water-accessible defects. Morphology is often shaped by process history, including solvent exchange rate, polymer concentration, surfactant system, and hardening conditions. For this reason, unexplained differences in release are often morphology problems rather than polymer problems. Good formulation work therefore treats morphology as a critical quality attribute that directly connects processing to long-acting behavior.
How Drug Loading and Release Work in Polymer Nanoparticles?
Drug loading in polymer nanoparticles is not just a matter of how much payload can be added. It determines where the payload sits, how stably it is retained, and which release pathway becomes dominant after formulation. A nanoparticle can only function as a long-acting platform if the drug remains associated with the structure strongly enough to avoid rapid loss, yet not so strongly that release becomes incomplete or impractical. Understanding how loading state translates into release logic is therefore central to rational design.
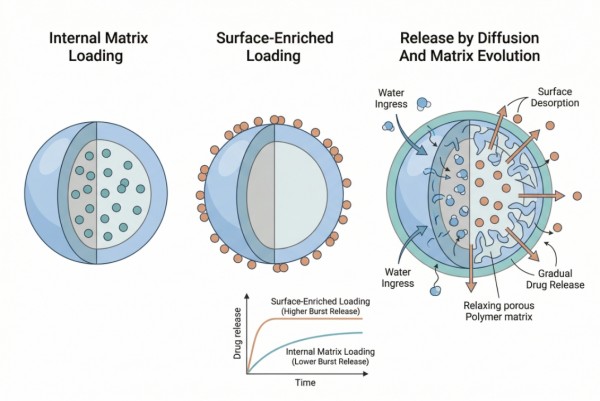 Fig. 2. Loading state, internal distribution, and matrix evolution jointly determine nanoparticle release behavior (BOC Sciences Authorized).
Fig. 2. Loading state, internal distribution, and matrix evolution jointly determine nanoparticle release behavior (BOC Sciences Authorized).
Loading Hydrophobic Drugs into Polymer Matrices
Hydrophobic drugs are commonly loaded by dissolving or dispersing them into the polymer phase during nanoparticle formation. In well-matched systems, the drug partitions into the core or matrix and remains retained until diffusion, swelling, or degradation gradually opens transport pathways. This approach is often effective with polyester-based or amphiphilic polymer nanoparticles because the matrix can provide a relatively favorable environment for poorly water-soluble compounds. The challenge is that apparent loading is not always equivalent to stable loading. A drug may be detected in the particle yet still be concentrated near the surface or in loosely trapped domains that produce rapid release after dispersion. For hydrophobic payloads, true long-acting performance depends on drug–polymer compatibility, internal distribution, matrix density, and how strongly the surrounding medium promotes partitioning out of the nanoparticle.
Loading Charged and Sensitive Payloads
Charged or structurally sensitive payloads often require association or complexation rather than simple matrix dissolution. In these systems, cationic or functional polymers may bind nucleic acids, peptides, or other charged molecules through electrostatic interactions, protective layering, or hybrid assembly. This can improve loading and shield the payload from degradation, especially in platforms related to polymers for nucleic acid delivery or protein and peptide delivery. For long-acting delivery, however, strong association is only useful if it remains reversible and structurally stable over time. If the payload is released too easily, the nanoparticle fails as a depot. If the binding is too strong, the payload may remain trapped or lose functionality. Selection must therefore balance protective association with controlled liberation, especially for systems designed for gene-related or macromolecular delivery.
Diffusion-Controlled and Degradation-Assisted Release
Release from polymer nanoparticles is commonly driven by diffusion, matrix relaxation, desorption, degradation, or a combination of these mechanisms. In hydrophobic polymer matrices, early release may be dominated by diffusion from accessible regions, while later release may become more dependent on polymer hydration or degradation. In other systems, especially those formed by ionic association or less dense assembly, release may remain dominated by diffusion and environmental displacement rather than by matrix erosion. Whether a nanoparticle behaves as a long-acting system depends on which of these mechanisms dominates across the relevant timescale. A particle that relies mainly on surface desorption is unlikely to sustain release meaningfully, while one that combines internal retention with gradual matrix evolution may offer better control. The release mechanism must therefore be matched intentionally to the formulation objective instead of inferred after the fact from cumulative data alone.
Surface Desorption and Burst Release in Nanoparticles
Burst release in nanoparticles often comes from drug associated with the outer shell, weakly bound at the interface, or trapped in water-accessible defects near the surface. Because nanoparticles have high surface area, even a modest amount of superficial payload can create a disproportionate early release event. This is especially problematic when the platform is intended for long-acting performance, because early loss can remove the very fraction needed to sustain the later profile. Surface desorption is not just a loading issue. It is often a formulation-history issue linked to process rate, solvent exchange, polymer precipitation path, and how the payload partitions during particle formation. Effective mitigation therefore requires both material understanding and process redesign. Reducing burst is rarely achieved by one variable alone if the underlying drug distribution problem has not been resolved.
How to Choose Materials and Processes for Nanoparticle Formulation?
Successful nanoparticle formulation requires material selection and process selection to be developed together. A well-chosen polymer can still fail if the particle formation method produces poor morphology, unstable interfaces, or surface-enriched drug. Likewise, an elegant process cannot compensate for a polymer that fundamentally mismatches the payload. Rational development therefore connects API properties, polymer behavior, and fabrication route from the beginning rather than optimizing each in isolation.
Matching Polymer Type to API Properties
The correct polymer type depends on how the payload behaves during and after particle formation. Hydrophobic drugs often require retentive matrix-forming polymers, whereas charged macromolecules may need functional or cationic systems that support association and protection. Structurally sensitive payloads may require mild aqueous or hybrid assembly rather than solvent-heavy encapsulation. Material choice should therefore begin with solubility, polarity, charge, size, and stability of the API, not with a default preference for one nanoparticle chemistry. For long-acting design, the critical question is whether the selected polymer can do more than merely entrap the payload. It must support the intended release mechanism and remain compatible with the process conditions required to build the particle. When polymer type and API behavior are misaligned, the result is often low loading, burst release, or poor batch reproducibility regardless of how much downstream optimization is attempted.
Choosing Nanoprecipitation or Emulsification Methods
Nanoprecipitation is often useful for relatively hydrophobic polymers and payloads because it allows rapid particle formation through solvent displacement. Emulsification-based methods can be more flexible for certain payload types, especially when internal aqueous phases or staged solidification are needed. Each route influences how quickly the polymer hardens, how the drug redistributes during formation, and how porous or dense the final particle becomes. The choice between methods should therefore reflect not only convenience, but the desired internal structure and loading logic. A method that forms particles easily may still be unsuitable if it leaves too much drug near the surface or creates morphology that supports fast release. Good process selection is always linked to how the final particle is expected to retain and release the payload.
Balancing Loading, Stability, and Release Duration
Nanoparticle development usually involves trade-offs. Increasing loading may destabilize the matrix or promote burst release. Increasing colloidal stability may introduce more hydrated shells that facilitate payload escape. Increasing matrix density may slow release but reduce processability or create incomplete release later. A useful formulation strategy therefore avoids optimizing any one parameter in isolation and instead balances the three linked goals of loading efficiency, storage stability, and long-acting behavior. The best design is rarely the one with the highest nominal loading. It is the one that retains enough payload in a stable internal state to support the intended release profile under realistic storage and use conditions. For that reason, loading data should always be interpreted together with release shape, morphology, and colloidal stability rather than used as a stand-alone success metric.
How Process Conditions Change Final Particle Behavior?
Process conditions often determine final particle behavior as strongly as polymer identity. Solvent selection, solvent exchange rate, polymer concentration, phase ratio, mixing intensity, stabilizer type, and solidification history all influence whether the resulting nanoparticle becomes dense, porous, smooth, phase-separated, or surface loaded. Small changes in these variables can produce large differences in release, even when the nominal composition remains constant. For long-acting formulations, this means process development must be treated as a structural design tool rather than a manufacturing afterthought. The goal is not simply to create nanoparticles, but to create nanoparticles with the internal organization needed for controlled and reproducible release. This is especially important when the platform will later be scaled or transferred, because process sensitivity often becomes the hidden cause of batch-to-batch inconsistency.
Common Problems in Polymer Nanoparticle Development
Polymer nanoparticle systems often fail for a relatively small number of recurring reasons: poor payload compatibility, unstable colloidal behavior, unfavorable internal drug distribution, or process-driven structural variability. These problems are especially important in long-acting delivery because minor defects that might be acceptable in a short-acting nanocarrier can completely undermine sustained release. Effective development therefore depends on identifying the dominant failure mode early and linking it back to material choice, process conditions, or loading logic.
- Low loading efficiency in nanoparticle systems: Low loading usually indicates weak drug–polymer compatibility, rapid diffusion loss during fabrication, or an API that prefers the external phase rather than the particle interior. Hydrophilic or charged payloads are especially prone to this problem in hydrophobic matrices. Effective solutions usually require changing the loading mechanism, modifying the polymer environment, or redesigning the process rather than simply increasing feed concentration.
- Aggregation and poor storage stability: Aggregation often arises from insufficient surface stabilization, unfavorable charge balance, salt sensitivity, or structural changes during storage. Even if release performance looks promising initially, aggregated nanoparticles often lose reproducibility and may alter payload distribution. Storage stability should therefore be treated as part of the release design problem, not as a separate packaging issue.
- Uncontrolled burst release after formulation: Burst release usually reflects surface-associated drug, porous morphology, rapid hydration, or weak internal retention. Because nanoparticles have high interfacial area, this problem can be difficult to suppress unless payload placement inside the matrix is controlled carefully. Material changes, process redesign, and better internal densification are often more effective than simply modifying the external medium.
- Batch variability during scale-up and transfer: Small differences in mixing, solvent removal, concentration, or stabilizer distribution can change particle size, morphology, and loading state. This means a formulation that appears robust at small scale may drift substantially during transfer or scale-up. A good development program links critical process variables directly to structural and release outcomes early enough to avoid hidden instability later.
- When nanoparticle complexity outweighs platform benefits: Some projects become unnecessarily complex when nanoparticles are used to solve a problem better addressed by another platform. If the payload cannot be retained sufficiently, if process sensitivity is too high, or if the desired release duration depends on a larger depot structure, then a nanoparticle may offer sophistication without real long-acting benefit. Recognizing this early is part of good formulation judgment, not a failure of the platform.
When Polymer Nanoparticles Are the Right Platform Choice?
The value of polymer nanoparticles depends on whether their specific strengths match the real formulation problem. They are not universally superior to larger depots, hydrogels, or other controlled-release systems. Their main advantages usually involve tunable nanoscale structure, payload protection, adaptable surface design, and the ability to combine transport control with a degree of release engineering. The decision to use them should therefore come from formulation objectives rather than from platform popularity.
When Nanoparticles Outperform Larger Depot Systems?
Nanoparticles often outperform larger depot systems when small carrier size is important for dispersion, transport, local penetration, or protection of payloads that are difficult to formulate in larger matrices. They can also be advantageous when the release goal depends on controlled nanoscale encapsulation rather than on a single large depot structure. In these cases, nanoparticles may provide a more practical balance between loading, stability, and controlled release than larger particulate or implantable formats. Their advantages are strongest when the intended long-acting behavior comes from matrix retention, protective association, or persistent localized nanoparticle residence rather than from very long bulk degradation alone. If the formulation logic depends mainly on nanoscale architecture, nanoparticles can offer capabilities that larger depot systems do not.
When Microspheres or Hydrogels May Be Better?
Microspheres or polymer hydrogels may be better when the project needs a larger and more structurally persistent depot, lower interfacial area, or stronger resistance to early release. Microspheres are often more naturally suited to prolonged matrix-driven release over longer timescales, while hydrogels can be advantageous for localized, water-rich depots and sensitive biomacromolecules. In contrast, nanoparticles may introduce unnecessary complexity if the same release target can be achieved more directly with a larger system. This comparison becomes especially important when high burst release, low loading, or process variability persist despite repeated nanoparticle optimization. In such cases, the issue may not be poor execution, but a mismatch between platform scale and delivery objective.
How to Choose Based on Formulation Goals?
Platform choice should begin with the actual delivery problem. If the goal is prolonged local retention with matrix-controlled release, a larger depot may be preferable. If the goal is nanoscale payload protection with controlled distribution and moderate sustained release, polymer nanoparticles may be the better fit. If the payload is sensitive, charged, or dependent on a protective nanostructure, the nanoparticle platform may offer unique advantages that larger systems cannot easily replicate. Good platform choice therefore links the desired release profile, payload type, route constraints, process feasibility, and material compatibility in one framework. The question is not whether nanoparticles are advanced, but whether they solve the dominant formulation challenge better than the alternatives.
How to Judge Whether a Nanoparticle Is Truly Long-Acting?
A nanoparticle is truly long-acting only when its structure meaningfully prolongs payload retention or release over the intended timeframe. Long circulation alone does not make it long-acting. Good colloidal stability alone does not make it long-acting. The key test is whether the loaded payload remains associated with the carrier and is released in a controlled way that matches the target product logic. In development, this judgment should be based on loading state, early burst, medium-term retention, structural evolution, and whether the nanoparticle remains functionally useful across the intended release period. If the payload escapes rapidly despite an apparently stable carrier, then the system is a nanocarrier, but not a meaningful long-acting nanoparticle platform.
Services
Advanced Polymer Nanoparticle Design and Formulation Services
BOC Sciences provides integrated support for polymer nanoparticle development, from polymer selection and nanoparticle synthesis to loading design, release evaluation, and formulation optimization. Our technical focus is on the variables that determine whether a nanoparticle becomes a useful long-acting platform, including polymer class, particle architecture, surface functionality, payload compatibility, colloidal stability, and structural control during fabrication. By linking material design to nanoparticle behavior, we help teams develop nanocarriers that are better aligned with long-acting delivery goals rather than generic nanoscale formulation alone.

Polymer Selection & Nanocarrier Design
- Screening of biodegradable, amphiphilic, cationic, and functional polymers.
- Selection of materials based on loading logic and release targets.
- Design of nanoparticle systems for small molecules and biomacromolecules.
- Support for long-acting and controlled-release nanocarrier strategies.

Polymer Nanoparticle Synthesis
- Custom polymer nanoparticle synthesis using tailored fabrication routes.
- Optimization of nanoprecipitation, emulsification, and related processes.
- Control of particle size, morphology, and colloidal behavior.
- Development of nanoparticles for sustained and localized delivery goals.

Loading and Release Evaluation
- Drug loading and encapsulation efficiency analysis.
- Assessment of burst release, sustained release, and retention behavior.
- Characterization of structure–loading–release relationships.
- Comparison of formulation variables that affect nanoparticle performance.

Formulation Optimization Support
- Reduction of aggregation and improvement of storage stability.
- Optimization of payload retention and controlled release behavior.
- Support for process transfer and batch consistency improvement.
- Integrated development from material screening to nanoparticle design.
Need Support with Polymer Nanoparticle Development?
Discuss polymer choice, nanoparticle synthesis, loading strategy, and long-acting release design with a team focused on advanced polymer-based drug delivery systems.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What makes polymer nanoparticles useful for long-acting drug delivery?
Polymer nanoparticles are useful when they can protect payloads, control distribution, and modulate release through matrix design, surface properties, or degradable structure. Their value in long-acting delivery depends on whether the particle actually slows release or prolongs retention rather than only improving transport or uptake.
-
Why are some polymer nanoparticles not truly long-acting systems?
Some polymer nanoparticles improve solubility, circulation, or cellular uptake but release their payload too quickly to function as true long-acting depots. A nanoparticle is not automatically long-acting just because it is stable. Long-acting performance depends on loading state, matrix retention, and how release is controlled over time.
-
How does particle size affect polymer nanoparticle performance?
Particle size influences surface area, colloidal stability, distribution behavior, and release rate. Smaller particles may improve dispersion and transport but can also increase burst release because more drug is closer to the particle surface. Size distribution is equally important because broad distributions often create inconsistent loading and release behavior.
-
What causes burst release in polymer nanoparticles?
Burst release usually comes from surface-associated drug, weak drug–polymer compatibility, porous particle structure, or rapid water penetration into the matrix. It can also result from process conditions that leave a large fraction of payload near the particle exterior rather than uniformly distributed within the polymer core.
-
How should polymer nanoparticle formulations be evaluated in development?
Evaluation should include particle size, size distribution, surface charge, morphology, loading efficiency, colloidal stability, and release profile. These data should be interpreted together, because a formulation with good size control may still fail if the payload is poorly retained, releases too quickly, or becomes unstable during storage.

 Fig. 1. Polymeric nanoparticles differ from micelles and microspheres in structure, scale, and release behavior (BOC Sciences Authorized).
Fig. 1. Polymeric nanoparticles differ from micelles and microspheres in structure, scale, and release behavior (BOC Sciences Authorized). Fig. 2. Loading state, internal distribution, and matrix evolution jointly determine nanoparticle release behavior (BOC Sciences Authorized).
Fig. 2. Loading state, internal distribution, and matrix evolution jointly determine nanoparticle release behavior (BOC Sciences Authorized).