Polymer-Based Gene Delivery Platforms for DNA, RNA, and Oligonucleotide Therapeutics
With the breakthrough advances in gene therapy and mRNA vaccine technologies, the safe and efficient delivery of nucleic acid therapeutics to target cells has become a central challenge in the biopharmaceutical field. Although viral vectors are widely used in clinical applications, issues such as immunogenicity, limited cargo capacity, and high manufacturing costs have driven the scientific community to explore non-viral alternatives. Among these, polymer-based gene delivery systems have emerged as a leading platform for next-generation nucleic acid delivery, owing to their structural design flexibility, low immunogenicity, and ability to accommodate high–molecular-weight genetic payloads.
Resources
Introduction to Polymer-Based Gene Delivery Systems
The core objective of gene therapy is to overcome multiple extracellular and intracellular barriers to transport therapeutic nucleic acids intact to their sites of action. Polymer carriers are not merely passive transport vehicles; rather, through precise chemical design, they emulate the high delivery efficiency of viruses while avoiding the associated safety risks.
Fundamentals of Gene Delivery and Polymeric Vectors
The essence of gene delivery lies in overcoming the physicochemical limitations of biological macromolecules. Naked DNA or RNA carries a high density of negative charges, making it highly susceptible to nuclease degradation in vivo and difficult to penetrate the negatively charged cell membrane. The primary mechanism of polymeric vectors is to condense negatively charged nucleic acids into nanoscale complexes (polyplexes) through electrostatic interactions.
An ideal polymeric vector must possess the following characteristics:
- Protection and Condensation: Protect nucleic acids from degradation during systemic circulation.
- Cellular Uptake: Enter cells via endocytosis.
- Endosomal Escape: This is the rate-limiting step, achieved by disrupting the endosomal membrane through the “proton sponge effect” or other mechanisms.
- Nuclear Localization / Cytoplasmic Release: Deliver DNA into the nucleus or release mRNA into the cytoplasm.
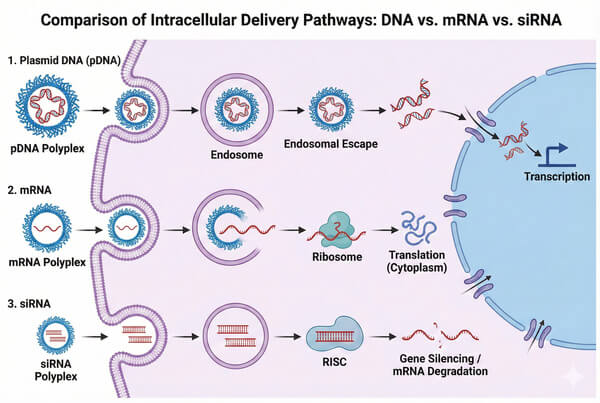 Fig. 1. Schematic of cellular uptake, endosomal escape via proton sponge effect, and nuclear entry (BOC Sciences Authorized).
Fig. 1. Schematic of cellular uptake, endosomal escape via proton sponge effect, and nuclear entry (BOC Sciences Authorized).
Advantages of Polymers over Viral Gene Delivery Systems
Compared with adeno-associated viruses (AAVs) or lentiviral vectors, polymer-based systems offer significant industrial and translational advantages:
- Low Immunogenicity: Synthetic polymers are far less likely to elicit strong host immune responses than viral proteins.
- No Cargo Size Limitation: Polymers can deliver a wide range of payloads, from short siRNA to large CRISPR-Cas9 plasmids.
- Structural Tunability: Chemists can precisely control biodistribution by adjusting molecular weight, side-chain modifications, and charge density.
- Scalable Manufacturing: Synthetic chemical processes are easier to scale up, are compatible with GMP standards, and are substantially more cost-effective than biological fermentation.
Design Requirements for Effective Polymer-Based Gene Delivery
The design of high-performance polymeric vectors is a multiparameter optimization process that must meet the following critical criteria:
- Biocompatibility: The carrier and its degradation products must be non-toxic or minimally toxic.
- Colloidal Stability: Complexes should not aggregate under physiological salt concentrations or in the presence of serum proteins.
- Targeting Capability: Ligand modification should enable preferential accumulation in specific tissues or cell types.
- Environmental Responsiveness: The carrier should respond to changes in pH, redox potential, or enzymatic conditions to enable controlled nucleic acid release.
Functional Roles of Polymers in Gene Delivery
The core value of polymer-based gene carriers lies in overcoming multiple biological barriers encountered by nucleic acid therapeutics during in vivo transport through precise molecular engineering. Functional design primarily focuses on improving colloidal stability, membrane penetration, endosomal escape efficiency, and intracellular release kinetics.
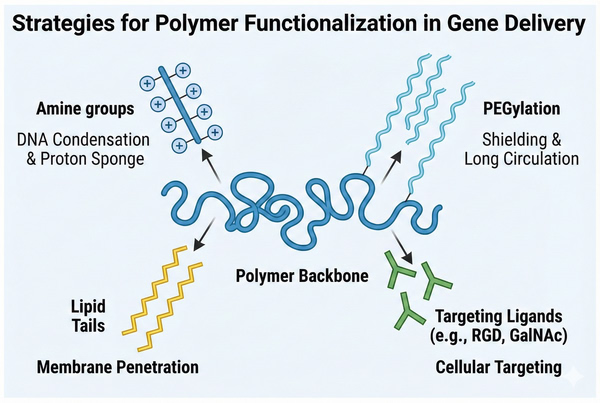 Fig. 2. Schematic illustration of common polymer chemical modification strategies (BOC Sciences Authorized).
Fig. 2. Schematic illustration of common polymer chemical modification strategies (BOC Sciences Authorized).
Modulation of Surface Charge
Surface charge is the primary physicochemical parameter in polymeric gene carrier design and is typically quantified by zeta potential. Its functional role extends far beyond simple electrostatic binding:
- Electrostatic Condensation and Protection: Cationic groups on the polymer backbone (such as primary, secondary, tertiary amines, or quaternary ammonium salts) interact strongly with the negatively charged phosphate backbone of DNA/RNA. This interaction drives polymer chains to condense extended nucleic acid molecules into polyplexes ranging from tens to hundreds of nanometers. Tight condensation not only reduces particle size for efficient endocytosis but also forms a physical barrier that protects the payload from serum nucleases.
- Interaction with the Cell Membrane: Mammalian cell membranes are rich in negatively charged proteoglycans. A moderate positive surface charge (typically between +10 mV and +30 mV) promotes nonspecific electrostatic adsorption of complexes onto the cell surface, representing the first step in adsorptive endocytosis.
- The Charge Dilemma and Dynamic Shielding: While positive charge is essential, excessively high zeta potential can cause erythrocyte aggregation, complement activation, and severe cytotoxicity. Therefore, modern polymer designs often incorporate charge-shielding strategies. For example, PEGylation or zwitterionic modification can mask positive charges during circulation, extending half-life. Alternatively, charge-reversal polymers can be designed to remain neutral or slightly negative at blood pH (7.4) and rapidly protonate under the acidic tumor microenvironment (pH 6.5–6.8), achieving “on-demand activation.”
Alteration of Topological Structures
Polymer topology—the spatial arrangement of molecular chains—directly determines nucleic acid binding modes, complex stability, and transfection efficiency.
- Linear Polymers: Examples include linear PEI or linear PLL. These polymers exhibit good chain flexibility and are easy to synthesize and characterize. However, linear structures often form relatively loose complexes with weaker resistance to dissociation. In certain mRNA delivery applications, this ease of dissociation can be advantageous for cytoplasmic release.
- Branched and Hyperbranched Structures: By introducing branching points, polymers form three-dimensional network structures. This architecture increases charge density and steric hindrance, resulting in more compact and stable nanoparticles. Branched polymers typically exhibit higher transfection efficiency than their linear counterparts due to improved resistance to serum protein displacement.
- Dendrimers: Such as PAMAM dendrimers, these represent the pinnacle of topological control, featuring monodispersity and precisely defined numbers of surface functional groups. Internal void spaces can physically encapsulate small-molecule drugs, while the multivalency effect enables the formation of highly uniform spherical complexes with nucleic acids. This dramatically improves batch-to-batch reproducibility, which is critical for GMP manufacturing.
- Star-Shaped Polymers: These structures consist of multiple polymer arms radiating from a central core. Star-shaped polymers are less prone to intermolecular aggregation at high concentrations, thereby maintaining excellent colloidal stability during formulation storage.
Introduction of Stimuli-Responsive Units
To resolve the inherent contradiction between extracellular protection and intracellular release, incorporating environmentally responsive chemical linkages is currently the most effective strategy.
- pH Responsiveness and Endosomal Escape (The Proton Sponge Effect): This is the most well-known mechanism of polymeric carriers, particularly PEI and its derivatives. Polymers containing large numbers of bufferable amine groups (pKₐ 5–7) absorb protons upon entering acidic endosomes (pH 5.0–6.0). To maintain charge balance, chloride ions influx increases osmotic pressure, ultimately causing endosomal membrane rupture and releasing nucleic acids into the cytoplasm.
- Redox Responsiveness: Intracellular glutathione (GSH) concentrations (approximately 2–10 mM), especially in tumor cells, are orders of magnitude higher than extracellular levels (2–10 μM). By introducing disulfide bonds into the polymer backbone or crosslinkers, redox-sensitive carriers can be constructed. Once inside the cytoplasm, high GSH levels reduce disulfide bonds, leading to rapid polymer degradation. This enables efficient nucleic acid release while significantly reducing the accumulation toxicity of high–molecular-weight polymers.
- Enzyme Responsiveness: By exploiting the overexpression of matrix metalloproteinases (MMPs) or lysosomal enzymes (such as cathepsin B) in tumor tissues, specific peptide linkers can be incorporated into polymer structures. When the carrier reaches target tissues or organelles, enzymatic cleavage triggers PEG shell shedding or carrier disassembly, enabling precise delivery.
Polymer Modification
Pristine synthetic polymers often suffer from limited functionality and poor biocompatibility. Through chemical modification, their physicochemical properties can be finely tuned.
- Hydrophobic Modification: Hydrophobic chains (such as lipid moieties, cholesterol, or stearic acid) are grafted onto hydrophilic cationic polymers.
- Mechanism of Action: Hydrophobic interactions stabilize the polyplex core and prevent premature dissociation in serum.
- Membrane Affinity: Hydrophobic groups can insert into the phospholipid bilayer of cell membranes, significantly enhancing membrane penetration. This is a key factor behind the success of many high-performance non-viral vectors, such as lipid–polymer hybrids.
- Fluorination: An emerging and highly effective strategy, fluorination introduces fluorine atoms (e.g., perfluoroalkyl groups) to lower surface energy and impart unique amphiphobic properties.
- Fluorine Effect: Fluorinated polymers significantly reduce serum protein adsorption (antifouling) while enhancing transmembrane transport. Studies have shown that moderate fluorination can dramatically improve endosomal escape efficiency.
- Targeting Ligand Functionalization: To achieve active targeting, ligands can be conjugated to polymer termini using techniques such as click chemistry.
- GalNAc (N-Acetylgalactosamine): Targets the ASGPR receptor on hepatocytes and represents the most successful delivery strategy for siRNA therapeutics to date.
- RGD Peptides: Target integrin receptors on tumor neovasculature endothelial cells.
- Folic Acid: Targets cancer cells that overexpress folate receptors.
Types of Polymers Used in Gene Delivery Applications
The selection of polymeric gene carriers depends on the physicochemical properties of nucleic acid payloads (such as the rigid structure of plasmid DNA versus the single-stranded and degradation-prone nature of mRNA), as well as the desired pharmacokinetic behavior. Based on chemical composition and functional characteristics, gene delivery polymers are generally classified into cationic polymers, biodegradable polymers, surface-shielded polymers, and stimuli-responsive polymers.
Cationic Polymers for DNA and RNA Delivery
- Polyethylenimine (PEI) : Polyethylenimine (PEI) is widely regarded as the “gold standard” for polymer-based transfection. Its molecular structure contains a high density of primary, secondary, and tertiary amine groups, which confer strong buffering capacity across a broad pH range. This unique chemical property enables a pronounced proton sponge effect, leading to endosomal swelling and rupture and thereby facilitating efficient nucleic acid escape. Although high–molecular-weight branched PEI (25 kDa) exhibits extremely high transfection efficiency, it is also associated with significant cytotoxicity. As a result, current research focuses more on crosslinked derivatives of low–molecular-weight PEI to balance delivery efficiency and safety.
- Poly-L-lysine (PLL): Poly-L-lysine (PLL) is one of the earliest studied polypeptide-based cationic polymers and has well-defined biodegradable products. While PLL can effectively condense DNA into stable nanoparticles, the high pKₐ of its side-chain amine groups causes it to remain fully protonated under both physiological and endosomal pH conditions. As a result, PLL lacks intrinsic buffering capacity, leading to very poor endosomal escape efficiency when used alone. Therefore, PLL is typically combined with endosomolytic agents (such as chloroquine) or fusogenic peptides, or employed as a cationic block within more complex delivery systems.
- Poly(2-dimethylaminoethyl methacrylate) (PDMAEMA): PDMAEMA is a methacrylate-based polymer synthesized via controlled radical polymerization techniques such as ATRP, offering excellent structural controllability. Its side chains contain tertiary amine groups with a pKₐ of approximately 7.5, allowing it to combine efficient nucleic acid condensation with PEI-like endosomal buffering capacity. As an intelligent polymer, PDMAEMA also exhibits temperature sensitivity and can be readily modified through copolymerization to adjust hydrophilic–hydrophobic balance, making it an ideal building block for multifunctional gene delivery systems.
- Poly(β-amino esters) (PBAEs): PBAEs are a class of biodegradable cationic polymers synthesized via Michael addition reactions between amines and diacrylates. Their backbones contain hydrolyzable ester bonds that rapidly degrade into non-toxic diacrylate and amino alcohol small molecules upon entering the aqueous cytoplasmic environment. This rapid degradation not only eliminates polymer accumulation–related toxicity in vivo but also actively promotes DNA or mRNA release through carrier disassembly.
- Polyesters (PLGA / PLA): Poly(lactic-co-glycolic acid) (PLGA) exhibits excellent biocompatibility and biodegradability and has been approved by the FDA. Because the PLGA backbone lacks cationic charges, it cannot directly condense nucleic acids via electrostatic interactions. In practice, nucleic acid aqueous solutions are typically encapsulated within polymer microspheres using the double-emulsion solvent evaporation method (W/O/W), or PLGA is blended with cationic lipids or polymers to form hybrid nanoparticles. These systems are primarily used for sustained gene release or as adjuvant delivery platforms for DNA vaccines.
- Chitosan : Chitosan is a natural cationic polysaccharide derived from chitin, featuring good biocompatibility and unique mucoadhesive properties. It can transiently open tight junctions between epithelial cells, making it particularly suitable for oral or intranasal gene delivery. However, unmodified chitosan exhibits poor solubility at neutral physiological pH, which limits its transfection efficiency. Quaternization or the introduction of hydrophilic groups (such as glycol chitosan) can significantly improve its solubility and gene delivery performance under physiological conditions.
PEGylated and Shielded Polymers for Improved Circulation
- PEGylated Polymers: To address rapid clearance by the reticuloendothelial system (RES) following systemic administration, PEGylation remains the most mature and widely adopted strategy. Grafting hydrophilic polyethylene glycol (PEG) chains onto polymer surfaces forms a dense hydration layer that produces a stealth effect, effectively preventing nonspecific adsorption of plasma opsonin proteins and significantly extending circulation half-life. To overcome the “PEG dilemma,” where PEG layers hinder cellular uptake, acid-sensitive or enzyme-sensitive linkers are often introduced to enable PEG detachment upon reaching target tissues.
- Zwitterionic Polymers: Zwitterionic polymers (such as poly(carboxybetaine) PCB and poly(phosphorylcholine) PMPC) carry both positive and negative charges within the same monomer unit and are overall electrically neutral. Compared with PEG, zwitterionic materials bind water molecules through ionic solvation, forming stronger and more stable hydration layers with exceptional antifouling properties. In addition, zwitterionic polymers exhibit extremely low immunogenicity and avoid the accelerated blood clearance (ABC) phenomenon associated with PEG, making them promising next-generation long-circulating stealth materials.
Looking for Custom Polymers for Gene Delivery?
Whether you need cationic polymers, biodegradable carriers, or stimuli-responsive platforms, our experts provide tailored synthesis and functionalization solutions to support your research and therapeutic development.
Polymer-Based Gene Delivery Technologies
Polymer-based gene delivery technologies achieve efficient encapsulation, protection, and delivery of DNA, RNA, and mRNA through precise control over polymer structure, self-assembly behavior, and surface engineering. With advances in nanotechnology and materials chemistry, a wide range of polymer delivery platforms have continuously emerged, providing scalable, customizable, and safer technological pathways for non-viral gene therapy.
Polyplex Formation and Optimization
Polyplexes are the most fundamental and widely used delivery form and are formed through the self-assembly of positively charged polymer solutions and negatively charged nucleic acid solutions. The key control parameter in this process is the N/P ratio (the molar ratio of polymer amine nitrogen to nucleic acid phosphate groups). Optimization of the N/P ratio aims to identify a balance point that ensures complete nucleic acid condensation and sufficient positive surface charge for cellular uptake, while avoiding excess free polymer that may induce cytotoxicity. In addition, mixing order, ionic strength, and stirring speed can significantly influence particle size distribution (PDI) and morphology.
Polymer Nanoparticles for Gene Delivery
Unlike polyplexes formed via electrostatic adsorption, polymer nanoparticles typically refer to solid particles in which nucleic acids are physically encapsulated within a polymer matrix. These structures are commonly prepared using nanoprecipitation or emulsion solvent evaporation methods and provide more robust physical protection against serum-mediated displacement during circulation. Polymer nanoparticles are particularly suitable for hydrophobically modified polymers or non-charged materials such as PLGA. Their release kinetics are primarily governed by polymer degradation or erosion rather than simple dissociation, making them well suited for sustained and long-acting gene release.
Polymer Micelles and Hybrid Delivery Systems
Polymeric micelles are formed by the self-assembly of amphiphilic block copolymers in aqueous environments and exhibit a core–shell structure. Nucleic acids are typically condensed within the hydrophilic core through electrostatic interactions or encapsulated within the hydrophobic core via hydrophobic modification, while the outer hydrophilic shell (commonly PEG) provides colloidal stability. Lipid–polymer hybrid nanoparticles (LPNs) combine the high membrane fusion capability of liposomes with the structural stability of polymers. For example, nucleic acids can be encapsulated within a PLGA core and coated with a lipid bilayer, forming a “sandwich” structure that significantly enhances in vivo delivery efficiency and biocompatibility.
Surface-Engineered Polymers for Targeted Gene Delivery
To enable precision medicine, surface engineering of polymer carriers is critical. By conjugating specific ligands to polymer surfaces through click chemistry or amide coupling reactions, active targeting can be achieved. For example, conjugation of N-acetylgalactosamine (GalNAc) enables specific targeting of the ASGPR receptor on hepatocytes; RGD peptide conjugation targets tumor neovasculature; and transferrin or folic acid conjugation targets cancer cells that overexpress the corresponding receptors. Surface engineering not only enhances drug accumulation at disease sites but also substantially reduces off-target toxicity to healthy tissues.
Application Scenarios of Polymer-Based Gene Delivery Systems
Polymer-based gene delivery systems have been widely applied in plasmid DNA expression, RNA interference, mRNA vaccines, oligonucleotide therapeutics, and gene editing. Their high degree of design flexibility enables polymer carriers to be optimized for specific nucleic acid types and therapeutic objectives, providing versatile and reliable solutions for basic research, clinical development, and industrial translation.
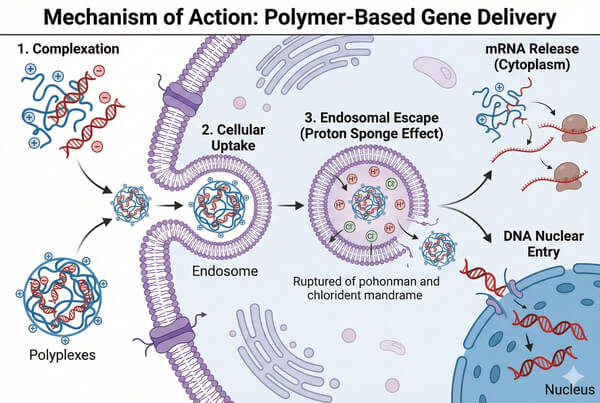 Fig. 3. Intracellular trafficking pathways comparison for plasmid DNA, mRNA, and siRNA therapeutics (BOC Sciences Authorized).
Fig. 3. Intracellular trafficking pathways comparison for plasmid DNA, mRNA, and siRNA therapeutics (BOC Sciences Authorized).
Polymer Systems for Plasmid DNA Delivery
Plasmid DNA (pDNA) delivery is a core technology for gene replacement therapy and DNA vaccine development but faces more stringent biological barriers than RNA delivery, as it must overcome both the cellular membrane and the nuclear envelope to initiate transcription. To address this challenge, high-efficiency polymer carrier systems often employ nuclear localization signal (NLS) peptide conjugation strategies to leverage active nuclear pore transport mechanisms. Alternatively, cell cycle–responsive smart polymers are designed to exploit nuclear envelope disassembly during mitosis to enable payload delivery. These strategies are critical for treating monogenic diseases or transfecting rapidly proliferating tumor cells.
Polymer-Based siRNA and miRNA Delivery
For RNA interference (RNAi) therapies targeting cancer and rare diseases, polymer delivery system design focuses on overcoming the short length, rigidity, and low charge density of siRNA and miRNA molecules. To address the instability of simple electrostatic adsorption, researchers often utilize hydrophobic interactions to construct self-assembled polymeric micelles or nanohydrogels. These systems maintain circulation stability while enabling precise accumulation in specific tissues, such as hepatocytes, through the introduction of targeting ligands like GalNAc. Because these systems function in the cytoplasm without requiring nuclear entry, they serve as highly effective tools for treating hypercholesterolemia and regulating oncogene expression.
Polymers for mRNA Vaccine and Therapeutic Delivery
With the rapid advancement of mRNA therapeutics in infectious disease vaccines, cationic polymers have emerged as strong competitors to lipid nanoparticles, aiming to overcome cold-chain storage limitations and patent barriers associated with lipid-based systems. Given that mRNA does not require nuclear entry but is highly susceptible to enzymatic degradation, next-generation polymers—such as charge-altering releasable transporters (CARTs)—are designed to provide robust physical protection during circulation while undergoing chemical degradation or charge neutralization upon cellular entry to release mRNA. This low-immunogenicity delivery strategy demonstrates unique clinical advantages in cancer immunotherapy vaccines and protein replacement therapies requiring repeated dosing.
Polymer Systems for Antisense Oligonucleotide (ASO) and Immunostimulant Delivery
Antisense oligonucleotides (ASOs), as single-stranded nucleic acid therapeutics, possess enhanced nuclease resistance through backbone chemical modifications (such as phosphorothioate or 2′-O-methylation). However, their clinical efficacy remains severely limited by poor membrane permeability and endosomal entrapment. Polymer carriers play a critical role as “intracellular enhancers” in ASO delivery by forming highly cell-affinitive nanocomplexes that significantly improve cellular uptake and endosomal escape. This enables ASOs to efficiently reach the nucleus and mediate RNase H–dependent target mRNA degradation or splicing modulation, which is of particular importance in treating neurodegenerative diseases such as spinal muscular atrophy (SMA). In addition, in immunotherapy applications, cationic polymers are widely used for co-delivery of CpG oligonucleotides (TLR9 agonists) and tumor antigens. Through self-assembly into pathogen-mimicking nanoparticles, these systems not only protect CpG from degradation but also promote lymph node drainage and uptake by antigen-presenting cells (APCs), thereby inducing robust antigen-specific immune responses.
Polymer Vectors for CRISPR and Genome Editing
In the delivery of CRISPR/Cas9 genome editing systems, direct delivery of ribonucleoprotein (RNP) complexes offers significant advantages in reducing off-target effects due to its “hit-and-run” mechanism. However, the large molecular weight of Cas9 protein and its complex surface charge distribution present substantial challenges for carrier design. To address these issues, polymer nanocapsules or in situ polymerization techniques have been developed to physically encapsulate Cas9 RNPs, forming stable nanocage structures. By incorporating redox-sensitive linkages, these smart carriers rapidly disassemble in response to high intracellular glutathione levels after cytoplasmic entry, releasing the editing machinery to perform precise genome surgery while maximizing biosafety and editing efficiency.
Formulation and Design Strategies for Polymer Gene Delivery Systems
Possessing a high-performance polymer material is only the starting point of drug development. Translating materials into clinically viable therapeutics requires a rigorous formulation development process. The core objective at this stage is to define critical quality attributes (CQAs) and identify an optimal balance among transfection efficiency, cytotoxicity, colloidal stability, and in vivo pharmacokinetics.
Polymer Molecular Weight and Charge Density Optimization
Polymer molecular weight (MW) and charge density represent a double-edged sword affecting both transfection efficiency and biosafety. In general, high–molecular-weight polymers provide stronger DNA binding and improved endosomal escape but are also associated with increased cytotoxicity and non-degradability risks. The central formulation strategy lies in identifying an “efficiency–toxicity” balance window. For linear polymers, molecular weights in the range of 5 kDa to 25 kDa are typically screened to ensure stable complex formation while allowing effective renal clearance or intracellular degradation. In addition, the spatial distribution of charge density is critical. Compared with densely packed positive charges on side chains, dispersing charges along the polymer backbone through copolymerization or diluting surface charge density by introducing hydrophilic blocks (such as PEG) can significantly reduce physical membrane damage and hemolytic effects, while maintaining sufficient proton-buffering capacity to support transfection.
Polymer–Nucleic Acid Compatibility Assessment
Different nucleic acid therapeutics possess distinct physicochemical properties, and formulation design must achieve structural matching between carrier and payload. Plasmid DNA (pDNA) is a rigid, double-stranded supercoiled structure that requires strong cationic polymers to induce conformational transitions from a loose coil to a compact globular state. In contrast, mRNA is a flexible, single-stranded molecule that is highly susceptible to nuclease degradation and must be released into the cytoplasm rather than transported into the nucleus. Therefore, the first step in formulation optimization is to determine the optimal N/P ratio. This parameter not only defines the mixing ratio but also governs surface charge, particle size (typically targeted at 50–150 nm), and encapsulation efficiency. Using gel retardation assays and ethidium bromide displacement experiments, scientists must precisely quantify polymer–nucleic acid binding affinity to ensure sufficient stability during circulation while enabling responsive disassembly upon cellular entry.
Stability and Storage of Polymer Gene Delivery Formulations
Liquid nanocomplex formulations often face risks of Ostwald ripening or irreversible aggregation during long-term storage, leading to particle size growth, precipitation, and loss of transfection activity. To meet commercial distribution requirements, the development of robust lyophilization processes represents the industry-standard solution. Incorporation of appropriate cryoprotectants, such as trehalose, sucrose, or mannitol, enables the formation of a glassy matrix during freezing, effectively isolating nanoparticles and preventing mechanical damage caused by ice crystal growth.
In Vitro and In Vivo Performance Evaluation
The transition from in vitro cell screening to in vivo animal studies represents the decisive stage of formulation validation. In vitro evaluation should not be limited to easily transfected model cell lines (such as HEK293), but should also include hard-to-transfect primary cells or stem cells relevant to the therapeutic indication. Key metrics include the ratio of transfection efficiency (measured using luciferase or GFP reporter genes) to cellular metabolic activity (assessed by MTT or CCK-8 assays). Upon entering the in vivo stage, biodistribution and pharmacokinetics (PK) become central evaluation parameters. Using in vivo imaging techniques, the accumulation of fluorescently labeled polymer complexes in major organs (liver, spleen, lung, kidney, and tumors) must be tracked to verify targeting capability and long-circulation behavior. In parallel, rigorous immunogenicity assessments are required, including measurement of serum inflammatory cytokines (such as IL-6 and TNF-α) and liver enzyme levels, to ensure that polymer carriers do not induce acute immune responses or organ toxicity, thereby establishing a comprehensive safety and efficacy profile.
Services
Custom Polymer Services for Gene Delivery at BOC Sciences
Leveraging extensive expertise in polymer chemistry and biomedical materials, BOC Sciences provides one-stop polymer-based gene delivery solutions for DNA, RNA, and mRNA therapeutics—from material design to delivery system construction. Our services span polymer molecular design, functional modification, delivery system development, and comprehensive characterization, supporting both translational research and industrial applications.

Polymer Material Design, Screening, and Synthesis
- Polymer structure design based on the distinct properties of DNA, siRNA, and mRNA
- Customizable linear, branched, hyperbranched, and block copolymer architectures
- Precise control over polymer molecular weight, distribution, and charge density
- Screening and optimization of cationic, zwitterionic, or neutral functional polymers

Polymer Modification and Functionalization for Nucleic Acid Delivery
- PEGylation to improve blood circulation time and reduce immune clearance
- Incorporation of targeting ligands (e.g., saccharides, small molecules, or peptides) for cell-specific delivery
- Construction of pH-, redox-, or enzyme-responsive functional units for controlled intracellular release
- Regulation of surface hydrophobic/hydrophilic balance to optimize self-assembly and nanostructure stability

Development of Polymer–Nucleic Acid Delivery Systems
- Construction and optimization of polymer–DNA/RNA/mRNA complexes (polyplexes)
- Development of polymer nanoparticles, micelles, and hybrid delivery systems
- Optimization of polymer/nucleic acid ratios, assembly conditions, and size distribution
- Systematic design tailored to in vitro transfection or in vivo delivery requirements

Morphological Characterization, Loading Capacity, and Release Performance Evaluation
- Particle size, distribution, and morphology analysis (DLS, TEM/SEM, etc.)
- Surface charge (zeta potential) and stability assessment
- Evaluation of nucleic acid loading efficiency, binding affinity, and protective capability
- In vitro release profiles and stimuli-responsive release performance studies
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
What are polymer-based gene delivery systems?
Polymer-based gene delivery systems are non-viral platforms that use synthetic or natural polymers as carriers. Through electrostatic interactions, positively charged polymers condense negatively charged nucleic acids (such as DNA, mRNA, and siRNA) into nanoscale complexes known as polyplexes. These systems offer low immunogenicity, high structural tunability, and ease of large-scale production, making them key tools for overcoming in vivo delivery barriers of nucleic acid therapeutics.
How does polymer-based gene delivery work?
The core mechanism involves four steps: “condensation–protection–entry–release.” First, polymers condense nucleic acids into stable nanoparticles to protect them from enzymatic degradation. Next, the complexes enter cells via adsorption-mediated or receptor-mediated endocytosis. Subsequently, endosomal escape is achieved through the proton sponge effect or related mechanisms. Finally, the carrier disassembles in response to intracellular conditions, releasing the active payload in the cytoplasm (for RNA) or nucleus (for DNA).
How do polymers deliver DNA and RNA into cells?
Polymers primarily cross cellular barriers by mimicking the physicochemical features of viral surfaces. Cationic polymers electrostatically interact with negatively charged glycoproteins on the cell membrane, inducing clathrin-mediated or caveolae-mediated endocytosis. In addition, by conjugating specific ligands (such as GalNAc, RGD peptides, or folic acid), polymer carriers can recognize target cell receptors and achieve efficient active targeting delivery.
What are the advantages of non-viral gene delivery polymers?
Compared with viral vectors, polymer carriers offer superior safety, with minimal risk of severe immune responses or oncogenicity. They are largely unrestricted by cargo size, enabling delivery of large plasmids or CRISPR complexes. Moreover, their chemical structures are highly tunable, allowing versatile functionalization (e.g., PEGylation for stealth properties or stimuli responsiveness). Mature chemical synthesis processes also make them cost-effective and well suited for GMP-scale manufacturing.
How are polymers selected for gene delivery applications?
Selection strategies depend on the nucleic acid type and therapeutic goal. For example, plasmid DNA delivery often requires polymers that facilitate nuclear localization, whereas mRNA delivery emphasizes cytoplasmic release and protection. The administration route is also critical: intravenous delivery typically favors PEGylated or zwitterionic polymers to prolong circulation, while local administration prioritizes tissue retention. Balancing biodegradability (e.g., PBAEs) with transfection efficiency is another key consideration during formulation development.

 Fig. 1. Schematic of cellular uptake, endosomal escape via proton sponge effect, and nuclear entry (BOC Sciences Authorized).
Fig. 1. Schematic of cellular uptake, endosomal escape via proton sponge effect, and nuclear entry (BOC Sciences Authorized). Fig. 2. Schematic illustration of common polymer chemical modification strategies (BOC Sciences Authorized).
Fig. 2. Schematic illustration of common polymer chemical modification strategies (BOC Sciences Authorized). Fig. 3. Intracellular trafficking pathways comparison for plasmid DNA, mRNA, and siRNA therapeutics (BOC Sciences Authorized).
Fig. 3. Intracellular trafficking pathways comparison for plasmid DNA, mRNA, and siRNA therapeutics (BOC Sciences Authorized).