Polymer Materials for Drug Delivery: Controlled Release, Targeting, and Formulation Solutions
Polymer materials play a critical role in modern drug delivery systems, enabling precise control over drug release, enhanced targeting, and advanced formulation strategies. Biodegradable, stimuli-responsive, and functionalized polymers allow for tailored therapeutic profiles, improving drug stability, solubility, and bioavailability. These materials support a wide range of delivery platforms, including nanoparticles, micelles, hydrogels, and polymer–drug conjugates, and are applicable to small molecules, proteins, peptides, and nucleic acid therapeutics. By combining material design with controlled release mechanisms, polymer-based systems offer versatile solutions to address the challenges of effective and targeted drug delivery.
Resources
Introduction to Polymers for Drug Delivery
The landscape of pharmaceutical formulation has fundamentally shifted towards advanced macromolecular architectures to overcome the pharmacokinetic limitations of complex active pharmaceutical ingredients (APIs). By leveraging engineered synthetic polymers, formulators can design sophisticated delivery vehicles that precisely dictate payload stability, target tissue accumulation, and release kinetics. These customized structural frameworks bridge the gap between early-stage molecular discovery and highly effective, translatable therapeutic formulations.
Definition and Scope of Polymeric Drug Delivery Systems
Polymeric drug delivery systems represent a diverse class of engineered platforms, ranging from nanoscale carriers such as polymeric micelles and nanoparticles to macroscopic hydrogels and implantable depots, all designed to encapsulate, protect, and transport therapeutic agents through complex physiological environments. By tailoring polymer composition, molecular weight, architecture, and surface functionality, these systems can enhance solubility of poorly water-soluble drugs, protect sensitive biomolecules from enzymatic or chemical degradation, and enable controlled or sustained release profiles. Functionalization with targeting ligands or stimuli-responsive groups further allows site-specific delivery and adaptive release in response to pH, temperature, or enzymatic cues. Applicable to small molecules, peptides, proteins, and nucleic acids, polymeric carriers facilitate multiple administration routes—oral, parenteral, transdermal, or mucosal—while overcoming biological barriers such as rapid clearance, membrane transport limitations, and gastrointestinal degradation, making them versatile tools for precise and efficient therapeutic delivery.
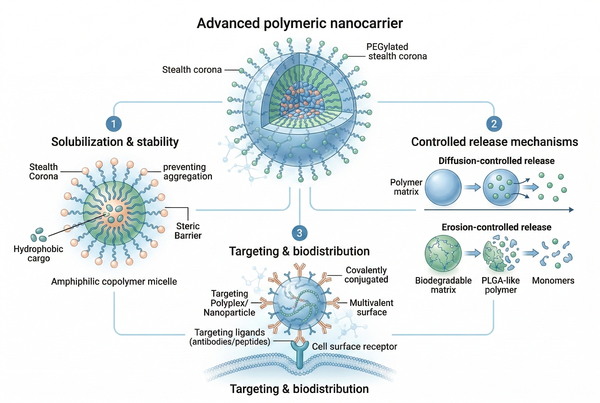 Fig. 1. Advanced polymeric nanocarrier mechanisms for targeted drug delivery systems (BOC Sciences Authorized).
Fig. 1. Advanced polymeric nanocarrier mechanisms for targeted drug delivery systems (BOC Sciences Authorized).
Evolution of Polymers in Pharmaceutical Drug Delivery
The role of polymer chemistry in pharmaceutical sciences has progressed from the use of simple, inert excipients to sophisticated, multifunctional macromolecular carriers that actively influence drug pharmacokinetics and biodistribution. Early controlled-release systems predominantly relied on bulk-degrading macroscopic polyesters to provide basic sustained diffusion, offering limited tunability and negligible interaction with the biological milieu. Advances in polymerization techniques, particularly precision living polymerizations such as Atom Transfer Radical Polymerization (ATRP) and Reversible Addition-Fragmentation Chain Transfer (RAFT) Polymerization, now enable the synthesis of sequence-defined copolymers with exact molecular weights, ultra-low dispersity, and programmable architectures. These innovations facilitate the design of stimuli-responsive carriers that can undergo conformational changes, trigger site-specific drug release, or modulate cellular uptake in response to pH, redox conditions, temperature, or enzymatic activity. Modern polymeric systems can also incorporate targeting ligands, bioactive side chains, or imaging moieties, transforming them into intelligent, multifunctional delivery platforms capable of addressing complex therapeutic challenges, including the delivery of nucleic acids, proteins, and poorly soluble small molecules. The evolution of polymer design thus represents a paradigm shift from passive excipients to active participants in precision medicine.
Key Performance Requirements for Drug Delivery Polymers
Selecting the optimal polymeric backbone requires a rigorous, multidimensional evaluation of both physicochemical properties and biological compatibility. To ensure formulation viability, the polymer must meet several critical performance criteria:
- Biocompatibility and Clearance: The polymer and its degradation byproducts must be strictly non-immunogenic and safely eliminated from the body.
- High Loading Capacity: The polymer should efficiently accommodate a significant fraction of the active pharmaceutical ingredient (API) to minimize excipient burden.
- Mechanical and Chemical Integrity: The polymer must maintain stability during processing and under physiological conditions.
- Controlled Release Capability: The polymer should enable predictable and tunable release kinetics, including sustained, extended, or targeted drug release profiles.
- Solubility and Processability: The polymer must exhibit appropriate solubility, viscosity, and thermal behavior to facilitate formulation methods such as nanoparticle formation, micelle assembly, or hydrogel fabrication.
- Degradability and Metabolic Fate: For biodegradable systems, the rate of polymer degradation must match therapeutic requirements, producing non-toxic metabolites compatible with biological pathways.
- Stability and Shelf-Life: The polymer must resist premature hydrolysis, oxidation, or aggregation to ensure long-term stability of the drug formulation.
- Targeting and Functionalization Potential: The polymer should allow chemical modification or conjugation with ligands, antibodies, or targeting moieties to achieve site-specific delivery.
- Compatibility with Other Formulation Components: The polymer must be chemically and physically compatible with excipients, solvents, and active ingredients without inducing precipitation or adverse reactions.
Functional Roles of Polymers in Drug Delivery Systems
Within advanced formulation architectures, polymers serve highly specialized bio-physicochemical functions that directly dictate therapeutic success. They are engineered to interface dynamically with both the encapsulated active pharmaceutical ingredient (API) and the complex physiological milieu. By precisely tailoring monomeric sequences, molecular weights, and side-chain functionalities, these macromolecules actively resolve critical biopharmaceutical bottlenecks and transform unstable compounds into viable delivery platforms.
- Polymeric Solubilization and API Stability Enhancement: Hydrophobic compounds frequently suffer from poor aqueous solubility, limiting systemic bioavailability. Amphiphilic block copolymers self-assemble above their critical micelle concentration (CMC) to form nanoscopic core-shell structures. The lipophilic core sequesters the poorly soluble agent, while the hydrophilic corona provides steric stabilization, dramatically increasing solubility and physical stability without harsh co-solvents.
- Mechanisms of Controlled and Sustained Drug Release: Polymer matrices modulate release kinetics to reduce toxic burst effects and maintain optimal drug levels over time. Controlled release is achieved through diffusion-controlled networks or progressive erosion of biodegradable polymers such as poly(lactic-co-glycolic acid) (PLGA). Adjusting lactide-to-glycolide ratios, crystallinity, and molecular weight allows precise tuning of hydrolytic degradation and release profiles.
- Targeted Delivery and Systemic Biodistribution Control: Polymers modify systemic biodistribution by shielding the API from immune recognition. Surface PEGylation creates a hydration shell that reduces opsonization and clearance, extending circulation half-life. Functional end-groups enable covalent attachment of targeting ligands for receptor-mediated uptake at specific cellular sites.
- Protecting Sensitive Therapeutic Payloads from Degradation: Fragile peptides and nucleic acids are prone to enzymatic degradation and aggregation. Polymers provide complexation or encapsulation to shield these molecules. Cationic polymers, for instance, form electrostatic polyplexes with negatively charged genetic materials, condensing them into stable nanoparticles that protect the payload during extracellular transit and enhance intracellular delivery.
Types of Polymer Materials for Drug Delivery
The diverse landscape of polymer chemistry offers a vast array of materials, each presenting unique structural, degradation, and functional advantages for formulation development. By categorizing these macromolecular architectures according to origin, biodegradability, physicochemical properties, and environmental responsiveness, formulators can rationally select optimal polymer backbones tailored to specific therapeutic payloads and pharmacokinetic objectives. This classification framework is critical for aligning material properties—such as hydrophobicity, charge density, and degradation rate—with the requirements of small molecules, biologics, and nucleic acid-based therapeutics.
Biodegradable polymers are central to controlled drug delivery systems that require transient structural support and predictable degradation without the need for surgical removal. Aliphatic polyesters such as polylactic acid (PLA) and poly(caprolactone) (PCL) degrade primarily through hydrolytic cleavage of ester bonds, generating biocompatible metabolites that enter natural metabolic pathways. By modulating copolymer composition, crystallinity, and molecular weight, researchers can finely tune degradation kinetics and drug release profiles, enabling sustained delivery ranging from days to months. These materials are widely employed in microspheres, implants, and injectable depots for long-acting therapeutics.
Natural and Semi-Synthetic Polymers for Biomedical Use
Natural polymers provide intrinsic biological compatibility, biodegradability, and biofunctionality that are difficult to replicate with fully synthetic systems. Polysaccharides such as Dextran, alginate, and chitosan exhibit unique physicochemical properties, including hydrophilicity, gel-forming capability, and bioadhesion. Chitosan, in particular, carries a positive charge under physiological conditions, enabling strong electrostatic interactions with negatively charged biological membranes and nucleic acids, thereby enhancing mucoadhesion and facilitating paracellular transport. Semi-synthetic derivatives further expand functionality by introducing tailored substituents, improving solubility, stability, and drug loading efficiency for advanced biomedical applications.
Synthetic Polymers in Advanced Drug Delivery Systems
Synthetic polymers offer unparalleled control over molecular architecture, enabling the rational design of drug delivery systems with precise physicochemical properties. Through advanced polymerization techniques, parameters such as chain length, block composition, branching, and functional group density can be systematically engineered to optimize drug encapsulation, circulation time, and release kinetics. Amphiphilic block copolymers, for example, self-assemble into micellar or vesicular nanostructures that enhance the solubility of hydrophobic drugs, while cationic polymers facilitate complexation with nucleic acids for gene delivery. This level of structural precision allows for the fine-tuning of interactions between the polymer carrier, the drug payload, and the biological environment.
Stimuli-responsive polymers, often referred to as "smart" polymers, are designed to undergo physicochemical transformations in response to specific internal or external triggers, enabling spatially and temporally controlled drug release. pH-responsive systems exploit ionizable groups that alter polymer solubility or swelling behavior in acidic microenvironments, such as tumor tissues or endosomes. Redox-responsive polymers incorporate disulfide linkages that are selectively cleaved in the reductive intracellular environment, while temperature- and enzyme-responsive systems provide additional layers of control. These dynamic responses allow for precise activation of drug release only at the target site, minimizing systemic exposure and improving therapeutic efficacy.
Functionalized Polymers for Specialized Applications
Functionalized polymers are engineered with reactive groups or bioactive moieties that enable advanced functionalities such as targeted delivery, multivalent binding, and high-density drug conjugation. Polyamino acids, including polylysine and Poly-γ-glutamic acid, are widely used for constructing electrostatic complexes with nucleic acids, forming stable polyplexes for gene delivery. Meanwhile, highly branched architectures such as dendrimers offer monodisperse structures with a high density of surface functional groups, enabling simultaneous drug loading, targeting ligand attachment, and imaging integration. These multifunctional systems are particularly valuable in precision medicine, where combined therapeutic and diagnostic (theranostic) capabilities are increasingly required.
Looking for Custom Polymers for Drug Delivery?
Whether you need cationic polymers, biodegradable carriers, or stimuli-responsive platforms, our experts provide tailored synthesis and functionalization solutions to support your research and therapeutic development.
Formulation Development & Polymeric Drug Delivery Technologies
Translating a raw polymer and active pharmaceutical ingredient (API) into a highly functional delivery system requires rigorous formulation engineering and a deep understanding of polymer physicochemistry. Key parameters—including polymer molecular weight, glass transition temperature (Tg), hydrophilic–lipophilic balance, and chain architecture—directly influence processing methods, carrier morphology, and drug–polymer interactions. The selection of fabrication techniques such as solvent evaporation, nanoprecipitation, or crosslinking determines encapsulation efficiency, particle uniformity, and scalability. Moreover, thermodynamic stability and kinetic trapping of drug molecules within polymer matrices must be carefully balanced to prevent premature leakage while enabling controlled release. Robust formulation strategies are therefore essential to preserve payload integrity, especially for labile biomolecules, and to achieve reproducible, tunable pharmacokinetic profiles.
Polymer–drug conjugates represent a highly controlled delivery strategy in which APIs are covalently linked to polymer backbones through cleavable spacers, forming macromolecular prodrugs with defined stoichiometry. The choice of linker chemistry—such as ester, hydrazone, disulfide, or enzyme-sensitive peptide bonds—governs the release mechanism and site specificity, enabling triggered drug liberation in response to pH gradients, redox conditions, or enzymatic activity. This conjugation approach enhances aqueous solubility, prolongs systemic circulation, and reduces off-target toxicity by limiting passive diffusion. Additionally, polymer conjugation can alter cellular uptake pathways, favoring endocytosis and intracellular trafficking, which is particularly advantageous for cytotoxic agents and nucleic acid therapeutics.
Polymeric hydrogels are three-dimensional crosslinked networks capable of absorbing large volumes of water while maintaining structural integrity, making them ideal for localized and sustained drug delivery. These systems can be engineered for in situ gelation through physical (e.g., temperature or ionic interactions) or chemical (e.g., covalent crosslinking) mechanisms, allowing minimally invasive administration. The mesh size of the hydrogel network, governed by crosslink density, directly controls diffusion rates of encapsulated therapeutics. Furthermore, stimuli-responsive hydrogels can dynamically alter swelling behavior in response to environmental cues, enabling on-demand release. Their high biocompatibility and tunable mechanical properties make them particularly suitable for tissue engineering, regenerative medicine, and localized depot formulations.
Polymeric nanoparticles, including nanospheres and nanocapsules, provide versatile platforms for systemic and intracellular drug delivery. Fabrication methods such as nanoprecipitation, emulsification–solvent evaporation, and microfluidic-assisted synthesis allow precise control over particle size, surface charge, and morphology. These parameters critically influence biodistribution, cellular uptake pathways, and endosomal escape efficiency. Surface modification strategies, including PEGylation or ligand conjugation, further enhance circulation time and targeting specificity. For nucleic acid delivery, cationic polymers facilitate electrostatic complexation, forming stable nanostructures that protect genetic payloads from nuclease degradation and promote efficient transfection. In particular, polymer nanoparticles for gene delivery provide enhanced structural stability, tunable particle size, and optimized surface charge, enabling improved cellular uptake, prolonged circulation, and more efficient intracellular trafficking compared to conventional polyplex systems.
The delivery of structurally complex and highly hydrophilic glycans and polysaccharides presents unique challenges due to rapid enzymatic degradation and poor membrane permeability. Polymeric carriers designed for these biomolecules often rely on electrostatic interactions, hydrogen bonding, or encapsulation within hydrophilic matrices to enhance stability and prolong circulation time. Tailored polymer architectures can reduce renal clearance and protect against enzymatic hydrolysis, while facilitating receptor-mediated uptake or endocytic internalization. These strategies are particularly important for applications in immunomodulation, vaccine adjuvants, and glycan-based therapeutics.
Polymeric micelles are self-assembled nanostructures formed from amphiphilic block copolymers, characterized by a hydrophobic core and hydrophilic shell. Their exceptionally low critical micelle concentration (CMC) ensures structural stability under physiological dilution, preventing premature disassembly. The hydrophobic core efficiently encapsulates poorly water-soluble drugs, while the hydrophilic corona provides steric stabilization and reduces protein adsorption. Advanced micelle systems can be functionalized with targeting ligands or designed to respond to environmental triggers, enabling site-specific drug accumulation and controlled release. Their dynamic assembly behavior also allows for reversible drug loading and release, making them highly adaptable carriers for precision therapeutics.
Polymer microspheres, typically ranging from 1 to 1000 µm in diameter, function as long-acting depot systems capable of delivering drugs over extended periods. Fabrication techniques such as double emulsion (W/O/W), spray drying, and microfluidics enable precise control over particle size distribution and internal morphology. Drug release from microspheres is governed by a combination of diffusion, polymer degradation, and matrix erosion, allowing for finely tuned release kinetics. By adjusting polymer composition, porosity, and surface characteristics, these systems can minimize initial burst release and achieve sustained therapeutic levels, making them particularly valuable for chronic disease management and long-term injectable formulations.
Drug Encapsulation and Release Mechanisms in Polymeric Systems
The core utility of any polymeric carrier lies in its capacity to securely entrap a payload and selectively release it in a controlled, predictable manner. Understanding the thermodynamic principles of initial encapsulation and the complex transport phenomena governing subsequent release is absolutely critical for formulation success. Formulators meticulously manipulate polymer chemistry to dictate these mechanistic pathways, balancing structural stability during systemic transit with highly efficient offloading at the desired physiological locus.
- Passive and Active Targeting Mechanisms: Passive targeting predominantly exploits specific pathophysiological phenomena, such as the Enhanced Permeability and Retention (EPR) effect, where precise size profiles (typically 20-200 nm) allow engineered nanoparticles to extravasate through leaky neovasculature. Active targeting refines this by conjugating high-affinity targeting ligands to the polymer surface, promoting precise, receptor-mediated internalization into specific cell populations.
- Sustained and Extended Release Strategies: Achieving a predictable zero-order release profile minimizes peak-to-trough plasma fluctuations. This advanced kinetic profile is engineered utilizing surface-eroding polymers or highly crosslinked structural diffusion barriers that mathematically restrict mass transport mechanisms out of the carrier, effectively extending the therapeutic window.
- Influence of Polymer Molecular Weight and Matrix Crosslinking: Higher molecular weights significantly increase polymer chain entanglement and internal matrix viscosity, which severely retards the aqueous diffusion of the encapsulated API. Similarly, increasing the crosslinking density within polymeric hydrogels or solid microspheres drastically decreases the internal mesh size, tightly restricting the egress of large biomolecular payloads.
- Surface Modification for Release Control: The interfacial properties of the nanocarrier dictate its biological fate. Surface modification via extensive PEGylation forms a dense hydrophilic corona that not only imparts essential "stealth" properties to evade macrophage clearance but also creates a robust steric barrier. This intentionally slows the diffusion of water into the lipophilic core, actively modulating hydrolytic degradation rates.
- Nanocarrier Optimization: While cationic surface charges strongly enhance cellular internalization via electrostatic interactions, excess charge density can induce severe cellular toxicity. Formulators must meticulously optimize the zeta potential and the hydrophile-lipophile balance (HLB) of the constituent monomers to maximize structural stability and promote highly efficient interaction with target lipid membranes.
- Improving Drug Solubility and Bioavailability: By actively shifting the physical state of the API from a highly crystalline lattice to an amorphous state within a solid polymer dispersion, the apparent solubility and dissolution rate are significantly enhanced. The surrounding macromolecular matrix physically impedes molecular recrystallization, thermodynamically stabilizing the high-energy amorphous state.
- Controlling Drug Loading Efficiency and Release Rates: Maximizing initial encapsulation without inducing polymer precipitation is a critical engineering challenge. Disproportionately high drug-to-polymer ratios can lead to rapid, uncontrollable burst release. Formulators optimize the Flory-Huggins interaction parameters to ensure high thermodynamic compatibility between the API and the polymer core.
- Ensuring Polymer–Drug Compatibility: Successful, long-term formulation stability relies heavily on strong intermolecular interactions—such as hydrogen bonding, van der Waals forces, and hydrophobic interactions—between the target drug and the surrounding polymer matrix. Mismatched chemical polarities inevitably result in low initial loading efficiencies and rapid physical phase separation during storage.
Selecting the optimal nanocarrier platform is a critical inflection point in advanced formulation development. While lipid-based systems (such as LNPs) excel in specific nucleic acid applications and rigid inorganic carriers offer unique optical or magnetic properties for theranostics, polymer-based systems provide unmatched structural versatility and precisely tunable degradation kinetics. Understanding the distinct physicochemical trade-offs among these delivery platforms is essential for aligning the carrier matrix with the active pharmaceutical ingredient (API) to achieve the targeted pharmacokinetic profile.
| Carrier Platform | Structural Versatility | Release Kinetics | Physical Stability | Primary Application Strength |
|---|
| Polymeric Systems | Exceptionally high (customizable molecular weights, block ratios, and crosslinking) | Highly tunable; excellent for prolonged, zero-order sustained release | High; frequently compatible with lyophilization and ambient storage | Sustained depot delivery, complex API stabilization, and targeted targeted mucosal delivery |
| Lipid Systems (e.g., LNPs, Liposomes) | Moderate (reliant on natural or synthetic phospholipid combinations) | Often rapid or diffusion-controlled; prone to initial burst release | Lower; thermodynamically unstable, often requiring strict cold-chain logistics | Systemic nucleic acid (mRNA/siRNA) delivery and rapid intracellular transfection |
| Inorganic Systems (e.g., Mesoporous Silica) | Low (rigid crystalline or amorphous lattice structures) | Stimuli-responsive; relies on external triggers or porous diffusion | Highest; completely inert and highly resistant to chemical or thermal stress | Theranostics, photothermal therapy, and simultaneous diagnostic imaging |
When formulating highly hydrophobic APIs, differentiating between advanced polymeric micelles and conventional surfactant-based micelles is critical for ensuring systemic formulation integrity. Polymeric micelles, synthesized from well-defined amphiphilic block copolymers, exhibit an exceedingly low critical micelle concentration (CMC), which grants them exceptional thermodynamic and kinetic stability against rapid volumetric dilution upon intravenous administration. Conversely, conventional micelles assembled from low-molecular-weight surfactants exist in a highly dynamic equilibrium and are highly susceptible to premature dissociation in the bloodstream. Consequently, macromolecular polymeric architectures represent the superior, robust choice for facilitating targeted, long-circulating, and highly controlled hydrophobic drug delivery.
Applications of Polymer-Based Drug Delivery Systems
The intrinsic physicochemical versatility of polymeric materials allows them to be systematically engineered for a broad spectrum of therapeutic modalities. By precisely tuning parameters such as charge density, hydrophobic–hydrophilic balance, molecular weight, and degradation kinetics, polymer systems can be adapted to overcome key biological barriers including opsonization, enzymatic degradation, rapid renal clearance, and limited cellular internalization. Importantly, the structure–function relationship of polymers dictates not only drug loading efficiency but also intracellular trafficking pathways, endosomal escape efficiency, and ultimately therapeutic efficacy. As a result, polymer-based delivery platforms have become indispensable tools for enabling controlled release, tissue targeting, and intracellular delivery across small molecules, biologics, and advanced genetic therapeutics.
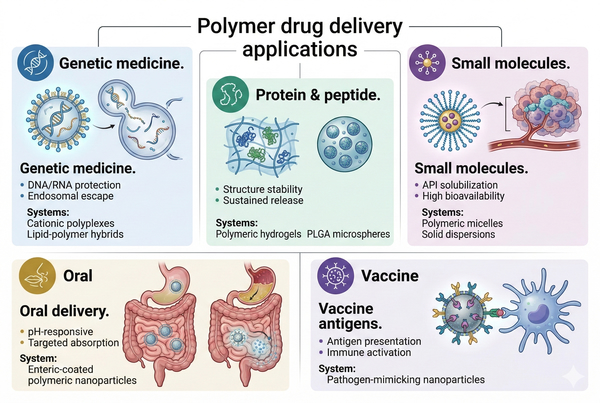 Fig. 2. Five key therapeutic applications of polymeric drug delivery systems (BOC Sciences Authorized).
Fig. 2. Five key therapeutic applications of polymeric drug delivery systems (BOC Sciences Authorized).
Cationic polymers are widely utilized for non-viral gene delivery due to their ability to electrostatically condense nucleic acids into nanoscale polyplexes with controlled size and surface charge. These complexes facilitate cellular uptake primarily via clathrin- or caveolae-mediated endocytosis. A major rate-limiting step in gene delivery is endosomal escape, which is effectively addressed through the "proton sponge" effect exhibited by polymers containing tertiary amines. This buffering capacity induces osmotic swelling and membrane destabilization, enabling cytosolic release of genetic cargo. Advanced systems further incorporate biodegradable backbones, PEGylation, and targeting ligands to balance transfection efficiency, circulation stability, and cytotoxicity.
Polymeric delivery systems for vaccines function at the interface of material science and immunology, acting both as carriers and immunomodulators. Nanoparticle size (typically 20–200 nm) and surface characteristics critically influence lymphatic drainage and uptake by dendritic cells. Once internalized, polymers can facilitate endosomal escape or controlled antigen release, promoting cross-presentation via MHC class I pathways, which is essential for cytotoxic T cell activation. Additionally, co-delivery of adjuvants such as Toll-like receptor (TLR) agonists within the same polymer matrix enables synchronized immune stimulation, significantly enhancing vaccine efficacy and durability.
Protein and peptide therapeutics present significant formulation challenges due to their structural fragility and susceptibility to aggregation, deamidation, and proteolysis. Polymeric carriers such as hydrogels and biodegradable microspheres provide hydrated, protective environments that stabilize secondary and tertiary structures. Release kinetics are governed by a combination of Fickian diffusion and polymer matrix degradation, which can be tuned by adjusting crosslink density and polymer composition. Additionally, surface modification strategies can reduce protein adsorption and immunogenicity, while enabling targeted or sustained delivery for chronic therapeutic applications.
Oral delivery systems must overcome multiple physiological barriers, including low gastric pH, digestive enzymes, and tight epithelial junctions. pH-responsive polymers enable enteric protection by remaining insoluble in acidic environments and dissolving at higher intestinal pH. Beyond protection, advanced polymer systems incorporate mucoadhesive properties and permeation enhancers that transiently modulate tight junction integrity, facilitating paracellular transport. Nanostructured polymer carriers can also promote transcytosis across intestinal epithelial cells, further improving systemic bioavailability of poorly absorbed drugs.
For poorly water-soluble small molecules, polymer-based delivery systems improve dissolution kinetics and bioavailability through mechanisms such as amorphous solid dispersion formation and nanoscale encapsulation. Polymers inhibit drug crystallization by stabilizing supersaturated states via intermolecular interactions, including hydrogen bonding and hydrophobic interactions. Additionally, nanoscale carriers alter biodistribution by exploiting the enhanced permeability and retention (EPR) effect in tumor tissues, enabling passive targeting and improved therapeutic index.
Advanced polymer systems for nucleic acid delivery are designed with stimuli-responsive architectures that enable precise intracellular release. Incorporation of pH-sensitive or redox-cleavable linkages allows carriers to remain stable in circulation while rapidly disassembling in endosomal or cytosolic environments. These systems must also address key delivery bottlenecks, including cellular uptake, endosomal escape, and nuclear transport (for DNA). Rational polymer design focuses on balancing stability and release kinetics to maximize gene expression while minimizing cytotoxicity and off-target effects.
mRNA delivery requires overcoming unique challenges related to molecular size, instability, and susceptibility to enzymatic degradation. Ionizable polymers, such as poly(beta-amino esters), are engineered to exhibit pH-dependent charge behavior, enabling efficient encapsulation under acidic conditions and reduced toxicity at physiological pH. Upon endosomal acidification, these polymers become protonated, promoting membrane destabilization and cytosolic release of mRNA. Compared to lipid-based systems, polymer-based carriers offer greater tunability in structure and degradation profiles, providing opportunities to optimize translation efficiency, tissue targeting, and dosing frequency in next-generation mRNA therapeutics.
Safety and Regulatory Considerations for Polymer-Based Drug Delivery
The successful translation of polymer-based drug delivery systems from early-stage design to clinical and commercial applications is governed by stringent safety, toxicological, and regulatory requirements. Beyond achieving therapeutic efficacy, the polymer carrier must demonstrate a well-defined safety profile throughout its entire lifecycle, including degradation, metabolism, and clearance. This necessitates a comprehensive evaluation of structure–property–toxicity relationships, alongside robust chemistry, manufacturing, and controls (CMC) documentation. Regulatory approval pathways require not only reproducible manufacturing processes but also detailed characterization of polymer composition, impurity profiles, and in vivo behavior to ensure long-term patient safety and product consistency.
Biocompatibility and Toxicity Evaluation of Polymers
Biocompatibility assessment of polymeric materials involves a multi-tiered evaluation strategy encompassing in vitro, ex vivo, and in vivo studies. Key parameters include cytotoxicity (e.g., MTT or LDH assays), hemocompatibility (hemolysis, coagulation, complement activation), and immunogenicity profiling through cytokine release and macrophage activation assays. In vivo studies further evaluate biodistribution, pharmacokinetics, and long-term accumulation, particularly within the reticuloendothelial system (RES) organs such as the liver and spleen. The degradation behavior of polymers must also be characterized, ensuring that resulting oligomers or monomers are non-toxic, non-bioaccumulative, and eliminated via renal or hepatic pathways without inducing chronic inflammation or organ-specific toxicity.
Regulatory Guidelines for Pharmaceutical Polymers
Regulatory evaluation of polymeric excipients follows a risk-based framework aligned with global guidelines, requiring detailed documentation of material composition, manufacturing processes, and safety data. Polymers with established regulatory acceptance—such as those listed in pharmacopeias or classified as Generally Recognized as Safe (GRAS)—offer a streamlined pathway for formulation development. In contrast, novel polymers must undergo extensive preclinical evaluation, including toxicology, genotoxicity, and degradation studies, supported by comprehensive CMC data. Critical aspects include demonstrating batch consistency, defining impurity thresholds, and validating analytical methods, as well as establishing clear correlations between polymer structure and functional performance in drug delivery systems.
Quality Assurance and Control in Polymer Production
Ensuring pharmaceutical-grade polymer quality requires a rigorous quality control framework supported by advanced analytical characterization techniques. Methods such as Gel Permeation Chromatography (GPC) are used to determine molecular weight distribution and dispersity, while Nuclear Magnetic Resonance (NMR) spectroscopy confirms chemical structure and copolymer composition. Additional techniques, including Fourier-transform infrared spectroscopy (FTIR), differential scanning calorimetry (DSC), and mass spectrometry, provide insights into functional groups, thermal properties, and impurity profiles. Strict control of residual monomers, catalysts, and solvents—often guided by ICH impurity limits—is essential to minimize toxicity risks. These analytical protocols ensure consistency, traceability, and compliance with regulatory expectations.
Risk Mitigation and Best Practices
Effective risk mitigation in polymer-based drug delivery development is achieved through the implementation of Quality by Design (QbD) principles, which emphasize a systematic understanding of the relationship between material attributes, process parameters, and product performance. By defining Critical Quality Attributes (CQAs) such as particle size, zeta potential, drug loading efficiency, and release kinetics, and linking them to Critical Process Parameters (CPPs), developers can establish a robust design space that ensures consistent product quality. Risk assessment tools, including failure mode and effects analysis (FMEA), are employed to identify and control potential failure points. This proactive, data-driven approach enhances process reliability, reduces variability, and supports regulatory approval by demonstrating a high level of process understanding and control.
Scale-Up and Manufacturing of Polymer Drug Delivery Systems
Transitioning polymeric drug delivery systems from laboratory-scale development to industrial manufacturing requires the integration of polymer chemistry, transport phenomena, and process engineering principles. At larger scales, subtle variations in mixing efficiency, solvent exchange kinetics, and thermal gradients can significantly alter nucleation, polymer self-assembly, and drug encapsulation behavior. Ensuring scalability therefore depends on maintaining consistent dimensionless parameters—such as Reynolds number, Peclet number, and mixing time—across scales, while preserving critical quality attributes (CQAs) including particle size, polydispersity index (PDI), drug loading, and release kinetics. Successful scale-up strategies must also consider regulatory compliance, process robustness, and cost-efficiency to enable reliable translation from research to commercial production.
Challenges in Scaling Laboratory Formulations
Laboratory-scale formulation methods, including nanoprecipitation, thin-film hydration, and probe sonication, are highly sensitive to local mixing conditions and often rely on rapid diffusion and turbulent microenvironments that are difficult to replicate at scale. As production volume increases, the relative influence of diffusion versus convection shifts, leading to non-uniform supersaturation, altered nucleation rates, and heterogeneous particle growth. These changes can result in broader particle size distributions, reduced encapsulation efficiency, and instability of polymeric assemblies. Additionally, heat accumulation and solvent evaporation rates differ significantly in large systems, further complicating process reproducibility and product consistency.
Process Optimization for Large-Scale Production
To address scale-dependent variability, advanced process engineering strategies are employed to achieve controlled and reproducible polymer assembly. Continuous flow reactors and microfluidic mixing systems enable precise control over mixing timescales, solvent gradients, and concentration profiles, thereby standardizing nanoparticle formation. Process optimization involves systematic adjustment of flow rates, solvent-to-antisolvent ratios, and shear conditions to maintain uniform supersaturation and nucleation kinetics. The integration of computational fluid dynamics (CFD) modeling further supports rational reactor design and scale translation, allowing prediction and mitigation of flow heterogeneity and mass transfer limitations.
Reproducibility and Batch-to-Batch Consistency
Achieving consistent product quality across batches is critical for regulatory approval and commercial viability. Variability in polymer raw materials, including molecular weight distribution, block composition, and residual monomer content, can significantly influence drug loading capacity and release behavior. Implementation of Process Analytical Technology (PAT) enables real-time monitoring of critical process parameters (CPPs) such as particle size, turbidity, and solvent composition. Coupled with advanced analytical techniques—such as dynamic light scattering (DLS), gel permeation chromatography (GPC), and differential scanning calorimetry (DSC)—these tools ensure tight control over CQAs and enable adaptive process control strategies for maintaining reproducibility.
Industrial Manufacturing Techniques
Industrial-scale production of polymer drug delivery systems relies on robust, scalable unit operations tailored to specific formulation requirements. High-pressure homogenization and microfluidization are commonly used to produce uniform nanoscale dispersions, while tangential flow filtration (TFF) enables efficient purification, solvent exchange, and concentration of nanoparticle suspensions. Controlled lyophilization processes, supported by optimized cryoprotectants, are essential for maintaining structural integrity during long-term storage. For larger depot systems such as polymer microspheres, techniques including spray drying, fluidized bed processing, and continuous double-emulsion solvent evaporation are employed to achieve precise control over particle morphology, porosity, and residual solvent levels. These integrated manufacturing strategies ensure scalability, product stability, and compliance with stringent pharmaceutical quality standards.
Services
Advanced Polymer Synthesis and Formulation Services
Advancing polymer-based drug delivery systems requires not only innovative material design but also integrated technical capabilities across synthesis, modification, and characterization. BOC Sciences provides comprehensive support spanning custom monomer development, precision polymer synthesis, and advanced functionalization to meet diverse formulation requirements. With expertise in controlling molecular weight distribution, polymer architecture, and surface chemistry, we enable the development of tailored macromolecules designed to address critical challenges such as drug solubility, stability, targeting, and controlled release. Our capabilities further extend to analytical characterization and scalable production strategies, ensuring that materials developed at the research stage can be reliably translated into practical and manufacturable drug delivery solutions.

Custom Polymer & Copolymer Synthesis
- Precision control over molecular weight and block architectures.
- Advanced living polymerization techniques (ATRP, RAFT, ROP).
- Design of multi-block, branched, and star-shaped macromolecular structures.
- Synthesis of stimuli-responsive and bio-reducible polymers.

Polymer Functionalization & Bioconjugation
- End-group modification for targeted ligand attachment.
- Covalent conjugation of peptides, small molecules, and targeting moieties.
- Cleavable linker engineering for controlled active payload release.
- Custom PEGylation strategies for circulation extension.

Nanocarrier Formulation Development
- Optimization of polymeric micelles, nanospheres, and polyplexes.
- Tuning of Critical Quality Attributes (CQAs) including size, PDI, and Zeta potential.
- Maximizing encapsulation efficiency for hydrophobic and biological payloads.
- Stability screening and microenvironmental responsiveness testing.

GMP-Ready Material Supply & Scale-Up
- High-purity supply of biodegradable polymers, dendrimers, and specialized monomers.
- Seamless transition from milligram research scales to kilogram production.
- Stringent analytical quality control (NMR, GPC, HPLC).
- Process optimization for robust batch-to-batch consistency.
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What is the most common polymer used in drug delivery?
One of the most widely used polymers in drug delivery is poly(lactic-co-glycolic acid) (PLGA), valued for its excellent biocompatibility and predictable biodegradation. It enables controlled drug release and is approved in multiple pharmaceutical products, making it a gold standard for sustained-release formulations and injectable delivery systems.
-
How do polymeric micelles improve drug solubility?
Polymeric micelles enhance drug solubility by forming core–shell nanostructures from amphiphilic block copolymers. The hydrophobic core encapsulates poorly soluble drugs, while the hydrophilic shell stabilizes the system in aqueous environments. This significantly improves dissolution, bioavailability, and systemic stability of hydrophobic therapeutic compounds.
-
What is the difference between bulk erosion and surface erosion in polymers?
Bulk erosion occurs when water penetrates the entire polymer matrix, causing uniform degradation throughout the material. In contrast, surface erosion is limited to the outer layer, leading to gradual mass loss. These mechanisms directly influence drug release kinetics, with bulk erosion often causing burst release and surface erosion enabling more controlled delivery.
-
Why is polymer molecular weight important for formulation?
Polymer molecular weight critically determines mechanical strength, degradation rate, and drug release behavior. Higher molecular weight polymers typically degrade more slowly and provide prolonged release, while lower molecular weight materials enable faster drug diffusion. Precise control ensures optimal balance between stability, processability, and therapeutic performance.
-
What are stimuli-responsive polymers?
Stimuli-responsive polymers, also known as “smart polymers,” undergo structural or chemical changes in response to environmental triggers such as pH, temperature, redox conditions, or enzymes. These materials enable site-specific and controlled drug release, improving therapeutic precision while minimizing systemic side effects in advanced drug delivery systems.
-
How do cationic polymers aid in gene delivery?
Cationic polymers facilitate gene delivery by electrostatically binding negatively charged nucleic acids to form stable polyplexes. These complexes protect genetic material from enzymatic degradation and promote cellular uptake. Additionally, buffering capacity enables endosomal escape, enhancing intracellular delivery efficiency and improving overall transfection performance.

 Fig. 1. Advanced polymeric nanocarrier mechanisms for targeted drug delivery systems (BOC Sciences Authorized).
Fig. 1. Advanced polymeric nanocarrier mechanisms for targeted drug delivery systems (BOC Sciences Authorized). Fig. 2. Five key therapeutic applications of polymeric drug delivery systems (BOC Sciences Authorized).
Fig. 2. Five key therapeutic applications of polymeric drug delivery systems (BOC Sciences Authorized).