Overcoming Oral Drug Delivery Challenges with Polymer Carriers
In the contemporary field of drug development, oral administration remains the preferred route of drug delivery due to its non-invasive nature, high patient compliance, and cost-effectiveness. However, with advances in modern medicinal chemistry, new molecular entities (NMEs) often exhibit poor solubility, low permeability, or instability within the gastrointestinal (GI) environment, posing severe challenges to conventional formulation technologies. Polymers, as versatile drug delivery platforms, provide powerful solutions to overcome these biological barriers through physicochemical modification and structural design. Customized polymer-based drug delivery strategies, biomimetic and biopolymer architectures, and drug-free macromolecular processing approaches have all been applied to address challenges in polymer drug delivery. Polymer drug delivery systems based on natural and synthetic polymers have been rapidly adopted across the pharmaceutical industry. Integrating perspectives from both artificial and biological disciplines will facilitate the development of innovative models for polymer-based therapeutic and nucleic acid delivery systems.
Resources
The Importance of Oral Drug Delivery in Modern Therapeutics
Oral Drug Delivery Systems (ODDS) account for the largest share of the global pharmaceutical market. Compared with intravenous injection or transdermal administration, oral delivery is more consistent with human physiology and is particularly critical for the long-term management of chronic diseases. However, to achieve effective therapeutic outcomes, drugs must successfully traverse a series of physiological barriers from ingestion to entry into systemic circulation.
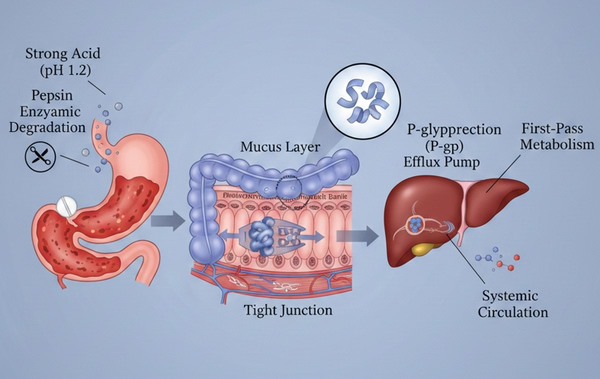 Fig. 1. Biological barriers in oral delivery: gastric acid, mucus, and first-pass metabolism (BOC Sciences Authorized).
Fig. 1. Biological barriers in oral delivery: gastric acid, mucus, and first-pass metabolism (BOC Sciences Authorized).
Limitations of Conventional Oral Drug Formulations
Conventional oral formulations, such as immediate-release tablets and hard capsules, rely heavily on the intrinsic physicochemical properties of the active pharmaceutical ingredient (API). According to the Biopharmaceutics Classification System (BCS), a large proportion of promising drug molecules exhibit extremely low aqueous solubility due to high crystal lattice energy (BCS Class II) or insufficient transmembrane flux caused by excessive molecular polarity (BCS Class IV), resulting in poor bioavailability and significant inter-individual variability in absorption. In addition, traditional formulations struggle to cope with the dramatic pH fluctuations and complex enzymatic environment of the gastrointestinal tract, causing many innovative small-molecule and biologic drugs to lose efficacy before reaching their absorption sites, thereby limiting their clinical applicability.
Why Advanced Delivery Technologies Are Needed for Oral Drugs?
Among drug candidates generated by modern drug discovery technologies such as combinatorial chemistry and high-throughput screening, approximately 70%–90% suffer from solubility limitations, while the emergence of macromolecular therapeutics—including peptides, proteins, and nucleic acids—poses unprecedented challenges to delivery systems. These drugs face not only chemical degradation by gastric acid and proteases but also substantial diffusion resistance and the epithelial barrier of the intestinal lining. Consequently, there is an urgent need to develop advanced material-based delivery technologies—particularly those employing functional polymer carriers—to precisely regulate the drug microenvironment and enable advanced functionalities such as supersaturation maintenance, site-specific release, and intracellular targeting. Such strategies are essential to transform therapeutically promising but physicochemically challenging molecules into safe and effective clinical drugs.
Key Challenges in Oral Drug Delivery
Before reaching their targets, orally administered drugs must pass through the complex physiological environment of the gastrointestinal tract. Each step in this process can become a rate-limiting factor for drug absorption. Understanding these physiological and biochemical barriers is a prerequisite for the rational design of effective polymer carriers:
- Poor Aqueous Solubility and Dissolution Rate: Solubility is a prerequisite for drug absorption. Many hydrophobic drugs exhibit extremely low equilibrium solubility in gastrointestinal fluids. According to the Noyes–Whitney equation, their dissolution rate limits absorption. When drugs exist in crystalline form, high lattice energy impedes dissolution, resulting in drug elimination via feces before absorption occurs.
- Low Intestinal Permeability and Limited Absorption: The intestinal epithelial layer constitutes the primary absorption barrier. Drug molecules must enter the bloodstream via passive diffusion, carrier-mediated transport, or endocytosis. Hydrophilic macromolecules or highly polar drugs struggle to penetrate the lipid bilayer, while tight junctions restrict paracellular transport. In addition, efflux transporters such as P-glycoprotein (P-gp) can pump drugs that have entered epithelial cells back into the intestinal lumen.
- Enzymatic Degradation in the Gastrointestinal Tract: The GI tract is rich in digestive enzymes, including pepsin, trypsin, chymotrypsin, and various peptidases. These enzymes are particularly destructive to protein and peptide drugs, which are often hydrolyzed into amino acids or short peptides before reaching absorption sites, completely losing their pharmacological activity.
- Instability Under Gastric pH and Harsh GI Conditions: The gastric environment is highly acidic (pH 1.0-2.5), while intestinal pH gradually increases to neutral or slightly alkaline levels. Many acid-labile drugs, such as proton pump inhibitors and certain antibiotics, undergo chemical degradation in gastric acid. This dramatic pH variation requires delivery systems capable of providing protection under specific pH conditions.
- First-Pass Metabolism and Reduced Bioavailability: After intestinal absorption, drugs first enter the liver via the portal vein. The liver's abundant cytochrome P450 (CYP450) enzyme system metabolizes drugs through first-pass metabolism, often allowing only a small fraction of the unchanged drug to reach systemic circulation, thereby significantly reducing oral bioavailability.
Polymer Carriers as Enabling Technologies for Oral Drug Delivery
Advances in polymer science provide a multidimensional toolbox to address the challenges described above. By precisely designing polymer molecular weight, hydrophilic–hydrophobic balance, charge, and functional groups, intelligent drug carrier systems can be constructed:
- Improving Solubility and Dissolution via Polymer-Based Systems: Polymers enhance the solubility of poorly soluble drugs through multiple mechanisms. Amphiphilic polymers can self-assemble into micelles in aqueous media, encapsulating hydrophobic drugs within their cores and increasing apparent solubility by several orders of magnitude. Polymers can also act as precipitation inhibitors in supersaturating drug delivery systems (SDDS), maintaining drugs in high-energy amorphous states and preventing recrystallization.
- Protecting Drugs from Acidic and Enzymatic Degradation: Enteric polymers can be used to coat or encapsulate drugs, effectively shielding them from gastric acid. These polymers typically contain carboxyl groups that remain unionized and insoluble in acidic environments but ionize and dissolve under intestinal pH conditions to release the drug. Against enzymatic degradation, polymer matrices provide steric hindrance that limits enzyme–substrate interactions.
- Enhancing Intestinal Transport and Cellular Uptake: Certain functional polymers, such as chitosan and its derivatives, can transiently open tight junctions between intestinal epithelial cells, promoting paracellular transport of hydrophilic macromolecules. Other polymers, such as Pluronics, have been shown to inhibit P-gp efflux activity, increasing intracellular drug accumulation.
- Controlled and Sustained Drug Release Profiles: Polymer carriers can achieve zero-order or first-order drug release kinetics. By adjusting polymer erosion rates or drug diffusion coefficients within polymer matrices, sustained-release formulations can be designed to maintain stable plasma drug concentrations and reduce dosing frequency.
- Reducing Variability in Oral Drug Absorption: Food intake, gastric emptying time, and inter-individual variability can cause fluctuations in drug absorption. Polymer carriers—particularly mucoadhesive systems—can prolong gastrointestinal residence time, making the absorption process more controllable and predictable.
Types of Polymer Carriers Used in Oral Drug Delivery
In the development of oral formulations, polymer selection directly determines the stability, release kinetics, and ultimate bioavailability of drug delivery systems. Based on source, chemical structure, and functional characteristics, polymers commonly used for oral carriers can be classified as follows:
Natural Polymers for Oral Drug Protection and Release
Natural polymers, such as polysaccharides (chitosan, alginate, pectin) and proteins (gelatin, albumin), are widely used due to their excellent biocompatibility, low immunogenicity, and extracellular-matrix-like properties. Chitosan and its derivatives, in particular, possess unique cationic characteristics that enable electrostatic interactions with negatively charged intestinal mucins, significantly prolonging residence time at absorption sites and opening tight junctions. In addition, these polymers typically exhibit mild gelation conditions, allowing drug encapsulation without organic solvents and thereby preserving the activity and conformational stability of biologic drugs such as peptides and proteins.
Synthetic Polymers with Tunable Physicochemical Properties
Compared with natural polymers, synthetic polymers such as polyethylene glycol (PEG), polyvinylpyrrolidone (PVP), and polyvinyl alcohol (PVA) offer superior batch-to-batch consistency and predictable physicochemical properties. Through precise control of polymerization processes, researchers can tailor molecular weight, branching degree, and hydrophilic–hydrophobic balance, enabling fine regulation of mechanical strength, degradation rate, and drug release profiles. This high degree of tunability makes synthetic polymers ideal backbone materials for complex formulation systems such as solid dispersions and polymeric micelles, effectively addressing poor solubility and physical instability.
Biodegradable Polymers for Safe and Efficient Oral Drug Delivery
Biodegradable polymers, including poly(lactic-co-glycolic acid) (PLGA), polylactic acid (PLA), and polycaprolactone (PCL), are among the most representative FDA-approved pharmaceutical polymers. These materials undergo ester bond hydrolysis under physiological conditions to form non-toxic metabolites such as lactic acid and glycolic acid, which are ultimately eliminated via the tricarboxylic acid cycle, eliminating the risk of polymer accumulation toxicity. Owing to their excellent film-forming and encapsulation capabilities, they are widely used to prepare nanoparticles or microspheres that resist gastric acid and enable long-acting sustained release.
pH-Responsive Polymers for Site-Specific Drug Release
pH-responsive polymers, primarily poly(methacrylic acid) derivatives such as the Eudragit® series, exploit the pronounced pH gradients along the gastrointestinal tract—from pH 1.2 in the stomach to above pH 7.0 in the colon—to achieve intelligent drug release. These polymers contain ionizable side-chain groups, such as carboxyl or amino groups, that undergo protonation or deprotonation at specific pH thresholds, resulting in abrupt changes in solubility. This mechanism enables formulations to function as molecular "switches," remaining intact in gastric acid to protect acid-labile drugs and rapidly dissolving upon reaching target pH environments in the intestine or colon for precise site-specific delivery.
Mucoadhesive Polymers for Prolonged Gastrointestinal Retention
Mucoadhesive polymers, including carbomers, hydroxypropyl methylcellulose (HPMC), and thiolated polymers, are designed to overcome gastrointestinal clearance caused by peristalsis by extending contact time at the site of action. These polymer chains adhere tightly to the intestinal mucosal layer through hydrogen bonding, van der Waals forces, or covalent disulfide bond formation with mucin glycoproteins. Prolonged intimate contact not only increases the concentration gradient driving transmembrane diffusion and shortens diffusion distances but also significantly improves the oral bioavailability of drugs with narrow absorption windows.
Amphiphilic Polymers for Solubilizing Poorly Soluble Drugs
Amphiphilic block copolymers, such as Poloxamers/Pluronics and PEG–lipid conjugates, contain both hydrophilic and hydrophobic segments that enable self-assembly into thermodynamically stable micelles in aqueous solutions. The hydrophobic core of these supramolecular structures provides an excellent solubilization microenvironment for poorly soluble drugs (BCS Class II and IV), increasing apparent solubility by several orders of magnitude. Meanwhile, the hydrophilic corona offers steric stabilization to prevent particle aggregation and can partially inhibit P-gp efflux activity, thereby synergistically enhancing drug absorption.
Looking for Custom Polymers for Oral Drug Delivery?
Whether you need cationic polymers, biodegradable carriers, or stimuli-responsive platforms, our experts provide tailored synthesis and functionalization solutions to support your research and therapeutic development.
Polymer-Based Oral Delivery Systems and Formulation Strategies
Transforming polymeric materials into highly efficient drug delivery carriers requires the application of advanced formulation technologies and precise structural design. The following are some of the most widely used polymer-based formulation strategies in current drug development:
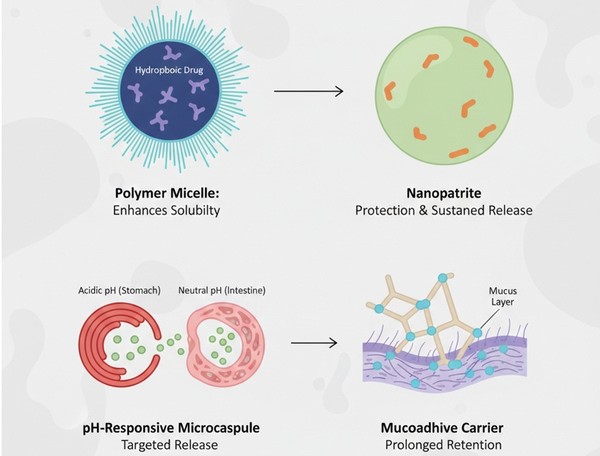 Fig. 2. Structural diversity of polymer carriers: micelles, nanoparticles, pH-responsive, and mucoadhesive systems (BOC Sciences Authorized).
Fig. 2. Structural diversity of polymer carriers: micelles, nanoparticles, pH-responsive, and mucoadhesive systems (BOC Sciences Authorized).
Polymer Micelles for Oral Delivery of Hydrophobic Drugs
Polymer micelles are nanoscale core–shell structures formed by the self-assembly of amphiphilic block copolymers in aqueous solutions. Their hydrophobic cores can efficiently encapsulate and solubilize hydrophobic drugs, significantly increasing the apparent solubility of poorly soluble compounds. The outer hydrophilic shell provides steric stabilization to the system, preventing clearance by the reticuloendothelial system, and facilitates penetration through the intestinal mucus layer via steric repulsion effects. In addition, certain micellar materials, such as Pluronics, have been shown to inhibit intestinal P-glycoprotein (P-gp) efflux pump activity, thereby further enhancing cellular uptake and oral bioavailability.
Polymeric Nanoparticles for Enhanced Oral Bioavailability
Polymeric nanoparticles, including nanospheres and nanocapsules, provide a physical barrier against gastric acid and enzymatic degradation by encapsulating or adsorbing drugs within a polymer matrix, thereby protecting labile active ingredients such as proteins, peptides, and nucleic acids. These submicron carriers can exploit specific transport mechanisms, such as M-cell-mediated endocytosis in Peyer's patches, to facilitate trans-epithelial transport of macromolecular drugs. Surface modification of nanoparticles with PEG or conjugation with targeting ligands can further extend systemic circulation half-life and enable precise delivery to diseased sites.
Solid Dispersions and Amorphous Polymer Matrices
Solid dispersion technology employs hydrophilic polymer carriers, such as PVPVA and HPMC-AS, to disperse poorly soluble drugs in a molecular or amorphous state, disrupting the drug's stable crystalline lattice and significantly reducing the energy barrier required for dissolution. During this process, the polymer matrix plays a critical anti-plasticization role by inhibiting drug recrystallization during storage and in vivo dissolution through intermolecular hydrogen bonding or steric hindrance. Maintenance of this high-energy state markedly improves drug wettability and dissolution rate, making it one of the most mature and commercially successful strategies for overcoming oral absorption challenges associated with BCS Class II drugs.
Functional Polymer Coatings for Controlled Release
Functional polymer coating technologies form a continuous semipermeable or pH-sensitive film on the surface of tablets, pellets, or granules to precisely regulate drug diffusion and release kinetics. Insoluble polymers such as ethyl cellulose can be used to construct reservoir-type sustained-release systems, while enteric acrylic resins enable site-specific release in the small intestine or colon based on environmental pH. This strategy not only masks undesirable taste and odor but also prevents gastric irritation, serving as a classic approach for achieving pulsatile or zero-order release profiles.
Polymeric Capsules for Improved Stability and Oral Drug Administration
Capsule shells based on plant-derived polymers such as hydroxypropyl methylcellulose (HPMC) overcome the limitations of conventional gelatin capsules, including cross-linking reactions and instability toward moisture-sensitive drugs, due to their low moisture content and chemical inertness. The mechanical strength of polymer capsules is not affected by low-humidity environments, effectively preventing brittleness, and making them particularly suitable for filling and storage of hygroscopic or hydrolysis-prone drugs. In addition, next-generation enteric polymer capsules enable intestinal targeting without the need for additional coating processes, simplifying manufacturing and improving batch-to-batch reproducibility.
Applications of Polymer Carriers in Oral Drug Delivery
The multifunctionality of polymer carriers confers significant clinical value in the delivery of a wide range of drug molecules. Through customized design based on specific physicochemical properties of drugs, polymer systems can substantially enhance therapeutic efficacy while reducing toxicity and adverse effects.
Oral Delivery of Poorly Soluble Small-Molecule Drugs
For poorly soluble small-molecule drugs that dominate modern drug discovery pipelines, polymer carriers effectively address dissolution limitations in gastrointestinal fluids by forming solid dispersions or polymeric micelles. Polymer matrices maintain drugs in a supersaturated state and act as precipitation inhibitors to prevent conversion to low-energy crystalline forms, thereby creating a sustained absorption driving force. This strategy has demonstrated excellent performance in the oral reformulation of anticancer agents, such as paclitaxel derivatives, and immunosuppressants, significantly improving bioavailability and reducing food-related absorption variability.
Polymer Strategies for Oral Peptide and Protein Delivery
Oral delivery of peptide and protein drugs faces multiple barriers, including extreme pH conditions, extensive proteolytic degradation, and limited trans-epithelial transport. Functional polymers, such as thiolated chitosan or pH-responsive hydrogels, can protect these biomacromolecules by forming protective barriers in the stomach and releasing them at specific sites in the small intestine. Furthermore, temporary modulation of intestinal tight junctions or promotion of M-cell-mediated endocytosis enables effective transport of macromolecules into lymphatic or systemic circulation, making oral delivery of drugs such as insulin and calcitonin feasible.
Oral Delivery Solutions for Highly Permeable but Unstable APIs
For active pharmaceutical ingredients (APIs) with high permeability but pronounced chemical instability under gastrointestinal physiological conditions, polymer encapsulation technologies provide indispensable stabilization solutions. By embedding APIs within hydrophobic polymer matrices or microcapsules, exposure to moisture, oxygen, and acidic gastric fluids is effectively minimized. This physical shielding extends the effective residence time of drugs in the GI tract and ensures that they reach absorption sites in their intact form, thereby preserving expected pharmacological activity and therapeutic concentrations.
Polymer-Based Approaches for Chronic Disease Therapies
In the long-term management of chronic diseases such as hypertension, diabetes, and cardiovascular disorders, patient compliance is a critical determinant of therapeutic success. Polymer-based osmotic pump tablets or multilayer matrix tablets enable constant drug release over 24 hours, significantly reducing peak–trough fluctuations in plasma drug concentrations and lowering dosing frequency and associated adverse effects. This controlled-release mechanism is independent of gastrointestinal pH or motility, ensuring safety and efficacy under complex physiological conditions and highlighting the central role of polymers in precision medicine.
Polymer-Based Dosage Forms for Advanced Drug Delivery
- Polymer Patches: Buccal mucoadhesive patches use bioadhesive polymers such as carbomers and sodium alginate to retain drugs on the buccal mucosa. This dosage form avoids first-pass metabolism and allows direct entry into systemic circulation, making it suitable for drugs requiring rapid onset or protection from gastrointestinal degradation.
- Polymer-Engineered Tablets: Matrix tablets are the most common sustained-release dosage form. Hydrophilic gel matrices, such as HPMC, swell upon hydration to form a gel layer that controls drug diffusion, while insoluble matrices, such as ethyl cellulose, regulate release through pore diffusion.
- Polymer Tapes: Polymeric drug-loaded tapes are an emerging dosage form typically composed of a backing layer, a drug-containing polymer layer, and an adhesive layer. Their flexibility allows localized administration to specific sites or use as a novel orally disintegrating delivery system.
- Semisolid Polymer Systems: Semisolid formulations such as gels and ointments utilize thermoresponsive polymers like Poloxamers, which remain liquid at low temperatures for easy swallowing or application and undergo sol–gel transition at body temperature to prolong mucosal residence time.
- Polymer Films: Oral thin films (OTFs) are fabricated from water-soluble polymers such as pullulan and HPMC. They dissolve within seconds when placed on the tongue without the need for water, greatly improving compliance in patients with swallowing difficulties, including pediatric and geriatric populations.
- Polymer Powders: Functionalized polymer powders can be used for direct compression, dry granulation, or as adsorption carriers. Porous polymer carriers can convert liquid drugs into solid powders, improving flowability and compressibility.
Key Design Considerations for Polymer Carriers in Oral Formulations
Developing a successful polymer-based oral delivery system requires not only a strong foundation in materials chemistry but also a deep understanding of biopharmaceutics. The design process must identify the optimal balance among drug properties, polymer characteristics, and physiological barriers.
Polymer Selection Based on Drug Properties
Polymer selection should begin with an in-depth analysis of the physicochemical properties of the API, particularly the compatibility between drug solubility parameters and those of the polymer. Flory–Huggins interaction parameter predictions can be used to assess molecular-level miscibility between drug and polymer, which is critical for preventing phase separation in solid dispersions during storage. In addition, the polymer glass transition temperature (Tg) should be significantly higher than the intended storage temperature to provide sufficient kinetic barriers that limit drug recrystallization.
Biocompatibility, Safety, and Regulatory Considerations
As components of oral delivery systems, polymers must exhibit excellent biocompatibility, and their degradation products must be demonstrated to be non-toxic, non-immunogenic, and eliminable through normal physiological pathways. Since oral formulations are often administered long term, potential irritation to gastrointestinal mucosa and effects on normal microbiota must be evaluated through toxicological studies. From a regulatory perspective, prioritizing excipients listed as Generally Recognized as Safe (GRAS) by the FDA or included in major pharmacopeias can significantly reduce regulatory risk for New Drug Applications (NDAs) or Abbreviated New Drug Applications (ANDAs).
Scalability and Manufacturing Feasibility
Innovations developed at the laboratory scale must consider feasibility for large-scale industrial production, including process robustness and cost-effectiveness. For example, although hot-melt extrusion (HME) is highly effective for producing solid dispersions, the thermal stability of the API and the melt-processability of the polymer must be carefully evaluated. Similarly, nanoparticle fabrication processes involving complex organic solvent removal or ultracentrifugation may be difficult to translate into commercial manufacturing due to equipment constraints or solvent residue concerns. In contrast, simple and continuous processes such as fluidized-bed coating often offer greater competitive advantages.
Stability and Shelf-Life of Polymer-Based Oral Formulations
Polymer-based formulation systems are thermodynamically unstable or metastable, and their physicochemical stability over shelf life is a core indicator of developability. Polymers may undergo physical aging, leading to matrix densification or changes in hygroscopicity, which can subsequently affect drug release behavior and cause dissolution drift. Formulation design must therefore consider moisture-protective packaging, such as aluminum–plastic blister packs, and the inclusion of appropriate antioxidants or desiccants. Long-term and accelerated stability studies should be conducted in accordance with ICH guidelines to ensure that critical quality attributes (CQAs) remain consistent throughout the product's shelf life.
Services
BOC Sciences' Polymer Solutions for Oral Drug Delivery
Leveraging extensive expertise in polymer materials and drug delivery, BOC Sciences provides customized polymer development and formulation support services tailored for oral drug delivery applications. Our polymer solutions are designed to overcome challenges such as gastrointestinal degradation, poor solubility, and limited bioavailability, enabling the oral development of small-molecule drugs, peptides, and emerging therapeutic modalities.

Custom Polymer Synthesis for Oral Delivery Applications
- Custom synthesis of polymer materials suitable for oral delivery, such as PLGA, PCL, and HPMC
- Precise control of molecular weight, hydrophobic/hydrophilic balance, and degradation behavior
- Design of enteric, sustained-release, or mucoadhesive polymer architectures
- Support for the construction of polymer–drug conjugates and encapsulation systems

Polymer Modification and Functionalization Services
- Surface modification to enhance mucoadhesion and prolong intestinal residence time
- Introduction of pH-responsive or enzyme-responsive moieties for site-specific drug release
- PEGylation or hydrophilic modification to improve solubility and dispersion
- Conjugation of permeation-enhancing groups to increase trans-epithelial absorption efficiency

Formulation Development and Optimization Support
- Development of oral dosage forms (microparticles, nanoparticles, capsules, tablets, etc.)
- Evaluation of polymer–drug compatibility and encapsulation efficiency
- Optimization of release profiles, dissolution behavior, and performance under simulated gastrointestinal conditions
- Excipient screening and optimization of formulation parameters (pH, ionic strength)

Analytical Characterization of Polymer-Based Delivery Systems
- Particle size, size distribution, and morphology analysis (DLS, SEM/TEM)
- Surface charge and dispersion stability evaluation
- Drug loading, release behavior, and degradation kinetics analysis
- Structural and compositional characterization (GPC, FTIR, NMR, etc.)
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
What are polymer carriers used for in oral drug delivery?
Polymer carriers serve as multifunctional platforms in oral drug delivery. Their core applications include significantly enhancing the solubility and dissolution rate of poorly soluble APIs by forming solid dispersions or micelles; protecting drugs from degradation by gastric acid and gastrointestinal proteases through physical encapsulation or chemical shielding; and enabling controlled release via matrix or osmotic pump systems. In addition, mucoadhesive properties can prolong drug residence time within the absorption window. Collectively, these strategies aim to substantially improve bioavailability and reduce dosing frequency.
How do polymers protect drugs from gastric degradation?
Polymeric protection is primarily achieved through two mechanisms. The first is pH-dependent barrier formation: enteric polymers, such as the Eudragit® series, remain non-ionized and insoluble in gastric acid (pH 1.2), forming a dense protective film. The second is steric hindrance, whereby polymer matrices or nanospheres embed drug molecules within crosslinked networks, preventing effective contact with digestive enzymes. For peptide drugs in particular, this physical shielding significantly reduces enzymatic degradation rates, ensuring that APIs reach the intestinal absorption site in their active form.
Can polymer carriers improve oral bioavailability of proteins or peptides?
Yes, polymer carriers are among the most promising approaches for enabling oral delivery of biomacromolecules. Functional polymers such as chitosan and its derivatives can transiently open tight junctions between intestinal epithelial cells, facilitating paracellular transport. Meanwhile, certain polymeric nanocarriers protect peptides from enzymatic degradation and exploit M-cell-mediated endocytosis to enter the lymphatic system. Although trans-epithelial transport efficiency remains a challenge, synergistic designs combining polymers with permeation enhancers or enzyme inhibitors have already led to significant improvements in the oral bioavailability of protein therapeutics.
How are polymers selected for oral drug delivery applications?
Polymer selection is a multidimensional decision-making process. First, the physicochemical properties of the API must be evaluated, with solubility parameters and Tg values calculated to assess polymer–drug compatibility and ensure physical stability. Second, regulatory requirements must be considered, prioritizing pharmaceutical excipients with established toxicological profiles and FDA/NMPA approvals. Finally, clinical needs—such as sustained or site-specific release—and industrial feasibility, including suitability for processes like hot-melt extrusion (HME), are integrated using Design of Experiments (DoE) approaches for screening and formulation optimization.
How do polymers protect drugs in the gastrointestinal tract?
Polymers form protective matrices or coatings that shield drugs from acidic gastric conditions and enzymatic degradation. pH-responsive and enteric polymers remain stable in the stomach and dissolve or swell in the intestine, enabling site-specific release. This protection helps preserve drug potency and improves consistency in oral drug absorption.
What types of polymers are used in oral drug delivery systems?
A wide range of polymers can be used, including synthetic polymers such as PEG, PVP, HPMC, and PLGA, as well as natural polymers like chitosan and alginate. The selection depends on drug properties, target release site, and formulation goals. Customization enables precise control over release behavior and stability.

 Fig. 1. Biological barriers in oral delivery: gastric acid, mucus, and first-pass metabolism (BOC Sciences Authorized).
Fig. 1. Biological barriers in oral delivery: gastric acid, mucus, and first-pass metabolism (BOC Sciences Authorized). Fig. 2. Structural diversity of polymer carriers: micelles, nanoparticles, pH-responsive, and mucoadhesive systems (BOC Sciences Authorized).
Fig. 2. Structural diversity of polymer carriers: micelles, nanoparticles, pH-responsive, and mucoadhesive systems (BOC Sciences Authorized).