Polymers for Small Molecule Drug Delivery: Formulation Strategies and Design Principles
In the field of modern drug development, despite the rapid rise of biologics, small molecule therapeutics continue to dominate the pharmaceutical market due to their well-defined chemical structures, mature synthetic processes, and ability to penetrate cell membranes to act on intracellular targets. However, with the advancement of high-throughput screening (HTS) and combinatorial chemistry, approximately 70%-90% of newly identified lead compounds fall into BCS Class II or IV, exhibiting low solubility or low permeability. Advances in polymer science offer revolutionary tools to address these pharmacological challenges. Functional polymers are no longer merely inert excipients; they have become key materials capable of precisely modulating drug release, improving pharmacokinetic (PK) profiles, and enabling targeted delivery.
Resources
Overcoming Bioavailability Challenges in Small Molecule Therapeutics
The clinical translation of small molecule drugs is often limited by their inherent physicochemical properties. Bioavailability refers to the extent and rate at which the active drug ingredient is released from a formulation and absorbed into systemic circulation, serving as a critical determinant of therapeutic efficacy. Many promising small molecule candidates are eliminated because they cannot cross physiological barriers to achieve effective therapeutic concentrations. The introduction of polymer carriers aims to modify the drug microenvironment at the molecular level, thereby overcoming intrinsic bioavailability barriers.
Solubility Limitations of Small Molecule Drugs
According to the Noyes-Whitney equation, the dissolution rate of a drug is proportional to its saturated solubility. Many hydrophobic small molecules exhibit extremely low solubility in the aqueous environment of the gastrointestinal (GI) tract, resulting in limited dissolution and insufficient concentration gradients to drive transmembrane transport. Excessive lattice energy caused by hydrophobic interactions is the primary thermodynamic reason. Polymers can significantly enhance apparent solubility by disrupting crystal structures, forming amorphous solid dispersions, or solubilizing drugs through micelle formation.
Chemical and Enzymatic Instability
Small molecule drugs face a complex biochemical environment in vivo. The highly acidic gastric environment (pH 1–3) can hydrolyze or degrade acid-sensitive drugs, while abundant enzymes in the intestine and bloodstream, such as esterases and peptidases, can rapidly metabolize drugs before they reach target tissues. In addition, oxidative reactions are a common cause of drug inactivation.
Poor Bioavailability and Absorption Issues
Beyond solubility, permeability is also a critical limiting factor. Many drugs are actively effluxed by transport proteins such as P-glycoprotein (P-gp) in intestinal epithelial cells. Moreover, hepatic first-pass metabolism can significantly reduce the amount of active drug entering systemic circulation. Drug molecular weight, lipophilicity (LogP), and polar surface area (PSA) must be carefully balanced within narrow ranges to achieve optimal absorption.
Need for Targeted and Controlled Delivery
Traditional formulations often lead to fluctuating plasma drug concentrations ("peaks and troughs"), which can trigger systemic toxicity or insufficient efficacy. For small molecules with narrow therapeutic windows, such as anticancer drugs, non-specific systemic distribution can result in severe off-target toxicity. Therefore, developing intelligent systems capable of maintaining steady-state plasma concentrations and precisely delivering drugs to diseased tissues is critical.
Functional Roles of Polymers in Small Molecule Drug Delivery
The role of polymers in drug delivery systems (DDS) has evolved from structural support to functional execution. Through precise chemical synthesis, polymers can be tailored in terms of molecular weight, hydrophilicity/hydrophobicity, charge, and degradation kinetics, enabling them to perform specific functions at each stage of drug delivery.
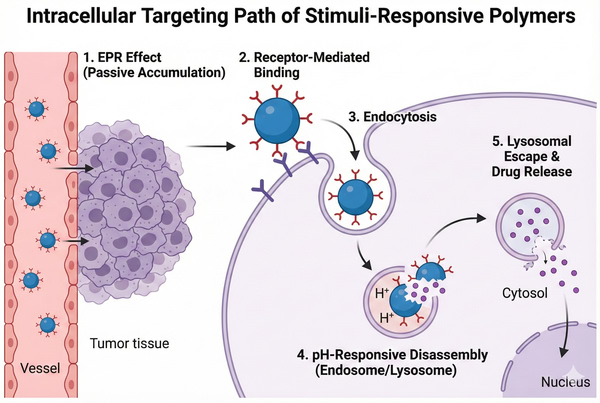 Fig. 1. Illustration of intracellular trafficking and pH-responsive drug release in tumor cells (BOC Sciences Authorized).
Fig. 1. Illustration of intracellular trafficking and pH-responsive drug release in tumor cells (BOC Sciences Authorized).
- Solubility Enhancement for Poorly Soluble Drugs: Amphiphilic polymers can self-assemble into micelles in aqueous solutions, with hydrophobic cores encapsulating poorly soluble drugs and hydrophilic shells ensuring colloidal stability. Hydrophilic polymers such as PVP and HPMC can act as anti-nucleating agents in solid dispersions, increasing the system's glass transition temperature (Tg) and forming intermolecular hydrogen bonds to inhibit drug recrystallization, maintaining a high-energy amorphous state.
- Controlled and Sustained Release of Therapeutics: Polymer matrices regulate drug release via diffusion-controlled and degradation-controlled mechanisms. In non-degradable systems, drugs diffuse through polymer mesh pores, while in biodegradable systems (e.g., PLGA), polymer backbone hydrolysis and drug release occur simultaneously. This approach can extend drug release from hours to months, significantly improving patient compliance.
- Protection Against Degradation and Metabolism: Polymer networks provide a physical barrier against water, protons, and enzymatic attack. For instance, enteric polymers (e.g., Eudragit® series) remain protonated and insoluble at low pH, protecting acid-sensitive drugs through the stomach and releasing them in the neutral pH of the intestine. PEGylation can create a steric hindrance layer, reducing opsonization and avoiding clearance by the reticuloendothelial system (RES).
- Targeted Delivery and Tissue-Specific Release: Through the enhanced permeability and retention (EPR) effect in solid tumors, nanoscale polymer carriers passively accumulate in tumor tissues. Further, conjugating specific ligands (e.g., folate, antibodies, RGD peptides) to polymer termini enables receptor-mediated active targeting, promoting specific endocytosis into target cells.
- pH-Responsive Release for Targeted Delivery: By exploiting pH differences between pathological tissues (e.g., tumor microenvironment pH ~6.5, lysosome pH ~5.0) and normal tissues, polymers containing ionizable groups (e.g., amino, carboxyl) or acid-sensitive bonds (e.g., hydrazone, acetal) undergo conformational changes or chemical bond cleavage under specific pH conditions, triggering burst release of the drug.
- Strategies to Extend Drug Half-Life: Small molecules are often rapidly cleared by the kidneys, resulting in short half-lives. Conjugating drugs to high-molecular-weight polymers (e.g., PEG, PAS, XTEN) can significantly increase hydrodynamic radius beyond the glomerular filtration threshold (~40–60 kDa), extending circulation half-life and enabling long-acting therapy.
Types of Polymers Used for Small Molecule Drug Delivery
Selecting the appropriate polymer carrier is central to DDS design. It depends not only on the physicochemical properties of the drug (e.g., lipophilicity, molecular weight, pKa) but also on the desired release kinetics and biodistribution for the therapeutic indication. Based on origin, degradation mechanism, and responsiveness, commonly used pharmaceutical polymers can be categorized as follows, each serving indispensable roles in specific formulation contexts.
Natural Polymers for Biocompatible Carriers
Natural polymers are favored in small molecule delivery for their excellent biocompatibility, biodegradability, and unique bioactive functions. They often act not only as inert carriers but also actively participate in physiological processes.
- Chitosan: The only naturally occurring cationic polysaccharide, derived by deacetylating chitin. Its amino groups (-NH₂) protonate in mildly acidic environments, conferring positive charges. This mucoadhesiveness allows electrostatic interaction with negatively charged mucosal proteins, prolonging drug retention in the nasal cavity or intestine. Chitosan can also transiently open epithelial tight junctions, significantly enhancing paracellular transport of hydrophilic small molecules.
- Alginate: An anionic polysaccharide extracted from brown algae, composed of guluronic (G) and mannuronic (M) acid units that crosslink with divalent cations (e.g., Ca²⁺) via ionic gelation to form "egg-box" hydrogels. This property is ideal for pH-sensitive oral microspheres, protecting drugs in gastric acid and swelling to release them in the alkaline intestine.
- Hyaluronic Acid (HA): A major extracellular matrix component, HA is highly hydrophilic and minimally immunogenic. Importantly, it is a natural ligand for the CD44 receptor, overexpressed on many solid tumors. HA-modified nanocarriers enable active tumor targeting, delivering chemotherapeutic drugs (e.g., paclitaxel, doxorubicin) via receptor-mediated endocytosis.
Synthetic Polymers with Tunable Properties
Compared to natural polymers, synthetic polymers offer higher batch-to-batch consistency and allow precise design of chemical structure, molecular weight distribution (PDI), and topology.
- Poly(vinyl pyrrolidone) (PVP): Highly hygroscopic and amphiphilic, PVP serves as an amorphous carrier in solid dispersions, forming hydrogen bonds to inhibit drug nucleation and maintain high-energy amorphous states, significantly enhancing dissolution rates. Different K-values (molecular weights) of PVP can be used to modulate tablet disintegration and drug release.
- Poly(vinyl alcohol) (PVA): Commonly used as an emulsifier/stabilizer in PLGA microsphere preparation via solvent evaporation. Its partially hydrolyzed acetate groups provide excellent interfacial activity, preventing nanoparticle aggregation during formulation and storage.
- Dendrimers: Such as polyamidoamine (PAMAM), these highly branched, monodisperse spherical polymers possess abundant surface functional groups (e.g., amino, hydroxyl) for high-density drug conjugation or surface modification, with internal cavities capable of physically encapsulating small molecules. Generation determines size and surface charge density, making dendrimers ideal for precision delivery.
Aliphatic polyesters are the most widely FDA-approved biodegradable implant materials, degrading via hydrolysis of ester bonds into non-toxic acidic metabolites, subsequently entering the tricarboxylic acid (TCA) cycle.
- PLGA (Poly(lactic-co-glycolic acid)): The "gold standard" in drug delivery, PLGA degradation rate and release kinetics can be precisely controlled by adjusting the lactide:glycolide (L:G) ratio and molecular weight. A 50:50 ratio degrades fastest (~1–2 months), whereas lactide-rich PLGA degrades more slowly due to increased hydrophobicity.
- PLA (Polylactic acid): With an additional methyl side chain, PLA is more hydrophobic than PGA and exists as optical isomers (PLLA, PDLA). Semi-crystalline PLLA degrades very slowly (up to several years) and possesses high mechanical strength, suitable for orthopedic fixation or long-term drug-eluting scaffold coatings.
- PCL (Polycaprolactone): With a low melting point (~60°C) and excellent rheology, PCL is compatible with hot-melt extrusion. Its high permeability allows small molecules to diffuse readily. Extremely slow degradation (>2 years) makes it suitable for long-acting implants (e.g., Implanon) or sustained-release drug reservoirs.
- Polyanhydrides: Unlike bulk erosion of PLGA, polyanhydrides undergo surface erosion, where only the polymer surface degrades over time while geometry decreases layer by layer. This allows near-zero-order release, ideal for drugs with strict release rate requirements (e.g., Gliadel Wafer).
PEGylated and Surface-Modified Polymers
A major obstacle for nanocarriers in vivo is clearance by the reticuloendothelial system (RES). Surface modification techniques aim to alter carrier interface properties to prolong circulation half-life.
- PEGylation: Attaching PEG chains to the carrier surface is the most established strategy for imparting "stealth" properties. The highly hydrated PEG layer provides substantial steric hindrance, preventing adsorption of opsonins (e.g., IgG, C3b) and thereby evading immune phagocytosis.
- Zwitterionic Polymers: As PEG alternatives, polymers such as poly(carboxybetaine) (PCB) or poly(phosphorylcholine) (PMPC) form stronger hydration layers through ionic solvation, offering ultra-low fouling characteristics and typically avoiding the accelerated blood clearance (ABC) phenomenon, making them a research focus for next-generation long-circulating carriers.
Stimuli-Responsive Polymers for Site-Specific Delivery
These smart polymers can sense subtle physiological or pathological changes and undergo dramatic physicochemical transformations, triggering site-specific drug release.
- pH-Responsive: Drugs conjugated to polymers via hydrazone or acetal bonds remain stable at physiological pH but rapidly hydrolyze in acidic lysosomes (pH 4.5–5.0) within tumor cells. Additionally, polymers like polyhistidine or sulfonamide derivatives protonate in the slightly acidic tumor microenvironment (pH ~6.5), causing solubility changes or a "proton sponge effect," facilitating lysosomal escape.
- Temperature-Responsive: Polymers such as poly(N-isopropylacrylamide) (PNIPAM) exhibit a lower critical solution temperature (LCST) around 32 °C. By copolymerization, the LCST can be adjusted slightly above body temperature (e.g., 40–42 °C), allowing local hyperthermia to induce phase transition and contraction at the tumor site, "squeezing out" the drug for targeted release.
- Redox-Responsive: Intracellular glutathione (GSH) concentrations (~10 mM) are much higher than extracellular (~10 μM). Disulfide-containing polymers can be reduced upon cellular entry, causing carrier breakdown and intracellular drug release.
- Enzyme-Responsive: Tumor tissues often overexpress matrix metalloproteinases (MMPs) or cathepsins. By incorporating specific peptide sequences in the polymer backbone or side chains, enzymatic cleavage at the tumor site triggers polymer degradation or ligand exposure, releasing the encapsulated drug.
Looking for Polymers for Small Molecule Drug Delivery?
Whether you need cationic polymers, biodegradable carriers, or stimuli-responsive platforms, our experts provide tailored synthesis and functionalization solutions to support your research and therapeutic development.
Key Formulation Strategies Utilizing Functional Polymers
Transforming small molecule APIs into clinically effective therapeutics relies on precisely designed formulation strategies. Functional polymers act as bridges between drug molecules and physiological environments, fundamentally altering solubility, biodistribution, and metabolic kinetics through physical encapsulation, chemical conjugation, or molecular dispersion. The following are core polymer-based formulation strategies widely applied in both industry and academia:
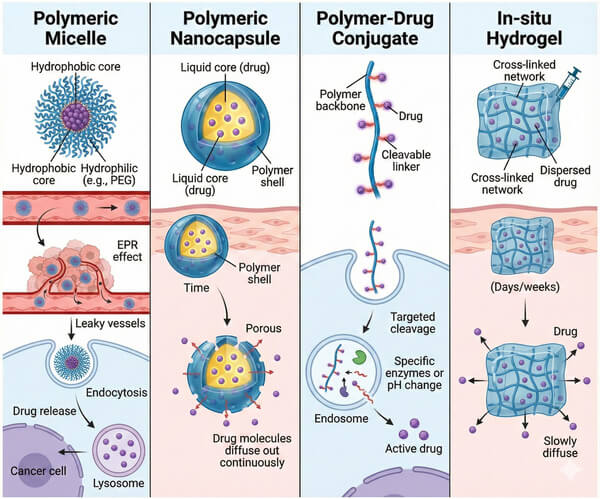 Fig. 2. Schematic comparison of polymeric micelles, nanocapsules, conjugates, and hydrogel drug carriers (BOC Sciences Authorized).
Fig. 2. Schematic comparison of polymeric micelles, nanocapsules, conjugates, and hydrogel drug carriers (BOC Sciences Authorized).
Polymer-Drug Conjugates for Sustained Release
Based on the classic Ringsdorf model, polymer-drug conjugates (SMDCs) represent a precise strategy to optimize drug pharmacokinetics via covalent modification. In these systems, drugs are not physically entrapped but attached to a water-soluble polymer backbone through a specific chemical linker, forming a macromolecular prodrug. Linker design is critical—for example, pH-sensitive linkers using hydrazone or cis-aconitic anhydride remain stable in circulation but rapidly cleave in acidic endosomes or lysosomes, releasing the parent drug. This approach significantly prolongs half-life and alters cellular uptake pathways, bypassing P-glycoprotein-mediated efflux to overcome multidrug resistance (MDR), though limited drug loading remains a focus of polymer chemistry research.
Nanoparticle-Based Drug Delivery Systems
Polymeric nanoparticles are submicron colloidal carriers, typically categorized as nanospheres or nanocapsules, each with distinct release kinetics. Nanospheres are solid matrices with drugs uniformly dispersed, releasing drugs via combined Fickian diffusion and matrix erosion, often prepared via nanoprecipitation for hydrophobic drugs. Nanocapsules feature a core-shell architecture, where a liquid core is encapsulated by a polymer shell, forming a reservoir system that improves drug loading efficiency and enables near-zero-order release when the shell remains intact. Techniques such as double emulsion solvent evaporation allow precise control of particle size distribution, influencing biodistribution and cellular uptake.
Polymeric Micelles for Hydrophobic Drugs
For BCS Class II and IV poorly soluble drugs, polymeric micelles provide a highly effective solubilization approach based on amphiphilic block copolymer self-assembly (e.g., PEG-b-PLA). In aqueous media, these copolymers spontaneously form core-shell structures, with the hydrophobic core acting as a micro-reservoir to enhance drug solubility hundreds-fold, while the hydrophilic PEG shell provides a hydrated layer that prevents aggregation and evades RES clearance. Compared to low-molecular-weight surfactants, polymeric micelles exhibit extremely low critical micelle concentrations (CMC), maintaining structural integrity even upon dilution in circulation, thereby minimizing premature drug leakage. Clinical success of Genexol-PM® exemplifies this strategy.
Hydrogel Systems for Local and Sustained Release
Polymeric hydrogels are 3D networks of physically or chemically crosslinked polymers. In situ gelling systems are attractive for minimally invasive delivery. These smart polymer solutions remain low-viscosity liquids at room temperature for injection and undergo sol-gel transition in response to stimuli such as body temperature (e.g., ReGel® system) or shear stress, forming a semi-solid drug depot at the administration site. Drug molecules then slowly diffuse through the polymer network pores, and release rates can be precisely tuned by controlling mesh size, enabling sustained therapeutic concentrations for several weeks.
Polymeric Solid Dispersions for Solubility Enhancement
A mature industrial approach to improving small molecule solubility, polymeric solid dispersions use hot-melt extrusion (HME) or spray drying to molecularly disperse and immobilize drugs within a polymer carrier, forming high-energy amorphous states. Solubility enhancement relies on generating and maintaining supersaturation: high-energy amorphous drugs dissolve rapidly in bodily fluids, while polymers like HPMC-AS or PVP act as precipitation inhibitors, adsorbing to drug surfaces via hydrogen bonding to prevent nucleation and crystal growth, maintaining supersaturation long enough for transmembrane absorption, thereby converting thermodynamic instability into kinetic stability and greatly improving bioavailability.
Polymer Coatings and Matrix Systems for Oral and Injectable Delivery
This foundational technology supports the transition from conventional to advanced formulations, encompassing oral film coatings to long-acting injectable microspheres. Oral formulations employ acrylic resins (e.g., Eudragit®) for pH-dependent coatings to protect acid-sensitive drugs or enable colon-targeted delivery. In long-acting injectables (LAIs), PLGA microsphere matrices dominate; by controlling porosity, polymer degradation rate, and drug distribution, formulators balance diffusion and erosion to minimize burst release and achieve steady drug release over weeks to months, improving adherence in therapies for schizophrenia or hormonal treatment.
Applications of Polymer-Based Delivery Systems
Polymer technologies have penetrated multiple clinical applications, significantly improving patient outcomes:
Oral Delivery of Poorly Soluble Drugs
For BCS Class II and IV drugs, amorphous solid dispersions (ASDs) prepared via HME or spray drying are the industrial standard to enhance oral bioavailability. Polymers inhibit nucleation, significantly increasing apparent solubility and dissolution in GI fluids while maintaining drugs in a high-energy supersaturated state to drive transmembrane absorption. This approach also mitigates food effect variability, ensuring consistent plasma concentrations and reproducible therapeutic outcomes.
Injectable Formulations with Improved Pharmacokinetics
Polymeric micelles and PEGylation in intravenous formulations not only act as solubilizers but also fundamentally alter hydrophobic drug PK profiles. By replacing toxic surfactants (e.g., Cremophor EL), biocompatible polymer carriers reduce hemolysis and hypersensitivity risks, allowing higher maximum tolerated doses (MTD). Nanoscale carriers evade RES clearance through steric hindrance, extend circulation half-life, and exploit the EPR effect for passive tumor accumulation.
Topical and Transdermal Polymer-Based Systems
Pressure-sensitive adhesive polymer matrices provide physical adhesion and regulate drug penetration through the stratum corneum via thermodynamic activity gradients, achieving sustained systemic delivery. Emerging dissolvable polymer microneedle arrays rapidly release encapsulated drugs upon skin insertion, overcoming the stratum corneum barrier while eliminating injection pain. This non-invasive delivery is ideal for chronic self-administration or vaccination.
Pulmonary and Mucosal Drug Delivery
For pulmonary delivery, polymer microspheres with optimal aerodynamic diameters (1–5 μm) bypass upper airway deposition and reach deep alveolar regions, enabling local high-concentration therapy or rapid systemic absorption. Thiolated polymers (thiomers) in nasal or ocular delivery form covalent disulfide bonds with mucins, enhancing mucoadhesion, resisting mucociliary clearance, prolonging residence time, and improving hydrophilic small molecule permeability.
Polymer-Based Implants and Depot Systems
Biodegradable implants and in situ gels based on PLGA or PCL provide sustained drug release over weeks to months via polymer backbone hydrolysis. This approach achieves near-zero-order release kinetics, eliminating plasma "peak-trough" fluctuations and improving patient compliance. It has been successfully applied in long-acting therapies for prostate cancer (e.g., leuprolide microspheres) and schizophrenia, significantly improving clinical outcomes.
Design Considerations for Polymer-Based Formulations
In the development of polymeric drug delivery systems, adhering to the Quality by Design (QbD) principle is critical to ensure successful clinical translation. This requires not only a thorough understanding of the physicochemical properties of polymeric materials but also precise control over the thermodynamic and kinetic interactions between the drug molecule and the polymer matrix. The following are core factors formulation scientists must consider during the design phase.
Selection of Polymer Type and Molecular Weight
The chemical structure and molecular weight of a polymer are fundamental parameters that determine the performance of a delivery system, directly influencing degradation rate, mechanical strength, and drug release behavior.
- Matching Degradation Kinetics: The polymer degradation timeline must align with the intended therapeutic window. For microspheres requiring multi-month release, high molecular weight polymers (Mw > 50 kDa) or hydrophobic-rich monomers (e.g., high-lactide PLGA) are preferred. For fast-acting carriers, lower molecular weight or more hydrophilic polymers (e.g., PEG-PLA) are more suitable.
- Polydispersity Index (PDI): Narrow PDI ensures consistent degradation behavior across batches.
- Crystallinity and Tg: Semi-crystalline polymers (e.g., PLLA) generally degrade slower than amorphous polymers (e.g., PDLLA) due to limited water penetration into crystalline domains. Additionally, the polymer's glass transition temperature (Tg) should exceed body temperature (37 °C) to maintain structural integrity in vivo and prevent premature drug diffusion.
Optimization of Drug Loading Efficiency
High drug loading is key to reducing excipient load, minimizing administration volume, and controlling production costs, but it is often limited by drug-polymer compatibility.
- Thermodynamic Compatibility: Hansen Solubility Parameters (δ) or Flory-Huggins interaction parameters can predict drug-polymer miscibility. When the δ values of the drug and polymer are similar, the Gibbs free energy of the mixture is lower, allowing higher drug concentrations without phase separation.
- Impact of Preparation Methods: During nanoprecipitation or emulsification, selecting appropriate organic solvents and surfactants, and adjusting the oil-to-water ratio, can maximize the drug's partitioning into the polymer phase, thereby improving encapsulation efficiency.
Controlling Release Kinetics
An ideal formulation provides predictable, physiologically appropriate release profiles, eliminating uncontrolled fluctuations.
- Suppressing Initial Burst Release: Early-stage drug release often arises from drug adsorbed on the carrier surface. Optimizing surface washing, increasing polymer matrix density, or using core-shell designs can limit burst release to safe levels.
- Regulating Release Mechanisms:
- Diffusion-Controlled: Suitable for non-degradable or slow-degrading systems, where release follows Fickian diffusion, influenced by polymer mesh size and the drug's hydrodynamic radius.
- Erosion-Controlled: Applicable to biodegradable systems, where drug release occurs concurrently with polymer degradation (bulk or surface erosion).
- Mathematical Modeling: Fitting release data with zero-order, Higuchi, or Korsmeyer-Peppas models helps infer microscopic release mechanisms and guide formulation optimization.
Balancing Biocompatibility and Stability
Safety and stability are decisive factors for market approval.
- Microenvironment Control of Degradation Products: Polyester polymers such as PLGA degrade into acidic monomers, causing a sharp internal pH drop (as low as pH 2–3), which can inactivate acid-sensitive drugs or trigger local inflammation. Incorporating basic additives (e.g., Mg(OH)₂, ZnCO₃) as pH buffers is a common mitigation strategy.
- Physical Aging: Polymers undergo structural relaxation during storage, reducing free volume and altering drug release rates.
- Sterilization Compatibility: Gamma or electron-beam sterilization may cause polymer chain scission (reduced molecular weight) or crosslinking, affecting final formulation performance. For sensitive systems, aseptic processing is often the only viable option.
Services
Custom Polymer Solutions for Small Molecule Drug Delivery
BOC Sciences specializes in tailored polymer delivery solutions for small molecule therapeutics, aiming to improve solubility, bioavailability, and targeting. Our services cover polymer design, functional modification, delivery system development, and characterization optimization, supporting rapid translation from R&D to clinical and commercial applications.

Polymer Screening, Design, and Synthesis for Small Molecules
- Screening polymeric materials suitable for small molecule drugs
- Customizing polymer molecular weight, hydrophobic/hydrophilic balance, and degradation rate
- Optimizing polymer structure to enhance drug solubility and stability
- Supporting linear, block, or branched polymer design and synthesis

Polymer Modification and Functionalization for Targeted Delivery
- Surface modification to enhance tissue or cellular targeting
- Incorporation of pH-, temperature-, or enzyme-responsive units for controlled release
- PEGylation or hydrophobic/hydrophilic tuning to improve solubility and stability
- Support for conjugation with small molecule drugs, targeting ligands, or permeation enhancers

Polymer–Drug Compatibility and Formulation Support
- Polymer–drug compatibility analysis and complex stability evaluation
- Assessment of drug loading, release characteristics, and stability
- Characterization of particle size, distribution, and surface properties
- Optimization of formulation conditions (pH, solvent, excipients)

Polymeric Small Molecule Drug Delivery System Development
- Construction of polymer–drug nanoparticles, microparticles, and hybrid systems
- Suitable for oral, intravenous, or local delivery of small molecules
- Optimization of delivery efficiency, release profiles, and targeting
- In vitro and in vivo performance evaluation (solubility, stability, bioavailability)
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
What are polymer-based small molecule drug delivery systems?
Polymer-based delivery systems use natural or synthetic polymers to encapsulate or conjugate small molecule drugs. They improve drug solubility, stability, and bioavailability, while enabling controlled release. These systems protect drugs from premature degradation, enhance therapeutic efficacy, and can be tailored for targeted delivery, reducing side effects and improving patient compliance.
What are polymer solutions used for in small molecule delivery?
Polymer solutions serve as multifunctional carriers in small molecule drug delivery. They address key biopharmaceutical challenges, including enhancing solubility and bioavailability of poorly soluble drugs, prolonging circulation half-life via steric hindrance, and enabling targeted delivery to specific tissues or cells. Additionally, polymers act as physical barriers to protect labile drugs from enzymatic or chemical degradation in vivo.
How do polymers enhance solubility and stability of small molecules?
Polymers enhance solubility via two main mechanisms: amphiphilic block copolymers self-assemble into micelles, encapsulating hydrophobic drugs in their cores; and amorphous solid dispersion techniques restrict molecular mobility to prevent drug crystallization, maintaining high-energy soluble states. For stability, the polymer matrix isolates drugs from water and oxidants and buffers microenvironment pH, preventing hydrolytic or oxidative degradation.
Can polymer carriers provide controlled release for small molecule drugs?
Yes, controlled release is a core advantage of polymer carriers. By tuning monomer ratios, molecular weight, and crystallinity of biodegradable polymers (e.g., PLGA), or leveraging diffusion limitations in non-degradable polymers, scientists can precisely program drug release kinetics. This enables release over hours to months, eliminates plasma peak-trough fluctuations, and supports zero-order or pulsatile delivery, significantly improving efficacy and patient compliance.
How are polymers selected for specific small molecule therapeutics?
Polymer selection is guided by the drug's physicochemical properties (e.g., LogP, molecular weight) and thermodynamic compatibility, which determines loading efficiency and phase stability. Expected clinical release profiles dictate polymer degradation kinetics and molecular weight—for example, high-molecular-weight PLGA for long-acting release. The administration route is also critical: injectable formulations require biodegradable materials, whereas oral formulations may benefit from pH-responsive or mucoadhesive polymers.
Are polymer-based delivery systems safe?
Most polymers used for drug delivery are biocompatible and biodegradable, breaking down into non-toxic metabolites. Regulatory-approved polymers like PLGA, PEG, and chitosan have established safety profiles. Proper formulation and dosing ensure minimal toxicity, while the polymer matrix can reduce drug-related side effects by enabling controlled or targeted release.
Which polymers are commonly used for small molecule drugs?
Commonly used polymers include biodegradable options like PLGA, PLA, PCL, and natural polymers like chitosan or alginate. PEG is widely used for solubility enhancement and stealth properties. Selection depends on drug properties, release profile, administration route, and desired biocompatibility, allowing highly customizable delivery solutions.

 Fig. 1. Illustration of intracellular trafficking and pH-responsive drug release in tumor cells (BOC Sciences Authorized).
Fig. 1. Illustration of intracellular trafficking and pH-responsive drug release in tumor cells (BOC Sciences Authorized). Fig. 2. Schematic comparison of polymeric micelles, nanocapsules, conjugates, and hydrogel drug carriers (BOC Sciences Authorized).
Fig. 2. Schematic comparison of polymeric micelles, nanocapsules, conjugates, and hydrogel drug carriers (BOC Sciences Authorized).