Polymer–Drug Conjugates for Drug Delivery: Design Strategies, Materials, and Applications
Polymer–drug conjugates (PDCs) have emerged as an important strategy for improving the delivery of therapeutic molecules, particularly compounds with poor solubility or limited stability in aqueous environments. In these systems, drug molecules are covalently attached to polymer carriers, allowing the physicochemical properties of the polymer to enhance the overall performance of the drug formulation. The polymer component can improve dispersion in aqueous media, protect the drug from premature degradation, and influence release behavior through carefully designed linker chemistry. The effectiveness of polymer–drug conjugates largely depends on rational molecular design. Factors such as polymer type, molecular weight, linker structure, and conjugation strategy play critical roles in determining drug loading capacity, stability, and release kinetics. With advances in polymer synthesis and bioconjugation technologies, a wide variety of polymer backbones and functional materials can now be engineered to construct highly tailored conjugate systems for modern drug delivery research.
Resources
What Are Polymer–Drug Conjugates?
Polymer–drug conjugates are covalent assemblies in which drug molecules are tethered to a polymeric carrier via stable or cleavable chemical linkers, sometimes augmented with targeting moieties or solubilizing segments. This modular format allows developers to tune physicochemical properties (hydrophilicity, size, charge, conformation) and to program drug liberation through specific bond-cleavage mechanisms rather than relying solely on passive diffusion of a free drug.
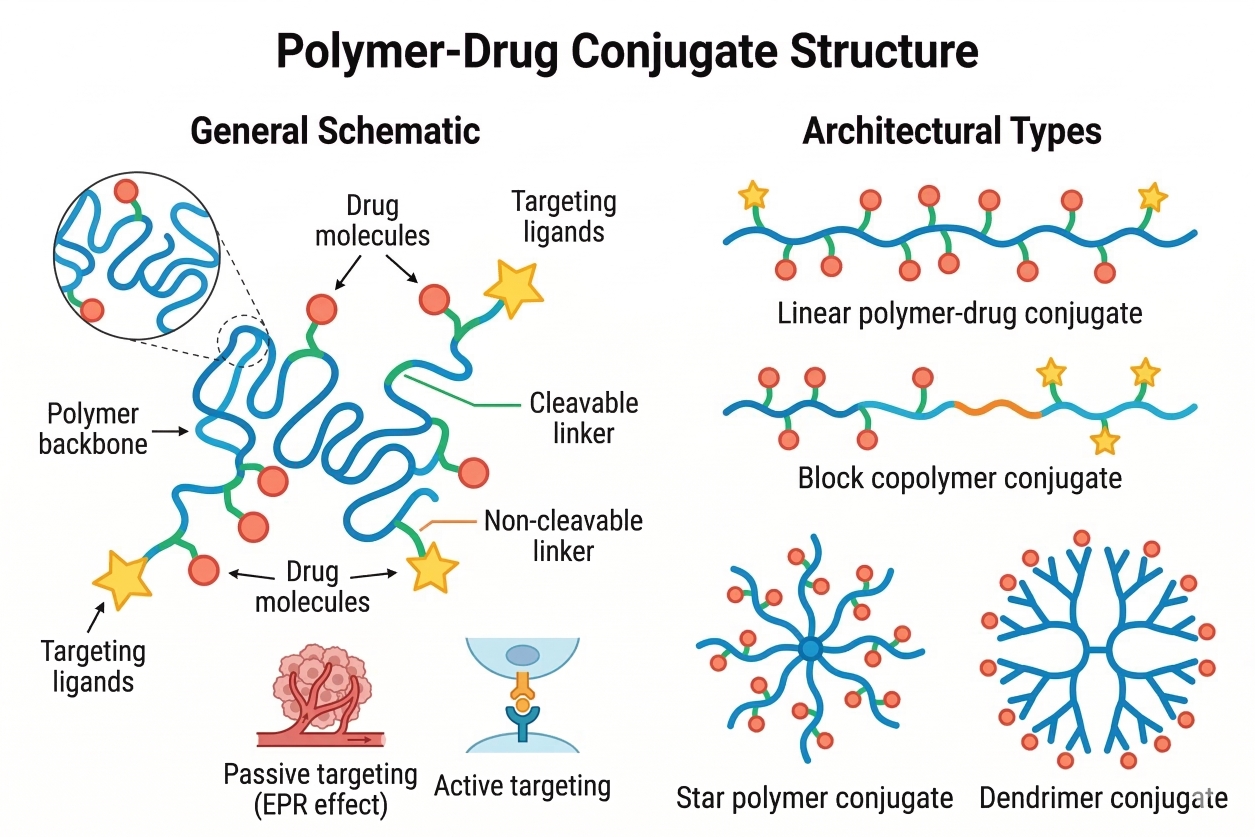 Fig. 1. Schematic overview of polymer-drug conjugate components and architectural types (BOC Sciences Authorized).
Fig. 1. Schematic overview of polymer-drug conjugate components and architectural types (BOC Sciences Authorized).
A PDC is best understood as a system with four essential elements and a fifth optional one. The polymer carrier defines size, solubility, flexibility, and degradability. The drug payload contributes potency and chemical functionality but is also a structural modifier because loading level can drive self-assembly or aggregation. The linker is the control point for stability versus release, dictating whether the conjugate behaves like a long-lived prodrug or a rapidly shedding carrier. Functional handles (reactive groups, chain ends, orthogonal tags) enable synthesis, purification, and analytics. Many PDCs further incorporate targeting moieties such as peptides, sugars, antibodies, or small-molecule ligands to bias uptake or localization.
Advantages of Polymer–Drug Conjugates over Conventional Drugs
Polymer–drug conjugates offer several structural and functional advantages compared with conventional small-molecule drug formulations. Through covalent attachment of therapeutic molecules to polymer backbones, these systems enable improved physicochemical properties, controlled delivery behavior, and tunable pharmacological profiles. Key advantages include:
- Improved aqueous solubility of poorly soluble drugs: Many bioactive compounds exhibit limited solubility in physiological environments. Conjugation with hydrophilic polymers significantly enhances water compatibility, facilitating formulation development and improving dispersion in biological systems.
- Enhanced stability of sensitive drug molecules: Covalent attachment to a polymer backbone can protect drugs from premature chemical degradation, hydrolysis, or enzymatic metabolism. This protective effect helps maintain structural integrity during circulation and storage.
- Extended systemic circulation time: Polymer conjugation increases the hydrodynamic size of drug molecules, reducing rapid renal filtration and slowing systemic clearance. As a result, the drug remains available in circulation for longer durations.
- Controlled and tunable drug release profiles: Incorporation of specific linker chemistries allows the release rate of the active molecule to be carefully engineered. Hydrolytic, enzymatic, or environmental triggers can regulate when and where the drug is released.
- Reduced off-target distribution: The macromolecular nature of polymer–drug conjugates can influence biodistribution patterns, reducing nonspecific diffusion into unintended tissues and improving delivery selectivity.
- Higher drug loading capacity: Polymer architectures containing multiple functional groups allow attachment of multiple drug molecules per polymer chain, increasing payload density and improving delivery efficiency.
- Modular structural design: Polymer backbones, molecular weight, architecture, and functional groups can be tailored to optimize physicochemical properties, stability, and release behavior according to the specific drug and delivery objective.
- Potential for multifunctional systems: Polymer carriers can incorporate additional functionalities such as targeting ligands, imaging moieties, or responsive groups, enabling multifunctional therapeutic platforms.
Types of Polymers Used in Polymer–Drug Conjugates
Polymer selection is not a cosmetic choice—it is the dominant design lever that determines degradability, conjugation chemistry options, stealth behavior, interaction with proteins, and ultimately how reproducibly a conjugate can be synthesized and characterized. In practice, polymer families are chosen to balance synthetic control, functionalizability, biological tolerance, and manufacturability, with the polymer architecture (linear, branched, star, brush, block copolymer) often mattering as much as monomer identity.
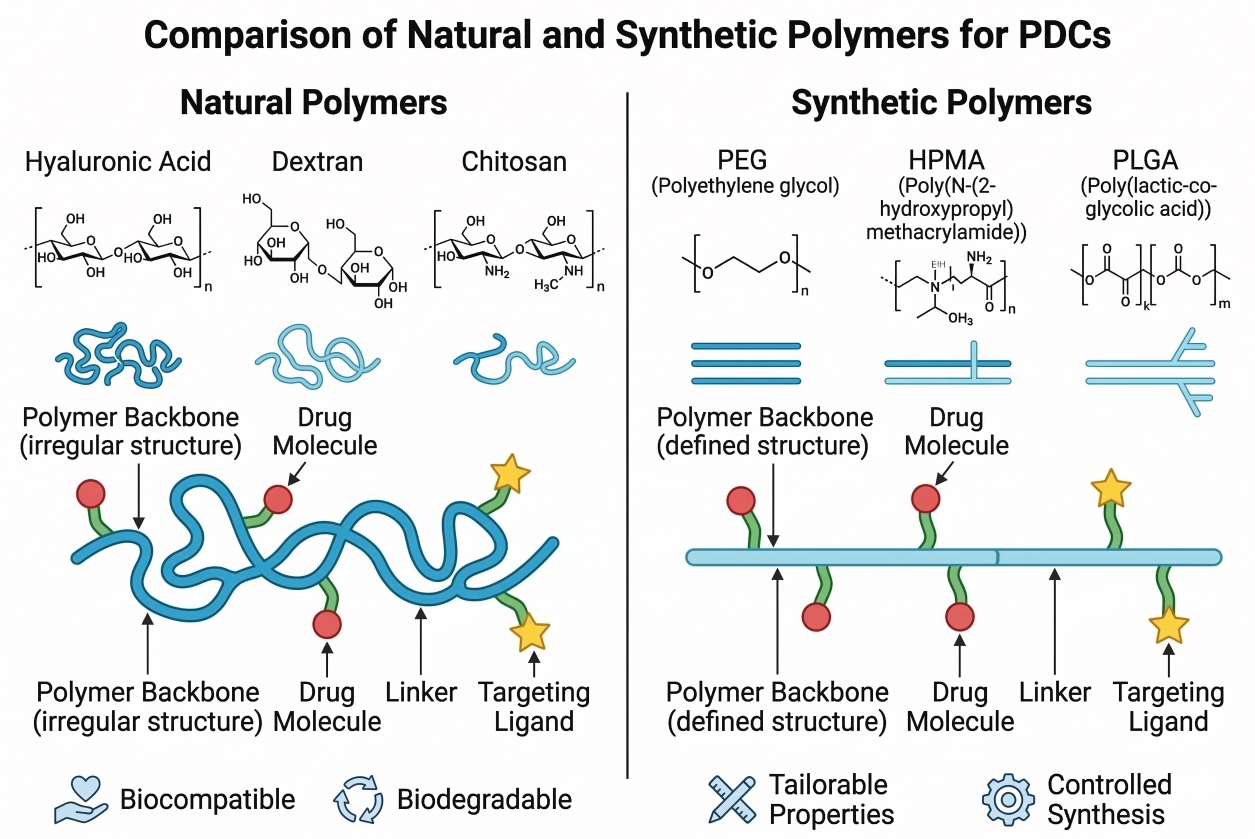 Fig. 2. Comparison of natural and synthetic polymers used in PDCs (BOC Sciences Authorized).
Fig. 2. Comparison of natural and synthetic polymers used in PDCs (BOC Sciences Authorized).
Synthetic Polymers in Drug Conjugation
Synthetic polymers are widely used because their monomer feed, topology, and functional group placement can be controlled with comparatively high precision. Classic water-soluble backbones include PEG and HPMA-based copolymers, which have a long history in polymer therapeutics research and can be prepared with reactive handles for drug attachment. Poly(2-oxazoline)s represent another versatile class built by cationic ring-opening polymerization, offering tunable side chains and end-group functionalization that support both conjugation and self-assembly approaches. Across these families, the key advantage is reproducible structure-property tuning: chain length distribution, pendant functional density, hydrophilic–hydrophobic balance, and targeting-ligand presentation can be engineered rather than inherited from natural variability.
Natural and Biopolymer-Based Drug Carriers
Natural polymers and biopolymers are attractive when developers want inherent biodegradability, biological recognition motifs, or abundant functional groups for derivatization. Dextran is a prominent example: it can be chemically modified into multiple derivatives and formulated into diverse delivery systems with tunable size and surface charge. Hyaluronic acid is valued for biocompatibility and rich derivatization chemistry through hydroxyl and carboxyl groups, enabling broad functionalization strategies. Chitosan offers cationic character and accessible amino groups that support conjugation of antimicrobial or other actives. Poly-γ-glutamic acid provides a biodegradable poly(amino acid) platform available in different isoforms, also supporting conjugation and delivery design. The tradeoff across many biopolymers is batch-to-batch variability and a potentially narrower processing window, making robust analytics and specification setting particularly important.
Biodegradable Polymer–Drug Conjugates
Biodegradable PDCs aim to avoid indefinite persistence of high-molecular-weight materials by embedding cleavable bonds either in the backbone, in side chains, or in the drug-linker junction. Common strategies include hydrolysable polyesters, enzyme-degradable peptide linkers, and bioresponsive polycarbonates or polypeptide-derived systems. A particularly chemistry-forward direction is the development of polyprodrugs, where drug-bearing monomers are polymerized so that degradation or bond cleavage releases active molecules over time; this approach emphasizes monomer design and degradable linkages as primary control parameters. Biodegradability, however, must be engineered with realism: faster degradation can improve clearance but may compromise storage stability or cause premature release if the cleavage chemistry is overly labile under manufacturing or shelf conditions.
PEGylated and Surface-Functionalized Conjugates
PEGylation remains a foundational strategy for increasing hydrophilicity, reducing nonspecific interactions, and improving circulation behavior of macromolecular constructs. PEG-based polymer–drug conjugates are extensively studied with established design rules for linker selection, conjugation methods, and characterization. At the same time, anti-PEG immune responses and accelerated blood clearance phenomena have been increasingly discussed in the literature, prompting more careful risk evaluation and, in some programs, consideration of alternative stealth polymers or mixed-corona approaches. From a design perspective, the key lesson is not that PEG is unsuitable, but that surface chemistry should be treated as a variable requiring evidence-based selection, especially when repeat exposure or high systemic presentation is expected in the intended use scenario.
Hydrophilic Polymers for Improved Pharmacokinetics
Hydrophilic polymers are frequently used to increase hydrodynamic size and reduce rapid filtration while also resisting protein adsorption that can accelerate clearance. Beyond PEG, several alternative hydrophilic and stealth-like materials are actively explored. Poly(2-oxazoline)s have been positioned as flexible, modular platforms with strong potential for bioconjugation and delivery applications. Polysarcosine has also been reviewed as a PEG alternative for modifying biotherapeutics and nanocarriers. Zwitterionic polymers, including polybetaines such as carboxybetaine-based materials, are widely studied for antifouling behavior and long-circulation potential because strongly hydrated surfaces can reduce nonspecific protein adsorption. The practical implication for PDC design is that stealth behavior is not a single-material property; it is an emergent result of hydration, chain mobility, charge distribution, and the specific protein environment encountered in vivo.
Stimuli-Responsive Polymers for Smart Drug Delivery
Stimuli-responsive PDCs incorporate chemical triggers so that drug release is accelerated or activated under defined conditions such as acidic microenvironments, reductive conditions, elevated reactive oxygen species, specific enzyme activity, temperature shifts, or externally applied stimuli. In practice, this responsiveness is most often implemented at the linker level (pH-labile, redox-labile, enzyme-cleavable bonds) but can also be embedded in the polymer backbone by using self-immolative or degradable segments that amplify release once triggered. A critical design requirement is kinetic alignment: the cleavage rate must be slow enough to prevent premature release during storage and circulation, yet fast enough to achieve meaningful release at the intended site and timescale.
Looking for Custom Polymers for Conjugate Development?
Whether you need functional polymers, linker-enabled backbones, or tailored conjugation strategies, our experts provide customized synthesis and modification solutions to support advanced drug delivery research.
Fundamental Design Principles of Polymer–Drug Conjugates
A high-performing PDC is rarely achieved by optimizing only one variable. Polymer identity, architecture, linker chemistry, and drug loading interact nonlinearly to determine solubility, aggregation, stability, and release. As a result, PDC design is best treated as a structured engineering problem with explicit design targets (solubility, size range, release half-life, storage stability) and a characterization plan capable of confirming that the intended molecular structure has actually been built.
Drug Selection and Conjugation Considerations
Not every drug is a good candidate for polymer conjugation. The payload should have a functional group that enables stable attachment (or a feasible derivatization route) without destroying core activity once released. Drugs with poor aqueous solubility, rapid degradation, or problematic biodistribution are common motivations for exploring conjugation, but the chemistry must be honest about tradeoffs: attaching a bulky polymer often reduces immediate potency because the free drug is masked until cleavage occurs. In addition, the drug can change the polymer's behavior: hydrophobic drugs at higher loading can drive self-assembly into nanoparticles or cause aggregation, shifting the delivery mechanism from single-chain macromolecules to supramolecular structures.
Choice of Polymer Backbone and Architecture
Backbone chemistry and architecture determine how a conjugate occupies space in solution and how it interacts with biological barriers. Linear polymers can be simpler to characterize and may show predictable size scaling with molecular weight. Branched, multi-arm, or brush architectures can reach larger hydrodynamic sizes at lower contour length, potentially improving exposure without excessively increasing viscosity or synthesis complexity. Block copolymers can form micelles or polymersomes, enabling a hybrid strategy where some drug is covalently bound while additional cargo can be physically encapsulated. The expanding diversity of polymer–drug conjugate materials—spanning water-soluble macromolecules, nanoparticles, micelles, polymersomes, and hydrogels—highlights how architecture is used to tailor both delivery route and release profile.
Linker Chemistry and Cleavable Bond Design
Linker design is the primary determinant of when and where free drug is regenerated. Broadly, linkers fall into stable bonds (intended to keep the drug attached for prolonged periods) and cleavable bonds (engineered to break under defined triggers). Common cleavable motifs include acid-labile hydrazones, acetals/ketals, enzyme-cleavable peptides, redox-labile disulfides, and hydrolysable esters or carbonates. Rational linker selection should be guided by measurable hydrolysis or cleavage kinetics and by compatibility with polymer synthesis, purification, and storage. Comparative studies of acid-cleavable linkages, for example, emphasize that small structural changes can meaningfully alter hydrolysis rates, reinforcing the need for data-driven rather than purely literature-traditional choices.
Controlling Molecular Weight and Drug Loading
Molecular weight and drug loading affect nearly every downstream attribute: clearance rate, tissue penetration, viscosity, aggregation, and batch reproducibility. In general, increasing hydrodynamic size can reduce renal clearance and extend circulation time; multiple studies across macromolecular systems show strong molecular-weight dependence of clearance and exposure. Yet larger is not automatically better: overly large or poorly degradable constructs can raise long-term persistence concerns or impair penetration into certain tissues. Drug loading also requires balance. Too low, and the polymer is mostly carrier with limited payload efficiency; too high, and the conjugate may become poorly soluble, self-assemble unpredictably, or release too quickly because of destabilized linker microenvironments.
Stability, Sterility, and Storage Considerations
PDC stability is governed by the weakest chemical link: hydrolysable linkers, oxidation-prone groups, or backbone segments susceptible to chain scission. Storage risks include gradual linker hydrolysis, oxidation of sensitive moieties, and physical instability such as aggregation or precipitation—especially for amphiphilic conjugates that can undergo slow structural reorganization. Sterility strategy depends on the product form. For solution-state materials, sterile filtration and aseptic processing are commonly used in sterile manufacturing contexts, with regulatory guidance emphasizing filter integrity controls for sterile boundaries. For solid or device-associated polymer systems, irradiation or ethylene oxide may be considered, but literature on sterilization of polymeric materials and nanoparticles repeatedly shows that sterilization can alter molecular weight, morphology, and release behavior, so method selection must be validated for each specific polymer system rather than assumed.
Pharmacokinetics and Drug Release Mechanisms of Polymer–Drug Conjugates
PDC pharmacokinetics and release should be approached as coupled phenomena: the polymer scaffold changes distribution and clearance, while linker chemistry and polymer degradation control the time course of free drug availability. The most reliable PDC programs define target ranges for size, charge, and release kinetics early, then confirm these through orthogonal characterization and relevant in vitro or in vivo models that reflect the intended use environment.
Improved Circulation Time and Reduced Clearance
Increasing apparent size is a central mechanism by which polymer conjugation can reduce rapid clearance. Renal filtration is strongly influenced by the properties of the glomerular filtration barrier, and multiple approaches to pharmacokinetic extension aim to remain above rapid filtration thresholds via increased hydrodynamic size or optimized surface properties. Empirical studies of pharmacokinetic modifying anchors demonstrate clear molecular weight dependence in clearance reduction, reinforcing why molecular weight control and dispersity management are not just synthetic preferences but functional requirements.
Enhanced Permeability and Retention Effect
The enhanced permeability and retention effect describes preferential accumulation of macromolecules and nanoparticles in certain tumor tissues due to leaky vasculature and impaired lymphatic drainage, an idea strongly associated with foundational work by Maeda and colleagues. At the same time, modern reviews emphasize heterogeneity: permeability and retention vary widely by tumor type, lesion microenvironment, vascular state, and model system, meaning EPR should be treated as a context-dependent opportunity rather than a guaranteed mechanism. Workshop and review discussions in the field further highlight that successful macromolecular delivery often requires complementary strategies beyond assuming uniform permeability.
Enhanced Solubility and Stability of Drugs
Conjugating hydrophobic drugs to hydrophilic polymers is a direct route to improving apparent solubility and formulation practicality. Additionally, the polymer environment can shield sensitive drugs from premature degradation or nonspecific interactions, depending on how exposed the payload remains and whether the conjugate self-assembles into a core–shell structure. Reviews of PEG-based and broader PDC systems repeatedly frame solubilization and stability as recurring drivers for adopting polymer conjugation, but they also emphasize the importance of verifying that the intended stability improvements do not come at the cost of incomplete release or uncontrolled aggregation.
Controlled and Sustained Drug Release
Controlled release in PDCs is a chemically programmed event rather than a purely diffusion-driven one. Release can occur by linker cleavage (pH, redox, enzyme) or by polymer degradation that exposes or liberates drug-bearing fragments. Advanced systems can incorporate self-immolative behavior or cascade mechanisms to amplify release once triggered. Because release is bond-governed, developers can often map release kinetics to measurable chemical transformations, such as hydrolysis rates of acid-labile bonds or enzymatic cleavage rates of peptide linkers—making analytical chemistry and mechanism studies central to PDC optimization.
Targeted Delivery to Specific Cells or Tissues
Targeting can be passive (size- and permeability-driven localization) or active (ligand-mediated binding and uptake). The conceptual model of polymer therapeutics explicitly includes optional targeting groups, reflecting long-standing interest in steering macromolecules toward specific cell types or tissue microenvironments. In modern design, targeting is often combined with cleavable linkers so that localization and release are coordinated: a targeting ligand improves enrichment near the site of interest, while microenvironment-responsive cleavage enables drug liberation under locally distinct conditions. Reviews of polymer–drug conjugate therapeutics consistently emphasize that targeting performance depends on the full construct—ligand density, spacer length, polymer corona, and particle size—not simply the identity of a targeting ligand.
Therapeutic Applications of Polymer–Drug Conjugates
PDCs have been explored across many therapeutic areas because polymer conjugation is a general-purpose strategy: it can increase solubility, reshape distribution, and implement controlled release in ways that are difficult to achieve with small molecules alone.
Cancer Therapy and Targeted Chemotherapy
Oncology has been a major driver for PDC research because many cytotoxic payloads suffer from narrow tolerability margins and poor selectivity when administered as free drugs. Polymer conjugation enables multiple complementary strategies: prolonged circulation to increase tumor exposure time, passive enrichment mechanisms such as EPR in responsive tumor models, and cleavable linkers that bias drug release toward intracellular compartments or tumor-associated microenvironments. Critically, the field also recognizes that tumor targeting is heterogeneous, so successful constructs often rely on multi-factor optimization rather than a single mechanism.
Polymer–Antibiotic Conjugates for Infectious Diseases
Polymer–antibiotic conjugation is an active area of exploration for improving local retention, reducing premature deactivation, and enabling sustained exposure in difficult environments such as biofilms. Chitosan-based conjugates are a well-reviewed platform in this area, supported by accessible amino and hydroxyl groups for derivatization and inherent cationic character that can interact with microbial membranes. Literature also describes covalent linking approaches where antibiotics are tethered to chitosan or related oligomers to address biofilm-associated infections, illustrating how polymer conjugation can be built around the physical biology of infection rather than only the pharmacology of a free drug.
Polymer Conjugates in Anti-Inflammatory Therapy
Inflammatory microenvironments can differ from healthy tissues in vascular permeability, enzyme activity, oxidative stress levels, and pH, creating opportunities for responsive polymer–drug designs. Some delivery concepts originally discussed in the context of macromolecular accumulation note that enhanced permeability phenomena can appear not only in tumors but also in inflamed tissues, motivating research into macromolecular delivery for anti-inflammatory applications. In parallel, stimuli-responsive linker strategies designed around ROS, enzyme activity, or pH shifts provide a chemistry-based route to bias drug release toward inflammatory sites, provided the trigger differences are large enough to produce practical selectivity.
Emerging Applications in Gene and Protein Delivery
Polymer therapeutics extends beyond small molecules into protein and nucleic-acid delivery, where polymers can act as conjugation partners, stabilizing chains, or complexing agents. Reviews of polymer therapeutics explicitly include polymer–protein conjugates and polyplexes for DNA delivery, reflecting the broad applicability of polymer chemistry to biologic modalities. Poly(2-oxazoline)-based systems, for example, are reviewed across drug, protein, and gene delivery contexts. For nucleic acids, polymer design often centers on balancing binding strength (for protection and complexation) against release, while minimizing nonspecific interactions through hydrophilic or zwitterionic shielding segments.
Services
Polymer–Drug Conjugate Development Support Servies at BOC Sciences
BOC Sciences provides comprehensive technical support for the development of polymer–drug conjugate systems by integrating advanced polymer chemistry, functional material design, and drug conjugation technologies. Our team supports projects ranging from custom polymer synthesis and functionalization to conjugation process development and formulation optimization, enabling researchers to design polymer carriers with tailored molecular structures and controlled drug release profiles.

Custom Polymer Synthesis for Conjugate Formulations
- Custom design and synthesis of functional polymers for polymer–drug conjugate systems.
- Structural design and molecular weight control of PEG, HPMA, PLA, and PCL polymers.
- Support for controlled polymerization techniques, including RAFT, ATRP, and ring-opening polymerization (ROP).
- Development of functionalized polymers with reactive groups for drug conjugation.

Functionalization for Targeted and Controlled Delivery
- Targeting ligand modification services for active drug delivery systems.
- Conjugation of folic acid, peptides, antibody fragments, and aptamers.
- Design of pH-responsive, redox-responsive, and enzyme-cleavable linkers.
- PEGylation and surface functionalization to improve stability and circulation time.

Drug Conjugation Process Development and Optimization
- Development of chemical conjugation processes between drugs and polymers.
- Design and optimization of ester, amide, and hydrazone linkages.
- Optimization of conjugation efficiency and reaction conditions.
- Development of polymer conjugation systems for diverse drug molecules.

Polymer–Drug Compatibility and Formulation Optimization
- Evaluation of polymer–drug compatibility to ensure conjugate stability.
- Optimization of polymer structure and drug loading ratios.
- Assessment of drug release behavior and delivery performance.
- Formulation optimization for polymer–drug conjugate systems.
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What are polymer–drug conjugates used for in drug delivery?
They are used to reshape how a drug behaves in the body by covalently attaching it to a polymer carrier, commonly to improve solubility, extend circulation time, reduce rapid clearance, and implement controlled release through engineered linker cleavage or polymer degradation. Because the polymer is modular, additional functions such as targeting ligands or stealth segments can be incorporated when supported by the design constraints of the payload and intended use scenario.
-
How do polymer–drug conjugates improve drug stability and efficacy?
PDCs can improve stability by shielding the drug from premature degradation or unfavorable interactions and by preventing rapid precipitation of hydrophobic payloads through hydrophilic polymer attachment. Whether overall performance improves depends on correct kinetic design: the conjugate must remain stable during storage and circulation while still enabling timely drug release at the intended site.
-
Can polymer–drug conjugates provide targeted and sustained release?
Yes, but only when targeting and release are engineered as system properties. Targeting can be passive (size- and permeability-driven enrichment in certain tissues) or active (ligand-mediated binding and uptake), while sustained release is implemented through cleavage or degradation chemistry. Many modern designs combine both: a targeting element or size regime biases localization, and a cleavable linker provides site- or microenvironment-biased drug liberation, with responsiveness tuned by the chemical stability of the bond under different conditions.
-
How are polymer–drug conjugates customized for specific therapeutics?
Customization usually begins with payload constraints (functional groups, solubility limits, stability), then proceeds to polymer selection (synthetic vs biopolymer, degradable vs persistent), architecture choice (linear, branched, block copolymer), and linker selection (stable vs trigger-cleavable). The final step is analytic confirmation: verifying molecular weight distribution, drug loading, residual impurities, and release kinetics using orthogonal methods.

 Fig. 1. Schematic overview of polymer-drug conjugate components and architectural types (BOC Sciences Authorized).
Fig. 1. Schematic overview of polymer-drug conjugate components and architectural types (BOC Sciences Authorized). Fig. 2. Comparison of natural and synthetic polymers used in PDCs (BOC Sciences Authorized).
Fig. 2. Comparison of natural and synthetic polymers used in PDCs (BOC Sciences Authorized).