Stimuli-Responsive Polymer Drug Delivery Systems for Targeted Drug Release
Targeted drug delivery has become a central focus in modern pharmaceutical and biomedical research, as conventional drug administration often suffers from limited therapeutic efficiency, systemic toxicity, and poor control over drug release profiles. To address these challenges, stimuli-responsive polymer drug delivery systems—also known as smart polymer systems—have emerged as an innovative strategy for achieving controlled and site-specific drug release. These advanced materials are designed to respond to specific environmental triggers, enabling therapeutic agents to be released only when and where they are needed. Stimuli-responsive polymers can undergo physical or chemical changes when exposed to particular internal stimuli (such as pH variations, enzymatic activity, or redox conditions) or external stimuli (including temperature, light, magnetic fields, ultrasound, or electric signals). These responsive behaviors allow polymer carriers to precisely regulate drug release kinetics, improve drug stability, and enhance the accumulation of therapeutic molecules at target tissues. As a result, smart polymer-based delivery systems have attracted significant interest in areas such as cancer therapy, gene delivery, inflammatory disease treatment, and regenerative medicine.
Resources
What Are Stimuli-Responsive Polymers?
Stimuli-responsive polymers, frequently referred to as smart polymers, are macromolecules capable of undergoing rapid and significant physicochemical transitions in response to minute changes in their surrounding environment. These transitions can manifest as alterations in solubility, shape, volume, or structural integrity. The environmental changes triggering these responses can be physiological variations, such as shifts in acidity or enzyme concentrations, or externally applied physical forces like light or magnetic fields. The core utility of these materials lies in their ability to act as intelligent sensors and actuators at the molecular level.
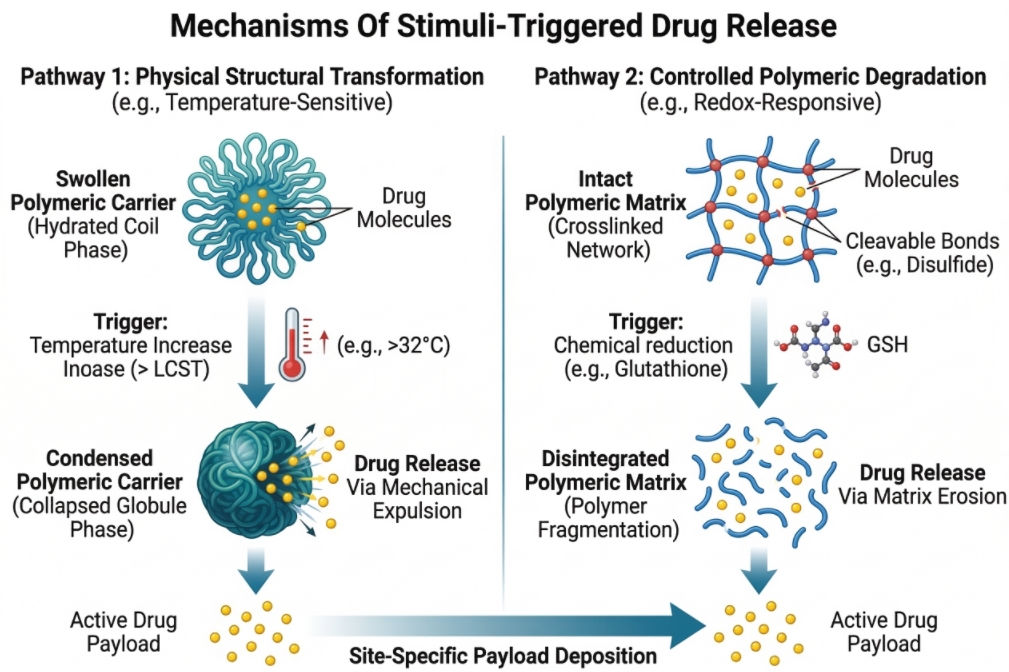 Fig. 1. Illustration of physical and chemical stimuli-triggered drug release mechanisms (BOC Sciences Authorized).
Fig. 1. Illustration of physical and chemical stimuli-triggered drug release mechanisms (BOC Sciences Authorized).
Why Targeted Drug Release Is Critical in Modern Therapeutics?
Systemic drug administration often faces severe limitations, primarily poor biodistribution, rapid clearance, and off-target toxicity. Targeted drug release is fundamentally critical because it maximizes the therapeutic index of a pharmacological agent. By sequestering the active pharmaceutical ingredient within a polymeric carrier until it reaches the intended physiological site, researchers can achieve high localized drug concentrations while simultaneously minimizing exposure to healthy tissues. This spatial and temporal control is particularly vital for highly potent molecules, such as cytotoxic antineoplastic agents or delicate biologic therapeutics like nucleic acids and proteins.
Key Advantages Over Conventional Drug Delivery Systems
Conventional delivery systems typically rely on passive diffusion or simple, continuous degradation, resulting in a predictable but uncontrollable release profile. In contrast, stimuli-responsive platforms offer several distinct advantages:
- On-Demand Release: Payload discharge is suppressed during systemic circulation and exclusively activated at the target site.
- Protection of Cargo: Sensitive molecules are shielded from enzymatic degradation and immune clearance until reaching the target microenvironment.
- Enhanced Intracellular Delivery: Certain responsive mechanisms facilitate endosomal escape, ensuring drugs reach specific subcellular compartments like the cytosol or nucleus.
- Tailorable Kinetics: The release rate can be precisely tuned by modifying the polymer backbone, molecular weight, and the ratio of responsive functional groups.
Mechanisms of Stimuli-Triggered Drug Release
Understanding the fundamental mechanisms that govern payload discharge is essential for engineering effective polymeric carriers. These release mechanisms dictate exactly how a polymer network interacts with encapsulated molecules upon encountering specific physiological or external triggers. The design process requires a rigorous understanding of polymer physics and thermodynamic principles to ensure that the transition is both reliable and biologically compatible.
Polymer Structural Changes Under External or Internal Stimuli
The primary mechanism of action for most smart polymers involves macroscopic or microscopic structural transformations. When exposed to a stimulus, the polymer chain may undergo a phase transition, such as the coil-to-globule transition seen in thermoresponsive materials. Alternatively, crosslinked polymer networks like hydrogels may experience massive swelling or deswelling as the thermodynamic affinity between the polymer strands and the aqueous solvent shifts. These conformational changes physically squeeze the drug out of the matrix or increase the pore size to allow rapid diffusion.
Controlled Drug Diffusion and Polymer Degradation
Drug release from polymeric carriers is heavily governed by diffusion kinetics and degradation pathways. In a responsive system, the stimulus often accelerates degradation by cleaving specific covalent bonds within the polymer backbone or crosslinking agents.
- Bulk Degradation: The entire polymer matrix degrades simultaneously, leading to a massive burst release of the encapsulated drug.
- Surface Erosion: The polymer degrades solely at the solid-liquid interface, providing a more sustained, zero-order release profile. The responsive trigger alters the permeability of the matrix, fundamentally shifting the diffusion coefficient of the drug molecules.
Reversible vs. Irreversible Stimuli-Responsive Mechanisms
The nature of the chemical response dictates whether the delivery system can cycle through multiple release phases or if the release is a singular event.
- Reversible Mechanisms: These rely on non-covalent interactions or physical phase transitions. For example, temperature-induced swelling and collapsing can be cycled repeatedly, allowing for pulsatile release profiles corresponding to rhythmic stimulus application.
- Irreversible Mechanisms: These involve the permanent cleavage of covalent bonds, such as ester hydrolysis in acidic environments or disulfide bond reduction. Once the structural integrity is compromised, the polymer cannot return to its original state, making this approach ideal for complete and rapid drug offloading at a terminal target.
Molecular Design Strategies for Precise Drug Release Control
Achieving precise control over the release profile requires meticulous molecular engineering. Researchers manipulate the hydrophilic-lipophilic balance by carefully selecting appropriate functional monomers. Strategic placement of responsive linkages—whether in the main polymer backbone, as pendent side chains, or within the crosslinking nodes—determines the sensitivity and rate of the response. Furthermore, varying the molecular weight and grafting density allows scientists to fine-tune the exact threshold at which the polymer system activates.
Types of Stimuli Used in Responsive Polymer Drug Delivery
The architectural versatility of smart polymers stems from their capacity to be precisely engineered to respond to a vast and diverse array of specific triggers. In the context of macromolecular design, these triggers are systematically categorized into endogenous stimuli, which exploit the natural biochemical and thermodynamic gradients present in highly specific biological microenvironments, and exogenous stimuli, which involve the controlled application of external physical energy fields. Selecting and integrating the appropriate stimulus-responsive moiety is the foundational step in designing a targeted therapeutic carrier, dictating both the synthetic strategy and the ultimate release kinetics of the payload.
pH-Responsive Polymer Drug Delivery Systems
Pathological microenvironments frequently exhibit pronounced pH dysregulation compared to the physiological pH of 7.4 found in standard systemic circulation. For instance, specific disease models and localized inflammatory sites often present a mildly acidic extracellular pH ranging from 6.5 to 6.8. Furthermore, intracellular trafficking pathways involve significant acidification, with endosomes dropping to a pH of 5.5 to 6.0 and lysosomes reaching a pH of 4.5 to 5.0. To exploit these gradients, polymer chemists employ two primary structural strategies:
- Protonable Networks: Polymers incorporating ionizable functional groups, such as tertiary amines (e.g., poly(N,N-dimethylaminoethyl methacrylate)) or carboxylic acids (e.g., poly(acrylic acid)). As the environmental pH drops below the polymer pKa, these groups undergo rapid protonation. This induces massive electrostatic repulsion along the polymer backbone, leading to globule-to-coil transitions, micellar dissociation, or profound hydrogel swelling.
- Acid-Labile Linkages: This approach utilizes covalent bonds that are stable at neutral pH but rapidly hydrolyze under acidic conditions. Common moieties include hydrazones, acetals, ketals, orthoesters, and imines. Integrating these linkages into the main polymer backbone results in bulk degradation, while placing them as tethering arms between the polymer and the active pharmaceutical ingredient ensures controlled, site-specific cleavage.
Temperature-Sensitive Polymer Platforms
Temperature represents one of the most extensively researched stimuli due to the fundamental thermodynamic principles governing polymer chain hydration. Thermoresponsive polymers are characterized by a critical solution temperature, dictating their phase behavior in aqueous media.
- Lower Critical Solution Temperature (LCST): Polymers such as poly(N-isopropylacrylamide) or poly(oligo(ethylene glycol) methacrylate) exhibit an LCST. Below this threshold, hydrogen bonding between the polymer and water molecules is thermodynamically favorable, resulting in a soluble, extended coil state. As the temperature rises above the LCST, the entropic penalty of water organization drives the rapid dehydration and collapse of the polymer into an insoluble, hydrophobic globule, aggressively expelling any encapsulated payload.
- Upper Critical Solution Temperature (UCST): Less common but highly valuable, UCST polymers rely on supramolecular interactions like hydrogen bonding or electrostatic interactions between polymer chains. They remain collapsed at lower temperatures and dissolve upon heating. Through strategic copolymerization, synthetic chemists can meticulously tune these transition temperatures to align seamlessly with localized hyperthermic conditions or external heat application.
Enzyme-Responsive Polymer Carriers
Enzymatic dysregulation is a definitive biomarker for numerous specific physiological states. Certain proteases, lipases, esterases, or glycosidases are massively overexpressed in highly localized tissue microenvironments. Enzyme-responsive polymers represent the pinnacle of biological specificity, utilizing complex peptide sequences or highly specific ester linkages that act as exclusive substrates for these target biocatalysts.
- Protease Cleavage: Systems often integrate specific oligopeptide sequences—such as GFLG (Gly-Phe-Leu-Gly) for Cathepsin B—into the polymer architecture. Upon encountering the localized enzyme concentration, the biocatalyst physically cleaves the polymer matrix or the drug-polymer tether.
- Matrix Degradation: In crosslinked systems, enzymatic degradation of the crosslinking nodes leads to a complete breakdown of the three-dimensional network, shifting the release profile from slow diffusion to rapid, localized burst release.
Redox-Responsive Polymer Systems for Intracellular Drug Release
The redox potential presents a massive concentration gradient between the extracellular matrix and the highly reducing intracellular space. This gradient is primarily maintained by glutathione (GSH). While extracellular GSH concentrations are typically in the low micromolar range (approximately 2 to 20 µM), cytosolic levels are exponentially higher, typically between 2 and 10 mM.
- Disulfide Linkages: The most prevalent strategy involves integrating disulfide bonds into the polymer backbone, crosslinkers, or pendant side chains. These bonds demonstrate excellent stability during systemic transport but undergo rapid, thiol-disulfide exchange reactions upon cellular internalization, causing architectural collapse and payload discharge directly into the cytoplasm.
- Reactive Oxygen Species (ROS): Conversely, certain pathological tissues exhibit elevated oxidative stress. Polymers incorporating thioether, thioketal, or selenium/tellurium-containing domains are engineered to undergo rapid phase transitions or degradation when exposed to high localized concentrations of hydrogen peroxide or hydroxyl radicals.
Photo-Responsive Polymer Drug Delivery Systems
Light application provides an unparalleled degree of spatiotemporal control, allowing researchers to dictate both the exact geographical location and the precise chronological moment of payload discharge. Photo-responsive polymers incorporate specific light-sensitive chromophores that undergo structural alterations upon absorbing specific wavelengths of electromagnetic radiation.
- Photo-Isomerization: Chromophores like azobenzene or spiropyran undergo reversible structural shifts (e.g., trans-to-cis isomerization) when irradiated, significantly altering the hydrophilic-lipophilic balance and local geometry of the polymer network.
- Photo-Cleavage: Moieties such as o-nitrobenzyl groups undergo irreversible photolysis upon UV irradiation, triggering matrix degradation or the severing of drug-tethering bonds.
- Near-Infrared (NIR) Activation: Because UV light suffers from poor tissue penetration and potential phototoxicity, advanced systems utilize NIR-responsive agents or upconverting nanoparticles that translate deep-penetrating NIR light into localized UV/Visible emissions or localized photothermal heat, indirectly triggering release.
Magnetic-Responsive Polymer Drug Delivery Platforms
Magnetic-responsive systems are sophisticated organic-inorganic hybrid nanomaterials, fundamentally combining responsive polymeric matrices with superparamagnetic iron oxide nanoparticles (SPIONs). This dual-component architecture provides multiple mechanisms for payload delivery when subjected to an external alternating magnetic field.
- Magneto-Thermal Release: Under an alternating magnetic field, SPIONs dissipate energy through Néel and Brownian relaxation processes, generating intense localized thermal energy. This targeted hyperthermia can directly trigger a secondary thermoresponsive polymer transition (such as an LCST collapse) to expel the drug.
- Magneto-Mechanical Disruption: The physical oscillation and alignment of the incorporated magnetic nanoparticles generate microscopic shear forces. These mechanical stresses transiently disrupt the polymer matrix, increasing porosity and accelerating the diffusion kinetics of the encapsulated agents, completely independent of thermal effects.
Acoustic-Responsive Polymer Drug Delivery Systems
Ultrasound serves as an exceptional exogenous stimulus, offering strictly non-invasive, deep tissue penetration with highly tunable frequencies and intensities. Acoustic-responsive polymer systems—frequently engineered as nanodroplets, microbubbles, or specific micellar architectures—are designed to interact with focused ultrasound waves.
- Acoustic Cavitation: The primary release mechanism relies on the mechanical energy derived from inertial cavitation. The rapid formation, growth, and violent collapse of microscopic gas bubbles generate intense localized shockwaves, microstreaming, and profound shear forces.
- Mechanophore Activation: These extreme mechanical forces can physically tear apart the supramolecular assembly of the polymer carrier, transiently increasing localized cell membrane permeability (sonoporation), and even cleaving specific mechanically labile covalent bonds (mechanophores) embedded within the polymer backbone.
Electric-Responsive Polymer Drug Delivery Systems
Electric-responsive architectures are fundamentally constructed using intrinsically conducting polymers, such as polyaniline, polypyrrole, or polythiophene derivatives. These conjugated macromolecules possess the unique ability to undergo reversible redox cycling when subjected to an external electrochemical potential.
- Electrochemical Actuation: The application of a precise, low-voltage electrical field induces dramatic shifts in the polymer oxidation state. This electrochemical transition forces the influx or efflux of dopant ions to maintain charge neutrality, resulting in massive alterations to the polymer network volume, charge density, and internal porosity.
- Pulsatile Control: This dynamic actuation selectively expels charged pharmaceutical molecules bound within the matrix. By manipulating the frequency and amplitude of the applied current, researchers can achieve highly precise, on-off pulsatile dosing profiles, a concept heavily investigated for next-generation implantable biomaterials and transdermal iontophoretic devices.
Design and Synthesis of Stimuli-Responsive Polymers
Designing a stimuli-responsive polymeric system requires an extraordinary level of precision to construct complex, highly defined macromolecular architectures. The selection of exact synthetic routes directly determines the polymer molecular weight, dispersity, compositional uniformity, and the precise spatial arrangement of responsive functional domains. This architectural control is paramount, as even minor deviations in the polymer backbone or side-chain topology can drastically alter the thermodynamic phase transitions, degradation kinetics, and overall stimuli sensitivity of the resulting nanocarrier.
Functional Monomers Used in Smart Polymer Design
The genesis of any stimuli-responsive material lies in the strategic selection and purification of functional monomers. These low-molecular-weight building blocks dictate the macroscopic behavior of the final polymer network through their inherent physicochemical properties.
- Vinyl and (Meth)acrylate Derivatives: For pH and thermoresponsive systems, monomers such as N-isopropylacrylamide, N,N-dimethylaminoethyl methacrylate, and various acrylic acids are foundational. The precise carbon chain length and the hydrophobicity of the substituent groups on these monomers allow chemists to finely tune the critical solution temperatures and pKa values of the resulting macromolecules.
- Cyclic Monomers: For biodegradable responsive systems, cyclic esters like lactide, glycolide, and epsilon-caprolactone are heavily utilized.
- Custom Functional Monomers: Advanced designs often require the custom synthesis of monomers bearing specific cleavable linkages, such as disulfide bonds for redox responsiveness or photolabile o-nitrobenzyl groups for photo-responsiveness. These are synthesized with polymerizable vinyl groups to allow direct incorporation into the polymer backbone.
Controlled Polymerization Techniques for Responsive Polymers
Historically, conventional free-radical polymerization yielded polymers with broad molecular weight distributions and poor architectural control, which are entirely unsuitable for precise therapeutic delivery. The advent of reversible deactivation radical polymerization techniques revolutionized the synthesis of smart polymers, enabling the creation of materials with highly predictable molecular weights and incredibly low dispersity.
- Atom Transfer Radical Polymerization (ATRP): This technique utilizes a transition metal catalyst, most commonly copper, complexed with a suitable ligand to establish an equilibrium between active and dormant propagating polymer chains via a reversible halogen transfer. ATRP is highly versatile, allowing for the synthesis of well-defined star polymers, brush polymers, and highly dense grafting architectures.
- Reversible Addition-Fragmentation Chain Transfer (RAFT) Polymerization: RAFT relies on a thiocarbonylthio chain transfer agent to mediate the polymerization process. Unlike ATRP, RAFT is highly tolerant to a vast array of functional groups, including unprotected acids and amines, making it exceptionally powerful for synthesizing highly functionalized, stimuli-responsive macromolecules directly without the need for complex protecting group chemistry.
- Ring-Opening Polymerization (ROP): For the synthesis of biodegradable responsive polyesters and polypeptides, controlled ROP of cyclic monomers is the gold standard, often utilizing organocatalysts to achieve high conversion rates and narrow molecular weight distributions while completely avoiding heavy metal contamination.
Copolymer and Block Polymer Engineering
Integrating multiple, distinct functionalities within a single delivery vehicle—such as combining stealth properties with specific stimuli-responsiveness—necessitates the sophisticated engineering of copolymers. Block copolymers are characterized by distinct, continuous sequences of different monomeric units linked together.
- Thermodynamic Assembly: By adjoining a permanently hydrophilic block, such as poly(ethylene glycol), with a stimuli-responsive block, chemists engineer macromolecules that reliably self-assemble into complex nanostructures like micelles or polymersomes in aqueous media. This assembly is driven by the thermodynamic incompatibility between the blocks, governed by the Flory-Huggins interaction parameters.
- Architectural Variations: Sequential monomer addition using controlled polymerization techniques allows for the creation of AB diblock, ABA triblock, and even complex multiblock architectures. The specific volume fraction of each block directly dictates the ultimate morphological geometry of the assembled nanocarrier, ranging from spherical micelles to cylindrical worms or planar lamellae.
Surface Functionalization and Ligand Modification Strategies
Beyond the fundamental stimuli-responsive behavior embedded within the polymer core or backbone, achieving absolute site-specific cellular targeting requires extensive surface engineering. Post-polymerization modification is a critical synthetic step, ensuring that sensitive targeting ligands are attached without compromising their biological activity.
- Orthogonal Click Chemistry: Researchers heavily rely on highly efficient, orthogonal click chemistry reactions for surface modification. Techniques such as copper-catalyzed azide-alkyne cycloaddition, strain-promoted alkyne-azide cycloaddition, and robust thiol-ene radical reactions offer near-quantitative yields under mild, physiological conditions.
- Spacer Engineering: The conjugation of specific antibodies, aptamers, or small-molecule ligands is rarely done directly to the polymer backbone. Instead, ligands are tethered via hydrophilic spacer arms, typically poly(ethylene glycol) chains. This strategic spacer placement overcomes steric hindrance, ensuring the ligand extends beyond the nanoparticle hydration layer, allowing the nanocarrier to bind with high avidity to specific receptors overexpressed on target cell membranes.
Common Stimuli-Responsive Polymer Materials
The translation of theoretical drug delivery concepts into robust pharmacological research tools relies on a well-established repertoire of responsive materials. Certain classes of polymers have emerged as foundational pillars in the field due to their reliable phase transitions, well-understood pharmacokinetics, and biocompatibility profiles. These materials serve as the structural basis upon which more complex, multi-responsive systems are continually iterated.
Thermoresponsive Polymers (e.g., PNIPAM-Based Systems)
Thermoresponsive materials are characterized by their ability to undergo dramatic hydration state changes in response to temperature fluctuations, driven by complex thermodynamic shifts in polymer-solvent interactions. Poly(N-isopropylacrylamide), often abbreviated as PNIPAM, remains the absolute gold standard in this category. In aqueous solutions, PNIPAM exhibits a sharp Lower Critical Solution Temperature at approximately 32 degrees Celsius.
Below this threshold, the polymer forms stable intermolecular hydrogen bonds with surrounding water molecules, adopting a relaxed, hydrophilic coil conformation. As the temperature breaches the transition point, the entropic penalty of maintaining this highly ordered water structure forces a rapid shift; the polymer chains undergo entropy-driven dehydration, favoring intramolecular hydrogen bonding and collapsing into a dense, hydrophobic globule. Synthetic chemists exploit this behavior by copolymerizing PNIPAM with varying ratios of hydrophilic or hydrophobic comonomers, precisely tuning the transition point to align exactly with standard physiological body temperature or localized hyperthermic parameters.
pH-Sensitive Polymeric Carriers
Materials exhibiting pronounced structural or solubility shifts across physiological pH gradients form the fundamental basis of pH-sensitive delivery. These polymers are heavily engineered to leverage their specific acid dissociation constants (pKa) against the surrounding environmental pH.
Basic polymers, such as those bearing tertiary amine groups like poly(beta-amino esters) or poly(L-histidine), remain uncharged and hydrophobic at neutral physiological pH. However, upon encountering mildly acidic environments—such as those found in ischemic tissues or endolysosomal compartments—these basic moieties undergo rapid protonation. This sudden influx of positive charge induces massive electrostatic repulsion along the polymer backbone, causing catastrophic swelling, micellar disassembly, and facilitating the highly sought-after proton sponge effect to rupture endosomal membranes. Conversely, acidic polymers like poly(acrylic acid) derivatives remain tightly coiled and protective in highly acidic environments, making them exceptional candidates for oral delivery applications where they shield payloads from gastric acid before swelling and releasing their cargo in the neutral environment of the intestinal tract.
Biodegradable Stimuli-Responsive Polymers
To prevent long-term systemic accumulation and potential foreign body responses, integrating complete biodegradability with stimuli-responsiveness is an absolute necessity for advanced carrier design. Aliphatic polyesters, notably poly(lactic-co-glycolic acid) and poly(caprolactone), serve as exceptionally reliable, biocompatible hydrophobic backbones.
However, these traditional polyesters exhibit slow, bulk-erosion degradation profiles. To transform them into smart materials, researchers chemically engineer their architecture by inserting stimuli-labile linkages directly into the main polymer chain or by grafting highly responsive pendant groups along the backbone. For example, inserting rapidly hydrolyzable orthoester or acetal linkages creates a platform that maintains structural integrity during systemic circulation but rapidly fragments in targeted acidic microenvironments. This strategic synthesis ensures that the carrier executes a precise, stimulus-initiated release profile and subsequently degrades into harmless, easily excretable metabolic byproducts like lactic and glycolic acid.
Amphiphilic Block Copolymers for Responsive Drug Delivery
Amphiphilic block copolymers, which perfectly balance hydrophilic and hydrophobic macromolecular segments, are essential for formulating and transporting poorly water-soluble hydrophobic pharmaceutical agents. In aqueous media, these copolymers spontaneously self-assemble into core-shell nanostructures, driven by the thermodynamic necessity to shield their hydrophobic blocks from water, establishing a critical micelle concentration.
The structural brilliance of smart amphiphilic systems lies in engineering the hydrophobic block to be explicitly stimuli-responsive. The hydrophilic corona—most frequently composed of poly(ethylene glycol)—provides vital steric stabilization and prevents rapid immune clearance. When the nanocarrier reaches the target microenvironment and encounters the triggering stimulus, the responsive core-forming block undergoes a rapid phase transition from hydrophobic to hydrophilic. This transition completely destroys the delicate hydrophilic-lipophilic balance, drastically shifting the critical micelle concentration and forcing the immediate, thermodynamic dissociation of the micellar structure, resulting in rapid and localized cargo offloading.
Looking for Stimuli-Responsive Polymers for Drug Delivery?
Whether you need pH-responsive polymers, thermo-responsive carriers, or multi-stimuli smart systems, our experts provide tailored synthesis and functionalization solutions to support your research and therapeutic development.
Nanostructures in Stimuli-Responsive Polymer Drug Delivery
The macroscopic efficacy of a polymer matrix is dictated by its nanoscale organization. Macromolecular engineering allows for the spontaneous or directed assembly of polymers into distinct nanostructures. The geometric conformation, core-shell ratios, and surface area of these nanoparticles drastically influence drug loading capacity, circulation half-life, and interaction kinetics with cellular membranes and physiological barriers.
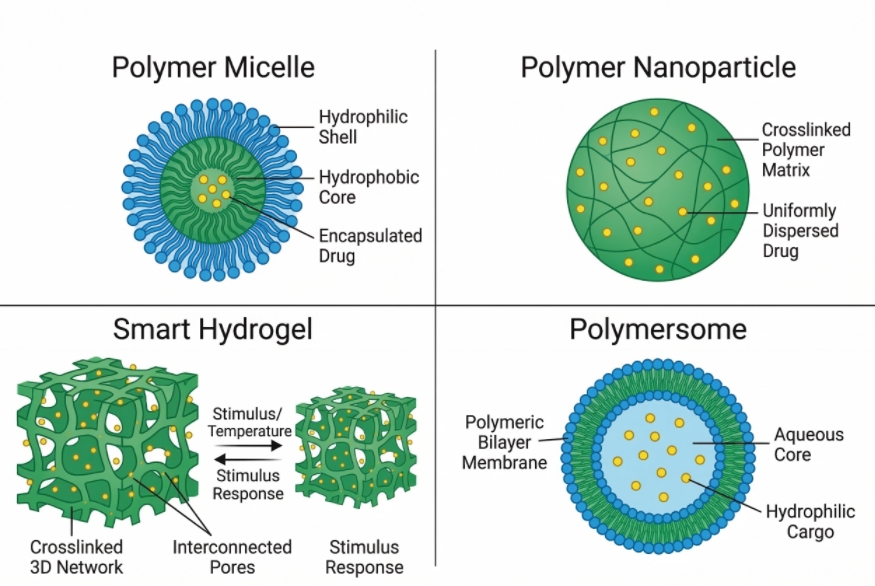 Fig. 2. Common stimuli-responsive polymer nanostructures for targeted drug delivery systems (BOC Sciences Authorized).
Fig. 2. Common stimuli-responsive polymer nanostructures for targeted drug delivery systems (BOC Sciences Authorized).
Polymer Micelles for Triggered Drug Release
Polymer micelles are core-shell nanostructures spontaneously formed by the self-assembly of amphiphilic block copolymers in aqueous solutions. The hydrophobic core serves as an excellent reservoir for lipophilic pharmaceutical agents, while the hydrophilic corona maintains colloidal stability. In a stimuli-responsive micelle, either the core-forming block is engineered to turn hydrophilic under specific conditions, causing immediate dissociation, or the corona is designed to shed, exposing targeting ligands or facilitating rapid membrane fusion.
Stimuli-Responsive Polymer Nanoparticles
Unlike micelles, which are dynamic assemblies, polymer nanoparticles are solid, matrix-type structures where the drug is uniformly dispersed or dissolved within a crosslinked polymer network. These robust structures offer superior stability against premature dilution in the bloodstream. Responsive nanoparticles often utilize degradation-based mechanisms, where environmental triggers cleave crosslinkers, initiating matrix erosion and facilitating a highly controlled, diffusion-mediated payload release over an extended period.
Smart Hydrogels for Controlled Drug Release
Smart hydrogels are macroscopic or microscopic three-dimensional crosslinked polymer networks capable of imbibing massive amounts of water or biological fluids without dissolving. Their high water content mimics native extracellular matrix, conferring excellent biocompatibility. Stimuli-responsive hydrogels function as intelligent drug depots. Environmental triggers induce massive volumetric changes, physically expelling the encapsulated water-soluble drugs or biologics, making them ideal candidates for localized, sustained release via subcutaneous or intramuscular injection.
Polymer Vesicles and Polymersomes
Polymersomes are artificial vesicles formed from amphiphilic block copolymers, structurally analogous to biological liposomes but characterized by vastly superior mechanical stability and lower membrane permeability. They possess a unique bilayer architecture enclosing an aqueous core, allowing for the simultaneous encapsulation of hydrophilic drugs within the core and hydrophobic agents within the membrane wall. Stimuli-responsive polymersomes are designed to undergo programmed bilayer disruption, inversion, or pore formation when exposed to physiological triggers, offering highly compartmentalized and precise delivery mechanisms.
Biomedical Applications of Stimuli-Responsive Drug Delivery Systems
The advanced material properties of smart polymers enable diverse applications across the spectrum of biomedical research. While these systems require rigorous optimization and extensive preclinical evaluation, their conceptual frameworks address some of the most critical challenges in modern pharmacology. By overcoming biological barriers and ensuring precise spatial-temporal drug distribution, these platforms pave the way for next-generation therapeutic modalities.
Targeted Cancer Therapy
In oncology research, stimuli-responsive polymers are engineered to exploit the unique physicochemical properties of the tumor microenvironment. Nanocarriers are designed to accumulate via the enhanced permeability and retention effect, subsequently utilizing the locally low pH, high concentrations of specific proteases, or elevated reactive oxygen species to trigger payload release. This localized activation restricts the exposure of highly toxic chemotherapeutic agents to malignant tissues, conceptually reducing severe systemic adverse effects common in traditional antineoplastic regimens.
Gene and Nucleic Acid Delivery
Delivering delicate genetic materials, such as small interfering RNA, messenger RNA, or plasmid DNA, requires overcoming significant physiological hurdles, including rapid enzymatic degradation and complex intracellular trafficking. Cationic stimuli-responsive polymers complex with negatively charged nucleic acids to form stable polyplexes. These systems are strategically designed to respond to the endolysosomal pH drop, triggering the proton sponge effect or membrane destabilization, facilitating endosomal escape and releasing the genetic payload directly into the cytosol or nucleus for efficient transfection.
Controlled Release for Chronic Diseases
Managing chronic conditions necessitates maintaining therapeutic drug concentrations within a narrow systemic window over extended periods. Smart polymeric depots can be designed as implantable or injectable formulations that respond dynamically to physiological biomarkers. For instance, advanced research explores glucose-responsive polymer matrices that autonomously regulate insulin release in precise accordance with fluctuating blood glucose levels, mimicking the physiological function of pancreatic beta cells and potentially revolutionizing endocrinology management.
Localized Drug Delivery for Inflammatory Diseases
Chronic inflammatory diseases, such as rheumatoid arthritis or inflammatory bowel disease, present localized microenvironments rich in specific biomarkers like reactive oxygen species or overexpressed proteolytic enzymes. Researchers develop stimuli-responsive hydrogels and nanoparticles that remain inert systemically but rapidly degrade and release potent anti-inflammatory agents or immunosuppressants upon direct interaction with these specific inflammatory markers, concentrating the therapeutic effect precisely at the site of tissue damage.
Smart Drug Delivery in Tissue Engineering and Regenerative Medicine
In the realm of regenerative medicine, stimuli-responsive polymers are frequently utilized as dynamic scaffolds that support cellular adhesion, proliferation, and differentiation. These smart scaffolds can be engineered to sequentially release specific growth factors, cytokines, or morphogens in response to cellular traction forces or the expression of matrix metalloproteinases during the tissue remodeling process. This spatiotemporally controlled release acts to orchestrate complex biological signaling cascades, accelerating functional tissue repair and regeneration.
Services
Custom Polymer Development and Support Services for Drug Delivery Research
BOC Sciences has extensive expertise in functional monomer design, polymer synthesis, and nanocarrier development, providing comprehensive support for the development of stimuli-responsive polymer drug delivery systems. To address the need for site-specific and controlled drug release triggered by environmental cues such as tumor microenvironments or physiological conditions, we offer integrated services covering stimuli-responsive monomer development, functional polymer design, drug carrier construction, and system characterization and optimization. By precisely tailoring polymer structures and response mechanisms (e.g., pH, temperature, and redox conditions), BOC Sciences helps clients develop smart drug delivery systems with environmental responsiveness, targeted release capability, and enhanced stability.

Stimuli-Responsive Functional Monomer Design and Synthesis
- Custom design and synthesis of pH-responsive, thermo-responsive, and redox-responsive monomers.
- Development of enzyme-responsive and photo-responsive monomers for smart delivery systems.
- Synthesis of functional monomers containing dynamic bonds or cleavable linkages.
- Supply of functional monomers for the construction of stimuli-responsive polymers.

Smart Polymer Design and Controlled Polymerization
- Custom design and synthesis of stimuli-responsive polymers.
- Development of amphiphilic block copolymers and functional polymers.
- Support for controlled polymerization techniques, including RAFT, ATRP, and ring-opening polymerization (ROP).
- Development of multi-responsive and biodegradable polymer materials.

Stimuli-Responsive Drug Delivery System Construction
- Development of stimuli-responsive nanocarriers and polymer micelle systems.
- Support for encapsulation of hydrophobic drugs and responsive release system design.
- Design of multi-stimuli-responsive drug delivery systems.
- Optimization of drug release behavior and responsiveness.

Characterization and Performance Evaluation
- Polymer structure and molecular weight analysis.
- Particle size and morphology characterization of nanocarriers.
- Evaluation of stimuli-responsive behavior and release kinetics.
- Assessment of stability and drug delivery performance.
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
How do targeted polymers improve drug delivery efficiency?
Targeted polymers enhance drug delivery efficiency by enabling controlled and site-specific release of therapeutic agents. By responding to biological or external stimuli, these systems improve drug accumulation at target tissues, reduce systemic toxicity, and increase the overall therapeutic efficacy of the treatment.
-
What polymers are commonly used in targeted delivery?
Common polymers used in targeted drug delivery include PEG-based polymers, PLGA, polycaprolactone (PCL), and stimuli-responsive materials such as PNIPAM and pH-sensitive copolymers. These polymers offer tunable properties, good biocompatibility, and versatile structures suitable for nanoparticles, micelles, and hydrogels.
-
Are These systems safe for clinical use?
Many polymer-based drug delivery systems use biocompatible and biodegradable materials that have been widely studied for biomedical applications. Polymers such as PEG, PLGA, and PCL have demonstrated favorable safety profiles and are used in several FDA-approved formulations and clinical drug delivery platforms.
-
What are the current challenges and limitations?
Key challenges include complex polymer synthesis, scalability of manufacturing, and ensuring consistent drug release behavior in vivo. Additional concerns involve long-term biocompatibility, regulatory approval processes, and maintaining stability and responsiveness of the polymer carriers under physiological conditions.

 Fig. 1. Illustration of physical and chemical stimuli-triggered drug release mechanisms (BOC Sciences Authorized).
Fig. 1. Illustration of physical and chemical stimuli-triggered drug release mechanisms (BOC Sciences Authorized). Fig. 2. Common stimuli-responsive polymer nanostructures for targeted drug delivery systems (BOC Sciences Authorized).
Fig. 2. Common stimuli-responsive polymer nanostructures for targeted drug delivery systems (BOC Sciences Authorized).