How Polymer Particles Improve Polysaccharide & Glycan Delivery Efficacy?
The successful delivery of complex biomolecules remains a primary hurdle in modern formulation science. While polysaccharides and glycans possess immense potential for advanced therapeutics, their inherent physicochemical properties often limit their bioavailability and targetability. Polymer particles have emerged as a sophisticated solution, providing a customizable, highly engineered vehicle to encapsulate, protect, and deliver these complex carbohydrates. By leveraging advanced polymer chemistry, researchers can dramatically improve the pharmacokinetic profiles of glycans, ensuring they reach their intended biological targets with high efficiency.
Resources
Introduction to Polymer Particles for Polysaccharide & Glycan Delivery
Polymers for polysaccharide & glycan delivery are engineered carrier systems designed to overcome the inherent challenges of delivering large, complex carbohydrate molecules. Polysaccharides and glycans often face enzymatic degradation, poor solubility, rapid clearance, and limited bioavailability, which significantly restrict their therapeutic potential. Polymer particles—ranging from nanoparticles and microparticles to micelles and hydrogel-based multicompartment structures—offer versatile solutions by encapsulating these molecules, protecting them from degradation, and enabling controlled or stimuli-responsive release. Through careful selection of biodegradable and biocompatible polymers such as PLGA, PLA, PEG, and PCL, combined with surface functionalization or ligand conjugation, these carriers can be tailored for targeted delivery, enhanced cellular uptake, and optimized biodistribution.
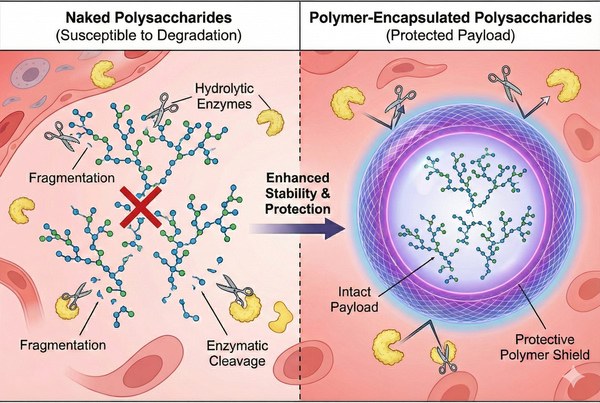 Fig. 1. Polymer nanoparticles protect fragile polysaccharides from rapid enzymatic degradation (BOC Sciences Authorized).
Fig. 1. Polymer nanoparticles protect fragile polysaccharides from rapid enzymatic degradation (BOC Sciences Authorized).
Applications span pharmaceutical development, including vaccines, immunomodulatory therapies, and cancer treatments, as well as nutraceuticals and functional foods, where precise release and improved bioavailability are critical. Advanced formulation strategies, such as emulsion–solvent evaporation, self-assembly, and spray-drying, allow fine control over particle size, morphology, and surface properties, while rigorous characterization using techniques like NMR, GPC, and FTIR ensures reproducibility, stability, and functional performance. Overall, polymer particles represent a powerful platform to expand the therapeutic and functional potential of polysaccharides and glycans in both research and clinical applications.
The Challenges of Polysaccharide and Glycan Therapeutics
Developing effective glycan-based formulations requires overcoming a series of physiological and structural hurdles that naturally degrade or eliminate these molecules before they can exert their biological effects.
- Structural Complexity of Polysaccharides and Glycans: Polysaccharides and glycans exhibit high molecular heterogeneity, characterized by varying chain lengths, branching patterns, and charge densities. This complex architecture complicates uniform formulation. Their high molecular weight and hydrophilicity naturally restrict their ability to passively diffuse across lipid bilayer membranes, resulting in extremely low intracellular concentration without an optimized delivery vehicle.
- Biological Barriers to Oral and Systemic Delivery: Upon administration, naked glycans face immediate physiological defense mechanisms. In the gastrointestinal tract, extreme pH variations and high concentrations of hydrolytic enzymes rapidly degrade carbohydrate structures. Systemically administered glycans are highly susceptible to enzymatic cleavage by glycosidases in the bloodstream. Furthermore, their foreign structural motifs often trigger rapid recognition and immune clearance by the reticuloendothelial system, preventing them from accumulating at the target tissue.
- Stability and Shelf-Life Limitations: Beyond biological barriers, polysaccharides suffer from inherent chemical instabilities during storage. Hydrolysis and oxidative degradation can easily compromise their structural integrity. Maintaining stability over an extended shelf-life typically requires strict environmental controls, making the formulation process technically demanding and costly without protective carrier matrices.
- Need for Advanced Carrier Systems: To transition glycans from promising biochemical compounds to viable therapeutic candidates, advanced carrier systems are strictly required. Polymer particles serve as protective shields and functional transporters, bridging the gap between the fragile nature of polysaccharides and the robust requirements of in vivo biological environments.
How Polymer Particles Overcome Glycan Delivery Barriers?
Polymer particles neutralize physiological barriers through targeted encapsulation and intelligent surface engineering, transforming fragile carbohydrate molecules into robust delivery systems.
- Biocompatibility and Biodegradability: The foundation of any successful delivery system lies in its interaction with human physiology. Advanced delivery systems utilize polymers specifically selected for their high biocompatibility and predictable degradation pathways. As these polymers break down into non-toxic metabolic byproducts, they ensure the safe release of the glycan payload without eliciting chronic inflammatory responses or long-term tissue toxicity.
- Physical Encapsulation and Steric Shielding: By entrapping polysaccharides within a dense polymeric matrix, the carrier system provides a physical barrier against circulating degrading enzymes. This steric shielding prevents premature degradation, ensuring the molecular payload remains entirely intact during transit through hostile physiological environments like the gastrointestinal tract or the systemic circulation.
- Enhancing Cellular Uptake and Endosomal Escape: Polymers can be engineered to interact favorably with cellular membranes. Cationic polymers, for example, facilitate electrostatic interaction with negatively charged cell surfaces, promoting rapid endocytosis. Once inside the cell, specific polymer formulations utilize the proton sponge effect to rupture endosomal vesicles, allowing the encapsulated glycans to escape into the cytoplasm before being destroyed in acidic lysosomes.
- Pharmacokinetic Modulation via PEGylation: Adding polyethylene glycol to the surface of polymer nanoparticles creates a hydrophilic hydration shell around the carrier. This modification fundamentally alters the pharmacokinetic profile by preventing protein adsorption and subsequent recognition by circulating macrophages. The resulting prolonged circulation time significantly increases the probability of the glycan cargo reaching its intended target site.
Mechanisms of Enhanced Delivery Using Polymer Particles
The enhancement of delivery efficacy is not a passive process but rather the result of highly engineered functional mechanisms designed into the polymer matrix.
- Nano/Microencapsulation Strategies: The formation of polymer particles dictates the final distribution and protection of the glycan. Emulsification techniques are highly effective for creating uniform microspheres, while nanoprecipitation allows for the rapid generation of sub-micron particles with tightly controlled size distributions. Self-assembly of amphiphilic block copolymers presents another elegant method for forming core-shell micelles that are highly effective at protecting hydrophilic carbohydrate payloads.
- Surface Modification and Functionalization: Beyond passive PEGylation, polymer particles can be actively functionalized to direct biological traffic. Conjugating specific targeting ligands, antibodies, or peptides to the polymer surface allows the particle to bind exclusively to specific cellular receptors. Additionally, fine-tuning the surface charge can dictate exactly how the particle interacts with mucosal surfaces or cellular barriers, driving enhanced cellular uptake.
- Stimuli-Responsive Release Mechanisms: Modern polymer engineering allows for the creation of smart delivery vehicles that only release their payload when triggered by specific microenvironmental cues. These triggers include a drop in pH characteristic of tumor microenvironments or endosomes, temperature fluctuations, or the presence of specific enzymes that cleave the polymer matrix, resulting in a highly localized, controlled release of the glycan.
- Improved Bioavailability and Pharmacokinetics: Through a combination of prolonged circulation, enhanced mucosal penetration, and protection from premature metabolism, polymer particles exponentially increase the overall bioavailability of polysaccharides. They reduce rapid renal clearance and enhance tissue penetration via the enhanced permeability and retention effect, specifically beneficial in targeted tissue accumulation.
Strategic Selection of Polymer Matrices for Polysaccharide and Glycan Delivery
The foundational success of any carbohydrate-based delivery system relies entirely on the strategic selection of the underlying polymer matrix. Because polysaccharides and glycans exhibit vast structural diversity, high molecular weights, and extreme hydrophilicity, a one-size-fits-all approach is practically impossible. Formulation scientists must carefully match the physicochemical properties of the polymer to the specific structural characteristics of the glycan payload. This selection dictates the encapsulation efficiency, the structural integrity of the carbohydrate during transit, the precise release kinetics, and the ultimate biological fate of the delivery vehicle. By leveraging distinct polymer architectures, researchers can engineer highly customized microenvironments that protect and propel these fragile biomolecules.
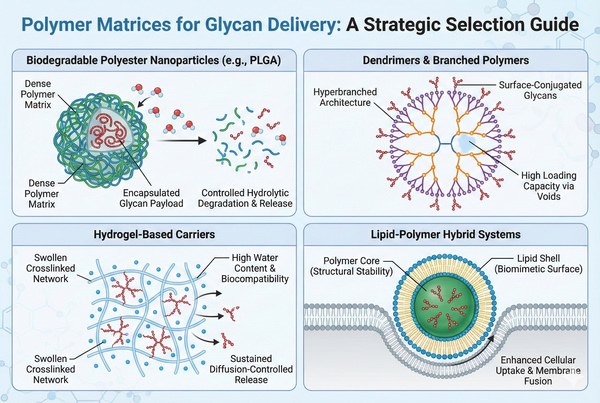 Fig. 2. Structural comparison of four main polymer matrices for glycan delivery (BOC Sciences Authorized).
Fig. 2. Structural comparison of four main polymer matrices for glycan delivery (BOC Sciences Authorized).
Aliphatic polyesters, primarily poly-lactic acid (PLA), poly-lactic-co-glycolic acid (PLGA), and polycaprolactone (PCL), serve as the foundational gold standard for systemic particulate delivery. These materials are deeply valued for their well-characterized and highly predictable degradation profiles.
- Mechanism of Action: They degrade primarily through the hydrolysis of their ester bonds in aqueous environments, breaking down into safe, natural metabolic byproducts like lactic and glycolic acids.
- Customizable Release: By altering the specific lactide-to-glycolide ratio within PLGA formulations, scientists can precisely tune the degradation rate from a few weeks to several months.
- Glycan Compatibility: For polysaccharide delivery, these dense hydrophobic matrices are exceptionally adept at completely shielding water-soluble carbohydrate payloads from circulating hydrolytic enzymes, preventing premature degradation during systemic circulation.
When the delivery strategy requires localized administration and sustained, long-term release, highly crosslinked polymer hydrogels offer an unparalleled structural solution.
- Biomimetic Environment: Hydrogels possess a three-dimensional macromolecular network capable of retaining massive amounts of water. This high hydration state closely mimics natural physiological tissue, providing a thermodynamically favorable environment that prevents the structural denaturation of complex, heavily branched glycans.
- Diffusion Control: The release of the polysaccharide is governed by the mesh size of the polymer network. By precisely controlling the crosslinking density during polymer synthesis, formulation engineers can tailor the spatial gaps within the gel.
- Stimuli Responsiveness: Advanced hydrogel systems can be engineered using smart polymers that swell or deswell in response to local microenvironmental triggers like pH shifts or temperature changes, allowing for on-demand glycan release exactly at the biological target site.
Lipopolymer Conjugates and Hybrid Systems
Highly complex delivery challenges often require integrating the distinct advantages of multiple material classes. Lipid-polymer hybrid nanoparticles represent a sophisticated convergence of biomimetic lipids and robust synthetic polymers.
- Core-Shell Architecture: These systems typically feature a solid polymeric core that securely entraps the hydrophilic polysaccharide, providing high structural stability and controlled release kinetics. Surrounding this core is a lipid monolayer or bilayer shell.
- Enhanced Interfacial Properties: The lipid shell drastically reduces the interfacial tension between the aqueous biological environment and the polymer core, significantly minimizing the risk of premature payload leakage.
- Membrane Interaction: Because the lipid shell structurally resembles biological cell membranes, these hybrid particles exhibit vastly superior cellular internalization rates compared to bare polymer particles, making them exceptionally effective for driving large glycans into the intracellular space.
Transitioning away from physical encapsulation, polymer-drug conjugates utilize direct covalent chemistry to physically bind the glycan to a polymer backbone, creating highly stable macromolecular prodrugs.
- Absolute Payload Retention: By forming strong chemical bonds (often via click chemistry, esterification, or amidation), this strategy completely eliminates the problem of burst release—a common phenomenon where physically encapsulated drugs leak out immediately upon administration.
- Stoichiometric Precision: Covalent conjugation allows for exact control over the carbohydrate-to-polymer ratio, ensuring highly uniform loading capacity across the entire formulation batch.
- Triggered Cleavage: The chemical linkers connecting the polymer and the glycan are often designed to be biologically responsive. They remain intact in systemic circulation but are rapidly cleaved by specific enzymes once internalized by the target cell, ensuring the glycan is only released exactly where it is biologically active.
Dendrimers and Branched Polymers
For applications demanding immense precision and multi-valent presentation, dendrimers provide a structurally perfect, highly symmetrical, and heavily branched polymeric architecture.
- High-Density Encapsulation: Synthesized through a controlled, stepwise process resulting in distinct generations, dendrimers feature spacious internal cavities that are highly effective at entrapping smaller glycan structures or specific carbohydrate active sites.
- Surface Functionalization: The defining feature of dendrimers is the exponential number of functional groups located on their exterior surface. This allows formulation scientists to conjugate an massive number of targeting ligands, shielding molecules like polyethylene glycol, or even the active glycans themselves.
- Multivalent Effects: By densely packing glycans on the surface of a dendrimer, researchers can exploit the multivalent effect, where simultaneous interaction with multiple cellular receptors exponentially increases binding affinity and targeted cellular uptake compared to monovalent delivery methods.
Looking for Custom Polymers for Glycan Delivery?
Whether you need cationic polymers, biodegradable carriers, or stimuli-responsive platforms, our experts provide tailored synthesis and functionalization solutions to support your research and therapeutic development.
Design Considerations for Optimal Delivery Efficacy
The transition of a polymer-glycan formulation from a conceptual design to a highly effective in vivo delivery system depends entirely on the meticulous engineering of its physical and chemical parameters. Because polysaccharides are inherently fragile and possess complex architectural motifs, the polymer carrier cannot be a generic vessel. Formulation scientists must rigorously tune the geometry, surface chemistry, and internal matrix mechanics of the particle to perfectly match the specific biological barriers it will encounter. These foundational design considerations directly dictate the loading capacity, the structural stability of the carbohydrate payload, and the ultimate pharmacokinetic success of the delivery vehicle.
Particle Size, Shape, and Polydispersity
The geometric profile of a polymer particle is the primary determinant of its biological fate, governing everything from circulation half-life to cellular internalization pathways.
- Size Optimization: For systemic administration, precise size control is critical. Polymer nanoparticles must typically be engineered to fall between 50 and 200 nanometers. Particles within this specific dimensional window are large enough to avoid rapid renal clearance but small enough to evade detection and filtration by the reticuloendothelial system in the liver and spleen.
- Morphological Influence: While spherical particles are the standard due to thermodynamic stability during formulation, non-spherical geometries, such as rods or discs, are increasingly utilized. These anisotropic shapes fundamentally alter fluid dynamics within blood vessels, promoting superior margination—the process by which particles drift toward the blood vessel walls—thereby enhancing tissue accumulation.
- Polydispersity Index: Maintaining a low polydispersity index is non-negotiable for formulation consistency. A narrow size distribution ensures that every particle within the dose behaves identically in a biological environment, providing highly predictable release kinetics and reproducible biodistribution profiles.
Polymer Chemistry and Functionalization
The foundational chemistry of the polymer matrix must be chemically compatible with the highly hydrophilic and structurally diverse nature of polysaccharides.
- Hydrophilic and Hydrophobic Balance: Since glycans are exceptionally water-soluble, encapsulating them within a purely hydrophobic polymer matrix often results in low loading efficiencies. Formulation engineers must utilize amphiphilic block copolymers or carefully blend hydrophilic excipients to create an internal microenvironment that readily accommodates the carbohydrate payload without compromising the structural integrity of the outer particle shell.
- Surface Charge Tuning: The zeta potential, or surface charge, of the polymer particle heavily influences biological interactions. Cationic polymers are frequently selected to create a net-positive surface charge, which facilitates strong electrostatic interactions with negatively charged cellular membranes, driving rapid endocytosis and enhancing mucosal adhesion in gastrointestinal or respiratory delivery routes.
- Molecular Weight and Branching: The architectural complexity of the polymer itself—specifically its molecular weight and degree of branching—directly controls the degradation rate of the matrix. Higher molecular weights generally result in denser, slower-degrading particles, allowing researchers to precisely dial in the release profile from days to several months.
Loading Efficiency and Encapsulation Stability
Maximizing the amount of active glycan securely stowed within the polymer matrix is a primary objective in formulation science, directly impacting the overall safety and efficacy of the system.
- Maximizing Payload Capacity: Achieving high encapsulation efficiency minimizes the total mass of the polymer carrier required per dose. This reduction in excipient burden is crucial for preventing potential material toxicity and ensuring the physiological clearance mechanisms are not overwhelmed by degraded polymer byproducts.
- Preserving Structural Integrity: The encapsulation process itself poses a significant threat to delicate polysaccharides. Techniques involving high shear stress, extreme temperature fluctuations, or harsh organic solvents can cause irreversible denaturation or cleavage of the glycan chains. Formulators must select gentle assembly methods, such as nanoprecipitation or self-assembly, to maintain the absolute structural fidelity of the carbohydrate during particle formation.
- Preventing Burst Release: A poorly designed polymer matrix will exhibit a massive initial release of the payload immediately upon administration. By optimizing the crosslinking density and ensuring the glycan is deeply embedded within the core rather than adsorbed on the surface, scientists can mitigate this burst effect and achieve a steady, zero-order release profile.
In Vitro and In Vivo Evaluation Parameters
Rigorous analytical and biological evaluations are required to validate the physicochemical design and confirm that the polymer particle effectively enhances the delivery of the polysaccharide.
- Physicochemical Characterization: Essential analytical techniques include dynamic light scattering to verify particle size and polydispersity, electrophoretic light scattering for zeta potential, and advanced electron microscopy to confirm particle morphology and structural uniformity.
- Release Kinetics Profiling: In vitro release assays utilize specialized dialysis methods in physiologically simulated fluids. These tests monitor the exact diffusion rate of the glycan out of the polymer matrix under varying pH and temperature conditions, ensuring the release trigger functions exactly as engineered.
- Cytotoxicity and Biodistribution: Preclinical biological assessments are strictly required. Comprehensive in vitro cytotoxicity panels across multiple cell lines ensure the polymer matrix and its degradation products are entirely biocompatible. Subsequently, advanced in vivo biodistribution studies utilizing fluorescent or radiolabeled tracking confirm that the particles successfully navigate physiological barriers and accumulate efficiently at the targeted tissue sites.
Application Scenarios of Polymer Particles in Glycan Delivery
The successful integration of advanced polymer matrices with complex polysaccharides has fundamentally transformed the landscape of formulation science. By overcoming the inherent physicochemical limitations of naked glycans, researchers can now deploy these biomolecules across a wide spectrum of advanced pharmaceutical research areas. These sophisticated delivery vehicles allow scientists to harness the immense biological potential of carbohydrates, translating them from highly unstable raw materials into robust, precisely targeted developmental therapeutics.
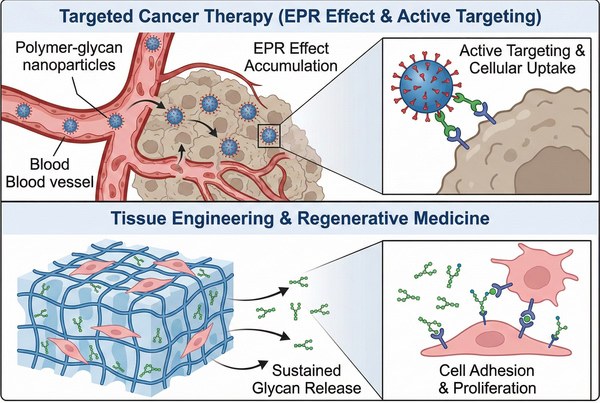 Fig. 3. Polymer-glycan applications in targeted cancer therapy and tissue engineering (BOC Sciences Authorized).
Fig. 3. Polymer-glycan applications in targeted cancer therapy and tissue engineering (BOC Sciences Authorized).
Glycan-Based Vaccines and Adjuvants
Polymer particles are currently revolutionizing the development of advanced vaccine platforms by providing structurally stable delivery vehicles for carbohydrate-based antigens and immune adjuvants.
- Antigen Protection: Carbohydrate antigens are notoriously susceptible to rapid enzymatic degradation upon injection. Encapsulating them within a biodegradable polymer matrix ensures they remain structurally intact until they reach the lymphatic system.
- Enhanced Presentation: Polymers can be explicitly engineered to target antigen-presenting cells, such as dendritic cells and macrophages. By tuning the particle size to mimic the dimensions of natural pathogens, these formulations stimulate robust phagocytosis, leading to superior antigen processing and presentation in developmental immunology models.
- Controlled Adjuvant Release: Formulators frequently co-encapsulate polysaccharides with specific immune adjuvants. The polymer matrix allows for the synchronized, sustained release of both components, generating a highly potent, durable immune response without causing severe localized inflammation.
Targeted Cancer and Anti-Inflammatory Therapies
In oncology and inflammatory disease research, polymer-glycan nanocarriers provide an unprecedented mechanism for achieving highly localized, targeted accumulation of active compounds.
- Exploiting the EPR Effect: Solid tumors and heavily inflamed tissues characteristically exhibit highly permeable, leaky vasculature and poor lymphatic drainage. Formulators design polymer nanoparticles within a specific size range (typically 50 to 150 nanometers) to exploit this enhanced permeability and retention effect, allowing the polysaccharide payload to passively accumulate exactly at the disease site.
- Active Targeting Ligands: Beyond passive accumulation, polymer surfaces can be functionalized with specific targeting moieties. For example, grafting hyaluronic acid onto a polymer carrier actively directs the nanoparticle to CD44 receptors, which are heavily overexpressed on numerous cancer cell lines, ensuring highly specific intracellular delivery.
- Microenvironment Triggers: Advanced formulations utilize stimuli-responsive polymers that remain completely stable in systemic circulation but rapidly degrade and release their glycan payload when exposed to the acidic pH or specific enzymatic profiles characteristic of tumor microenvironments.
Immunomodulation and Macrophage Targeting
Certain polysaccharides possess profound immunomodulatory capabilities, capable of upregulating or suppressing specific immune pathways. Polymer delivery systems are engineered to direct these effects with high precision.
- Receptor-Mediated Uptake: Macrophages heavily express specific surface receptors, such as mannose and scavenger receptors. By formulating polymer particles with mannosylated surfaces or specific charge densities, researchers can actively drive the accumulation of immunomodulatory glycans directly into these critical immune cells.
- Macrophage Polarization: In preclinical research targeting autoimmune conditions or tissue repair, polymer-delivered polysaccharides are utilized to force macrophage polarization. By delivering specific glycan payloads, researchers can effectively switch local macrophage populations from a pro-inflammatory state to a tissue-healing, anti-inflammatory state.
- Systemic Toxicity Reduction: Because naked immunomodulators can cause severe systemic cytokine storms if administered freely, encapsulating them within targeted polymer nanoparticles ensures that the immune activation remains strictly localized to the intended cellular targets.
Tissue Engineering and Regenerative Medicine
The intersection of polymer chemistry and structural glycans has created a highly fertile ground for advancements in regenerative medicine and synthetic tissue scaffolding.
- Biomimetic Scaffolds: Naturally occurring glycosaminoglycans are essential components of the human extracellular matrix. Formulation scientists create advanced hybrid hydrogels combining synthetic polymers with these structural glycans to fabricate artificial tissue scaffolds that precisely mimic the mechanical and biological properties of native tissue.
- Sustained Bioactive Release: These polymeric scaffolds do not merely provide structural support; they act as massive, localized drug depots. They are engineered to slowly degrade over weeks or months, continuously releasing bioactive polysaccharides that promote cellular adhesion, drive proliferation, and guide the differentiation of seeded stem cells in developmental models.
- Cartilage and Bone Regeneration: Specific polymer-glycan composites are heavily utilized in orthopedic research. By tuning the compressive strength of the polymer and incorporating chondrogenic or osteogenic polysaccharides, researchers can significantly accelerate the regeneration of articular cartilage and bone defects in specialized in vivo models.
Oral and Injectable Formulations
Converting highly unstable glycans into viable, scalable formulation formats requires extensive polymer engineering to overcome strict anatomical barriers.
- Overcoming Gastrointestinal Barriers: Oral delivery of polysaccharides is notoriously difficult due to extreme stomach acidity and poor intestinal permeability. Researchers utilize pH-sensitive, enteric-coated polymer particles that remain completely insoluble in the stomach but rapidly dissolve in the neutral pH of the intestines.
- Mucoadhesive Technologies: To enhance oral absorption, polymers like chitosan or thiolated derivatives are employed. These materials exhibit strong mucoadhesive properties, anchoring the nanoparticle to the intestinal lining and dramatically increasing the residence time and local concentration gradient required for passive diffusion or active transport.
- Injectable Depot Systems: For long-term systemic delivery, scientists formulate in situ forming polymer implants or precisely engineered microparticles. Upon subcutaneous or intramuscular injection, these polymer systems establish a localized depot that steadily biodegrades, providing tightly controlled, zero-order release of the polysaccharide payload over several months, entirely eliminating the need for frequent dosing in preclinical testing regimens.
Choosing the Right Polymer for Your Polysaccharide Delivery Needs
Selecting the optimal polymer matrix is the most consequential decision in the entire formulation development pipeline. Because polysaccharides and glycans exhibit vast structural diversity, variable molecular weights, and complex charge profiles, no single polymer can serve as a universal carrier. Formulation scientists must deliberately match the physicochemical properties of the synthetic or natural polymer to the specific structural demands of the carbohydrate payload. This strategic alignment dictates the initial encapsulation efficiency, governs the precise release kinetics in physiological environments, and ultimately determines the success of the targeted delivery strategy. The selection process requires a rigorous evaluation of thermodynamic properties, biological interactions, and practical manufacturing constraints.
- Biodegradable vs Non-Biodegradable Polymers: The fundamental degradation profile of the carrier system dictates its utility in preclinical research models. Biodegradable polymers, such as poly-lactic-co-glycolic acid or polycaprolactone, are overwhelmingly preferred for systemic administration and internal tissue targeting. They undergo controlled hydrolytic or enzymatic cleavage, breaking down into safe metabolic byproducts that are easily cleared by regular cellular respiration. Non-biodegradable polymers, conversely, are typically restricted to localized, permanent implants or external ex vivo applications where maintaining absolute, long-term structural mass is required and physiological clearance is not the primary objective.
- Molecular Weight and Polymer Chain Architecture: The macromolecular design of the polymer backbone directly controls the mechanical strength and degradation rate of the delivery vehicle. High molecular weight linear polymers form dense, highly entangled matrices that slow water penetration and dramatically extend the release duration of the encapsulated glycan. Alternatively, modifying the chain architecture to create branched, star-shaped, or heavily crosslinked networks allows researchers to manipulate the internal free volume of the particle. These architectural modifications alter fluid dynamics within the matrix, providing precise control over the diffusion rate of complex, heavily branched polysaccharides.
- Surface Functionalization for Targeted Delivery: The exterior chemistry of the polymer particle determines how the formulation interacts with biological barriers. Bare polymer surfaces are often subjected to rapid opsonization and clearance by the reticuloendothelial system. By selecting polymers with readily accessible reactive end-groups, formulators can easily attach hydrophilic shielding molecules like polyethylene glycol to extend circulation time. Furthermore, these reactive sites allow for the precise conjugation of specific targeting ligands, such as peptides or aptamers, directing the biologically active glycan exclusively toward specifically engineered cellular receptors.
- Hydrophilic vs Hydrophobic Polymer Characteristics: Matching the hydrophilic-lipophilic balance between the polymer and the payload is critical for maximizing encapsulation efficiency. Because polysaccharides are intensely hydrophilic, utilizing a purely hydrophobic polymer matrix often forces the carbohydrate to partition out of the particle during the formulation process. To resolve this, formulation scientists heavily rely on amphiphilic block copolymers. These highly engineered materials self-assemble in aqueous environments, creating a thermodynamically stable core-shell structure that firmly sequesters the hydrophilic glycan within a protected aqueous core while presenting an isolating hydrophobic barrier to the external environment.
- Stimuli-Responsive and Smart Polymers: Advanced targeted delivery relies heavily on stimuli-responsive materials that remain structurally dormant during systemic transit but activate precisely at the biological target. These smart polymers undergo rapid phase transitions, swelling, or structural collapse in response to specific microenvironmental triggers. By integrating pH-sensitive linkages, thermosensitive blocks, or redox-responsive disulfide bonds into the polymer backbone, researchers can ensure the polysaccharide payload is only released when it encounters the acidic environment of an endolysosomal compartment or the highly reductive environment of a targeted cellular cytoplasm.
- Safety, Biocompatibility, and Regulatory Considerations: Before a polymer formulation can advance through preclinical testing regimens, its underlying material must demonstrate unquestionable safety. Researchers must prioritize polymers with well-documented biocompatibility profiles to ensure that neither the intact matrix nor its degradation byproducts elicit unintended localized inflammation or systemic cytotoxicity. Utilizing materials that are generally recognized as safe streamlines the research and development process, ensuring that the foundational carrier system adheres to strict foundational safety parameters necessary for advancing highly complex glycan therapeutics.
- Polymer Selection for Specific Polysaccharide Types: The intrinsic electrostatic charge and three-dimensional conformation of the specific glycan heavily influence polymer selection. For intensely negatively charged carbohydrates, such as specific glycosaminoglycans or heparin derivatives, cationic polymers like chitosan or polyethylenimine are highly favored. These oppositely charged materials spontaneously form dense polyelectrolyte complexes through strong electrostatic interactions, yielding exceptionally stable nanoparticles. Conversely, for neutral, massively branched glycans, researchers often rely on physical entrapment within porous hydrogels or precisely engineered aliphatic polyester microspheres.
- Scalability and Manufacturing Feasibility: A conceptually brilliant polymer matrix is biologically useless if it cannot be reproducibly synthesized and formulated at scale. Formulation scientists must evaluate the ease of chemical synthesis, the requirement for highly toxic organic solvents during particle assembly, and the long-term stability of the final product during rigorous lyophilization processes. Partnering with specialized chemical providers ensures access to high-purity, batch-consistent polymer materials, which is strictly required to minimize variability and guarantee reproducible pharmacokinetic results across extensive research studies.
Services
Tailored Polymeric Particles for Efficient Polysaccharide & Glycan Delivery
Delivering polysaccharides and glycans effectively requires carriers that balance stability, loading efficiency, and controlled release—challenges that many researchers face. At BOC Sciences, we provide end-to-end polymeric particle solutions designed to meet these exact needs. From selecting the right polymer materials and customizing particle architecture to optimizing encapsulation strategies and surface functionality, our services help maximize payload protection and bioavailability. Whether your goal is oral delivery, targeted cellular uptake, or sustained release, we work closely with you to translate your concept into reproducible, application-ready polymeric particles that perform in real-world biological settings.

Polymer Materials Supply for Glycan & Polysaccharide Delivery
- Providing PLGA, PLA, PEG, PCL, and functionalized copolymers
- Customizable molecular weight, end groups, branching, and functional moieties
- Incorporation of PEG, targeting ligands, or stimuli-responsive groups (pH/enzymes/ROS)
- Optimizing hydrophilic–hydrophobic balance to improve polysaccharide encapsulation and delivery

Polymeric Particle Formulation Development
- Fabrication of nano/microparticles, micelles, or multicompartment structures
- Supporting self-assembly, emulsion–solvent evaporation, spray-drying, and other processes
- Control over particle size, polydispersity index, surface charge, and morphology
- Surface charge and hydrophobicity tuning to enhance biodistribution

Polysaccharide & Glycan Loading and Encapsulation
- Applicable to natural polysaccharides, oligosaccharides, and glycan derivatives
- Enhancing encapsulation efficiency, loading capacity, and uniform distribution
- Controlling release rates and enabling stimuli-triggered delivery
- Customizing particle structure and release strategy for specific applications

Characterization & Quality Evaluation
- Analysis of particle size, PDI, surface charge, and morphology
- Polymer and particle structural characterization (NMR, GPC, FTIR)
- Assessment of polysaccharide encapsulation efficiency, loading capacity, and release profiles
- Evaluation of stability, batch-to-batch consistency, and functional performance
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
Why use polymer particles for polysaccharide delivery?
Polysaccharides inherently suffer from severe physical and biological limitations when administered in their naked form. They are highly susceptible to rapid enzymatic degradation by glycosidases, exhibit poor membrane permeability due to their immense hydrophilicity, and face swift immune clearance. Polymer particles act as highly engineered, protective vehicles that physically shield the carbohydrate payload from hydrolytic enzymes, prevent premature structural breakdown, and significantly enhance cellular membrane penetration, ensuring the biomolecules reach their functional destination intact.
How do polymer particles enhance glycan stability and bioavailability?
Polymer particles fundamentally enhance stability by encasing the fragile glycan within a robust, dense matrix, effectively isolating it from harsh pH environments and circulating degrading enzymes. Bioavailability is exponentially increased because the polymer surface can be precisely engineered to prolong systemic circulation through hydrophilic shielding. Furthermore, these carrier systems facilitate active transport across strict mucosal barriers and promote targeted cellular internalization via specific endocytic pathways, preventing the rapid clearance that typically eliminates unencapsulated carbohydrates.
Can polymer particles provide controlled and sustained release?
Yes. The highly customizable nature of polymer chemistry allows for precise control over payload release kinetics. By utilizing biodegradable aliphatic polyesters or by designing specific cross-linked hydrogel networks, the degradation rate of the matrix can be mathematically tuned. This controlled degradation mechanism allows for the steady, sustained diffusion of the encapsulated glycan over predetermined periods, ranging from several hours to multiple months, which is strictly required for long-term preclinical efficacy studies.
How are polymer particles customized for specific glycan therapeutics?
Customization occurs directly at the macromolecular level to match the specific physicochemical traits of the payload. Formulation scientists can adjust the polymer molecular weight, alter the hydrophilic-to-hydrophobic ratio to accommodate highly water-soluble glycans, and attach precise targeting ligands like peptides to the particle exterior. Additionally, formulators can incorporate stimuli-responsive chemical bonds that react exclusively to the target tissues local microenvironment, such as localized pH drops or specific enzymatic concentrations, triggering highly customized release profiles.
What types of polymer particles are commonly used for glycan delivery?
The selection depends entirely on the required release kinetics and structural characteristics of the payload. The most utilized systems in advanced research include biodegradable polyesters for systemic nanoparticles, highly crosslinked natural and synthetic hydrogels for localized depots, amphiphilic block copolymer micelles for core-shell protection, lipid-polymer hybrids for enhanced membrane fusion, and highly symmetrical branched structures like dendrimers for multivalent surface presentation.
What are the main mechanisms by which polymer particles enhance bioavailability?
The primary mechanisms include steric shielding, often achieved via surface functionalization with hydrophilic polymers, to evade rapid macrophage clearance and extend blood half-life. Other critical mechanisms involve the use of localized cationic charges to trigger robust receptor-mediated endocytosis, specialized endosomal escape strategies like the proton sponge effect to prevent lysosomal destruction, and the creation of an impermeable physical barrier against aggressive enzymatic cleavage in the gastrointestinal tract and blood plasma.
Are polymer particles safe for in vivo research and formulation development?
Yes. Many advanced polymer systems have a long, well-documented history of excellent biocompatibility and highly predictable, non-toxic degradation pathways. Specifically, aliphatic polyesters break down into natural metabolic byproducts like lactic and glycolic acids, which are easily processed and eliminated by normal cellular respiration cycles. This established safety profile makes them highly favored materials in rigorous in vivo formulation research and advanced preclinical modeling.
How can researchers choose the optimal polymer particle system for polysaccharide delivery?
Selecting the optimal matrix requires a rigorous, data-driven assessment of the glycans exact molecular weight, electrostatic charge, and solubility profile, combined with the intended route of administration. Researchers must continuously balance high loading efficiency requirements with the desired temporal release kinetics. This process heavily relies on empirical in vitro release assays and structural characterization techniques to finalize the exact polymer chemistry and particle geometry necessary for their specific biological target.

 Fig. 1. Polymer nanoparticles protect fragile polysaccharides from rapid enzymatic degradation (BOC Sciences Authorized).
Fig. 1. Polymer nanoparticles protect fragile polysaccharides from rapid enzymatic degradation (BOC Sciences Authorized). Fig. 2. Structural comparison of four main polymer matrices for glycan delivery (BOC Sciences Authorized).
Fig. 2. Structural comparison of four main polymer matrices for glycan delivery (BOC Sciences Authorized). Fig. 3. Polymer-glycan applications in targeted cancer therapy and tissue engineering (BOC Sciences Authorized).
Fig. 3. Polymer-glycan applications in targeted cancer therapy and tissue engineering (BOC Sciences Authorized).