Choosing the Right Drug Delivery System: Polymer vs Lipid vs Inorganic Carriers
As the industry moves from small, soluble molecules to complex biologics—including proteins, peptides, monoclonal antibodies, and nucleic acids—the limiting factor in clinical success is often no longer the potency of the active pharmaceutical ingredient (API), but its bioavailability. A drug delivery system (DDS) is defined not merely as a vehicle, but as a specialized interface between the drug and the biological environment. Its primary function is to modulate the pharmacokinetics (PK) and pharmacodynamics (PD) of the therapeutic agent. By controlling the rate, time, and place of release, a well-engineered DDS can transform a potent but toxic or unstable molecule into a viable clinical therapy. The modern approach to DDS design focuses on overcoming the body's natural defense mechanisms while ensuring the cargo reaches its intracellular or extracellular target in a bioactive state.
Resources
Overview of Polymer, Lipid, and Inorganic Drug Carriers
Current nanotechnology research categorizes delivery vehicles into three dominant classes, each defined by distinct physicochemical properties and assembly mechanisms:
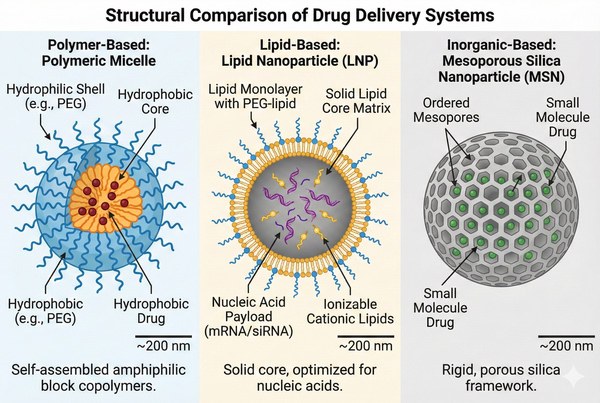 Fig. 1. Cross-sectional structural comparison of polymeric micelles, lipid nanoparticles, and mesoporous silica nanoparticles (BOC Sciences Authorized).
Fig. 1. Cross-sectional structural comparison of polymeric micelles, lipid nanoparticles, and mesoporous silica nanoparticles (BOC Sciences Authorized).
- Polymer-Based Systems: These are perhaps the most versatile class, ranging from natural biopolymers (e.g., Chitosan, Alginate) to synthetic macromolecules (e.g., PLGA, PEG, PCL). Polymers offer unparalleled control over architecture—allowing for the creation of dendrimers, hydrogels, and polymeric micelles. Their primary strength lies in chemical flexibility, enabling precise tuning of molecular weight, degradation kinetics, and surface functionality for ligand conjugation.
- Lipid-Based Systems: Comprising liposomes, Solid lipid nanoparticles (SLNs), and nanostructured lipid carriers (NLCs), these systems mimic biological membranes. Their amphiphilic nature allows for the encapsulation of both hydrophilic drugs (in the aqueous core) and hydrophobic drugs (in the lipid bilayer). They are currently the gold standard for nucleic acid delivery, as demonstrated by the global success of LNP-based mRNA vaccines.
- Inorganic Systems: This category includes mesoporous silica nanoparticles (MSNs), gold nanoparticles (AuNPs), iron oxide nanoparticles (SPIONs), and carbon nanotubes. Unlike organic carriers, inorganic systems provide a rigid structural framework that resists environmental stress. They are unique in their intrinsic physical properties—such as superparamagnetism or surface plasmon resonance—which enables theranostic applications (simultaneous therapy and imaging).
Key Challenges in Drug Delivery
The human body possesses sophisticated barriers designed to exclude foreign substances, presenting a series of biological hurdles that a carrier must overcome sequentially:
- Solubility and Stability: Approximately 40% of marketed drugs and 90% of drugs in the discovery pipeline are poorly water-soluble (BCS Class II and IV). Furthermore, biologics are prone to enzymatic degradation (by proteases or nucleases) and physical instability (aggregation) in physiological fluids.
- The Reticuloendothelial System (RES): Upon intravenous administration, opsonins (serum proteins) adsorb onto the surface of nanoparticles. This signals macrophages in the liver (Kupffer cells) and spleen to rapidly clear the carriers from circulation, drastically reducing the drug's half-life.
- Blood-Brain Barrier (BBB): The tight junctions of the BBB exclude 98% of small molecule drugs and nearly 100% of large molecules, making CNS drug delivery a significant challenge.
- The Tumor Microenvironment (TME): Solid tumors possess a dense interstitial matrix and high interstitial fluid pressure (IFP), which restricts the penetration of nanocarriers into the tumor core.
- Intracellular Trafficking: Even if a carrier reaches the target cell, it must undergo cellular uptake (endocytosis) and, crucially, endosomal escape. Failure to escape the endosome results in the lysosomal degradation of the payload, rendering the therapy ineffective.
Importance of Carrier Selection for Therapeutic Success
The selection of a drug carrier is a strategic decision that dictates the Therapeutic Index (TI) of the final formulation. The carrier must be matched to the drug's physicochemical identity and the disease pathology.
- Improving Bioavailability: For hydrophobic drugs, the right carrier (e.g., a polymeric micelle or lipid nanoparticle) can increase apparent solubility by orders of magnitude, enhancing absorption and bioavailability.
- Modulating Pharmacokinetics: Carrier selection determines whether a drug exhibits a "burst release" (useful for acute pain) or "sustained release" (vital for hormonal therapies or chronic inflammation), thereby maintaining plasma concentrations within the therapeutic window for extended periods.
- Targeting Efficiency:
- Passive Targeting: Utilizing the enhanced permeability and retention (EPR) effect, where carriers naturally accumulate in the leaky vasculature of tumors.
- Active Targeting: Conjugating specific ligands (antibodies, aptamers, peptides) to the carrier surface to bind receptors overexpressed on diseased cells (e.g., Folate or Transferrin receptors), minimizing off-target toxicity to healthy tissues.
Ultimately, the choice between a polymer, lipid, or inorganic carrier is a balance between loading capacity, stability, biocompatibility, and manufacturing scalability. Understanding these trade-offs is the first step toward successful clinical translation.
Polymer-Based Drug Delivery Systems
Polymer therapeutics has evolved from simple extended-release formulations to sophisticated nanomedicines capable of sensing and responding to the biological environment. Unlike lipids or inorganic carriers, polymers offer virtually infinite structural diversity. By manipulating monomer composition, molecular weight, and topology (linear, branched, cross-linked), researchers can engineer smart vehicles that precisely control drug solubility, stability, and release kinetics.
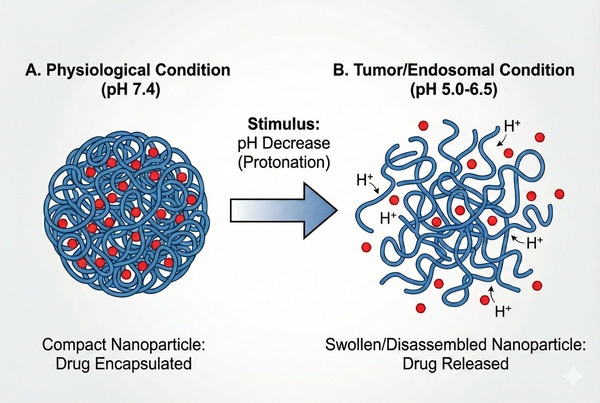 Fig. 2. Schematic of pH-triggered drug release from a polymeric nanoparticle in acidic tumor microenvironments (BOC Sciences Authorized).
Fig. 2. Schematic of pH-triggered drug release from a polymeric nanoparticle in acidic tumor microenvironments (BOC Sciences Authorized).
Functional Roles of Polymers in Drug Delivery
In a modern drug delivery system (DDS), the polymer is rarely just an inert matrix. It performs three critical bio-physicochemical functions:
- Stealth and Circulation Extension (PEGylation): The rapid clearance of nanoparticles by the reticuloendothelial system (RES) is a major barrier. Hydrophilic polymers, most notably Polyethylene Glycol (PEG), are conjugated to the surface of carriers to form a hydration shell. This steric barrier prevents the adsorption of opsonins (serum proteins that tag particles for phagocytosis), effectively masking the carrier from the immune system and significantly extending its plasma half-life (t1/2).
- Solubilization of Hydrophobic APIs: Many potent drug candidates (BCS Class II and IV) fail clinically due to poor water solubility. Amphiphilic block copolymers self-assemble into polymeric micelles, sequestering hydrophobic drugs within a non-aqueous core while presenting a hydrophilic shell to the aqueous environment. This can increase the apparent solubility of a drug by orders of magnitude.
- Active Targeting via Ligand Conjugation: Polymers provide abundant functional groups (hydroxyl, carboxyl, amine, thiol) at their termini or along the backbone. These sites are chemically accessible for the conjugation of targeting ligands—such as antibodies, peptides (e.g., RGD), or small molecules (e.g., folic acid)—facilitating site-specific accumulation via receptor-mediated endocytosis.
Types of Polymers Used in Therapeutics
The choice of polymer dictates the degradation profile and biocompatibility of the system.
Synthetic Biodegradable Polymers:
- PLGA (Poly(lactic-co-glycolic acid)): The gold standard for biodegradable devices. PLGA undergoes hydrolysis in the body to produce lactic acid and glycolic acid, two endogenous metabolic byproducts, ensuring minimal toxicity. Its degradation rate can be tuned from weeks to months by adjusting the lactate:glycolate ratio.
- PCL (Polycaprolactone): A semi-crystalline polymer with a slower degradation rate than PLGA, making it ideal for long-term implants (1+ years) and contraceptive delivery.
- Polyanhydrides: Known for surface erosion (as opposed to bulk erosion), allowing for zero-order drug release kinetics, crucial for delivering sensitive proteins like insulin or growth factors.
Natural Polymers:
- Chitosan: A cationic polysaccharide derived from chitin. Its positive charge allows it to adhere to negatively charged mucosal surfaces (mucoadhesion) and transiently open tight junctions, enhancing the paracellular transport of drugs across the intestinal or nasal epithelium.
- Hyaluronic Acid (HA): A naturally occurring glycosaminoglycan. Beyond its biocompatibility, HA targets CD44 receptors, which are often overexpressed on the surface of various tumor cells, providing intrinsic active targeting.
Complex Architectures:
- Dendrimers (e.g., PAMAM): These are hyper-branched, monodisperse macromolecules with a precise number of generation layers. Their multivalent surface allows for high-density drug loading and the simultaneous attachment of imaging agents and therapeutics.
Formulation Strategies and Controlled Release Mechanisms
The release of an active agent from a polymeric carrier is governed by specific physicochemical mechanisms, which can be engineered during synthesis:
Diffusion-Controlled Release: The drug permeates through the polymer matrix. In reservoir systems, the release rate is determined by the thickness and permeability of the polymer membrane. In matrix systems, it follows the Higuchi equation, dependent on the drug's diffusivity and the matrix porosity.
Erosion-Controlled Release:
- Bulk Erosion (e.g., PLGA): Water enters the entire polymer matrix, causing hydrolysis throughout. This often leads to a burst release followed by a sustained phase.
- Surface Erosion (e.g., Polyanhydrides): The polymer degrades only at the surface-water interface. The carrier becomes smaller over time while maintaining its geometric shape, protecting the inner drug payload from hydrolysis until release.
Stimuli-Responsive Release:
- pH-Responsive: Polymers containing ionizable groups (e.g., poly(acrylic acid) or poly(beta-amino esters)) change their conformation or solubility based on pH. This is utilized to trigger drug release in the acidic tumor microenvironment (pH ≈ 6.5) or to facilitate endosomal escape (pH ≈ 5.0) via the proton sponge effect.
- Redox-Responsive: Polymers cross-linked with disulfide bonds remain stable in the bloodstream but rapidly degrade inside cells due to the high intracellular concentration of glutathione (GSH), releasing the drug specifically within the cytosol.
Application Scenarios: Proteins, Peptides, Small Molecules, and Nucleic Acids
- Polymer for Small Molecules: Polymeric micelles and nanoparticles are extensively used to deliver chemotherapeutics (e.g., Paclitaxel, Doxorubicin), reducing systemic toxicity (like cardiotoxicity) and improving accumulation in tumor tissues.
- Polymer for Proteins and Peptides: Hydrogels are the preferred vehicle. Their high water content and soft network structure mimic the native extracellular matrix, stabilizing the tertiary structure of proteins and preventing denaturation during storage and release.
- Polymer for Nucleic Acids (Gene Delivery): Cationic polymers (e.g., PEI, Poly-L-lysine) form electrostatic complexes with negatively charged DNA or siRNA, known as polyplexes. These protect the genetic material from nuclease degradation in the serum and facilitate cellular uptake. However, balancing the high transfection efficiency of cationic polymers with their potential cytotoxicity remains a key formulation challenge.
Looking for Custom Polymers for Drug Delivery?
Whether you need cationic polymers, biodegradable carriers, or stimuli-responsive platforms, our experts provide tailored synthesis and functionalization solutions to support your research and therapeutic development.
Lipid-Based Drug Delivery Systems
Lipid-based systems are often considered the gold standard for clinical translation due to their biomimetic nature. They are composed of amphiphilic lipids that self-assemble into vesicles.
Lipid Nanoparticles, Liposomes, and Lipid Complexes
- Liposomes: Vesicular structures with an aqueous core surrounded by one or more lipid bilayers. They can encapsulate hydrophilic drugs in the core and hydrophobic drugs in the bilayer.
- Lipid Nanoparticles (LNPs): Unlike liposomes, LNPs possess a solid lipid core matrix. They are the preferred vehicle for nucleic acid delivery (siRNA, mRNA) and typically contain four components: ionizable lipids, helper lipids (phospholipids), cholesterol, and PEG-lipids.
- Solid Lipid Nanoparticles (SLNs): Composed of solid lipids at room temperature, offering improved physical stability compared to liquid emulsions.
Advantages of Lipid Carriers in Drug Delivery
The dominance of lipid carriers in clinical oncology and infectious disease is driven by several inherent advantages:
- Biocompatibility and Biodegradability: Composed of naturally occurring phospholipids (or their biodegradable analogs), lipid carriers metabolize into non-toxic byproducts, minimizing the risk of chronic accumulation or inflammatory responses common with inorganic carriers.
- High Transfection Efficiency for Nucleic Acids: The inclusion of ionizable lipids allows LNPs to encapsulate negatively charged mRNA or siRNA with high efficiency (>90%). Upon cellular uptake via endocytosis, the pH-triggered protonation of these lipids disrupts the endosomal membrane (the proton sponge effect or hexagonal phase transition), releasing the genetic cargo into the cytoplasm for translation.
- Versatility in Solubility Enhancement: Lipid systems are amphiphilic. They can solubilize lipophilic drugs (BCS Class II/IV) within the lipid acyl chains, significantly enhancing oral bioavailability and reducing variability in absorption caused by food effects.
Limitations and Challenges of Lipid-Based Systems
Despite their clinical success, developers must navigate significant physicochemical hurdles:
- Physical Instability: Lipid dispersions are thermodynamically unstable systems. They are prone to aggregation, fusion, and Ostwald ripening (growth of larger particles at the expense of smaller ones) over time. Furthermore, phospholipids can undergo chemical hydrolysis or oxidation (peroxidation) during storage, necessitating strict cold-chain management (often -20°C to -80°C).
- Burst Release Phenomenon: Upon intravenous administration, interaction with serum proteins (e.g., albumin) can destabilize the lipid membrane, causing a rapid, uncontrolled release of the drug before it reaches the target tissue. This is particularly problematic for small molecules encapsulated in the aqueous core of liposomes.
- Manufacturing Scalability: Transitioning from laboratory-scale thin-film hydration to industrial-scale production requires sophisticated technologies like microfluidic mixing or high-pressure homogenization. Achieving consistent particle size distribution (Polydispersity Index, PDI < 0.2) and high encapsulation efficiency at a large scale remains a complex engineering challenge.
Application Scenarios: mRNA Vaccines, Hydrophobic Drugs, and Targeted Therapy
- Genetic Medicine (mRNA/siRNA): The most prominent application is the delivery of mRNA vaccines (e.g., Comirnaty®, Spikevax®) and siRNA therapeutics (e.g., Onpattro® for hATTR amyloidosis). LNPs protect the fragile RNA backbone from ubiquitous RNases in the blood and facilitate cytosolic delivery.
- Oncology and Stealth Chemotherapy: PEGylated liposomes exploit the EPR effect to accumulate in tumor tissues. By encapsulating cytotoxic agents like doxorubicin or paclitaxel, lipid carriers alter the drug's biodistribution—significantly reducing cardiotoxicity and nephrotoxicity while maintaining anti-tumor efficacy.
- Active Targeting: Lipid surfaces can be easily functionalized with ligands (e.g., Transferrin, Folate, or cRGD peptides) to target specific receptors overexpressed on cancer cells or the blood-brain barrier. This active targeting strategy aims to further increase the therapeutic index by enhancing cellular uptake via receptor-mediated endocytosis.
Inorganic Drug Delivery Systems
While organic systems dominate the current clinical landscape, Inorganic drug delivery systems occupy a unique niche due to their intrinsic physical properties—specifically their magnetic, optical, and electronic characteristics. Unlike soft organic carriers which are susceptible to environmental degradation (hydrolysis or oxidation), inorganic nanoparticles (INPs) provide a rigid, chemically inert scaffold. This structural robustness makes them ideal candidates for theranostic applications, where simultaneous diagnostic imaging and therapeutic delivery are required in a single platform.
Types of Inorganic Carriers (Silica, Gold, Magnetic Nanoparticles)
The diversity of inorganic materials allows for distinct mechanisms of action:
- Mesoporous Silica Nanoparticles (MSNs): MSNs (e.g., MCM-41, SBA-15) are defined by their ordered, honeycomb-like porous structure. They possess an exceptionally high specific surface area (>1000 m2/g) and large pore volume (>0.9 cm3/g). This allows for the high-capacity loading of both small hydrophobic molecules and larger biologics within the pore channels, protecting the payload from enzymatic degradation without chemical modification.
- Gold Nanoparticles (AuNPs): AuNPs are synthesized in various geometries (spheres, rods, nanoshells, cages). They are biologically inert but chemically reactive at the surface, allowing for easy functionalization via thiol-gold (Au-S) chemistry. Their defining feature is surface plasmon resonance (SPR)—the coherent oscillation of conduction electrons upon light irradiation—which can be tuned to the near-infrared (NIR) window for deep tissue penetration.
- Magnetic Nanoparticles (SPIONs): Superparamagnetic iron oxide nanoparticles consist of a magnetic core (magnetite, Fe3O4, or maghemite, γ-Fe2O3) usually coated with a biocompatible polymer (PEG or Dextran) to prevent aggregation. Unlike ferromagnets, SPIONs exhibit magnetism only in the presence of an external magnetic field, preventing vessel embolization when the field is removed.
Functional Roles in Drug Stabilization and Targeting
Inorganic carriers offer unique advantages in how they stabilize drugs and interact with biological targets:
- Gatekeeper Controlled Release (MSNs): The pores of MSNs can be capped with gatekeepers (e.g., nanoparticles, polymers, or macrocyclic rings) that block drug exit. These gates can be engineered to open only in response to specific stimuli, such as the acidic pH of the tumor microenvironment (pH ≈ 6.5) or the presence of intracellular enzymes (e.g., esterases), achieving zero premature leakage.
- Magnetic Targeting (SPIONs): By applying an external magnetic field gradient over the target tissue (e.g., a solid tumor), SPIONs circulating in the bloodstream can be physically guided to and retained at the disease site. This magnetic drug targeting (MDT) significantly enhances local drug concentration while minimizing systemic exposure.
- Active Targeting via Surface Functionalization: The surface chemistry of inorganic particles is highly versatile.
- Silanization: The surface silanol groups (Si-OH) on MSNs allow for the covalent attachment of targeting ligands (antibodies, peptides, aptamers) using organosilanes (e.g., APTES).
- This precise orientation of ligands enhances the avidity of the nanoparticle for cell-surface receptors, facilitating receptor-mediated endocytosis.
Limitations and Safety Considerations
Despite their potential, the clinical translation of inorganic carriers faces significant hurdles related to ADME (absorption, distribution, metabolism, excretion) profiles:
- Non-Biodegradability and Clearance: Unlike PLGA or lipids, noble metals (Gold) and Carbon nanotubes are non-biodegradable. They tend to accumulate in the reticuloendothelial system (RES)—primarily the liver (Kupffer cells) and spleen. Long-term retention raises concerns about chronic toxicity, inflammation, and potential interference with organ function.
- Renal Clearance Threshold: Nanoparticles smaller than the renal filtration threshold (<5.5 nm) are rapidly excreted by the kidneys, limiting their circulation time. However, particles larger than this threshold are cleared by the liver. Balancing size to maximize circulation while ensuring eventual clearance is a critical design challenge.
- Reactive Oxygen Species (ROS) Generation: Certain inorganic materials (e.g., Zinc Oxide, Titanium Dioxide, and some formulations of SPIONs via the Fenton reaction) can induce oxidative stress by generating ROS, leading to cellular damage or apoptosis in healthy tissue if not properly coated.
Application Scenarios: Imaging, Cancer Therapy, and Combination Therapeutics
- Photothermal and Photodynamic Therapy: Inorganic nanosystems, particularly gold and carbon-based materials, leverage unique surface plasmon resonance and light-harvesting properties to achieve precise tumor ablation. Gold nanorods and nanoshells efficiently convert near-infrared (NIR) light into localized hyperthermia, physically destroying malignant cells while minimizing collateral damage to healthy surrounding tissue. When integrated with photosensitizers, these inorganic platforms can simultaneously execute photodynamic therapy by generating reactive oxygen species (ROS) under laser excitation, providing a dual-pronged oxidative and thermal attack that is far more potent than monotherapy.
- Magnetic Drug Targeting and Imaging: SPIONs represent a sophisticated class of inorganic agents that bridge the gap between diagnosis and targeted intervention. By exploiting their magnetic susceptibility, these particles can be actively steered to specific physiological sites using external magnetic field gradients, ensuring that therapeutic payloads are concentrated at the tumor site rather than circulating systemically. Beyond drug delivery, their intrinsic magnetic properties allow them to function as high-performance contrast agents for magnetic resonance imaging (MRI), enabling clinicians to monitor drug biodistribution and therapeutic progress in real-time.
- Hard-Tissue Engineering and Bone Regeneration: Inorganic carriers such as calcium phosphate, hydroxyapatite, and bioactive silica are indispensable in regenerative medicine due to their structural similarity to the mineral phase of human bone. These materials provide more than just a mechanical scaffold; they offer high osteoconductivity, facilitating the attachment, proliferation, and differentiation of osteoblasts. Mesoporous silica carriers, in particular, allow for the sustained and controlled release of osteogenic growth factors and antimicrobial agents, promoting rapid bone healing while maintaining the structural integrity required for load-bearing orthopedic applications.
- Multi-Modal Combination Therapeutics: By engineering a single inorganic core to carry chemotherapeutic drugs, respond to magnetic guidance, and generate photothermal heat, researchers can bypass the biological barriers that often lead to multi-drug resistance (MDR). This synergistic approach enhances the overall efficacy of the treatment, as the physical disruption of the tumor environment via heat or radiation often sensitizes the remaining cells to chemical agents, leading to more comprehensive clearance of the primary lesion.
Comparative Analysis: Polymer vs. Lipid vs. Inorganic Carriers
Selecting the optimal drug delivery system is an exercise in balancing trade-offs. While lipid-based systems currently dominate the landscape for genetic medicines (due to the success of mRNA vaccines), polymer-based systems remain unrivaled for sustained release and protein stabilization, and inorganic systems offer unique properties for theranostic applications.
The following table provides a direct comparison of physicochemical properties, biological performance, and manufacturing considerations.
| Feature / Metric | Polymer-Based Systems | Lipid-Based Systems | Inorganic Systems |
|---|
| Primary Structure | Macromolecular chains (synthetic or natural) | Amphiphilic lipid bilayers or solid lipid matrices | Rigid crystalline or amorphous lattice |
| Biocompatibility & Toxicity | Variable. Natural polymers (Chitosan) are safe; cationic polymers (PEI) can be toxic. PLGA is FDA-approved & biodegradable. | Excellent. Biomimetic; degrades into natural metabolites. Low immunogenicity (especially if PEGylated). | Low to Moderate. Often non-biodegradable. Concerns regarding long-term accumulation in RES (liver/spleen). |
| Drug Loading Capacity | High. Excellent for hydrophobic drugs (micelles) and biologics (hydrogels). | Moderate to High. High for nucleic acids (LNPs) and hydrophobic drugs; lower for hydrophilic small molecules. | High. High surface area and pore volume (MSNs) allow substantial loading of diverse payloads. |
| Release Kinetics | Highly Tunable. Can be engineered for sustained release (months) via hydrolysis or erosion. | Fast to Moderate. Often diffusion-controlled or triggered by membrane fusion. Difficult to sustain for long periods. | Stimuli-Responsive. Release often triggered by external stimuli (magnetic field, light, pH) or diffusion through pores. |
| Stability (Storage) | High. Can often be lyophilized (freeze-dried) and stored at room temperature or 4°C. | Low. Prone to hydrolysis, oxidation, and aggregation. Often requires ultra-cold storage (-20°C to -80°C). | Highest. Rigid structure resists environmental stress; thermally and chemically stable. |
| Targeting Capabilities | Versatile. Abundant functional groups for covalent attachment of ligands (antibodies, peptides). | Good. Ligands attached via PEG-lipids. Excellent for passive targeting (EPR effect). | Precise. Rigid surface allows for controlled orientation of ligands; magnetic targeting possible (SPIONs). |
| Scalability & Cost | Scalable. Standard chemical synthesis. Moderate cost. | Complex. Requires specialized equipment (microfluidics) and expensive GMP-grade lipids. | Scalable. generally low cost of raw materials, but size control at scale is challenging. |
| Best Application | Sustained release depots, oral delivery, protein stabilization. | mRNA/siRNA delivery, systemic chemotherapy, vaccines. | Theranostics (Imaging + Therapy), Photothermal therapy, deep tissue targeting. |
Key Factors When Selecting a Carrier for Your API
The transition from a promising drug candidate to a viable commercial product hinges on the marriage between the API and its delivery vehicle. There is no universal carrier; a system that excels for a small hydrophobic molecule may be disastrous for a fragile protein. Selecting the optimal DDS requires a systematic evaluation of three interacting dimensions: the drug's identity, the biological route, and the regulatory landscape.
Physicochemical Properties of the Drug Candidate
The intrinsic molecular characteristics of the API act as the primary filter for carrier selection. A comprehensive pre-formulation study must evaluate the following parameters to ensure encapsulation efficiency and drug stability:
- Solubility and Partition Coefficient (LogP): Highly hydrophobic drugs (BCS Class II/IV) require carriers with a lipophilic core, such as polymeric micelles or lipid nanoparticles, to achieve clinically relevant concentrations, whereas hydrophilic drugs are best entrapped within the aqueous core of liposomes or the matrix of hydrogels.
- Molecular Weight and Size: Small molecules (<1000 Da) can be easily loaded into the pores of MSNs or conjugated to polymer backbones, while large biologics (monoclonal antibodies, proteins) require soft carriers like PLGA microspheres or large unilamellar vesicles (LUVs) that preserve their tertiary structure.
- Surface Charge and Zeta Potential: Nucleic acids (DNA, mRNA, siRNA) possess a strong negative charge and require cationic carriers (e.g., ionizable lipids or PEI) for electrostatic complexation to form stable polyplexes or lipoplexes that prevent nuclease degradation.
- Chemical Stability and pKa: Labile drugs prone to hydrolysis or oxidation require carriers that provide a robust steric barrier or a water-free microenvironment, such as solid lipid nanoparticles (SLNs) or hydrophobic polymer matrices, to extend their half-life.
- Crystallinity and Polymorphism: The carrier must be compatible with the drug's physical state; for instance, amorphous solid dispersions are often required to prevent the recrystallization of poorly soluble drugs during storage.
Target Site and Route of Administration
The carrier must be engineered to navigate specific physiological barriers and leverage transport mechanisms unique to the intended route of administration:
- Intravenous (IV) Administration Requirements: Carriers intended for systemic circulation must be PEGylated (surface modified with polyethylene glycol) to form a hydration shell that prevents opsonization and rapid clearance by the reticuloendothelial system (RES) in the liver and spleen.
- Oral Delivery Challenges: Formulations must withstand the harsh acidic environment of the stomach (pH 1.5–3.5) and enzymatic degradation in the intestine, making enteric-coated polymeric nanoparticles (e.g., Eudragit® or Alginate) superior to standard liposomes which are susceptible to bile salts and lipases.
- Blood-Brain Barrier (BBB) Penetration: Delivering drugs to the CNS requires carriers smaller than 100 nm functionalized with specific ligands (e.g., Transferrin or Lactoferrin) to facilitate receptor-mediated transcytosis across the tight junctions of the endothelial cells.
- Tumor Microenvironment (TME) Accumulation: Carriers targeting solid tumors should be sized between 20 nm and 200 nm to exploit the EPR effect, while also requiring surface modification to penetrate the dense interstitial collagen matrix.
- Intracellular Trafficking and Endosomal Escape: For cytosolic targets (like mRNA translation), the carrier must possess a mechanism such as the proton sponge effect (in polymers) or pH-dependent membrane fusion (in ionizable lipids) to disrupt the endosome and prevent lysosomal degradation.
Regulatory Considerations and Clinical Translation
The scientific efficacy of a carrier must be balanced against its development risks, manufacturing complexity, and the regulatory pathway required for approval:
- GRAS Status and Excipient Safety: Prioritize carriers composed of materials generally recognized as safe (GRAS) by the FDA, such as PLGA, PLA, or naturally occurring phospholipids, to significantly reduce the burden of toxicological testing compared to novel synthetic materials.
- Biodegradability and Clearance Profiles: Ensure the carrier material degrades into non-toxic metabolites (e.g., lactic acid, fatty acids) or is below the renal filtration threshold (<5.5 nm) to avoid chronic accumulation and potential long-term toxicity, a common concern with inorganic carriers.
- Manufacturing Scalability (CMC): Evaluate whether the synthesis method (e.g., microfluidics, nanoprecipitation, or thin-film hydration) can be scaled from milligram-level R&D batches to kilogram-level GMP production without compromising particle size uniformity (polydispersity index < 0.2).
- Cost-Effectiveness of Raw Materials: Analyze the cost of goods sold (COGS), especially for lipid nanoparticles which may require expensive proprietary ionizable lipids, versus polymer systems which often utilize more affordable, off-the-shelf precursors.
Services
Custom Synthesis and Conjugation Services at BOC Sciences
At BOC Sciences, we provide integrated polymer solutions to support the design, development, and optimization of advanced drug delivery systems. Our capabilities span from high-quality polymer materials supply and custom polymer synthesis to polymer–drug conjugation, nanocarrier formulation development, and controlled release system engineering. Leveraging strong expertise in polymer chemistry and pharmaceutical materials science, we help our partners precisely tailor molecular structures, fine-tune delivery performance, and address key challenges such as solubility, stability, targeting, and release kinetics. From early-stage research to scale-up support, our services are designed to accelerate the translation of innovative drug delivery concepts into robust and reproducible delivery platforms.

Polymer Materials Supply for Drug Delivery
- Available materials: PEG, PLGA, PLA, PCL, PBAE, biodegradable copolymers, functionalized polymers, and more.
- Controlled molecular weight with customizable end groups (OH/COOH/NH₂/maleimide/azide, etc.).
- Supply from research scale to production scale (mg → kg).
- Complete documentation: COA, MSDS, and batch-to-batch consistency control.

Custom Polymer Synthesis & Modification
- Tailor-made design: molecular weight, dispersity, block architectures, branched/star-shaped structures.
- Functional modifications: PEGylation, incorporation of targeting ligands, pH/ROS/enzyme-responsive moieties.
- Copolymer design: diblock, triblock, and gradient copolymers.
- Comprehensive characterization: NMR, GPC, FTIR, and more.

Polymer–Drug Conjugation Services
- Covalent conjugation of polymers with small molecules, peptides, proteins, or nucleic acids.
- Cleavable linker design available (pH-sensitive, reductive, enzyme-cleavable, etc.).
- Support for DAR control, conjugation efficiency optimization, and stability evaluation.
- Applications include polymer prodrugs, PEGylated drugs, and long-circulating formulations.

Polymeric Nanocarrier Formulation Development
- System types: polymeric nanoparticles/micelles, nanospheres/nanocapsules, polymer–lipid hybrid systems.
- Support for encapsulation efficiency optimization, particle size control, PDI tuning, and stability screening.
- Cargo options: hydrophobic small molecules, proteins, peptides, siRNA/mRNA.
- Characterization: particle size, PDI, zeta potential, electron microscopy, encapsulation efficiency, and drug loading.
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
How to precisely tune the degradation rate of PLGA carriers?
The degradation kinetics of PLGA are primarily governed by the lactide-to-glycolide (L:G) ratio and the molecular weight (Mw). A higher glycolide content increases hydrophilicity, accelerating hydrolysis (weeks), while a higher lactide content or molecular weight creates a more hydrophobic, crystalline matrix that degrades slowly (months). Selecting the specific copolymer ratio allows you to align the release profile with your required therapeutic window.
What formulation strategies minimize the burst release effect?
The initial burst release is often caused by drug adsorbed on the nanoparticle surface. To mitigate this, increase the polymer concentration to densify the matrix or employ a double-emulsion solvent evaporation technique to better entrap the drug. Alternatively, coating the nanoparticle with a hydrophilic shell (e.g., Chitosan) or using higher molecular weight polymers can retard the initial diffusion rate.
Which chemical strategies are best for ligand conjugation?
For precise targeting, utilize pre-functionalized block copolymers (e.g., PLGA-PEG-Maleimide or PLGA-PEG-NHS) rather than physical adsorption. Maleimide groups react efficiently with thiol-modified ligands (antibodies/peptides), while NHS esters react with primary amines. This post-formulation conjugation approach preserves ligand bioactivity and ensures optimal surface orientation for receptor binding.
How to improve the loading efficiency of hydrophobic drugs?
For BCS Class II drugs, polymeric micelles formed from amphiphilic block copolymers (e.g., PEG-b-PCL) are the optimal choice. The key is to match the hydrophobicity of the polymer's core block with that of the drug. Additionally, optimizing the drug-to-polymer ratio during the precipitation phase can prevent drug crystallization and significantly enhance encapsulation efficiency.
How can cationic polymer toxicity be reduced for gene delivery?
High charge density in polymers like unmodified PEI leads to cytotoxicity via membrane disruption. To improve safety, mask the positive charge by PEGylating the surface or use biodegradable cationic polymers (e.g., poly(beta-amino esters)). This maintains the proton sponge effect necessary for endosomal escape while minimizing systemic toxicity and immune activation.

 Fig. 1. Cross-sectional structural comparison of polymeric micelles, lipid nanoparticles, and mesoporous silica nanoparticles (BOC Sciences Authorized).
Fig. 1. Cross-sectional structural comparison of polymeric micelles, lipid nanoparticles, and mesoporous silica nanoparticles (BOC Sciences Authorized). Fig. 2. Schematic of pH-triggered drug release from a polymeric nanoparticle in acidic tumor microenvironments (BOC Sciences Authorized).
Fig. 2. Schematic of pH-triggered drug release from a polymeric nanoparticle in acidic tumor microenvironments (BOC Sciences Authorized).