Polymers for Nucleic Acid Delivery: Strategies, Technologies, and Applications
With the breakthrough advances in gene therapy, mRNA vaccines, and CRISPR/Cas gene-editing technologies, nucleic acid therapeutics have emerged as a frontier of modern biomedicine. However, the clinical application of nucleic acids as therapeutic agents is primarily limited by inefficient in vivo delivery. Naked nucleic acid molecules are highly susceptible to enzymatic degradation by nucleases in biological fluids, and their strong negative charge and hydrophilic nature severely hinder cellular membrane penetration. As a result, the development of safe and efficient delivery carriers is critical to unlocking the full potential of gene-based therapies. Among various delivery platforms, polymers have become a central pillar of non-viral gene delivery systems due to their highly tunable chemical structures, favorable biocompatibility, and excellent nucleic acid loading capacity.
Resources
What Are Polymers for Nucleic Acid Delivery?
Polymers for nucleic acid delivery refer to a class of polymeric materials, obtained through chemical synthesis or natural extraction, that can form complexes with nucleic acid molecules via electrostatic interactions, hydrophobic interactions, or covalent conjugation. These polymer–nucleic acid complexes facilitate the intracellular delivery of nucleic acids and enable their biological functions. Unlike small-molecule drugs, nucleic acid therapeutics are large, negatively charged, and inherently unstable, and therefore require carrier systems for protection. Polymer carriers play a critical role in cellular uptake and endosomal escape through mechanisms such as the "proton sponge effect."
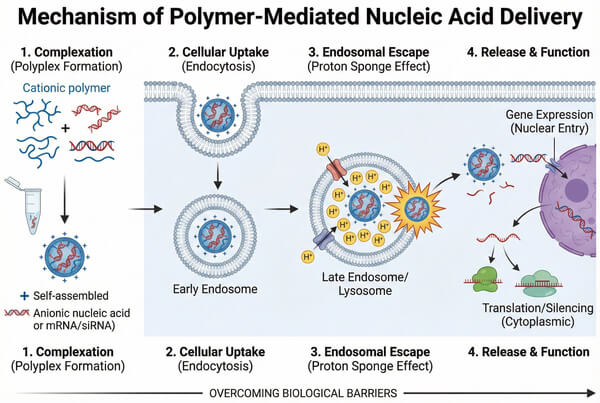 Fig. 1. Schematic showing polyplex formation, cellular uptake, endosomal escape, and gene expression (BOC Sciences Authorized).
Fig. 1. Schematic showing polyplex formation, cellular uptake, endosomal escape, and gene expression (BOC Sciences Authorized).
Types of Nucleic Acids Delivered: DNA, mRNA, siRNA, miRNA, Oligonucleotide
The design of polymer carriers is highly dependent on the type of nucleic acid being delivered. The major classes of nucleic acids currently delivered include:
- DNA (Plasmid DNA): Typically very large in molecular weight and requires polymers to condense it into compact nanoparticles for efficient nuclear entry.
- mRNA (Messenger RNA): A prominent therapeutic modality in recent years that does not require nuclear entry and is translated directly in the cytoplasm. However, its single-stranded structure is extremely unstable, demanding highly protective carrier systems.
- siRNA & miRNA (Small Interfering RNA & MicroRNA): Short double-stranded RNAs primarily used for gene silencing. Due to their short length and relatively high rigidity, the main challenge in complex formation lies in preventing premature dissociation.
- Oligonucleotides (Antisense Oligonucleotides, ASOs): Single-stranded DNA or RNA sequences used to modulate gene expression.
Comparison with Viral and Lipid-Based Delivery Platforms
- Viral Vectors: While viral vectors exhibit exceptionally high transfection efficiency, they suffer from significant limitations, including strong immunogenicity, risks of insertional mutagenesis, limited cargo capacity, and high manufacturing costs.
- Lipid Nanoparticles (LNPs): LNPs are the dominant delivery system for current mRNA vaccines; however, they face challenges such as poor thermal stability, short circulation time in vivo, and potential hepatotoxicity.
- Polymer Vectors: In contrast, polymer-based carriers offer lower immunogenicity, higher thermal stability, and easier large-scale manufacturing (scale-up). Moreover, their structures can be rationally designed to enable extrahepatic targeting, effectively bridging the gap between viral and lipid-based delivery systems.
Key Challenges in Nucleic Acid Delivery and How Polymers Address Them
Successful intracellular delivery of exogenous nucleic acids requires overcoming a series of complex physiological and cellular barriers, a process commonly referred to as the transfection cascade. Polymer carriers are specifically designed to address these biological challenges step by step.
- Protecting Nucleic Acids from Enzymatic Degradation: Blood and extracellular matrices are rich in nucleases, and naked RNA or DNA can be degraded within minutes after administration. Cationic polymers (such as PEI and PLL) bind to the negatively charged phosphate backbone of nucleic acids through electrostatic interactions, condensing and encapsulating them into nanoparticle cores (polyplexes). This physical barrier effectively shields nucleic acids from nuclease attack.
- Overcoming Cellular Uptake and Endosomal Escape Barriers: The negatively charged cell membrane naturally repels nucleic acid molecules. Even after internalization via endocytosis, carriers are often trapped within endosomes and exposed to degradation in acidic lysosomal environments. The positive surface charge of polymer complexes promotes adsorption onto the cell membrane and facilitates adsorption-mediated endocytosis. For endosomal escape, polymers with buffering capacity—such as those containing secondary or tertiary amine groups—utilize the proton sponge effect. In the acidic endosomal environment, these polymers absorb protons, leading to chloride ion influx and increased osmotic pressure, ultimately causing endosomal membrane rupture and release of nucleic acids into the cytoplasm.
- Balancing Transfection Efficiency and Cytotoxicity: Cationic polymers with high charge density typically exhibit high transfection efficiency but can also disrupt membrane integrity and induce significant cytotoxicity. To address this, researchers crosslink low-molecular-weight polymers using biodegradable linkers (such as disulfide or ester bonds) to construct degradable high-molecular-weight polymers. These polymers maintain high molecular weight prior to cellular entry to ensure efficient transfection, and subsequently degrade into low-toxicity small molecules inside the cell.
- Controlled and Sustained Nucleic Acid Release: Overly rapid or incomplete release of nucleic acids can compromise therapeutic efficacy. Polymers with tunable hydrolytic degradation kinetics (such as PLGA), or reduction-responsive polymers sensitive to high intracellular glutathione (GSH) concentrations, enable precise spatial and temporal control over nucleic acid release.
- Targeted Delivery to Specific Cells or Tissues: Nonspecific systemic distribution can lead to off-target effects and adverse reactions. By conjugating specific ligands—such as folic acid, RGD peptides, galactose, or antibodies—onto polymer surfaces, receptor-mediated endocytosis can be exploited to achieve active targeting of tumor cells or hepatocytes.
Major Polymer Classes Used for Nucleic Acid Delivery
The diversity of polymer chemistry provides a rich material library for nucleic acid delivery systems. From naturally derived materials to fully synthetic polymers, different classes exhibit distinct advantages in biocompatibility, transfection efficiency, and functionalization potential.
Natural Polymers for Biocompatible Nucleic Acid Delivery
Natural polymers are typically derived from biological sources and offer low toxicity, low immunogenicity, and excellent biodegradability, making them ideal for in vivo applications.
- Chitosan: A deacetylated derivative of chitin and the only naturally occurring cationic polysaccharide. Its strong mucoadhesive properties make it particularly suitable for intranasal or oral nucleic acid delivery. By adjusting the degree of deacetylation and molecular weight, its nucleic acid binding affinity and solubility can be optimized.
- Cyclodextrins (CDs): Cyclic oligosaccharides with a hydrophobic inner cavity and a hydrophilic exterior. Although inherently non-ionic, they are often chemically modified (e.g., cationized) or incorporated into polymer backbones. Their host–guest chemistry helps reduce carrier toxicity and enhance local drug loading.
- Hyaluronic Acid (HA): A naturally occurring anionic glycosaminoglycan with excellent biocompatibility. HA can specifically bind to CD44 receptors overexpressed on many tumor cells and is therefore widely used as a surface modifier for polymer complexes to achieve tumor-targeted delivery.
- Atelocollagen: A form of collagen processed enzymatically to remove antigenic telopeptides. It exists as a liquid or gel in vivo and has been successfully used for localized siRNA delivery, demonstrating minimal immune response and favorable sustained-release properties.
- Pullulan: A non-ionic extracellular polysaccharide. After cationic modification, it can bind nucleic acids and, due to its affinity for the asialoglycoprotein receptor (ASGPR), is widely studied for liver-targeted gene therapy.
Cationic Polymers for Electrostatic Complexation
This is the most extensively studied class of polymers, relying primarily on high-density positive charges to condense large nucleic acids and promote membrane adsorption and endocytosis.
- Polyethylenimine (PEI): Often regarded as the "gold standard" for polymer-based transfection. Branched 25 kDa PEI exhibits extremely high charge density and a strong proton sponge effect, resulting in high transfection efficiency but also notable cytotoxicity. Current research focuses on PEGylation or hydrophobic modification to mitigate toxicity.
- Poly-L-lysine (PLL): One of the earliest studied cationic peptides. While it efficiently condenses DNA into nanoparticles, it lacks sufficient buffering capacity for endosomal escape and often requires endosomolytic agents (such as chloroquine) or combination with buffering polymers to enhance efficiency.
- PAMAM Dendrimers: Highly defined, monodisperse, spherical macromolecules. Their surfaces contain numerous primary amine groups for nucleic acid binding, while internal tertiary amines provide buffering capacity for endosomal escape. Their well-defined structures make PAMAM dendrimers ideal models for structure–activity relationship (SAR) studies.
- Poly(2-(dimethylamino)ethyl methacrylate) (PDMAEMA): A methacrylate polymer containing tertiary amine groups. It exhibits good buffering capacity as well as pH- and temperature-responsive behavior, making it a multifunctional smart delivery material.
- Poly-L-arginine: Rich in guanidinium groups, this polymer mimics the mechanism of cell-penetrating peptides (CPPs), such as HIV-1 Tat, enabling direct translocation across cell membranes and often used to enhance membrane permeability in other polymer systems.
To prevent long-term toxicity or inflammatory responses caused by polymer accumulation in vivo, polymers with specific degradation mechanisms are particularly important.
- Poly(lactic-co-glycolic acid) (PLGA): An FDA-approved biomedical polymer with excellent biosafety and tunable degradation rates (via adjustment of the LA/GA ratio). Although electrically neutral, PLGA is often combined with cationic polymers to form double-emulsion nanoparticles for sustained nucleic acid release.
- Poly(β-amino esters) (PBAEs): A class of novel hydrolytically degradable cationic polymers synthesized via Michael addition reactions. They are positively charged at physiological pH to bind nucleic acids but rapidly hydrolyze into non-toxic small molecules in the cytoplasm, achieving both high transfection efficiency and low cytotoxicity.
- Polycaprolactone (PCL): A hydrophobic, semicrystalline polymer with a slower degradation rate than PLGA, making it suitable for long-acting gene delivery systems such as implants or microneedles. It is commonly used as the hydrophobic block in block copolymers to form micellar cores.
- Polyphosphoesters (PPEs): Polymers with repeating phosphate ester bonds in the backbone. Their degradation products—phosphates, alcohols, and diols—are all endogenous metabolites, conferring excellent biosafety. Their side chains are readily modifiable, allowing fine control over hydrophilicity and hydrophobicity.
- Synthetic peptides: Such as poly(glutamic acid) (PGA) or poly(aspartic acid) (PASP) derivatives. These materials mimic natural protein structures and introduce cationic groups through side-chain functionalization. Their degradation products are amino acids, resulting in exceptionally high safety profiles.
PEGylated Polymers for Improved Stability and Circulation
PEGylation is a widely adopted strategy to address the short circulation time and rapid clearance of cationic polymers by the reticuloendothelial system (RES) in vivo.
- PEG Block Copolymers (Block Copolymers, e.g., PEG-b-PLGA, PEG-b-PLL): By covalently linking hydrophilic PEG segments with hydrophobic or cationic polymer blocks, core–shell micelles or nanoparticles are formed. The PEG "stealth layer" effectively prevents serum protein adsorption (opsonization).
- Graft Copolymers (Graft Copolymers, e.g., PEG-g-PEI): PEG chains are grafted onto the backbone of high-charge-density polymers such as PEI. This approach not only reduces erythrocyte aggregation toxicity caused by surface positive charges but also improves colloidal stability of the complexes in physiological saline.
- Poloxamers/Pluronics: A class of PEO–PPO–PEO triblock copolymers. In addition to PEG-like stealth properties, certain Pluronics have been shown to overcome multidrug resistance by inhibiting P-glycoprotein (P-gp) efflux pumps and can further enhance gene transfection efficiency.
- Functionalized PEG: PEG molecules bearing specific terminal groups (such as maleimide or NHS esters) enable convenient conjugation of antibodies, peptides, or aptamers while introducing a stealth layer, thereby achieving dual functionality of stealth and targeting.
Stimuli-Responsive Polymers for Smart Delivery Systems
These smart polymers respond to changes in the microenvironment, enabling spatiotemporally controlled nucleic acid release.
- pH-Responsive Polymers: These polymers exploit the acidic environments of tumors (pH 6.5–6.8) or endosomes/lysosomes (pH 4.5–5.5). Polymers such as poly(histidine) (PolyHis) undergo protonation-induced charge reversal or structural disassembly under acidic conditions, triggering nucleic acid release and facilitating endosomal escape.
- Redox-Responsive Polymers: This strategy leverages the significant difference in glutathione (GSH) concentration between intracellular environments (approximately 2–10 mM) and extracellular environments (approximately 2–10 μM). Disulfide bonds introduced into polymer backbones or crosslinkers are cleaved upon cellular entry, leading to carrier disassembly and nucleic acid release.
- Enzyme-Responsive Polymers: These polymers are designed to respond to enzymes overexpressed in tumor tissues, such as matrix metalloproteinases (MMPs) or cathepsins. Specific enzyme-cleavable peptide substrates are incorporated into the polymer, enabling enzymatic degradation at tumor sites to release the payload or expose previously shielded targeting ligands.
- Thermo-Responsive Polymers: A representative example is poly(N-isopropylacrylamide) (PNIPAM), which exhibits a lower critical solution temperature (LCST). Near physiological temperature, PNIPAM undergoes a hydrophilic–hydrophobic phase transition and can be used to construct thermally triggered gene-releasing hydrogels or nanoparticles.
- Light-Responsive Polymers: Polymers containing photosensitive moieties such as azobenzene or o-nitrobenzyl groups undergo structural changes upon irradiation at specific wavelengths. Near-infrared (NIR) light, with superior tissue penetration, enables non-invasive remote-controlled release of nucleic acids.
Looking for Polymers for Nucleic Acid Delivery?
Whether you need cationic polymers, biodegradable carriers, or stimuli-responsive platforms, our experts provide tailored synthesis and functionalization solutions to support your research and therapeutic development.
Polymer-Based Nucleic Acid Delivery Technologies
The convergence of materials science and nanotechnology has driven the development of diverse polymer-based delivery architectures. These technologies not only enhance nucleic acid stability in complex physiological environments but also enable precise in vivo distribution and spatiotemporal release through sophisticated structural control.
Polyplexes represent the most fundamental and widely applied platform in nucleic acid delivery. They are formed through spontaneous electrostatic self-assembly between cationic polymers and negatively charged nucleic acids in aqueous solutions. During this process, polymer chains neutralize charges and condense large nucleic acid molecules into compact nanoparticles, typically 50–200 nm in diameter. This dense encapsulation effectively shields nucleic acids from nuclease-mediated degradation. To further enhance transfection performance, modern research increasingly employs microfluidic technologies to precisely control fluid shear forces and mixing rates, producing engineered nanoparticles with narrow size distributions and highly uniform morphologies. This technological advancement overcomes batch-to-batch variability associated with conventional bulk mixing and significantly improves intracellular unpacking efficiency and transfection kinetics.
Polymer Micelles for Nucleic Acid Encapsulation
Polymer micelles are core–shell nanocarriers formed by the self-assembly of amphiphilic block copolymers in aqueous environments, driven by hydrophobic or electrostatic interactions. Their primary advantage lies in clear functional compartmentalization: the hydrophilic shell—typically composed of PEG—provides excellent colloidal stability and stealth behavior during systemic circulation, effectively avoiding RES uptake, while the inner core selectively encapsulates nucleic acids or co-loads hydrophobic chemotherapeutic agents through chemical modification. This co-delivery capability endows polymer micelles with unique value in combination cancer therapy, enabling synergistic effects between gene silencing and chemotherapy. Moreover, by tuning block ratios, researchers can precisely control the critical micelle concentration (CMC), ensuring structural integrity even under extreme dilution in the bloodstream.
Polymer Hydrogels for Sustained Nucleic Acid Delivery
Polymer hydrogels are three-dimensional network biomaterials that retain large amounts of water through physical entanglement or chemical crosslinking, creating a microenvironment that mimics the extracellular matrix. From a delivery perspective, hydrogel systems are primarily focused on localized administration and long-term sustained release. By physically entrapping polyplexes or naked nucleic acids within the gel matrix, controlled release can be achieved over days to months via polymer degradation or diffusion mechanisms. This delivery mode offers significant advantages in tissue regeneration (such as bone or cartilage repair) and localized tumor immunotherapy, maintaining therapeutic concentrations at the target site while minimizing systemic toxicity. In recent years, injectable, in situ-forming responsive hydrogels have gained attention, as they undergo sol–gel transitions in response to body temperature or specific enzymes, enabling minimally invasive administration and precise localization.
Polymer-Engineered Inorganic Nanoparticles
Polymer-engineered inorganic nanoparticles represent functional hybrids of organic and inorganic materials. Typically, gold nanoparticles, mesoporous silica, or magnetic iron oxide serve as the core, while polymer chains such as PEI or PEG derivatives are grafted or coated onto the inorganic surface. This hybrid architecture confers multiple functionalities: the inorganic core provides unique physical properties such as photothermal conversion, magnetic targeting, or imaging contrast, while the polymer shell ensures biocompatibility and supplies nucleic acid binding sites. For example, the photothermal effect of gold nanoparticles can induce localized heating under near-infrared irradiation, triggering polymer conformational changes that facilitate rapid endosomal escape. Such "theranostic" platforms greatly expand the role of nucleic acid delivery in precision medicine.
Polymer Microneedles for Transdermal Nucleic Acid Delivery
Polymer microneedle arrays are a breakthrough minimally invasive transdermal delivery technology composed of hundreds of micron-scale polymer needles capable of painlessly penetrating the stratum corneum. These needles are typically fabricated from water-soluble or biodegradable polymers such as hyaluronic acid or polyvinyl alcohol and are loaded with nucleic acid vaccines or therapeutic oligonucleotides. Upon insertion into the skin, the microneedles dissolve or swell in interstitial fluid, releasing nucleic acids directly into the dermis rich in immune cells such as Langerhans cells. Compared with conventional intramuscular injection, polymer microneedles significantly enhance immune responses while offering superior patient compliance and eliminating cold-chain requirements. This technology is particularly well suited for large-scale mRNA vaccine deployment and gene-based therapies for chronic skin diseases.
Applications of Polymer-Based Nucleic Acid Delivery Systems
With advances in molecular medicine, polymer delivery systems have progressed from fundamental research to clinical application. Their tunable physicochemical properties enable applications ranging from preventive vaccines to complex genetic disease repair. Through precise modulation of polymer architectures, nucleic acid therapeutics are reshaping the landscape of clinical treatment.
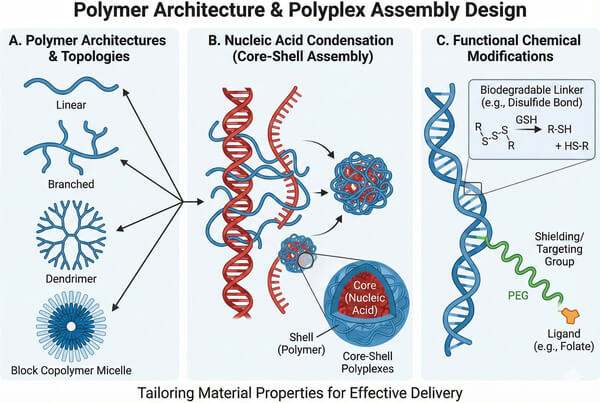 Fig. 2. Illustration of various polymer topologies and their self-assembly into core-shell polyplexes (BOC Sciences Authorized).
Fig. 2. Illustration of various polymer topologies and their self-assembly into core-shell polyplexes (BOC Sciences Authorized).
DNA Vaccines and Gene Therapy Delivery
In gene therapy, polymer carriers are primarily responsible for delivering functional plasmid DNA (pDNA) into the cell nucleus to restore missing protein expression or compensate for defective genes. Due to the large molecular weight and shear sensitivity of pDNA, polymer carriers must exhibit exceptional condensation capability, packaging DNA into highly stable nanocomplexes to withstand enzymatic degradation in the extracellular matrix. Polymer systems have demonstrated significant potential in localized treatments for genetic disorders such as cystic fibrosis and hemophilia. Examples include aerosolized delivery of CFTR gene-loaded polymer nanoparticles to the lungs or local injection of growth factor genes into fracture sites to promote healing. Compared with viral vectors, the non-immunogenic nature of polymers enables repeated dosing, which is critical for chronic diseases requiring sustained therapeutic levels.
mRNA Therapeutics and Vaccine Delivery
The explosive growth of mRNA technology has imposed stringent requirements on delivery systems. Since mRNA functions directly in the cytoplasm, polymer carrier design focuses on enhancing endosomal escape while protecting highly unstable single-stranded RNA. Although lipid nanoparticles (LNPs) currently dominate the market, emerging delivery systems based on amphiphilic polymers and polypeptoids are demonstrating superior stability compared to LNPs. Polymer carriers are widely applied not only in infectious disease vaccines but also in cancer immunotherapy, where they deliver mRNA encoding tumor-associated antigens (TAAs) to activate the patient's immune system in situ. Moreover, the excellent thermal stability of polymer carriers allows them to retain activity without strict cold-chain storage, which is essential for improving global vaccine accessibility.
siRNA, miRNA, and Antisense Oligonucleotide Delivery
For small interfering RNA (siRNA) and microRNA (miRNA) delivery, the core objective of polymer technology is precise gene silencing. Due to the rigidity of these short RNA molecules, conventional cationic polymers often struggle to form stable complexes. To address this, researchers have developed functionalized polymers—such as hydrophobically modified PEI or fluorinated polymers—to enhance hydrophobic interactions and improve loading stability. Clinically, polymer carriers deliver siRNA through systemic circulation to target organs such as the liver or tumor tissues, downregulating pathogenic protein expression via RNA interference. In treatments for amyloidosis or advanced solid tumors, polymer nanoparticles significantly improve oligonucleotide pharmacokinetics, extending half-life and reducing nonspecific systemic toxicity.
Genome Editing and CRISPR/Cas System Delivery
The advent of CRISPR/Cas9 technology has provided a powerful tool for curing genetic diseases, but its components—such as Cas9 protein complexed with sgRNA—are large and possess complex charge distributions, posing major delivery challenges. Polymer-based nanocapsule technologies mimic viral encapsulation mechanisms to efficiently package entire ribonucleoprotein (RNP) complexes. Compared with DNA-based expression strategies, this non-viral delivery approach enables transient expression, significantly reducing intracellular Cas9 residence time and thereby minimizing off-target effects. Polymer-mediated CRISPR delivery is now playing a central role in ex vivo and in vivo gene editing for Duchenne muscular dystrophy (DMD) and various rare hematological disorders.
Targeted Nucleic Acid Delivery for Oncology and Genetic Disorders
In oncology, polymer delivery systems fully exploit tumor physiological characteristics such as the enhanced permeability and retention (EPR) effect. By grafting specific ligands—such as folic acid, RGD peptides, or monoclonal antibodies against tumor-associated antigens—onto polymer surfaces, carriers can selectively bind to receptors on cancer cells and enter target cells via receptor-mediated endocytosis. This targeting capability increases the accumulation of therapeutic nucleic acids (such as the tumor suppressor gene p53) at tumor sites while protecting normal immune cells from off-target damage. Additionally, for certain metabolic genetic disorders, polymer carriers can be engineered to specifically target hepatic Kupffer cells or parenchymal cells, enabling precise regulation of lipid metabolism-related genes and providing a robust material foundation for personalized precision medicine.
How to Select the Right Polymer for Your Nucleic Acid Delivery Project?
A deep understanding of how polymer chemical structures influence their biological performance is the foundation for designing next-generation, high-efficiency delivery vectors. The physicochemical properties of a carrier determine its efficiency at every step of the transfection cascade, from nucleic acid condensation and stability in circulation to cellular uptake and intracellular release of the nucleic acid.
Factors to Consider: Nucleic Acid Type, Target Cells, Administration Route
- Nucleic acid type: mRNA is more sensitive than plasmid DNA and therefore requires carriers with stronger protective capability; siRNA is shorter in length and requires stronger binding affinity to prevent dissociation.
- Target cells: Primary cells and stem cells are generally more difficult to transfect than immortalized cell lines and may require specially optimized polymers (such as lipopolymers).
- Administration route: Intravenous administration requires carriers with long circulation times and serum resistance, whereas local administration places greater emphasis on tissue retention.
Polymer Molecular Weight and Charge Density Effects
- Molecular weight: Polymer chain length directly affects its ability to entangle and condense nucleic acids. In general, high-molecular-weight polymers (such as 25 kDa branched PEI) exhibit stronger multivalent electrostatic interactions, enabling the formation of more structurally stable, nuclease-resistant nanocomplexes and thus higher transfection efficiency. However, high-molecular-weight chains are often difficult for cells to metabolize and can cause irreversible membrane damage and mitochondrial stress, leading to significant cytotoxicity. Therefore, current research focuses on identifying the optimal "sweet spot," where sufficient nucleic acid stability is maintained while molecular weight is minimized or biodegradable linkages are introduced.
- Charge density: An appropriate positive charge density is essential for efficient nucleic acid condensation and for inducing adsorption-mediated endocytosis. In addition, amine groups on the polymer backbone (especially secondary and tertiary amines) provide critical buffering capacity, facilitating endosomal escape through the proton sponge effect. However, excessively high positive charge density can result in severe biocompatibility issues, such as erythrocyte aggregation, increased thrombosis risk, and strong inflammatory responses. Modulating charge density through copolymerization with neutral segments is a core strategy for improving safety.
Architecture Design: Linear, Branched, and Dendritic Polymers
- Linear architecture: Linear polymers (such as linear PEI or PLL) have regular structures and relatively narrow molecular weight distributions, making them attractive for studies on biodegradability. However, due to conformational limitations, linear chains often fail to tightly wrap large pDNA molecules, resulting in slightly reduced complex stability under physiological conditions compared to branched structures.
- Branched architecture: Branched polymers possess highly interconnected networks and a higher proportion of terminal amine groups. This three-dimensional structure provides strong charge accumulation effects and enhanced buffering capacity. For example, branched PEI typically exhibits significantly higher transfection efficiency than its linear counterpart. Branched architectures also offer more sites for chemical modification, facilitating the introduction of targeting ligands or hydrophobic moieties.
- Dendritic architecture: Dendrimers (such as PAMAM) are monodisperse, spherical macromolecules with precisely defined molecular structures. Owing to their highly controllable architecture, the size, surface charge number, and internal cavities of each generation can be predetermined, making dendrimers an ideal gold-standard model for studying structure–activity relationships (SAR). Surface engineering of dendrimers allows precise regulation of their interactions with biological membranes.
Influence of Hydrophobic/Hydrophilic Balance on Delivery Efficiency
- Role of hydrophobic modification: Introducing an appropriate amount of hydrophobic groups (such as lipid chains, cholesterol, or alkyl chains) into cationic polymers can significantly enhance their affinity for cell membranes. This amphiphilic design promotes membrane fusion and endosomal membrane disruption, representing an effective strategy to improve endosomal escape efficiency.
- Fine-tuning the HLB value: The hydrophilic–lipophilic balance (HLB) directly determines the colloidal stability of nanoparticles in aqueous environments. Excessive hydrophobicity can lead to particle aggregation, precipitation, and severe cytotoxicity, whereas insufficient hydrophobicity fails to effectively facilitate membrane penetration. Precise control over the proportion of hydrophobic segments enables an ideal state in which stability is maintained extracellularly while release is triggered intracellularly.
Surface Functionalization for Targeted Delivery
- Stealth properties and long circulation: Surface modification is key to improving the pharmacokinetics (PK) of polymer-based carriers. The introduction of PEG (polyethylene glycol) or zwitterionic polymers forms a hydrophilic barrier that significantly reduces opsonin adsorption and clearance by the mononuclear phagocyte system (MPS), thereby extending circulation time and reducing immunogenicity.
- Active targeting modification: Covalent conjugation of folic acid, RGD peptides, galactose, or monoclonal antibodies to polymer termini enables nanoparticles to specifically recognize diseased cells (such as tumor cells or hepatocytes). These interactions not only increase nucleic acid uptake in target tissues but also significantly reduce off-target effects on healthy tissues, thereby optimizing the therapeutic window.
Services
Custom Polymer Solutions for Nucleic Acid Delivery at BOC Sciences
BOC Sciences offers end-to-end custom polymer solutions for nucleic acid delivery, supporting a wide range of nucleic acid therapeutics including DNA, mRNA, siRNA, and oligonucleotides. Leveraging our extensive expertise in polymer chemistry and drug delivery systems, we design, synthesize, and optimize functional polymers tailored to specific biological barriers, administration routes, and therapeutic objectives, supporting projects from early research through preclinical development.

Custom Polymer Synthesis and Molecular Design
- Custom design of polymer carriers for DNA, siRNA, miRNA, and mRNA delivery
- Synthesis of linear, block, branched, or hyperbranched polymer architectures
- Precise control of molecular weight, molecular weight distribution, and charge density
- Design of cationic, amphiphilic, or biodegradable polymer systems

Polymer Modification and Functionalization Services
- Surface modification with targeting ligands, peptides, or glycans to achieve cell- or tissue-specific delivery
- PEGylation and hydrophilic modifications to extend in vivo circulation time and reduce immunogenicity
- Fine control of charge density and hydrophobicity to optimize nucleic acid binding and release behavior
- Customized conjugation and modification strategies compatible with diverse nucleic acid cargos

Formulation Optimization and Scale-Up Support
- Development of polymer–nucleic acid complexes, nanoparticles, and polyplex formulations
- Optimization of key parameters such as N/P ratio, particle size distribution, and formulation stability
- Optimization of delivery system stability, storage conditions, and lyophilization protocols
- Support for laboratory-scale, pilot-scale, and scale-up manufacturing process development

Analytical Characterization and Quality Control
- Characterization of molecular weight, distribution, and chemical structure (GPC, NMR, FTIR, etc.)
- Analysis of particle size, morphology, and surface charge (DLS, TEM/SEM, Zeta potential)
- Evaluation of nucleic acid loading efficiency, binding affinity, and protective capability
- Analysis of release behavior, degradation kinetics, and stability
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
What types of polymers are used for nucleic acid delivery?
Polymers used for nucleic acid delivery span a broad material library ranging from natural to synthetic systems. Major categories include cationic polymers such as PEI, PLL, and dendritic PAMAM, which condense nucleic acids through electrostatic interactions; biodegradable polymers such as PLGA, PCL, and poly(β-amino esters) (PBAEs), which emphasize controlled release and low-toxicity metabolism; as well as natural polysaccharides like chitosan with excellent biocompatibility. In recent years, PEGylated stealth polymers and stimuli-responsive smart polymers that respond to pH or redox potential have also become research hotspots, aiming to achieve more precise in vivo distribution.
How do polymers protect nucleic acids from degradation?
Polymers primarily protect nucleic acids through steric hindrance and charge neutralization mechanisms. When cationic polymers bind to nucleic acids, they condense long nucleic acid chains into compact nanoscale complexes (polyplexes), encapsulating the vulnerable phosphodiester bonds within a hydrophilic polymer matrix. This physical barrier effectively prevents contact between nucleases in biological fluids and the nucleic acid molecules, protecting them from degradation before reaching target cells. In addition, surface modifications such as PEGylation further reduce nonspecific serum protein adsorption, enhancing circulation stability and ensuring that genetic material enters cells in an intact molecular form.
Can polymer platforms enhance cellular uptake of DNA or RNA?
Polymer platforms significantly enhance cellular uptake through multiple mechanisms. First, polymers neutralize the negative charge of nucleic acids, imparting nanoparticles with a moderate positive surface charge that promotes electrostatic attraction to negatively charged cell membranes and triggers adsorption-mediated endocytosis. Second, grafting specific ligands such as RGD peptides, folic acid, or galactose onto polymer termini enables receptor-mediated endocytosis for active targeted uptake. In addition, certain hydrophobically modified polymers can promote membrane fusion through interactions with the lipid bilayer, allowing nucleic acids to bypass complex transport barriers and directly enter the cell interior, greatly enhancing transfection efficiency.
How are polymers selected for different nucleic acid therapeutics?
Polymer selection must be customized based on the physical properties of the nucleic acid and the therapeutic objective. For large plasmid DNA, cationic polymers with high charge density and nuclear entry–assisting capabilities are preferred; for highly unstable mRNA, amphiphilic polymers with strong endosomal escape capacity and robust protection are prioritized; for short-chain RNA such as siRNA, carriers with strong binding affinity and specific tissue-targeting capability are more suitable. The administration route is also a critical factor: systemic delivery emphasizes long-circulation stability, whereas local delivery focuses more on tissue retention and sustained release. The optimal polymer architecture is determined by balancing efficiency, toxicity, and degradation rate.
What is the Proton Sponge Effect and why is it important?
The proton sponge effect is a core mechanism by which amine-containing polymers (such as PEI) facilitate endosomal escape of nucleic acids. After nanocomplexes are internalized into endosomes, continuous proton pumping lowers endosomal pH. The abundant secondary and tertiary amine groups in the polymer absorb protons like a sponge, buffering the pH decrease. This process leads to an influx of chloride ions and water molecules, sharply increasing osmotic pressure and ultimately causing endosomal membrane rupture, releasing nucleic acids into the cytoplasm. This mechanism is critical for preventing acid-mediated degradation of nucleic acids in lysosomes and is a key biophysical process determining polymer transfection efficiency and overall bioavailability.
How to mitigate the cytotoxicity of cationic polymers?
The primary strategies for mitigating the toxicity of cationic polymers focus on balancing charge density and biodegradability. Researchers often introduce biodegradable linkages (such as disulfide or ester bonds) to construct degradable polymers that break down into low-toxicity small molecules after completing their delivery function. In addition, PEGylation can shield excess surface positive charge, reducing nonspecific membrane disruption. Another effective approach is to use naturally derived peptides or polysaccharides as polymer backbones, or to develop pH-responsive charge-reversal polymers that expose positive charge only within specific endosomal environments, thereby maintaining high transfection efficiency while minimizing interference with normal cellular functions.

 Fig. 1. Schematic showing polyplex formation, cellular uptake, endosomal escape, and gene expression (BOC Sciences Authorized).
Fig. 1. Schematic showing polyplex formation, cellular uptake, endosomal escape, and gene expression (BOC Sciences Authorized). Fig. 2. Illustration of various polymer topologies and their self-assembly into core-shell polyplexes (BOC Sciences Authorized).
Fig. 2. Illustration of various polymer topologies and their self-assembly into core-shell polyplexes (BOC Sciences Authorized).