How Polymer Carriers Improve Stability and Efficacy of Protein and Peptide Drugs?
Proteins and peptide drugs, with their high specificity and low toxicity, have become central to modern biopharmaceutical research. However, their intrinsic physicochemical instability and limitations in in vivo pharmacokinetics (PK) hinder clinical translation. Functional polymer carriers, as an advanced drug delivery strategy, can significantly improve the in vivo performance of biomacromolecules through mechanisms such as steric hindrance, surface modification, and structural encapsulation. This article explores, from both biochemical and materials science perspectives, the mechanisms by which polymer carriers overcome physiological barriers, the types of carriers available, and key design considerations in drug development.
Resources
Guide to Polymer-Based Delivery Systems for Proteins and Peptides
With the rapid advancement of biotechnology, biologics have demonstrated the potential to surpass traditional small-molecule drugs in treating cancer, metabolic disorders, and autoimmune diseases. Yet, converting inherently fragile proteins or peptides into effective therapeutics remains a major translational challenge. The introduction of polymer carriers offers a powerful solution to the delivery obstacles faced by biomacromolecules. Serving as a bridge between drug molecules and biological systems, polymer systems not only act as physical shields protecting the drug, but can also modulate in vivo distribution through chemical modification.
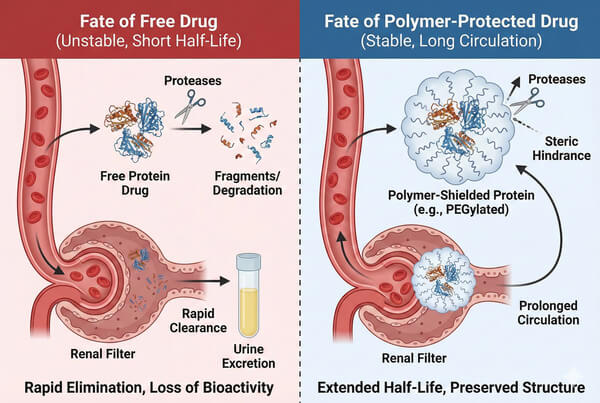 Fig. 1. Comparison showing how polymer shielding protects proteins from enzymatic degradation and extends circulation time (BOC Sciences Authorized).
Fig. 1. Comparison showing how polymer shielding protects proteins from enzymatic degradation and extends circulation time (BOC Sciences Authorized).
The Key Challenges in Protein and Peptide Drug Delivery
The properties of protein and peptide drugs are determined by their amino acid sequences and complex secondary and tertiary structures. While these structures confer high bioactivity, they also render the molecules highly susceptible to inactivation under physiological conditions. Drug delivery must overcome a series of biological barriers from the injection site to the target tissue.
- Overcoming Enzymatic Degradation and Short Circulation Half-Life: Proteases and peptidases, widely present in the body, pose the primary threat to biomacromolecular drugs. In blood circulation and interstitial spaces, unprotected peptide bonds are readily hydrolyzed, leading to rapid drug inactivation. For instance, many short peptides have plasma half-lives (t1/2) of only a few minutes. Polymer carriers can create physical barriers or alter the hydrodynamic volume of the drug, significantly reducing the likelihood of enzyme-substrate interactions.
- Preventing Aggregation, Denaturation, and Loss of Biological Activity: Protein bioactivity relies heavily on its correct three-dimensional folding. Environmental factors such as temperature fluctuations, pH changes, and even high shear stress during injection can induce denaturation or hydrophobic aggregation. Aggregates not only reduce efficacy but may trigger severe immune responses. Polymer matrices provide a buffered microenvironment that maintains protein conformational stability.
- Addressing Poor Pharmacokinetics and Limited Tissue Targeting: Due to lack of inherent targeting, free biomacromolecules often distribute systemically, accumulating non-specifically in organs like the liver and spleen. This reduces effective drug concentration at disease sites and may cause systemic toxicity. Functionalized polymer carriers, such as those conjugated with targeting ligands, can improve PK profiles and enable active delivery to specific tissues or cells.
- Overcoming Rapid Renal Clearance and Short Half-Life: The glomerular filtration threshold is approximately 5–7 nm in diameter (corresponding to 30–50 kDa molecular weight). Most peptides and low-molecular-weight proteins, such as cytokines or growth hormones, are smaller than this threshold, resulting in rapid renal clearance after entering circulation. Polymer modification, such as PEGylation, can increase the apparent molecular weight and hydrodynamic radius of the drug, allowing it to evade renal filtration and extend circulation time.
- Mitigating Immunogenicity and Hypersensitivity Reactions: Exogenous proteins can be antigenic, stimulating the host immune system to produce anti-drug antibodies (ADAs), which may neutralize the drug or trigger allergic reactions. Polymer chains on the protein surface form a "shield," masking epitopes and reducing immunogenicity, thereby improving drug safety.
Mechanisms of Action: How Polymers Stabilize Therapeutics
Polymer carriers are not merely inert excipients; they interact with drug molecules through multiple physicochemical mechanisms, reshaping their biological behavior at the molecular level. Understanding these mechanisms is essential for designing efficient delivery systems.
- Steric Shielding: Protecting against Proteolytic Attack: Steric hindrance is a core mechanism by which polymers protect proteins. For example, polyethylene glycol (PEG) forms a highly hydrated, flexible polymer cloud in solution. This "polymer corona" effectively prevents proteases from accessing the protein core, inhibiting enzymatic degradation.
- Preserving Structural Integrity and Therapeutic Function: Certain amphiphilic polymers or hydrogel systems stabilize the hydrophobic core of proteins through non-covalent interactions such as hydrogen bonding and hydrophobic interactions, preventing unfolding caused by polar solvents. This effect ensures that proteins maintain their native active conformation during storage and transport.
- Targeted Delivery via Enhanced Permeability and Retention (EPR) Effect: In tumors or inflamed tissues, endothelial gaps widen (200–800 nm), and lymphatic drainage is impaired. This phenomenon, known as the enhanced permeability and retention (EPR) effect, allows high-molecular-weight polymer nanocarriers to passively accumulate in these tissues, increasing local drug concentration and achieving passive targeting.
- Minimizing Aggregation During Processing and Storage: Processes like lyophilization and reconstitution can induce protein aggregation. Polymers, acting as cryoprotectants or fillers, can replace water molecules on the protein surface and restrict molecular motion through vitrification, preventing collisions and aggregation in the solid state.
- Enhancing Solubility and Formulation Stability: Many hydrophobic peptides have poor solubility in aqueous solutions. Amphiphilic block copolymers self-assemble into micelles, encapsulating hydrophobic drugs in the core, dramatically improving apparent solubility and enabling intravenous administration.
Types of Polymers for Protein & Peptide Drug Delivery
Selecting the right polymer matrix is critical when designing biomacromolecule delivery systems. Different polymer chemistries impart distinct biodegradability, immunogenicity, and pharmacokinetic profiles.
Polyethylene Glycol (PEG) and PEG Analogs
PEG, with its exceptional hydrophilicity and low immunogenicity, has long been considered the gold standard for improving protein drug PK. Its steric barrier reduces enzymatic degradation and renal clearance. However, clinical experience has revealed potential risks, including the accelerated blood clearance (ABC) phenomenon and anti-PEG antibody formation, prompting the search for alternatives. PEG analogs such as poly(2-oxazoline)s (POx) and polyglycerols (PG) offer superior chemical stability and tunable side chains. These emerging polymers retain PEG's long-circulation advantages while exhibiting lower oxidative sensitivity and better biocompatibility, providing opportunities for next-generation stealth carriers.
Biomimetic Polymers Inspired by Natural Systems
Inspired by the chemical properties of cell membranes, biomimetic polymers—particularly zwitterionic polymers (e.g., PCB, PMPC)—are redefining anti-fouling standards. Their polymer chains contain both positive and negative charges, which strongly bind water molecules to form a dense, thermodynamically stable hydration layer that resists nonspecific protein adsorption. Compared to traditional materials, biomimetic polymers more effectively evade immune recognition and clearance, extend circulation half-life, and significantly reduce inflammation from foreign-body responses, making them ideal for long-acting stealth delivery.
Biodegradable Polymers for Protein and Peptide Delivery
Biodegradable synthetic polymers such as PLGA, PLA, and PCL are core materials for constructing implants and microspheres for long-acting depot formulations. Their major advantage is that their final metabolites (e.g., lactic and glycolic acids) are non-toxic and fully metabolizable. By precisely tuning monomer ratios (e.g., lactide to glycolide) and polymer molecular weight, formulators can control degradation rates and porosity, enabling customizable drug release kinetics. This tunability makes them particularly suitable for peptide drugs requiring sustained blood levels over weeks or months, addressing patient compliance issues associated with frequent injections.
Stimuli-Responsive Polymers for Controlled Drug Release
Stimuli-responsive polymers are designed to sense specific physicochemical signals in pathological microenvironments, such as acidic pH, elevated temperature, redox potential changes, or overexpressed enzymes, and respond through conformational changes or chemical bond cleavage. For example, pH-sensitive polymers can undergo charge reversal in the slightly acidic tumor microenvironment (pH 6.5–6.8) or in endosomes, triggering nanocarrier disassembly or membrane fusion for rapid intracellular release and endosomal escape. This spatiotemporally controlled "on-demand release" significantly increases drug concentration at disease sites while minimizing off-target leakage, substantially improving the therapeutic window.
Looking for Polymers for Protein and Peptide Delivery?
Whether you need cationic polymers, biodegradable carriers, or stimuli-responsive platforms, our experts provide tailored synthesis and functionalization solutions to support your research and therapeutic development.
Classification of Polymeric Carriers for Protein and Peptide Delivery
Based on polymer molecular structure, assembly mode, and physicochemical properties, delivery systems can be constructed in various forms. Each carrier type is optimized for specific drug characteristics and clinical requirements to overcome particular in vivo barriers faced by biomacromolecules.
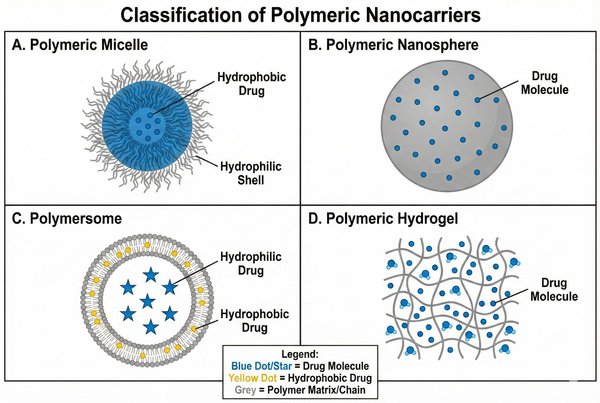 Fig. 2. Structural classification of polymeric nanocarriers including micelles, nanospheres, polymersomes, and hydrogels for drug delivery (BOC Sciences Authorized).
Fig. 2. Structural classification of polymeric nanocarriers including micelles, nanospheres, polymersomes, and hydrogels for drug delivery (BOC Sciences Authorized).
Polymeric Nanoparticles and Nanospheres
Polymeric nanoparticles generally refer to solid colloidal systems with sizes ranging from 10 to 1000 nm. Drug molecules can be either dissolved or embedded within the polymer matrix (nanospheres) or adsorbed/covalently attached to the particle surface (nanocapsules). This solid matrix structure provides a robust physical barrier for fragile proteins, effectively protecting them from proteolytic degradation and pH fluctuations in the external environment, thereby significantly enhancing drug stability. Additionally, hydrophilic surface modification or conjugation with targeting ligands can markedly improve pharmacokinetics, enabling prolonged circulation or targeted delivery to specific tumor tissues.
Polymeric Micelles for Protein and Peptide Encapsulation
Polymeric micelles are core–shell structures formed by the self-assembly of amphiphilic block copolymers in aqueous solution. Their hydrophobic core effectively solubilizes hydrophobic peptides, while the hydrophilic shell (typically PEG) provides steric stabilization and anti-fouling properties. Due to their small size (10–100 nm), micelles can efficiently penetrate tumor vasculature via the enhanced permeability and retention (EPR) effect, achieving passive targeting. Furthermore, smart micelles constructed from environmentally responsive materials can disassemble upon exposure to pH or enzymatic stimuli at the target site, enabling precise intracellular release and endosomal escape of the encapsulated drug.
Polymer Hydrogels and Nanogels for Local and Sustained Release
Polymer hydrogels are three-dimensional networks formed by hydrophilic polymer chains through physical or chemical crosslinking, exhibiting high water content and tissue-like viscoelasticity with excellent biocompatibility. Nanogels combine the high loading capacity of hydrogels with the flexibility of nanoparticles, allowing long-term zero-order or pulsatile release of protein drugs through network swelling or degradation mechanisms. These systems are particularly suitable for subcutaneous or local administration, effectively avoiding systemic side effects while maintaining therapeutic concentrations in the local microenvironment over extended periods.
Polymeric Microspheres for Controlled Protein Delivery
Polymeric microspheres are spherical carriers at the micron scale, typically prepared from biodegradable materials such as PLGA or PLA, and represent a core technology for long-acting depot formulations. By precisely controlling polymer molecular weight, monomer composition, and degradation kinetics, microspheres can extend the release period of peptide drugs to weeks or even months, reducing dosing frequency and improving patient compliance. This technology is widely applied in the delivery of growth hormones, GnRH agonists, and other chronic disease drugs, and demonstrates high maturity in scale-up and quality control.
Polymersomes as Vesicular Carriers for Biomacromolecules
Polymersomes are synthetic analogs of liposomes, assembled from amphiphilic block copolymers into bilayer or multilayer membrane structures, offering higher mechanical stability and lower membrane permeability than traditional liposomes. Their unique hydrophilic core and hydrophobic membrane allow simultaneous loading of water-soluble proteins (in the core) and hydrophobic drugs (in the membrane), making them ideal for combination therapy development. Furthermore, the membrane thickness and chemical properties are highly tunable, providing advantages in controlled drug release, enhanced in vivo stability, and surface functionalization.
Polymeric Nanocapsules for Enhanced Protein Protection
Polymeric nanocapsules possess a characteristic core–shell structure, typically consisting of a polymer shell encapsulating a liquid (aqueous or oily) core. This configuration isolates sensitive protein drugs from the external physiological environment. The polymer shell offers strong physical protection, effectively preventing denaturation, aggregation, or enzymatic degradation during storage, transport, and circulation, making it particularly suitable for highly unstable enzyme therapeutics. By adjusting shell thickness, porosity, and polymer degradation rate, drug diffusion can be precisely controlled, enabling sustained and constant release.
Polymer–Protein Conjugates for Improved Stability and Pharmacokinetics
Polymer–protein conjugates are not simple physical encapsulations but involve covalent attachment of water-soluble polymers (e.g., PEG, polyglutamic acid) to the protein surface, remodeling the physicochemical properties at the molecular level. This modification significantly increases the protein's hydrodynamic radius, effectively evading glomerular filtration, while the polymer chains shield surface epitopes to reduce immunogenicity. As one of the most clinically successful strategies (e.g., PEGylated proteins), conjugation has been proven to greatly extend half-life while maintaining bioactivity.
Application Scenarios of Polymer Carriers for Protein & Peptide Drugs
The high versatility of polymer carrier technologies enables them to meet therapeutic demands across various disease areas, from improving pharmacokinetics of biologics to enabling non-invasive administration routes.
Protein Therapeutics Delivery
Polymer carriers play an indispensable role in delivering large protein molecules such as cytokines (e.g., interferons), therapeutic enzymes (for enzyme replacement therapy), and growth factors. PEGylation or polymer nanocapsule encapsulation can significantly increase protein hydrodynamic volume, evading rapid renal clearance and maintaining stable plasma concentrations, effectively addressing the short half-life challenge. Moreover, the protective microenvironment provided by the polymer matrix prevents protein unfolding and aggregation under physiological conditions, ensuring that the therapeutic protein retains its native bioactivity at the target site, thereby improving bioavailability and safety.
Peptide Drug Delivery Systems
For short metabolic peptides such as insulin and GLP-1 receptor agonists, polymer systems aim to overcome intrinsic limitations of rapid in vivo clearance and enzymatic degradation. Long-acting release microspheres or in situ hydrogel implants constructed from biodegradable polymers (e.g., PLGA) can achieve steady zero-order release over weeks or months, avoiding the peaks and troughs associated with traditional injections. Smart stimuli-responsive polymers further enable peptides to be released in a closed-loop manner according to physiological signals (e.g., pH changes corresponding to blood glucose levels), providing a revolutionary approach for long-term management of diabetes and chronic metabolic disorders.
Oncology and Targeted Therapeutic Applications
In cancer therapy, polymer–drug conjugates and nanomicelles leverage the EPR effect to achieve passive accumulation of cytotoxic proteins or antibody drugs in tumor tissues. By modifying the carrier surface with specific ligands such as folate, transferrin, or RGD peptides, polymer systems can further enable active recognition and receptor-mediated endocytosis by cancer cells, significantly increasing intracellular uptake. This precise delivery mechanism maximizes antitumor efficacy while minimizing systemic toxicity to normal organs, particularly the heart and bone marrow.
Oral, Injectable, and Transdermal Formulations
Advances in polymer technologies have greatly expanded administration routes for biologics beyond traditional intravenous injection, improving patient compliance. For oral delivery, pH-sensitive and mucoadhesive polymers protect protein drugs from gastric acid and digestive enzymes while facilitating trans-epithelial absorption. For transdermal delivery, microneedle arrays made from soluble polymers can painlessly penetrate the stratum corneum and deliver drugs directly to the dermis. Injectable in situ hydrogels enable liquid formulations to rapidly transform into semi-solid depots post-injection, combining administration convenience with long-acting therapeutic effects.
Key Design Considerations for Protein and Peptide Delivery Polymers
Designing a successful polymer carrier requires comprehensive consideration of multiple factors to achieve optimal therapeutic index.
- Optimizing Polymer Molecular Weight and Architecture: Polymer molecular weight directly affects circulation time and renal clearance. Polymer topology (linear, branched, star-shaped, or dendritic) influences solution viscosity and interactions with cell membranes. Low polydispersity (PDI) is critical for batch-to-batch consistency.
- Balancing Polymer–Drug Interactions and Bioactivity: Interactions between the carrier and drug must be moderate. Excessive binding may hinder release or denature the protein, while insufficient binding may cause premature leakage (burst release) before reaching the target site.
- Enhancing Targeted Delivery and Cellular Uptake: For drugs requiring intracellular action (e.g., peptides targeting intracellular sites), carriers should incorporate cell-penetrating peptides (CPPs) or ligands to trigger endocytosis, while also considering endosomal escape mechanisms.
- Reducing Off-Target Effects and Immunogenic Responses: Although many polymers are considered biologically inert, their metabolites or surface charges (especially highly cationic polymers) may induce cytotoxicity or complement activation. Safety assessment should be integrated throughout development.
- Ensuring Scalability and Manufacturing Reproducibility: Scaling from lab to GMP production is often a major bottleneck. Design must account for synthetic simplicity, purification challenges, and supply chain stability of raw materials.
- Stability, Sterility, and Storage Considerations: Formulations must withstand sterilization (filtration or irradiation) while maintaining physicochemical stability during long-term storage. Lyophilization optimization often requires polymers with specific thermal transition properties.
How to Select the Right Polymer Carrier for Your Specific Drug?
The process of selecting a polymer is a multidimensional decision matrix that must be evaluated based on the intrinsic properties of the drug, the intended clinical treatment goals (Target Product Profile, TPP), and subsequent industrial feasibility.
Comparing Linear vs. Branched Polymers for Stability
Polymer topology directly determines hydrodynamic volume and steric shielding efficiency.
- Linear Polymers: Linear PEG, for example, generally exhibits a larger radius of gyration, providing excellent "cloud-like" steric protection. This makes it particularly suitable for preventing proteases from accessing the protein, thereby maximizing circulation half-life.
- Branched or Dendritic Polymers: Multi-arm PEG or PAMAM dendrimers offer a high density of surface functional groups. They are ideal for scenarios requiring high drug loading or multifunctional modifications for simultaneous drug and targeting ligand conjugation. Branched structures also tend to form tighter protective layers, enhancing thermal stability of the formulation.
Choosing Between Biodegradable vs. Non-Biodegradable Matrices
Polymer degradability must align with dosing frequency and treatment duration.
- Non-Biodegradable Polymers: For carriers below the renal threshold (<30–50 kDa), such as low-molecular-weight PEG, rapid renal clearance ensures high safety. These polymers are typically used for short- or medium-acting systemic delivery, focusing on improving solubility and circulation time.
- Biodegradable Polymers: For long-acting depot formulations or high-molecular-weight nanocarriers, degradable materials (e.g., PLGA, PCL, polyamino acids) are necessary to avoid accumulation in the reticuloendothelial system (RES) or lysosomes, which can lead to lysosomal storage diseases or chronic inflammation. Degradation rates should be precisely tuned to match the drug release window.
Matching Polymer Functionality with Protein Surface Chemistry
Successful conjugation or encapsulation relies on precise matching between polymer functional groups and protein surface chemistry.
- Site-Specificity: To preserve protein bioactivity, random modification near active sites should be avoided. For example, polymer derivatives targeting the N-terminal α-amino group (leveraging pKa differences) or introduced non-natural amino acids (e.g., azide groups) for click chemistry are preferable over conventional random lysine modification.
- Charge Interaction: In physical encapsulation systems, polymer charge density is critical. Cationic polymers can form polyplexes with negatively charged nucleic acids or certain proteins but may induce cytotoxicity. Therefore, matching isoelectric points (pI) or using charge-shielding strategies (e.g., PEGylated outer layers) is key to balancing drug loading efficiency and biocompatibility.
Services
Accelerate Your Drug Delivery with BOC Sciences' Polymer Solutions
Leveraging expertise in polymer chemistry, drug delivery, and biomacromolecule modification, BOC Sciences provides customized polymer solutions for protein and peptide drug development. Our services cover polymer synthesis, functional modification, delivery system design, and bioconjugation, enhancing drug stability, bioavailability, and therapeutic efficacy.

Custom Polymer Synthesis for Protein & Peptide Delivery
- Tailored synthesis of PEG, PLGA, PCL, PGA, and their copolymers.
- Precise control of molecular weight, molecular weight distribution, and hydrophilic/hydrophobic balance.
- Design of linear, block, or biodegradable polymers to accommodate diverse drug molecules.
- Support for constructing polymer–protein/peptide complexes or encapsulation systems.

Polymer Modification and Functionalization for Therapeutic Optimization
- PEGylation or other shielding modifications to extend circulation half-life.
- Introduction of targeting ligands or receptor-recognition groups for tissue- or cell-specific delivery.
- Construction of pH-, enzyme-, or redox-responsive functional units for controlled release.
- Tuning surface charge and hydrophilicity/hydrophobicity to reduce aggregation and non-specific adsorption.

Polymer–Drug Compatibility and Formulation Optimization
- Analysis of polymer–drug interactions and binding stability.
- Evaluation of encapsulation efficiency, protective ability, and release behavior.
- Systematic characterization of particle size, morphology, and surface charge.
- Optimization of formulation conditions (pH, ionic strength, excipients).

Site-Specific Bioconjugation Services for Proteins and Peptides
- Site-directed conjugation strategies targeting specific amino acid residues (e.g., Cys, Lys, N-terminal).
- Support for polymer–protein, polymer–peptide, and multifunctional bioconjugate construction.
- Precise control of conjugation sites and ratios to preserve bioactivity.
- Provision of conjugate purification, structural characterization, and functional validation.
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
Why use polymers for protein or peptide delivery?
Polymers protect proteins and peptides from enzymatic degradation and physical instability during storage and administration. By encapsulating or conjugating biomolecules, you can improve their solubility, stability, and bioavailability. Polymer-based systems also enable controlled and sustained release, helping maintain therapeutic levels over time and reduce dosing frequency.
How do polymer carriers improve protein stability?
Polymer carriers create a protective microenvironment that minimizes aggregation, denaturation, and degradation of proteins or peptides. By tuning polymer composition, molecular weight, and hydrophilicity, sensitive biomolecules can be stabilized against temperature, pH, and shear stress, ensuring preserved biological activity throughout formulation, delivery, and release.
What types of polymers are suitable for protein or peptide delivery?
Suitable options include biodegradable synthetic polymers such as PLGA, PLA, PCL, and PEG-based materials, as well as natural polymers like chitosan and gelatin. Selection depends on protein properties, delivery route, and desired release profile. Customization enables optimization of loading efficiency, stability, and biocompatibility for specific therapeutic applications.
How do polymers protect proteins and peptides from degradation?
Polymers primarily provide protection through steric shielding and physical isolation. Hydrophilic polymer chains (e.g., PEG) form a large hydration layer on the protein surface, effectively blocking proteases. Nanoparticles or micelles further encapsulate the drug in the core, isolating it from external physiological factors such as extreme pH, enzymes, and oxidants, thereby maintaining protein conformation and bioactivity.
Can polymer carriers control the release of protein therapeutics?
Yes. Polymer carriers are ideal platforms for controlled drug release. By adjusting molecular weight, copolymer composition, and crosslinking density of biodegradable polymers (e.g., PLGA), the dissolution rate of the matrix can be precisely tuned, enabling sustained release from days to months. Additionally, stimuli-responsive polymers can trigger pulsatile release in response to pathological signals, such as acidic pH or specific enzymes.
How are polymers selected for specific protein or peptide drugs?
Polymer selection should be tailored based on the drug's physicochemical properties (molecular weight, isoelectric point, hydrophilicity) and target product profile (TPP). For long-acting injectable microspheres, biodegradable materials like PLGA are preferred, whereas hydrophilic, non-degradable PEG is commonly used to extend plasma half-life. Comprehensive evaluation also includes polymer biocompatibility, metabolite toxicity, and compatibility of surface chemistry with drug conjugation sites.

 Fig. 1. Comparison showing how polymer shielding protects proteins from enzymatic degradation and extends circulation time (BOC Sciences Authorized).
Fig. 1. Comparison showing how polymer shielding protects proteins from enzymatic degradation and extends circulation time (BOC Sciences Authorized). Fig. 2. Structural classification of polymeric nanocarriers including micelles, nanospheres, polymersomes, and hydrogels for drug delivery (BOC Sciences Authorized).
Fig. 2. Structural classification of polymeric nanocarriers including micelles, nanospheres, polymersomes, and hydrogels for drug delivery (BOC Sciences Authorized).