Injectable Polymer Hydrogels for Sustained Drug Delivery and Controlled Release
The landscape of pharmaceutical formulation is undergoing a paradigm shift, moving away from systemic administration toward localized, smart delivery systems. Among the most promising advancements in this field are injectable polymer hydrogels. These stimuli-responsive biomaterials offer a unique solution to the historic challenges of bioavailability, patient compliance, and therapeutic precision. For researchers and formulators, understanding the physicochemical properties and rheological behaviors of these polymers is essential for designing next-generation drug delivery systems (DDS). This article explores the mechanisms, critical polymer classes, and design strategies behind injectable hydrogels.
Resources
What Are Injectable Polymer Hydrogels?
Injectable polymer hydrogels are three-dimensional, water-swollen networks of polymer chains that exhibit a unique phase transition. Unlike pre-formed solid implants that require surgical insertion, these systems are designed as free-flowing solutions or suspensions ex vivo, allowing for minimally invasive administration via a standard syringe. Upon reaching the target site, they undergo a rapid sol-gel transition to form a depot, responding to physiological stimuli such as temperature, pH, or ionic strength.
- Definition and Composition of Injectable Hydrogels
Fundamentally, an injectable hydrogel is a three-dimensional, hydrophilic polymer network capable of absorbing and retaining large amounts of water (often >90% by weight) while maintaining structural integrity.
- Structural Mimicry: Their high water content and porous architecture closely mimic the native extracellular matrix (ECM), reducing interfacial tension with surrounding tissues and minimizing inflammatory responses.
- Composition: These networks are constructed from homopolymers or block copolymers (e.g., PLGA-PEG-PLGA). The polymer backbone is typically engineered with hydrophilic segments (to ensure solubility) and hydrophobic or functional domains (to drive crosslinking). The mesh size of the resulting network is a critical parameter, acting as a molecular sieve that dictates the diffusion rate of encapsulated therapeutics, from small molecules to macromolecular biologics.
- Mechanisms of Sol-Gel Transition in Injectable Polymers
The efficacy of an injectable hydrogel hinges on its rheological behavior—specifically, the kinetics of the sol-gel transition. This phase change is driven by thermodynamic forces triggered by physiological stimuli.
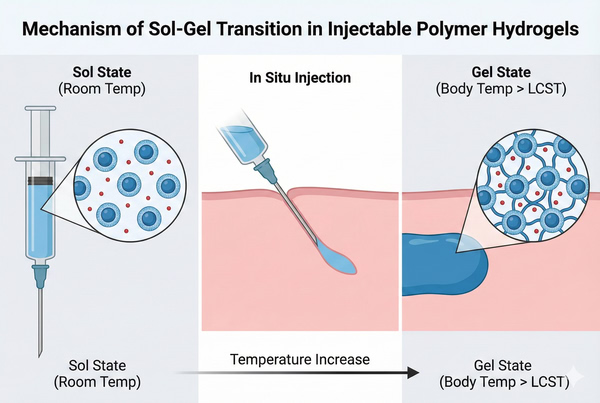 Fig. 1. Schematic illustration of thermosensitive sol-gel transition from liquid solution to solid state (BOC Sciences Authorized).
Fig. 1. Schematic illustration of thermosensitive sol-gel transition from liquid solution to solid state (BOC Sciences Authorized).
- Thermosensitive Transition (LCST/UCST): The most prevalent mechanism involves polymers exhibiting a lower critical solution temperature (LCST). Below the LCST, hydrogen bonding between water molecules and the polymer's hydrophilic groups dominates, maintaining a solution state. As the temperature rises to body temperature (37℃), hydrophobic interactions prevail. This entropic gain drives the dehydration of hydrophobic blocks, causing them to aggregate into micellar cores and form a physical gel network.
- pH-Responsive Gelation: Polymers containing ionizable groups (e.g., carboxylic acids or amines) undergo conformational changes based on the pKa of the pendant groups. A change from formulation pH to physiological pH (7.4) triggers deprotonation or protonation, altering the chain solubility and inducing gelation.
- Shear-Thinning (Thixotropy): Ideally, the precursor solution exhibits shear-thinning behavior—its viscosity decreases significantly under the high shear stress of injection (flowing easily through a needle) and instantly recovers to a high-viscosity gel state upon the cessation of shear at the injection site.
- Biocompatibility and Biodegradability Considerations
- Biocompatibility: The material must be chemically inert and non-immunogenic. The hydrogel surface should resist non-specific protein adsorption (fouling), which is the precursor to the foreign body response (FBR) and fibrous capsule formation.
- Biodegradability and Clearance: A definitive advantage of these systems is their ability to degrade into non-toxic metabolites.
- Hydrolysis: Synthetic polyesters (e.g., PLGA, PLA) degrade via the hydrolytic cleavage of ester bonds, producing lactic acid and glycolic acid, which enter the Krebs cycle and are eliminated as CO2 and water.
- Renal Clearance: The degradation products or the polymer residues must eventually fall below the renal filtration threshold (typically < 40-60 kDa) to ensure they are cleared from the body without accumulation in the liver or spleen.
Functional Roles of Polymer Hydrogels in Drug Delivery
The utility of polymer hydrogels extends beyond simple containment; they act as dynamic reservoirs that modulate the pharmacokinetics of the encapsulated payload.
- Controlled and Sustained Release of Therapeutics: The primary function of these systems is to prolong the therapeutic window. Release kinetics are governed by two main mechanisms: diffusion through the polymer matrix and erosion of the matrix itself. By tuning the cross-linking density and polymer concentration, formulators can achieve zero-order release profiles, mitigating the burst effect often seen in traditional formulations and maintaining plasma concentrations within the therapeutic range for days, weeks, or months.
- Protection of Drugs from Degradation: Biologics, such as proteins, peptides, and nucleic acids, are highly susceptible to enzymatic degradation and rapid renal clearance. The hydrogel matrix serves as a physical barrier, shielding these sensitive cargo molecules from proteases and nucleases in the physiological environment. This protection is critical for maintaining the bioactivity of the drug until it is released.
- Injectable Hydrogel Rheology and Tissue Compatibility: Rheological properties, specifically shear-thinning behavior, are critical for injectability. The formulation must possess low viscosity under high shear (during injection) to pass through fine-gauge needles but must rapidly recover high viscosity and elastic modulus (G') upon cessation of shear to localize at the injection site. This mechanical matching with soft tissue minimizes irritation and prevents the migration of the depot.
- Targeted Delivery and Localized Therapeutic Effect: Systemic administration often leads to off-target toxicity. Injectable hydrogels allow for the precise placement of high-concentration therapeutics directly at the disease site—such as a tumor margin or an articular joint. This localized approach significantly improves the therapeutic index by maximizing efficacy at the target while minimizing systemic exposure.
Critical Polymer Classes for Hydrogel Synthesis
The design of an effective injectable hydrogel begins with the selection of the appropriate polymer backbone. The choice between synthetic and natural polymers—or a hybrid of both—fundamentally dictates the hydrogel's gelation mechanism, degradation kinetics, mechanical stiffness, and biological interaction. For formulators, understanding the structure-property relationships of these materials is essential for tailoring the delivery system to the specific therapeutic window and target tissue.
Synthetic biodegradable block copolymers represent the frontier of thermosensitive hydrogel technology. These materials are favored for their batch-to-batch consistency and the ability to precisely tune their physicochemical properties through chemical synthesis.
- Thermosensitive Phase Transition Mechanisms: The most widely investigated systems are amphiphilic triblock copolymers, such as PLGA-PEG-PLGA (Poly(D,L-lactic-co-glycolic acid)-b-poly(ethylene glycol)-b-poly(D,L-lactic-co-glycolic acid)). These polymers exhibit a reverse thermal gelation behavior. At lower temperatures, the hydrophilic PEG blocks dominate, allowing the polymer to remain in a sol state. As the temperature rises to the lower critical solution temperature (LCST), the hydrophobic PLGA blocks undergo dehydration and associate into micellar cores. At critical concentrations, these micelles aggregate to form a supramolecular hydrogel network.
- Tunability of Degradation and Release: A distinct advantage of synthetic copolymers is the ability to modulate the Lactide:Glycolide (LA/GA) ratio. A higher glycolide content generally increases hydrophilicity and accelerates hydrolysis rates, whereas a higher lactide content prolongs the degradation profile. Furthermore, adjusting the molecular weight (Mw) of the PEG block impacts the gelation temperature and mechanical strength (G'), allowing researchers to customize the formulation for specific injection sites (e.g., subcutaneous vs. intra-articular).
Natural Polysaccharides (Chitosan, Alginate, Hyaluronic Acid)
Natural polysaccharides are inherently bioactive, often mimicking the mammalian ECM. They offer superior biocompatibility and unique cell-signaling capabilities, though their purification and modification require rigorous quality control to ensure consistency.
- Chitosan and pH-Responsiveness: Derived from the deacetylation of chitin, chitosan is a cationic linear polysaccharide composed of β-(1-4)-linked D-glucosamine and N-acetyl-D-glucosamine. Its amine groups (pKa ≈ 6.5) become protonated in acidic environments, making it soluble. Upon injection into a physiological pH environment (pH 7.4), deprotonation induces a sol-gel transition. The degree of deacetylation (DDA) is a critical quality attribute that influences its solubility, biodegradability, and immunogenicity. Chitosan is particularly noted for its mucoadhesive properties, making it ideal for transmucosal drug delivery.
- Alginate and Ionic Crosslinking: Alginate is an anionic polysaccharide composed of mannuronic acid (M) and guluronic acid (G) blocks. It undergoes rapid gelation in the presence of divalent cations (e.g., Ca2+, Mg2+) via the formation of "egg-box" junctions between G-blocks. This mild, non-covalent gelation process is exceptionally friendly to sensitive biologics and cells.
- Hyaluronic Acid (HA) and Chemical Modification: HA is a ubiquitous component of human connective tissue. While native HA degrades rapidly, it serves as an excellent backbone for chemical modification. Functionalization with methacrylate groups (e.g., HAMA) allows for photo-crosslinking, while Schiff-base reactions (e.g., oxidized HA with adipic acid dihydrazide) enable injectable, self-healing hydrogels. HA's natural affinity for CD44 receptors also allows for active targeting of CD44-overexpressing tumor cells.
Polypeptides and Protein-Based Polymers
Polypeptide hydrogels bridge the gap between synthetic precision and natural bioactivity. They degrade into endogenous amino acids, minimizing the risk of toxic accumulation during chronic administration.
- Self-Assembling Peptide Amphiphiles: These synthetic peptides are designed with specific sequences that trigger self-assembly into nanofibers or hydrogels in response to physiological ionic strength or pH. They offer a high degree of control over the epitope presentation, promoting specific cell interactions.
- Elastin-Like Polypeptides (ELPs): ELPs are recombinant biopolymers derived from the hydrophobic domain of tropoelastin. They exhibit a unique inverse transition cycling (ITC) behavior, transitioning from soluble to insoluble coacervates above a transition temperature (Tt). By genetically engineering the amino acid sequence, researchers can precisely control the Tt to coincide with body temperature, creating robust, highly elastic depots suitable for cardiovascular or load-bearing tissue engineering applications.
- Gelatin Methacryloyl (GelMA): While technically a modified protein derivative, GelMA has gained immense popularity. It combines the bioactivity of gelatin (RGD sequences for cell attachment) with the tunable mechanical properties of methacryloyl crosslinking, making it a versatile platform for both drug delivery and 3D bioprinting.
Looking for Custom Polymer Hydrogels?
Whether you need injectable hydrogels, stimuli-responsive systems, or functionalized networks, our experts deliver tailored synthesis and formulation solutions to support your research and development goals.
Classification of Polymer Hydrogels: A Structural and Functional Perspective
The versatility of polymer hydrogels lies in their diverse structural architectures and physicochemical properties. For formulation scientists, categorizing these biomaterials is not merely an academic exercise but a practical framework for selecting the appropriate matrix based on the desired sol-gel transition, degradation kinetics, and rheological behavior. Hydrogels are systematically classified based on four critical dimensions: polymer origin, crosslinking mechanism, stimuli-responsiveness, and network topology.
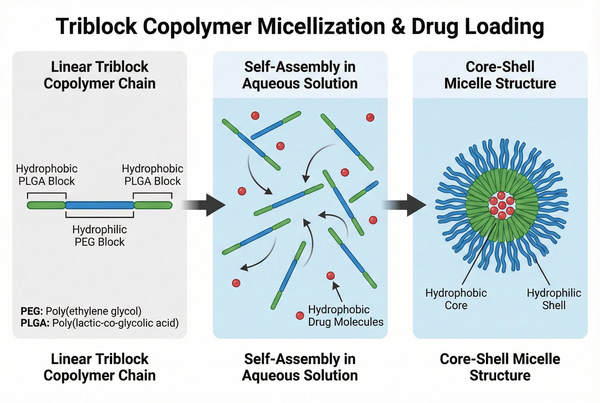 Fig. 2. Self-assembly of amphiphilic triblock copolymers into core-shell micelles for drug encapsulation (BOC Sciences Authorized).
Fig. 2. Self-assembly of amphiphilic triblock copolymers into core-shell micelles for drug encapsulation (BOC Sciences Authorized).
Based on Polymer Source: Biogenicity vs. Tunability
The origin of the polymer backbone dictates the inherent biological interaction and immunogenicity of the hydrogel system.
- Natural Polymer Hydrogels: Derived from biological sources, these polymers (e.g., Alginate, Chitosan, Collagen, Hyaluronic Acid) possess intrinsic biocompatibility and often contain cell-signaling domains (such as RGD sequences) that mimic the ECM. They are particularly advantageous for wound healing and tissue engineering, though they may suffer from batch-to-batch variability and rapid enzymatic degradation.
- Synthetic Polymer Hydrogels: Engineering polymers such as PEG, PVA, PLGA, and PCL offers superior control over molecular weight and polydispersity. Synthetic hydrogels provide reproducible mechanical properties and predictable degradation profiles, making them the preferred choice for strictly controlled drug delivery systems where long-term stability is required.
Based on Crosslinking Mechanism: Stability and Reversibility
The method of network formation determines the hydrogel's mechanical stiffness and its ability to respond to shear stress.
- Physically Crosslinked (Supramolecular) Hydrogels: These networks are formed via reversible non-covalent interactions, including hydrogen bonding, ionic interactions (e.g., Alginate-Ca2+), and hydrophobic association. They typically exhibit shear-thinning behavior, allowing the gel to flow during injection and recover its structure in situ. However, their mechanical strength is generally lower compared to chemical gels.
- Chemically Crosslinked Hydrogels: Formed through permanent covalent bonds via reactions such as Michael-type addition, Schiff base formation, or photopolymerization (e.g., PEG-Diacrylate). Chemical crosslinking allows for precise control over mesh size and swelling ratio, offering robust stability for implantable devices but requiring careful removal of unreacted monomers or catalysts.
Based on Stimuli-Responsiveness: The Smart Systems
Smart hydrogels undergo abrupt physiochemical changes in response to environmental triggers, enabling spatiotemporal control over drug release.
- pH-Sensitive Hydrogels: These polymers contain ionizable pendant groups (e.g., carboxylic acids or amines). Changes in pH cause protonation or deprotonation, leading to electrostatic repulsion and changes in hydrodynamic volume. This is critical for targeting the acidic tumor microenvironment or protecting drugs through the GI tract.
- Temperature-Sensitive Hydrogels: These systems exhibit a lower critical solution temperature (LCST). Polymers like Pluronic F127 or Poly(N-isopropylacrylamide) (PNIPAM) exist as sols at room temperature but undergo a phase transition to a gel state at physiological temperature due to increased hydrophobic interactions.
- Exogenous Stimuli-Responsive Hydrogels: Systems responsive to light, magnetic fields, or ultrasound allow for on-demand dosing, triggered by external equipment.
Based on Network Structure: Topology and Toughness
The internal topology affects the drug loading capacity and the mechanical durability of the depot.
- Single Network Hydrogels: Composed of a single polymer type. While simple to synthesize, they often lack high mechanical toughness.
- Interpenetrating Polymer Networks (IPN)/Double Network (DN): These consist of two independently crosslinked networks entangled at the molecular level. DN hydrogels are renowned for their high fracture toughness, where the first network (rigid) sacrifices itself to dissipate energy while the second network (soft) maintains structural integrity.
- Nanocomposite Hydrogels: The incorporation of nanomaterials (e.g., graphene oxide, silica nanoparticles, or nanoclays) into the polymer matrix enhances mechanical strength and can provide additional functionalities, such as electrical conductivity or photothermal properties.
Formulation and Design Strategies of Polymer Hydrogels
The transition from a promising polymer candidate to a functional injectable drug delivery system requires a rigorous formulation strategy. This phase demands a delicate balance between thermodynamic stability, rheological performance, and pharmacokinetic (PK) targets. Successful design hinges on optimizing the interplay between the drug's physicochemical properties and the hydrogel's network architecture.
Drug–Hydrogel Compatibility and Loading Strategies
Achieving high encapsulation efficiency and minimizing the burst release effect are the primary challenges in formulation. The loading strategy must be tailored to the drug's hydrophobicity and molecular weight.
Physical Entrapment vs. Chemical Conjugation:
- For hydrophilic macromolecules (e.g., proteins, antibodies), physical entrapment within the aqueous phase of the hydrogel is standard. However, this often relies solely on steric hindrance, leading to rapid diffusion.
- For hydrophobic small molecules, partitioning into the hydrophobic domains (e.g., the PLGA core in PLGA-PEG-PLGA micelles) significantly enhances solubility and retention.
- Chemical Conjugation: To prevent premature leakage, drugs can be covalently tethered to the polymer backbone via cleavable linkers (e.g., ester, hydrazone, or disulfide bonds). This converts the release mechanism from simple diffusion to a hydrolysis-controlled process.
Affinity-Based Loading:
- Advanced strategies involve incorporating affinity ligands, such as cyclodextrins or heparin, into the hydrogel network. These inclusions create non-covalent, reversible interactions with the drug payload, effectively slowing diffusion and linearizing the release profile without permanent chemical modification.
Controlling Release Kinetics and Duration
The release profile of an injectable hydrogel is governed by two competing mechanisms: Fickian diffusion and polymer erosion. Tuning these parameters allows for the customization of release duration from days to months.
Modulating Mesh Size (ξ):
- The mesh size of the hydrogel network acts as a molecular sieve. By increasing the crosslinking density or polymer concentration, the effective mesh size decreases, retarding the diffusion of the payload. This is particularly effective for macromolecular drugs where the hydrodynamic radius approaches ξ.
Erosion-Controlled Release:
- Bulk Erosion: Water penetrates the entire matrix, causing uniform degradation (typical of PLGA).
- Surface Erosion: Degradation occurs only at the polymer-water interface (typical of polyanhydrides), providing near zero-order release kinetics.
- Adjusting the Lactide: Glycolide ratio or the molecular weight allows formulators to synchronize the degradation rate with the desired therapeutic window.
Stability, Sterility, and Storage Considerations
Translating a hydrogel formulation from the bench to the clinic requires addressing critical stability and regulatory hurdles.
Lyophilization and Reconstitution:
- Aqueous polymer solutions, particularly those with ester linkages, are prone to hydrolysis during storage. Lyophilization (freeze-drying) is the standard approach to ensure long-term stability. However, the process requires the addition of cryoprotectants (e.g., trehalose, sucrose) to prevent polymer aggregation and maintain the porous structure upon reconstitution.
Sterilization Challenges:
- Gamma Irradiation: While effective, high-energy radiation often induces chain scission, significantly lowering the molecular weight and altering the sol-gel transition temperature and mechanical strength.
- Sterile Filtration: Suitable only for low-viscosity precursor solutions prior to gelation. For highly viscous systems, aseptic processing remains the only viable, albeit costly, option.
In Vitro and In Vivo Evaluation of Injectable Hydrogel Systems
Comprehensive characterization is essential to predict in vivo performance and ensure safety.
Rheological Profiling:
- Oscillatory rheometry is used to determine the viscoelastic properties. The crossover point where the Storage Modulus exceeds the Loss Modulus defines the gelation point. Key parameters include the Gelation Time (must be long enough to allow injection but short enough to prevent migration) and Yield Stress (indicating injectability).
In Vitro Release Assays:
- Standard dissolution methods (e.g., USP Apparatus 4 or dialysis bag methods) quantify drug release rates. These studies must simulate physiological conditions and often require sink conditions to accurately mimic in vivo clearance.
Biocompatibility and Degradation:
- Cytotoxicity: MTT or CCK-8 assays on relevant cell lines (e.g., fibroblasts, macrophages) to screen for leachable toxins.
- In Vivo Degradation: Subcutaneous or intramuscular injection studies in animal models to monitor the gel's resorption rate and the local inflammatory response (foreign body reaction) over time via histology.
Application Scenarios of Injectable Polymer Hydrogels
The versatility of injectable hydrogels has catalyzed their adoption across a broad spectrum of therapeutic areas. By transforming from a flowable solution to a robust depot in situ, these systems provide an effective intermediate solution between systemic administration and surgical implantation. They offer a platform to overcome critical pharmaceutical challenges, including poor bioavailability, rapid renal clearance, and off-target toxicity.
Sustained Delivery of Small Molecules
For hydrophobic small molecule drugs, solubility and retention at the target site are persistent hurdles. Injectable hydrogels, particularly amphiphilic block copolymers like PLGA-PEG-PLGA, address these by incorporating hydrophobic drugs into micellar cores within the gel network.
- Ocular Therapeutics: In the treatment of posterior segment eye diseases (e.g., age-related macular degeneration), hydrogels can maintain therapeutic drug levels in the vitreous humor for months, replacing frequent, invasive intravitreal injections.
- Local Anesthetics & Pain Management: Hydrogel depots loaded with anesthetics (e.g., Bupivacaine) provide prolonged nerve blockade for post-operative pain relief, significantly reducing opioid consumption.
- Mechanism: The release is governed by the diffusion of the drug through the water-filled pores and the simultaneous erosion of the polymer matrix, ensuring a linear release profile without the burst effect typical of simple suspensions.
Injectable Protein and Peptide Therapeutics
Biologics such as insulin, growth hormones, and monoclonal antibodies are notoriously unstable. They are prone to denaturation, aggregation, and enzymatic hydrolysis in physiological environments.
- Preservation of Bioactivity: Unlike hydrophobic microparticles that use organic solvents during fabrication (which can denature proteins), hydrogels provide a gentle, aqueous encapsulation environment. The high water content maintains the protein's native tertiary structure.
- Metabolic Disorders: Thermosensitive hydrogels are being developed for the basal delivery of insulin and GLP-1 receptor agonists. By forming a subcutaneous depot, these systems can mimic physiological basal secretion for weeks, improving patient compliance in diabetes management.
Localized Oncology Therapies
Systemic chemotherapy is often limited by dose-limiting toxicities (e.g., cardiotoxicity, neutropenia). Injectable hydrogels enable Intratumoral (IT) Administration, a strategy that maximizes the therapeutic index.
- Chemotherapy Depots: Injecting a drug-loaded hydrogel (e.g., Paclitaxel or Doxorubicin) directly into the tumor mass or the resection cavity post-surgery creates a high local concentration gradient. This ensures sustained cytotoxicity to residual cancer cells while keeping plasma concentrations negligible.
- Radio-immunotherapy: Hydrogels are increasingly used to retain radioisotopes or immunomodulators (e.g., checkpoint inhibitors like anti-PD-1/PD-L1 antibodies) within the tumor microenvironment. This localized retention potentiates the immune response and prevents the systemic autoimmune adverse events often associated with immunotherapy.
mRNA and Nucleic Acid Delivery
The rise of gene therapy and mRNA vaccines has created a demand for carriers that can protect nucleic acids from ubiquitous nucleases.
- Cationic Hydrogel Systems: Polymers with cationic functional groups, such as Chitosan or PEI-modified polypeptides, interact electrostatically with the negatively charged phosphate backbone of DNA/RNA.
- Polyplex Formation: This interaction forms stable polyplexes within the hydrogel matrix. Upon injection, the hydrogel facilitates cellular uptake via endocytosis and promotes endosomal escape—a critical step for the transfection efficiency of non-viral gene vectors. This approach is being explored for localized gene editing and angiogenesis induction in ischemic tissues.
Tissue Engineering and Regenerative Medicine Scaffolds
Beyond drug delivery, injectable hydrogels serve as bioactive scaffolds that support cell proliferation and tissue integration.
- Minimally Invasive Scaffolding: For irregular defects (e.g., traumatic brain injury cavities or cartilage defects), injectable hydrogels conform perfectly to the geometric complexity of the injury site—something pre-formed solid scaffolds cannot achieve.
- Co-delivery of Cells and Factors: These systems act as a dual-delivery vehicle, carrying both stem cells (e.g., MSCs) and osteoinductive/chondrogenic growth factors (e.g., BMP-2, TGF-β).
- Dynamic Remodeling: Ideally, the degradation rate of the polymer scaffold is synchronized with the rate of new tissue formation, ensuring that the mechanical load is gradually transferred from the polymer to the regenerating tissue.
Services
Polymer Hydrogel Custom Synthesis Services at BOC Sciences
BOC Sciences provides comprehensive polymer hydrogel solutions to support research, development, and application in drug delivery, tissue engineering, and bioactive material design. Our services cover high-quality polymer material supply, custom polymer synthesis and functional modification, hydrogel formulation and fabrication, stimuli-responsive design, drug or bioactive encapsulation, and full characterization and quality evaluation. By integrating expertise in polymer chemistry and hydrogel engineering, we help partners tailor mechanical properties, porosity, degradation, and release profiles to meet specific scientific and translational needs. From early-stage research to process scale-up, our solutions are designed to accelerate the development of robust, reproducible, and application-ready hydrogel systems.

Custom Polymer Synthesis & Modification
- Provides PEG, PVA, PCL, HA, and both natural and synthetic polymer raw materials
- Supports customization of molecular weight, end groups, and functional groups
- Tailored block, branched, or copolymer structures
- PEGylation, degradable linkers, and targeted modifications

Polymer Hydrogel Formulation & Fabrication
- Construction of chemically and physically crosslinked hydrogel systems
- Supports self-assembly, photo-crosslinking, and click chemistry preparation strategies
- Regulation of mechanical properties, porosity, and gelation rate
- Available in various forms such as microspheres, fibers, or 3D scaffolds

Stimuli-Responsive & Functional Hydrogel Design
- Design of pH-, temperature-, light-, enzyme-, and ROS-responsive hydrogels
- Controlled drug release rates and targeted, triggered delivery
- Functionalized hydrogels for cell culture or tissue engineering
- Incorporation of special functions such as adhesion, degradability, or conductivity

Drug/Bioactive Encapsulation & Delivery
- Encapsulation of small molecule drugs, peptides, proteins, and nucleic acids
- Optimization of encapsulation efficiency and drug loading
- Control of release profiles and release kinetics
- Enhancement of bioactivity and stability
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
What are injectable polymer hydrogels used for in drug delivery?
Injectable hydrogels are primarily used to create localized, sustained-release drug depots. They offer a minimally invasive alternative to surgical implants, allowing for the delivery of chemotherapeutics, proteins, and anti-inflammatory agents directly to target tissues (e.g., tumors, joints, or the posterior eye), thereby improving bioavailability and patient compliance.
How do hydrogels provide sustained drug release?
Hydrogels modulate release kinetics through two concurrent mechanisms: diffusion and erosion. The crosslinked polymer network acts as a physical barrier, retarding drug diffusion based on mesh size. Simultaneously, the biodegradable polymer backbone (e.g., PLGA or metabolic linkers) hydrolyzes over time, gradually releasing the entrapped payload in a controlled manner.
Can polymer hydrogels deliver proteins, peptides, or nucleic acids?
Yes, they are exceptionally well-suited for biologics. Unlike rigid hydrophobic implants, the high water content of hydrogels mimics physiological conditions, preventing protein denaturation and aggregation. Furthermore, cationic hydrogels can complex with anionic nucleic acids (DNA/RNA), protecting them from enzymatic degradation and facilitating cellular uptake for gene therapy applications.
How are hydrogels customized for specific therapeutic applications?
Researchers customize hydrogels by adjusting polymer parameters such as molecular weight, concentration, and the ratio of hydrophobic to hydrophilic blocks (e.g., Lactide:Glycolide ratio). These modifications precisely tune the sol-gel transition temperature, mechanical stiffness, and degradation rate to match the specific biological requirements of the target tissue and therapeutic window.

 Fig. 1. Schematic illustration of thermosensitive sol-gel transition from liquid solution to solid state (BOC Sciences Authorized).
Fig. 1. Schematic illustration of thermosensitive sol-gel transition from liquid solution to solid state (BOC Sciences Authorized). Fig. 2. Self-assembly of amphiphilic triblock copolymers into core-shell micelles for drug encapsulation (BOC Sciences Authorized).
Fig. 2. Self-assembly of amphiphilic triblock copolymers into core-shell micelles for drug encapsulation (BOC Sciences Authorized).