Polymer Nanoparticles for Gene Delivery: DNA, mRNA, siRNA and Oligo Therapeutics
The development of robust and efficient delivery vectors remains a cornerstone of modern nucleic acid research. While viral vectors have historically dominated early biological studies, synthetic non-viral systems, particularly polymer nanoparticles, have emerged as highly versatile alternatives. These macromolecular systems offer unparalleled chemical flexibility, allowing researchers to engineer custom carriers tailored to the specific physicochemical properties of the genetic payload. By neutralizing the negative charge of nucleic acids and condensing them into nanometric structures, polymers facilitate the safe transit of genetic material across complex biological barriers.
Resources
Introduction to Polymer Nanoparticles in Gene Delivery
Polymer nanoparticles used in gene delivery are sub-micron colloidal structures formed primarily through the electrostatic complexation of positively charged polymers with negatively charged nucleic acids. The resulting complexes, widely known as polyplexes, condense the genetic payload into dense, protective nanoparticles. Depending on the synthesis method and the polymer architecture, these nanoparticles can take the form of nanospheres, nanocapsules, or polymeric micelles, each offering distinct advantages for encapsulating hydrophilic biomolecules.
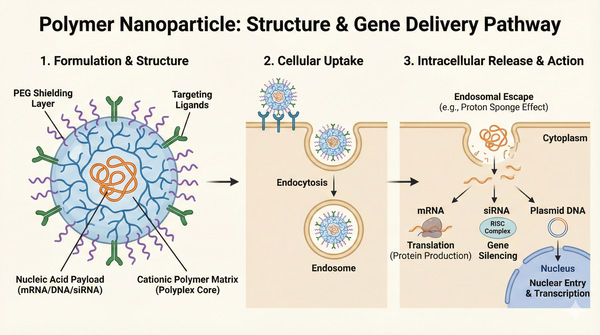 Fig. 1. Overview of polymer nanoparticle structure and intracellular gene delivery pathway (BOC Sciences Authorized).
Fig. 1. Overview of polymer nanoparticle structure and intracellular gene delivery pathway (BOC Sciences Authorized).
Why Polymers Are Ideal for Nucleic Acid Delivery?
Polymers provide a highly tunable platform for therapeutic development. Unlike rigid biological vectors, synthetic polymers can be precisely modified at the molecular level to adjust their molecular weight, charge density, degradation rate, and hydrophobic-hydrophilic balance. This profound level of chemical control allows formulation scientists to optimize the binding affinity between the carrier and the nucleic acid, ensuring stable encapsulation during transit and responsive unpacking once the target site is reached.
Key Advantages Over Viral and Lipid Systems
Polymer nanoparticles present several distinct advantages in preclinical research and therapeutic development. Compared to viral vectors, they exhibit significantly lower immunogenicity and lack the packaging constraints that limit the size of the delivered gene. Furthermore, their synthetic nature enables highly reproducible, large-scale manufacturing without the complex biological risks associated with viral production. When compared to basic lipid-based systems, certain polymer architectures offer superior structural stability in biological fluids and provide diverse conjugation sites for targeting ligands, creating a highly modular platform for advanced formulation design.
Core Challenges in Delivering DNA, mRNA, siRNA, and Oligos
Despite their immense potential, delivering naked nucleic acids into eukaryotic cells is hindered by a series of formidable biological barriers. A successful polymer nanocarrier must navigate an extracellular obstacle course, breach the cellular membrane, and survive the hostile intracellular environment.
- Stability and Degradation in Biological Environments: Unprotected nucleic acids are highly susceptible to enzymatic degradation. In systemic circulation or extracellular fluids, ubiquitous endonucleases and exonucleases rapidly cleave phosphodiester bonds, destroying the genetic blueprint before it reaches the cell. Additionally, interactions with serum proteins can lead to opsonization, causing premature clearance by the mononuclear phagocyte system. Polymer carriers must provide robust steric and electrostatic shielding to protect the payload from these destructive elements.
- Cellular Uptake and Endosomal Escape: Because both the cellular membrane and nucleic acids are strongly anionic, spontaneous cellular uptake is virtually nonexistent due to electrostatic repulsion. Polymer nanoparticles overcome this by maintaining a net positive surface charge, facilitating strong interactions with negatively charged proteoglycans on the cell surface. Following endocytosis, the critical bottleneck becomes escaping the endosome. If the polyplex remains trapped, it will be trafficked to the lysosome and degraded by acidic hydrolases.
- Targeting Specific Tissues and Cells: Achieving high accumulation at the desired biological site while minimizing off-target distribution is a primary objective in therapeutic development. While nanoscale particles can passively accumulate in certain microenvironments via the enhanced permeability and retention effect, true precision requires active targeting. Incorporating specific targeting strategies is vital to ensure the genetic payload reaches the correct cellular subpopulation without disrupting healthy tissues.
- Safety, Toxicity, and Immunogenicity Concerns: The use of highly cationic polymers can induce cytotoxicity by disrupting cellular membranes or interfering with critical intracellular processes. Furthermore, non-biodegradable polymers with high molecular weights can accumulate in reticuloendothelial organs over time. Designing polymers that balance optimal transfection efficiency with a favorable safety profile—such as incorporating biodegradable ester or disulfide linkages—is paramount for advancing from cellular models to complex in vivo studies.
Mechanistic Pathways: How Polymers Facilitate Intracellular Delivery
To engineer superior vectors, researchers must deeply understand the intricate intracellular journey of the polyplex. The transition from extracellular attachment to final intracellular unpackaging involves a cascade of highly dynamic biological and physical events.
- Cellular Internalization Routes: Polymer nanoparticles enter cells utilizing multiple endocytic pathways. The predominant mechanisms include clathrin-mediated endocytosis, caveolae-mediated uptake, and macropinocytosis. The specific internalization route is heavily dictated by the physicochemical properties of the nanoparticle, specifically its size, shape, and surface chemistry. Understanding these pathways is crucial, as the entry route often determines the subsequent intracellular trafficking and the overall efficiency of gene expression or silencing.
- The Proton Sponge Hypothesis: A primary mechanism utilized by many cationic polymers, notably polyethylenimine (PEI), to achieve endosomal escape is the proton sponge effect. These polymers possess a high buffering capacity due to secondary and tertiary amines. As the endosome attempts to acidify via ATPase proton pumps, the polymer absorbs the protons. This continuous influx of protons causes a parallel influx of chloride ions and water into the endosomal vesicle, leading to osmotic swelling and eventual physical rupture of the endosomal membrane, releasing the polyplex into the cytosol.
- Alternative Endosomal Escape Mechanisms: Beyond osmotic pressure, other structural phenomena drive endosomal escape. Certain polymers are engineered with hydrophobic domains that undergo conformational changes in acidic pH, exposing regions that directly interact with and destabilize the endosomal lipid bilayer. Additionally, fusogenic polymer behaviors can induce lipid mixing between the nanoparticle and the endosomal membrane, creating transient pores that allow the payload to slip into the safe confines of the cytoplasm.
- Intracellular Unpacking Kinetics: Once in the cytoplasm, the polyplex must disassemble to render the nucleic acid biologically active. The unpacking kinetics rely on a delicate balance: the binding affinity must be strong enough to survive extracellular transit but weak enough to dissociate intracellularly. This disassembly is often driven by interactions with intracellular polyanions, changes in pH, or the cleavage of responsive polymer bonds by high intracellular glutathione concentrations.
Polymer Materials Commonly Used for Gene Delivery Nanoparticles
The foundational success of any non-viral gene delivery vehicle hinges almost entirely on the selection and engineering of its constituent materials. The modern polymer chemistry landscape offers a vast and highly tunable library of macromolecules, enabling researchers to transition away from rigid, one-size-fits-all platforms toward highly specialized vectors. By manipulating parameters such as molecular weight, charge density, monomer sequence, and degradation kinetics, formulation scientists can precisely dictate how a nanoparticle interacts with nucleic acids, extracellular fluids, and intracellular compartments.
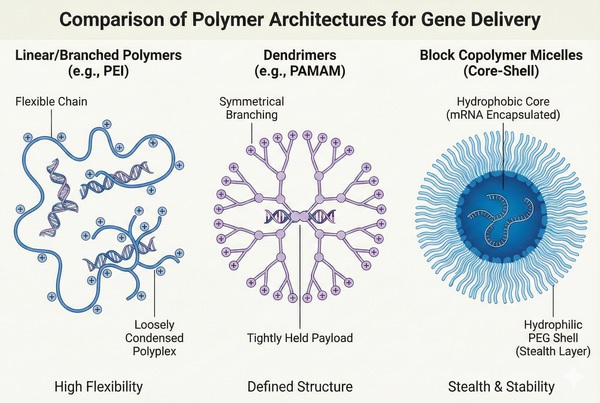 Fig. 2. Structural comparison of linear polymers, dendrimers, and block copolymer micelles (BOC Sciences Authorized).
Fig. 2. Structural comparison of linear polymers, dendrimers, and block copolymer micelles (BOC Sciences Authorized).
Cationic Polymers for Nucleic Acid Complexation
Cationic polymers represent the earliest and most extensively researched class of synthetic gene carriers. Their primary function relies on electrostatic interactions: the high density of positively charged amine groups along the polymer backbone binds spontaneously with the negatively charged phosphate backbone of DNA, mRNA, or siRNA. This interaction effectively neutralizes the charge and condenses the genetic material into tightly packed polyplexes.
- Polyethylenimine (PEI): Often considered the benchmark standard in in vitro transfection research, PEI possesses exceptional condensing capabilities and high buffering capacity. Its architecture significantly impacts its biological behavior. Branched PEI contains primary, secondary, and tertiary amines, offering massive proton sponge potential for endosomal escape, though often at the cost of higher cellular toxicity. Linear PEI, conversely, forms more flexible polyplexes with a generally more favorable biocompatibility profile, making it highly valuable for complex in vivo studies.
- Poly-L-lysine (PLL): As a synthetic polypeptide composed of basic amino acids, PLL offers excellent DNA condensation capabilities. Because it lacks tertiary amines, it does not possess inherent endosomal escape properties. Consequently, researchers frequently co-formulate PLL with fusogenic peptides or endosomolytic agents (like chloroquine) to prevent lysosomal degradation of the payload.
- Poly(amidoamine) (PAMAM) Dendrimers: Dendrimers are highly branched, star-shaped macromolecules synthesized with exact structural precision. Their generation number (e.g., G3, G4, G5) dictates their size, surface charge density, and internal cavity volume. PAMAM dendrimers offer a highly uniform structural platform, allowing for highly reproducible polyplex formation and extensive surface modification at their multivalent terminal groups.
Biodegradable and Biocompatible Polymer Systems
To address the inherent cytotoxicity and bioaccumulation risks associated with high-molecular-weight cationic polymers, the field has rapidly shifted toward designing fully biodegradable architectures. These systems are engineered to perform their delivery function and subsequently break down into small, easily metabolizable, or excretable byproducts.
- Poly(beta-amino ester)s (PBAEs): PBAEs have emerged as highly potent, biodegradable alternatives to PEI. Synthesized via a simple Michael addition reaction between bisacrylates and primary or secondary amines, PBAEs offer immense combinatorial flexibility. They rapidly degrade via ester hydrolysis in aqueous environments, resulting in minimal long-term cellular toxicity. Their fast degradation kinetics are particularly advantageous for the rapid intracellular release of delicate payloads like mRNA.
- Aliphatic Polyesters (PLGA and PLA): Poly(lactic-co-glycolic acid) and poly(lactic acid) are widely recognized biocompatible materials extensively utilized in drug delivery research. While they lack the inherent positive charge to condense nucleic acids effectively on their own, they are frequently blended with cationic lipids or conjugated to cationic polymers (like PEI-PLGA copolymers). This structural blend provides sustained release profiles and remarkable structural integrity for the nanoparticles.
- Natural Polysaccharides (Chitosan): Derived from the deacetylation of chitin, chitosan is a natural linear polysaccharide containing primary amine groups. It exhibits excellent biocompatibility, low toxicity, and unique mucoadhesive properties, making it an ideal candidate for mucosal gene delivery research, such as inhaled or oral formulation studies.
PEGylated and Stealth Polymer Nanoparticles
When transitioning from controlled in vitro environments to complex in vivo biological models, nanoparticles immediately face the reticuloendothelial system and blood serum proteins. Bare polyplexes are prone to opsonization—the binding of serum proteins that flags the particle for destruction by macrophages.
- The Stealth Effect: The covalent attachment of polyethylene glycol (PEG) to the surface of the polymer carrier creates a dense, highly hydrated corona around the nanoparticle. This steric barrier effectively masks the surface charge, prevents protein adsorption, prevents particle aggregation in high-salt biological buffers, and drastically extends the circulation half-life of the carrier.
- The PEG Dilemma: While PEGylation is crucial for extracellular survival, it severely hinders intracellular delivery. The steric shielding that prevents protein binding also prevents the nanoparticle from interacting with the target cell membrane and escaping the endosome.
- Sheddable PEG Architectures: To resolve this dilemma, advanced formulations utilize dynamically sheddable PEG chains. These systems incorporate stimuli-responsive linkers between the polymer core and the PEG corona. Once the nanoparticle reaches the target tissue or enters the mildly acidic tumor microenvironment, the linker cleaves, shedding the stealth layer. This exposes the underlying cationic surface, restoring strong cellular uptake and endosomal escape capabilities precisely when needed.
Stimuli-Responsive and Smart Polymers
The pinnacle of precision formulation lies in stimuli-responsive—or smart—polymers. These materials are rationally designed to undergo specific physicochemical transitions in response to endogenous biological triggers, providing spatial and temporal control over the unpacking of the genetic cargo.
- pH-Responsive Systems: The biological environment features distinct pH gradients, notably the transition from physiological pH (7.4) to the acidic environment of the endolysosomal pathway (pH 5.0 to 6.5). Polymers incorporating weak bases, such as poly(histidine) or specialized acetal linkages, undergo rapid protonation or hydrolysis in these acidic vesicles. This triggers immediate nanoparticle destabilization and promotes swift endosomal rupture.
- Redox-Responsive Systems: The intracellular cytosol contains significantly higher concentrations of reducing agents, specifically glutathione (GSH), compared to the extracellular fluid. Formulation scientists exploit this gradient by integrating disulfide bonds into the polymer backbone or crosslinking networks. Upon entering the cytosol, the high GSH concentration rapidly reduces and cleaves the disulfide bonds, dismantling the nanoparticle and releasing the naked nucleic acid directly to the cellular machinery.
- Enzyme-Responsive Linkers: Certain disease states or intracellular compartments overexpress specific enzymes. Integrating peptide sequences that are exclusively cleaved by target proteases (such as Cathepsin B within the lysosome or matrix metalloproteinases in specific tissue microenvironments) allows for highly targeted, bio-specific payload release, minimizing off-target genetic expression.
Looking for Nanoparticles for Gene Delivery?
From DNA and mRNA to siRNA and oligonucleotides, our team offers custom polymer synthesis, functionalization, and nanoparticle formulation to meet your research and therapeutic needs.
Design Strategies for High-Performance Polymer Gene Carriers
The transition from a basic polymer solution to a highly efficient gene delivery vector requires meticulous macromolecular engineering. Merely mixing cationic polymers with nucleic acids frequently yields heterogeneous aggregates with sub-optimal biological performance. To achieve high transfection efficiencies and robust structural stability in preclinical models, formulation scientists must employ sophisticated design strategies. By systematically manipulating the spatial arrangement, surface chemistry, and thermodynamic properties of the polymer complex, researchers can precisely dictate how the resulting nanoparticle interacts with physiological barriers and intracellular machinery.
Controlling Particle Size, Charge, and Morphology
The physicochemical attributes of a polyplex are the primary determinants of its biological fate. Strict control over these parameters is non-negotiable for achieving reproducible cellular uptake and desired biodistribution profiles.
- Dimensional Precision: The hydrodynamic diameter of the nanoparticle profoundly influences its cellular internalization pathway. Particles ranging from 50 to 150 nanometers are generally considered optimal. Particles smaller than 10 nanometers face rapid renal clearance, while those exceeding 200 nanometers are highly susceptible to phagocytic clearance by the reticuloendothelial system and struggle to penetrate dense extracellular matrices.
- Zeta Potential Management: The net surface charge, measured as zeta potential, must strike a delicate thermodynamic balance. A moderately positive charge (+10 to +30 mV) is essential for facilitating electrostatic attraction to anionic cellular membranes and driving endocytosis. However, highly positive zeta potentials often induce severe cytotoxicity by disrupting membrane integrity.
- Morphological Tuning: While spherical nanoparticles are the most common thermodynamic outcome of polymer-nucleic acid complexation, morphology actively influences cellular interactions. Through controlled microfluidic assembly or the use of specific rigid polymer backbones, researchers can engineer rod-like or worm-like micelles. These anisotropic shapes often exhibit unique flow dynamics in circulation and can present larger surface areas for receptor interaction, significantly altering uptake kinetics.
Optimizing Polymer Architecture
The topological structure of the polymer chain fundamentally alters how it condenses nucleic acids and shields the payload from enzymatic degradation. Formulation scientists actively select among various macromolecular architectures based on the specific modality being delivered.
- Linear Polymers: Linear architectures offer exceptional macromolecular flexibility. This allows the polymer backbone to easily thread into the major and minor grooves of large plasmid DNA, forming stable, discrete, and highly compact particles. Their flexible nature often results in a more favorable biocompatibility profile compared to highly branched counterparts.
- Branched and Dendritic Copolymers: Branched architectures provide a high localized density of functional groups. This localized charge density yields exceptional condensing power, making them highly suitable for small, rigid modalities like siRNA. Furthermore, the extensive internal cavities within perfectly structured dendrimers offer physical protection for the payload, while their dense array of multivalent terminal groups provides abundant sites for chemical modification.
- Amphiphilic Block Copolymers: By covalently linking distinct hydrophilic and hydrophobic polymer segments, researchers create block copolymers that undergo spontaneous self-assembly in aqueous environments. The hydrophobic segments associate to form a dense core that heavily shields the nucleic acid payload, while the hydrophilic segments (such as polyethylene glycol) project outward to form a protective corona. Adjusting the hydrophilic-lipophilic balance permits exact tuning of the nanoparticle stability and drug-release profile.
Surface Functionalization and Ligand Modification
To surpass the limitations of passive accumulation and achieve precision delivery to specific cellular subpopulations, active targeting strategies must be integrated into the nanoparticle design.
- Covalent Conjugation Chemistry: Surface functionalization relies on highly efficient, bio-orthogonal chemical reactions. Copper-free click chemistry, maleimide-thiol coupling, and localized EDC-NHS crosslinking are routinely employed to securely anchor targeting moieties to the nanoparticle corona without compromising the structural integrity of the underlying polyplex.
- Receptor-Mediated Endocytosis: By conjugating specific biological ligands, the nanoparticle can actively bind to cell surface receptors that are overexpressed in target tissues. Common targeting moieties include small molecules (such as folic acid), highly specific peptides (like the RGD sequence targeting integrins), functional aptamers, and monoclonal antibodies.
- Avidity vs. Affinity: Formulation optimization requires balancing the receptor binding affinity with the overall avidity, which is dictated by the ligand density on the nanoparticle surface. An excessively high ligand density can lead to steric hindrance or trigger premature clearance by the immune system, whereas a density that is too low will fail to initiate receptor-mediated cellular entry.
Enhancing Endosomal Escape and Intracellular Release
The most significant biological bottleneck in non-viral gene delivery is escaping the degradative environment of the endolysosomal pathway. Even if a nanoparticle achieves near-perfect cellular uptake, failure to exit the endosome results in complete therapeutic failure.
- Incorporating Fusogenic Elements: To augment base polymers that lack inherent escape mechanisms, researchers integrate fusogenic peptides or specific lipid-like hydrophobic domains into the polymer matrix. At physiological pH, these elements remain inert. However, upon exposure to the acidic environment of the endosome (pH 5.0 - 6.0), they undergo a conformational shift, transforming into amphiphilic alpha-helices that directly insert into and destabilize the endosomal lipid bilayer.
- Charge-Reversal Strategies: Advanced formulations utilize charge-reversal chemistry. These polymers are engineered with specific functional groups (such as citraconic anhydride derivatives) that maintain a neutral or anionic charge in the bloodstream to prevent protein fouling. Upon endosomal acidification, these groups rapidly cleave, exposing a highly cationic backbone that triggers the proton sponge effect and membrane rupture.
- Balancing the Dissociation Constant: Successful design requires optimizing the thermodynamic binding energy between the polymer and the nucleic acid. The dissociation constant must be carefully engineered: the electrostatic complexation must be energetically favorable enough to survive extracellular transit, yet weak enough to ensure timely intracellular unpacking. If the binding affinity is excessively strong, the polyplex remains intact within the cytosol, physically blocking the ribosomal machinery from accessing mRNA or preventing plasmid DNA from undergoing nuclear transcription.
Polymer Nanoparticles for Different Nucleic Acid Modalities
Nucleic acids are a structurally and functionally diverse class of macromolecules. Delivering a massive, double-stranded plasmid DNA requires vastly different macromolecular engineering compared to delivering a short, rigid siRNA duplex or a highly fragile, single-stranded mRNA. The physicochemical characteristics of the genetic payload—specifically its molecular weight, charge density, inherent stability, and required intracellular site of action—strictly dictate the architectural requirements of the polymer carrier. To achieve robust biological readouts in advanced preclinical research, formulation scientists must rationally tailor the polymer matrix to accommodate the unique vulnerabilities and biological routing of each specific modality.
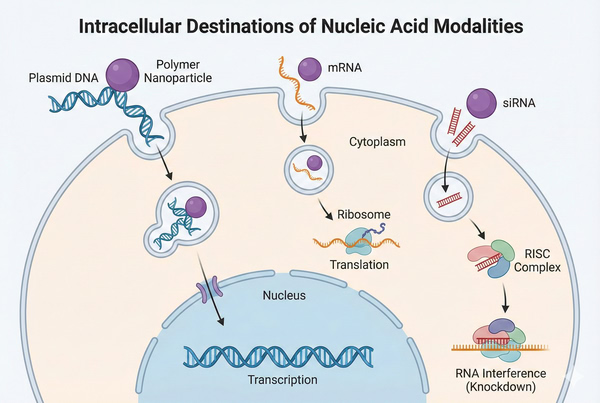 Fig. 3. Distinct intracellular delivery pathways for plasmid DNA, mRNA, and siRNA (BOC Sciences Authorized).
Fig. 3. Distinct intracellular delivery pathways for plasmid DNA, mRNA, and siRNA (BOC Sciences Authorized).
Polymer Nanoparticles for DNA Gene Delivery
Plasmid DNA represents one of the most mechanically challenging payloads in non-viral gene delivery due to its massive size, often comprising thousands of base pairs, and its strict biological destination. To induce successful gene expression, the delivered DNA must not only escape the endolysosomal pathway but also navigate through the dense cytoplasm and cross the highly restrictive nuclear envelope to access the host cell transcriptional machinery.
- Condensation Dynamics: Because plasmid DNA is large and flexible, polymer carriers must possess strong electrostatic binding affinities to condense these macromolecules into tight, sub-200 nanometer polyplexes. Highly branched cationic polymers are frequently utilized to achieve this profound volume reduction while neutralizing the extensive negative charge of the phosphate backbone.
- Nuclear Translocation Strategies: Overcoming the nuclear barrier is extremely difficult in non-dividing cells, where the nuclear membrane remains intact. Advanced polymer systems are specifically functionalized with nuclear localization signals. These short peptide sequences hijack the endogenous importin-based transport machinery of the cell, actively shuttling the polymer-DNA complex through the nuclear pore complex.
- Unpacking in the Nucleus: The dissociation constant of the polyplex must be precisely tuned so that the polymer matrix remains intact in the cytoplasm but rapidly disassembles upon entering the unique microenvironment of the nucleus, allowing RNA polymerase to access the uncoiled plasmid.
Polymer Carriers for mRNA Therapeutics
Messenger RNA has revolutionized functional genomics and basic biological research. However, it is inherently unstable and exquisitely sensitive to ubiquitous RNases present in almost all biological fluids. Unlike plasmid DNA, mRNA only needs to reach the cytoplasm to be translated by host ribosomes, completely bypassing the complex barrier of the nuclear membrane.
- Enzymatic Shielding: The primary objective of an mRNA polymer carrier is robust physical protection. Core-shell polymeric micelles and amphiphilic block copolymers are heavily favored, as they completely encapsulate the fragile single-stranded RNA within a dense hydrophobic core, physically denying nucleases access to the phosphodiester bonds.
- Rapid Cytosolic Release: Because mRNA degrades rapidly even within the cytoplasm, the polymer formulation must prioritize swift endosomal escape and immediate payload unpacking. Biodegradable systems, such as ester-based polymers, are highly advantageous. They maintain structural integrity during extracellular transit but undergo rapid hydrolysis in the cytosol, dumping the mRNA payload directly adjacent to the ribosomal machinery before structural degradation can occur.
- Lipid-Polymer Hybrids: To maximize transfection efficiency, formulation scientists often design hybrid nanoparticles. By blending degradable polymers with helper lipids, researchers achieve synergistic effects: the polymer provides structural stability and precise payload condensation, while the lipid components facilitate rapid membrane fusion and robust endosomal escape.
siRNA Delivery Using Polymeric Nanoparticles
Small interfering RNA operates via the endogenous RNA-induced silencing complex located in the cytoplasm to execute targeted gene knockdown. These molecules are extremely short, typically comprising only 20 to 25 base pairs, and possess a highly rigid double-stranded structure. This unique geometry makes stable electrostatic complexation surprisingly difficult compared to large, flexible plasmids.
- Multivalent Binding Requirements: Standard linear polymers often fail to condense siRNA effectively, resulting in loose polyplexes that easily fall apart in systemic circulation. Formulations strictly demand polymers with high local charge densities. Precisely engineered dendrimers and highly branched architectural topologies are necessary to provide the multivalent electrostatic interactions required to lock the rigid siRNA into stable nanostructures.
- Stoichiometric Optimization: Achieving uniform nanoparticle formation with siRNA requires meticulous tuning of the nitrogen-to-phosphate ratio. Optimization often involves utilizing specialized microfluidic mixing to ensure every siRNA duplex is uniformly coated and protected, minimizing the generation of empty polymer aggregates or unprotected payload.
- Integration with Silencing Machinery: The polymer carrier must not interfere with the biological activity of the payload. Once in the cytoplasm, the polymer must rapidly dissociate to allow the siRNA to seamlessly load into the Argonaute protein core of the RNA-induced silencing complex, enabling highly efficient, sequence-specific degradation of target messenger RNA.
Oligonucleotide and Antisense Delivery Platforms
Antisense oligonucleotides and other single-stranded oligo modalities present unique formulation challenges. They are primarily utilized to modulate RNA splicing or inhibit translation through direct steric hindrance. While backbone modifications—such as phosphorothioate linkages—greatly enhance their raw biological stability, polymeric carriers are strictly required to promote efficient cellular penetration and prevent rapid renal clearance in preclinical models.
- Size and Distribution Uniformity: Polymeric carriers for oligo delivery focus heavily on achieving highly monodisperse particle populations. Utilizing self-assembling block copolymers ensures that the resulting nanoparticles are exceptionally uniform in size, providing consistent and predictable pharmacokinetic profiles during complex in vivo biodistribution studies.
- Preventing Premature Release: Oligonucleotides are small enough to prematurely leak from porous delivery vehicles. Smart polymer systems, crosslinked via stimuli-responsive bonds, are deployed to physically lock the oligo within the carrier matrix during systemic circulation, ensuring zero-order release kinetics only upon cellular internalization.
- Enhancing Intracellular Routing: Depending on the specific mechanism of action, oligos may need to function in the cytoplasm or the nucleus. Polymer functionalization is highly customized here, directing the intracellular trafficking pathways to ensure the oligo reaches its exact site of action, thereby maximizing target engagement and robust biological readouts in disease-state models.
Manufacturing, Scale-Up, and Quality Considerations
For polymer-based therapeutics to transition from basic research to viable commercial products, the formulation process must be robust, scalable, and subject to stringent analytical oversight.
Reproducible Nanoparticle Formulation Methods
Traditional bulk mixing—where a polymer solution is rapidly pipetted into a nucleic acid solution—is notorious for causing extreme batch-to-batch variability. The localized concentration gradients created during manual mixing result in a highly polydisperse particle population, containing both massive aggregates and uncomplexed payload. To achieve precise reproducibility, the industry standard has decisively shifted toward microfluidic mixing technologies. Microfluidic cartridges utilize intricately designed microscopic channels to control the precise intersection of the aqueous nucleic acid stream and the polymer solvent stream. By manipulating fluid dynamics at a low Reynolds number, microfluidics ensures rapid, chaotic advection and uniform mixing times typically on the order of milliseconds. This highly controlled environment forces simultaneous nucleation across the entire reaction volume, consistently yielding highly monodisperse polyplexes with predictable encapsulation efficiencies.
Scale-Up from Research to GMP Production
Scaling up a nanocarrier formulation presents severe thermodynamic challenges. The heat and mass transfer properties that dictate particle self-assembly in a small vial behave fundamentally differently in a large-scale stirring tank. To bypass these limitations, modern polymer nanoparticle manufacturing relies on a continuous flow paradigm rather than batch processing. Instead of scaling up by increasing the size of the reaction vessel, manufacturers scale out. By running optimized microfluidic or impingement jet mixing systems continuously, or by operating multiple identical microfluidic chips in parallel, researchers can process liters of formulation without altering the fundamental mixing physics established at the bench scale. This continuous flow approach is essential for meeting GMP requirements for advanced preclinical toxicology studies, ensuring that critical quality attributes remain strictly uniform from the first milliliter to the last.
Analytical Characterization and Quality Control
Rigorous quality control requires a comprehensive suite of analytical techniques to validate both the physical structure and the chemical integrity of the nanoparticle. Assessing a formulation requires far more than a simple visual inspection.
- Physicochemical Profiling: Dynamic Light Scattering is the foundational tool for quantifying the hydrodynamic diameter and the polydispersity index of the nanoparticle suspension. Parallel electrophoretic light scattering determines the zeta potential, confirming the surface charge density. For precise morphological validation, Cryogenic Electron Microscopy is utilized to visualize the core-shell architecture and confirm the absence of amorphous aggregates.
- Payload Quantification: Formulations must be strictly evaluated for encapsulation efficiency. Gel retardation assays visually confirm that the polymer has completely immobilized the nucleic acid, preventing it from migrating through an electrical field. Concurrently, highly sensitive fluorometric assays quantify the exact percentage of unencapsulated payload remaining in the solvent.
- Purity and Integrity: High-Performance Liquid Chromatography combined with Mass Spectrometry is deployed to assess the chemical purity of the polymer backbone, quantify the exact degree of surface functionalization (such as PEG density), and verify that the sheer stress of the manufacturing process has not sheared or degraded the fragile nucleic acid payload.
Stability, Storage, and Shelf-Life Optimization
A structurally perfect nanoparticle is useless if it degrades before administration. Aqueous suspensions of polymer nanoparticles are inherently unstable over long periods; thermodynamic forces inevitably drive the particles to aggregate, while water facilitates the slow hydrolysis of biodegradable polymer backbones and the degradation of RNA. To secure a prolonged shelf life, formulations are typically subjected to lyophilization. Freeze-drying removes the aqueous solvent, locking the nanoparticles into a stable powder. However, the freezing and desiccation processes generate extreme osmotic and mechanical stresses that can easily rupture the polyplexes. Formulation scientists must rationally screen and integrate specific cryoprotectants and lyoprotectants—typically specific ratios of disaccharides like sucrose or trehalose. These excipients replace the hydrogen bonds previously provided by water, maintaining the three-dimensional architecture of the polymer network during storage and ensuring rapid, uniform reconstitution immediately prior to experimental application.
Applications in Research, Preclinical, and Therapeutic Development
As polymer nanoparticle technologies mature, their utility has expanded exponentially across diverse fields of biomedical research and preclinical therapeutic development. By overcoming the fundamental barriers of intracellular nucleic acid delivery, these synthetic vectors serve as indispensable tools for molecular biologists and formulation scientists. They enable the precise modulation of gene expression in complex biological models, facilitating breakthroughs in disease modeling, target validation, and the development of next-generation genetic interventions.
Gene Therapy and Genetic Disease Treatment
In the pursuit of correcting inherited genetic anomalies, polymer nanoparticles are extensively utilized to deliver functional gene copies or advanced gene-editing machinery in preclinical models. For monogenic disorders, optimized polyplexes can efficiently transport large plasmid DNA or formulated mRNA into target tissues to restore missing protein functions. Furthermore, polymers are uniquely suited for the co-delivery of complex payloads, such as CRISPR-Cas9 ribonucleoproteins or multiplexed combinations of Cas9 mRNA and single guide RNA. Their massive packaging capacity and customizable intracellular degradation profiles allow researchers to achieve transient, highly localized expression of editing nucleases. This thermodynamic control maximizes on-target genetic correction while rigorously minimizing off-target genomic alterations in sophisticated in vivo disease models.
Cancer Therapy and Immunotherapy
Oncology research represents one of the most prominent application areas for precisely engineered polymer delivery systems. Formulation scientists deeply exploit the unique physiological hallmarks of the tumor microenvironment—such as hypoxia, lowered extracellular pH, and the enhanced permeability and retention effect—to design smart, stimuli-responsive polymers that selectively accumulate and unpack within solid tumors. Beyond delivering specific siRNA to silence driver oncogenes or induce apoptosis, polymers are now at the vanguard of preclinical immunotherapy development. Researchers architect polymeric vectors to deliver mRNA encoding tumor-associated antigens or immunostimulatory cytokines directly to antigen-presenting cells. This targeted genetic payload successfully reprograms the immunosuppressive tumor stroma and stimulates robust, tumor-specific T-cell responses in advanced murine models, providing crucial validation data for next-generation immunotherapeutics.
Vaccines and Infectious Disease Applications
The rapid design and rigorous evaluation of nucleic acid-based vaccines rely heavily on versatile synthetic delivery platforms. Polymer nanoparticles offer distinct architectural advantages in preclinical infectious disease research by functioning simultaneously as a highly protective carrier and a potent immunological adjuvant. Certain cationic polymer backbones and carefully selected functional side chains possess inherent immunostimulatory properties. Upon endocytosis, these polymers can activate specific pattern recognition receptors, synergistically boosting the innate immune response to the delivered mRNA or plasmid DNA antigen. By carefully tailoring the molecular weight, branching architecture, and surface chemistry of the polymer, researchers can optimize the systemic biodistribution of the vaccine formulation to secondary lymphoid organs. This targeted delivery drives remarkably potent humoral and cellular immunity against diverse viral and bacterial pathogens in controlled laboratory settings.
Functional Genomics and Gene Silencing Studies
In basic biological research, target discovery, and early-stage drug development, polymer nanoparticles are essential synthetic reagents for elucidating complex genetic networks. The efficient and remarkably non-toxic intracellular delivery of customized siRNA or shRNA libraries via advanced polymeric vectors enables researchers to perform systematic, high-throughput gene silencing studies across varied cell lines. This robust transfection capability allows for the precise mapping of intricate metabolic and signaling pathways, the rigorous validation of novel therapeutic drug targets, and a profoundly deeper understanding of baseline cellular function. Because optimal polymer carriers are engineered to exhibit near-zero baseline cytotoxicity and excellent biocompatibility, they completely eliminate the confounding cellular stress responses often triggered by viral vectors or harsh commercial transfection reagents. This crucial biological neutrality ensures that the observed phenotypic readouts are strictly the result of the intended genetic modulation, granting high confidence to early-stage experimental data.
Services
Polymer Nanoparticle Services for Gene Delivery at BOC Sciences
Turning DNA, mRNA, or siRNA into reliable therapeutics depends on more than just having a carrier—it requires the right polymer design, stable nanoparticle formulation, and reproducible performance. At BOC Sciences, we provide integrated polymer nanoparticle solutions that help you move from concept to application-ready gene delivery systems with less trial and error. From tailored polymer materials and carrier design to formulation optimization, performance evaluation, and scale-up support, our services are built around your real development challenges—improving stability, boosting transfection efficiency, and ensuring consistency across batches—so you can focus on advancing your gene therapy and nucleic acid delivery programs with confidence.

Polymer Design & Materials Supply
- Supply PBAE, PEI derivatives, PLGA, PEGylated polymers, and biodegradable cationic polymers
- Customizable molecular weight, charge density, end groups, and functional moieties
- Scalable supply from research grade to larger quantities (mg → kg)
- Complete documentation including COA, MSDS, and batch-to-batch consistency

Polymeric Nanoparticle Carrier Development
- Design polymeric nanoparticles for DNA, mRNA, siRNA, and oligonucleotide delivery
- Support polyplex nanoparticles, polymeric micelles, and hybrid carrier systems
- Precise control of particle size, PDI, and zeta potential
- Carrier architecture optimized for both in vitro and in vivo delivery applications

Formulation & Delivery Performance Optimization
- Improve nucleic acid encapsulation efficiency, stability, and storage robustness
- Optimize preparation processes such as self-assembly, nanoprecipitation, and microfluidics
- Integrate strategies to enhance endosomal escape and intracellular release
- Boost transfection efficiency and gene expression performance

Characterization, Scale-Up & Translational Support
- Comprehensive characterization of particle size, morphology, structure, and stability
- Evaluation of nucleic acid binding, protection, and release behavior
- Process development support from small-scale to larger-scale production
- Deliver reproducible and scalable solutions for translational research
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
Why choose polymer nanoparticles for gene therapeutics?
Polymer nanoparticles offer exceptional chemical tunability, allowing researchers to precisely engineer molecular weight, charge, and degradation profiles. They provide a safer, less immunogenic alternative to viral vectors and feature highly scalable, reproducible synthetic manufacturing processes essential for advanced preclinical and formulation research.
How do polymer nanoparticles protect DNA, RNA, or mRNA?
Cationic polymers utilize strong electrostatic interactions to condense negatively charged nucleic acids into dense nanoscale structures. This robust physical encapsulation effectively shields the fragile genetic payload from ubiquitous endonucleases and exonucleases, preventing premature enzymatic degradation during complex in vivo extracellular transit.
Can polymer nanoparticles enhance cellular uptake and gene expression?
Yes. Naked nucleic acids experience strong electrostatic repulsion from anionic cell membranes. Polymer carriers maintain a net positive surface charge that facilitates robust electrostatic binding to cellular proteoglycans, actively driving endocytosis and significantly increasing the intracellular concentration of the genetic payload for efficient expression.
Which polymer system is best for DNA, mRNA, or siRNA delivery?
Selection depends entirely on the payload. Large plasmid DNA requires highly branched polymers for strong condensation. Fragile mRNA benefits from biodegradable ester-based systems for rapid cytosolic release. Conversely, short and rigid siRNA demands dense multivalent architectures, like dendrimers, to ensure stable encapsulation and targeted silencing.
How do polymer nanoparticles compare with lipid nanoparticles?
While lipid nanoparticles offer high transfection efficiencies for mRNA, polymer systems provide superior structural stability and far greater chemical modularity. Polymers allow for intricate architectural modifications, precise tuning of degradation kinetics, and highly specific ligand conjugation, making them highly adaptable for diverse preclinical research applications.
Are polymer gene delivery carriers safe and biodegradable?
Modern formulations prioritize exceptional biocompatibility. While early synthetic polymers exhibited some cytotoxicity, contemporary systems utilize sophisticated biodegradable backbones, such as poly(beta-amino ester)s. These advanced materials perform their delivery function and subsequently undergo rapid hydrolysis into non-toxic metabolites that biological systems easily clear.
How do you improve transfection efficiency with polymer nanoparticles?
Efficiency is optimized by meticulously tuning the nitrogen-to-phosphate ratio, selecting polymers with high buffering capacities to drive endosomal escape, and modifying the nanoparticle surface with cell-specific targeting ligands. Employing microfluidic formulation methods also ensures uniform particle size, which is critical for consistent cellular uptake.
Can polymer nanoparticles protect RNA from degradation?
Absolutely. Protecting RNA from highly active physiological RNases is a fundamental function of these carriers. The dense polymeric matrix completely envelopes the fragile single-stranded RNA, physically blocking nucleases from accessing and cleaving the phosphodiester backbone until the nanoparticle safely releases its cargo intracellularly.

 Fig. 1. Overview of polymer nanoparticle structure and intracellular gene delivery pathway (BOC Sciences Authorized).
Fig. 1. Overview of polymer nanoparticle structure and intracellular gene delivery pathway (BOC Sciences Authorized). Fig. 2. Structural comparison of linear polymers, dendrimers, and block copolymer micelles (BOC Sciences Authorized).
Fig. 2. Structural comparison of linear polymers, dendrimers, and block copolymer micelles (BOC Sciences Authorized). Fig. 3. Distinct intracellular delivery pathways for plasmid DNA, mRNA, and siRNA (BOC Sciences Authorized).
Fig. 3. Distinct intracellular delivery pathways for plasmid DNA, mRNA, and siRNA (BOC Sciences Authorized).