Overcoming mRNA Stability and Delivery Challenges with Polymers
The clinical validation of mRNA technology has ushered in a new era of medicine, yet the transition from bench to bedside remains hindered by the inherent fragility of the mRNA molecule. While lipid nanoparticles (LNPs) have established the foundational success for current vaccines, the field is increasingly pivoting toward polymer-based delivery systems to address limitations in thermal stability, immunogenicity, and tissue specificity. Synthetic polymers offer a broad chemical design space, allowing for precise control over molecular architecture and functionalization.
Resources
The Rapid Growth of mRNA Therapeutics and Vaccines
The success of mRNA vaccines against SARS-CoV-2 has accelerated the development pipeline for a diverse range of indications, including infectious diseases, cancer immunotherapy, and protein replacement therapies. The global market requires delivery vectors that can support not only prophylactic vaccines but also repeated dosing regimens required for chronic conditions. This expansion necessitates delivery vehicles that are less inflammatory and more chemically diverse than first-generation lipid systems.
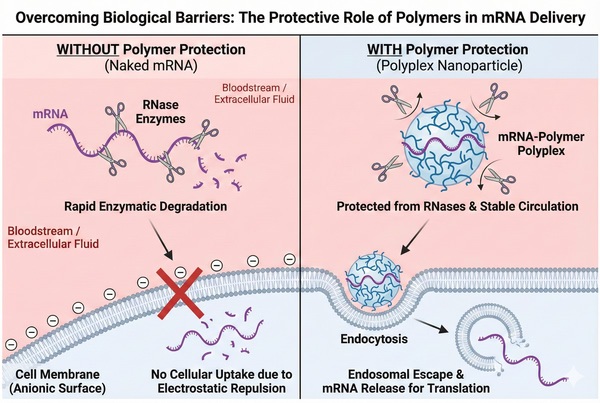 Fig. 1. Polymer encapsulation overcomes enzymatic degradation and cellular uptake biological barriers (BOC Sciences Authorized).
Fig. 1. Polymer encapsulation overcomes enzymatic degradation and cellular uptake biological barriers (BOC Sciences Authorized).
Key Barriers in mRNA Stability and In Vivo Delivery
Delivering a fragile macromolecule like mRNA in vivo involves navigating formidable biophysical hurdles. First, naked mRNA is rapidly degraded by ubiquitous ribonucleases (RNases) in the extracellular matrix, reducing its half-life to minutes. Second, its high polyanionic charge density creates strong electrostatic repulsion with the cell membrane, preventing passive diffusion. Finally, even after successful endocytosis, the mRNA faces the critical bottleneck of endosomal escape. If the carrier fails to translocate the payload into the cytosol promptly, the mRNA is destroyed within the acidic lysosome. Therefore, effective carriers must act dynamically: protecting the payload systemically, facilitating cellular entry, and actively disrupting the endosome.
Why Polymers Are Becoming Critical Delivery Enablers?
While traditional lipid systems rely heavily on the non-covalent self-assembly of hydrophobic components, synthetic polymers offer formulation scientists deterministic control through defined covalent chemistry. This synthetic flexibility allows for the precise engineering of molecular weight, architectural topology, and charge density (N/P ratio) to optimize mRNA complexation. Furthermore, polymers can be rationally designed with bio-cleavable linkages for predictable in vivo degradation. By integrating smart stimuli-responsive functionalities—such as protonatable amines for pH-triggered endosomal escape (the proton sponge effect)—polymers transform from simple protective carriers into highly adaptable, programmable nanomachines for next-generation mRNA delivery.
Fundamental Challenges in mRNA Stability and Delivery
To engineer effective delivery systems, one must first deconstruct the physicochemical and biological hurdles that mRNA faces. The instability of mRNA is twofold: chemical susceptibility to hydrolysis and enzymatic cleavage, and physical instability during storage and formulation.
- Enzymatic Degradation and Chemical Instability of mRNA: Messenger RNA is highly susceptible to hydrolysis catalyzed by RNases, which are present in blood and tissues. The phosphodiester backbone is prone to cleavage, and the molecule itself is sensitive to shear stress and temperature fluctuations. Without encapsulation, the half-life of naked mRNA in the bloodstream is measured in minutes, rendering it therapeutically useless without a protective vehicle.
- Poor Cellular Uptake and Endosomal Escape: The cell membrane presents a formidable barrier to mRNA due to electrostatic repulsion between the negatively charged phosphate backbone of the RNA and the anionic cell surface. While cationic carriers can facilitate uptake via endocytosis, the subsequent rate-limiting step is endosomal escape. If the mRNA remains trapped, the endosome matures into a lysosome, leading to the degradation of the payload in an acidic environment.
- Immune Activation and Toxicity Concerns: Exogenous mRNA is a potent pathogen-associated molecular pattern (PAMP) that can trigger toll-like receptors (TLRs), leading to systemic inflammation. Additionally, the delivery vehicle itself can induce toxicity. High charge density cationic lipids or polymers can disrupt cell membranes or cause complement activation. Balancing transfection efficiency with a favorable safety profile is a primary challenge in formulation.
- Storage, Formulation, and Scalability Issues: Thermodynamic instability often necessitates ultra-cold chain storage for mRNA formulations, which complicates logistics and distribution. Furthermore, scaling up the manufacturing of complex nanoparticles while maintaining consistent physicochemical properties (size, polydispersity index, and encapsulation efficiency) poses significant engineering challenges.
Why Polymers Are Ideal Materials for mRNA Delivery Systems?
From a materials science perspective, polymers offer an unparalleled toolkit for addressing the limitations of nucleic acid delivery. The synthetic flexibility of polymerization chemistry allows for the creation of vectors that are specifically tailored to the physicochemical properties of mRNA.
- Structural Versatility and Chemical Tunability: Polymers can be synthesized in various architectures—linear, branched, hyperbranched, or dendritic. This versatility allows researchers to optimize the spatial arrangement of functional groups. For instance, dendrimers provide a precise, monodisperse structure with a high density of surface groups for mRNA binding, while block copolymers can form core-shell micellar structures that protect the payload.
- Charge Modulation and mRNA Complexation Ability: Effective condensation of mRNA into polyplexes requires cationic groups. Polymers allow for the precise titration of charge density. By adjusting the Nitrogen-to-Phosphate (N/P) ratio, formulators can balance the binding strength required for protection against the dissociation required for intracellular release.
- Biodegradability and Biocompatibility Advantages: Modern polymer chemistry emphasizes biodegradable backbones. Polymers incorporating ester, disulfide, or phosphoester linkages can degrade into non-toxic metabolites under physiological conditions. This is crucial for reducing accumulation and long-term toxicity, particularly for therapies requiring repeated administration.
- Manufacturing Flexibility and Scalable Synthesis: Unlike the complex microfluidic mixing often required for lipid nanoparticles, many polymer systems can be synthesized using standard organic chemistry techniques and formulated via simple mixing or precipitation methods. This robustness simplifies the scale-up process and improves batch-to-batch consistency in GMP environments.
Key Types of Polymers Used in mRNA Delivery
Several classes of polymers have emerged as frontrunners in the race to optimize mRNA delivery, each offering distinct advantages regarding encapsulation and release mechanisms. The structural diversity of synthetic polymers provides formulation scientists with a vast chemical playground to design vectors that address the specific pharmacokinetic and pharmacodynamic requirements of mRNA therapeutics. Unlike lipid-based systems, which often rely on a relatively narrow set of ionizable lipid scaffolds, polymers can be engineered with infinite variations in molecular weight, monomer composition, branching, and end-group functionality.
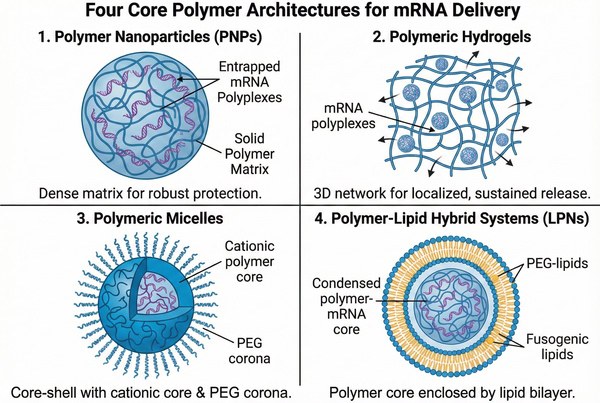 Fig. 2. Structural comparison of four core polymer architectures for mRNA delivery (BOC Sciences Authorized).
Fig. 2. Structural comparison of four core polymer architectures for mRNA delivery (BOC Sciences Authorized).
Cationic Polymers (e.g., PEI and Its Derivatives)
Cationic polymers represent the foundational class of non-viral nucleic acid vectors. Their primary function relies on a high density of primary, secondary, or tertiary amines that protonate at physiological pH. This positive charge facilitates robust electrostatic complexation with the polyanionic mRNA backbone, spontaneously forming nano-sized polyplexes.
- Polyethylenimine (PEI): Historically considered the gold standard for in vitro transfection, PEI exists in linear (lPEI) and branched (bPEI) architectures. Branched PEI (typically 25 kDa) exhibits exceptional endosomal escape capabilities due to the proton sponge effect driven by its diverse amine groups. However, its high charge density is inextricably linked to significant cellular toxicity and membrane disruption.
- Derivatization for Safety: To decouple efficacy from toxicity, modern research focuses on modifying low-molecular-weight PEI (e.g., 2 kDa or 5 kDa), which is natively non-toxic but a poor transfectant. By cross-linking these smaller chains with biodegradable linkages (such as disulfide or ester bonds) or conjugating them with lipophilic tails (lipid-polymer hybrids), researchers can synthesize vectors that condense mRNA effectively but rapidly degrade into non-toxic metabolites once inside the reducing environment of the cytosol.
- Poly(L-lysine) (PLL): Another classic polycation, PLL provides excellent mRNA compaction but lacks the intrinsic buffering capacity required for endosomal escape, typically necessitating the co-formulation of endosomolytic peptides.
Biodegradable Polyesters and Poly(β-amino esters)
The paradigm in polymer-based delivery has shifted heavily toward materials that balance robust extracellular stability with rapid intracellular clearance. Poly(β-amino esters) (PBAEs) have emerged as a leading platform in this category, offering a highly tunable and biodegradable alternative to traditional polycations.
- Combinatorial Synthesis: PBAEs are rapidly synthesized via Michael addition of primary or secondary amines to diacrylates. This highly scalable, step-growth polymerization allows for the creation of massive combinatorial libraries. Formulators can systematically screen thousands of structural variations to optimize the structure-activity relationship (SAR) for specific tissue targeting.
- Degradation Kinetics: The defining advantage of PBAEs is their hydrolytically cleavable ester backbone. While stable at the slightly alkaline pH of extracellular fluid, PBAEs undergo rapid hydrolysis in the aqueous cytoplasm. This degradation dismantles the polyplex, facilitating the immediate release of the mRNA payload to the ribosomes while simultaneously mitigating the long-term cytotoxicity associated with charge accumulation.
- Extrahepatic Targeting: Advanced PBAE formulations have demonstrated remarkable success in escaping the liver—a common bottleneck for lipid nanoparticles—enabling potent and highly specific mRNA delivery to the lungs (pulmonary endothelium) and the spleen for immunotherapeutic applications.
PEGylated Polymers for Stability and Circulation Time
Naked polyplexes frequently suffer from colloidal instability in protein-rich biological fluids. They are prone to aggregation, opsonization (the binding of serum proteins), and rapid clearance by the mononuclear phagocyte system (MPS). The integration of polyethylene glycol (PEG) into the polymer architecture is the primary strategy to overcome these systemic barriers.
- Steric Stabilization: Grafting or block-copolymerizing PEG onto a cationic polymer backbone creates a dense, hydrophilic hydration shell around the polyplex. This steric barrier significantly reduces protein fouling and prevents particle-particle aggregation, thereby enhancing the shelf-life and circulating half-life of the formulation.
- Cleavable PEG Systems: To resolve this, modern polymer designs utilize stimuli-responsive PEG-shedding mechanisms. PEG chains are attached via acid-labile (e.g., hydrazone or orthoester) or matrix metalloproteinase (MMP)-cleavable linkers. Upon reaching the acidic tumor microenvironment or the endosome, the PEG shell is jettisoned, exposing the underlying cationic core to facilitate cellular entry and endosomal rupture.
Stimuli-Responsive and Smart Polymer Systems
The ultimate goal of mRNA carrier design is to create smart nanoparticles that actively respond to the distinct physiological gradients between the extracellular matrix and the intracellular compartments. These stimuli-responsive polymers act as programmable nanomachines.
- pH-Responsive Polymers: Exploiting the pH gradient between the blood (pH 7.4) and the endo/lysosomal compartments (pH 6.0 to 4.5) is the most utilized strategy. Polymers incorporating specialized moieties, such as alkylacrylic acids or substituted amines, undergo a sharp conformational change or shift from hydrophobic to hydrophilic upon protonation, physically disrupting the endosomal membrane.
- Redox-Responsive Polymers: The intracellular environment is highly reducing compared to the extracellular space, primarily due to a 100-to-1000-fold higher concentration of glutathione (GSH) in the cytosol. By cross-linking polymer chains with bioreducible disulfide bonds, the carrier remains perfectly stable in the bloodstream but rapidly falls apart upon encountering cytosolic GSH, ensuring a burst release of the intact mRNA precisely where it is needed.
- Enzyme-Responsive Systems: Certain disease states, particularly solid tumors, overexpress specific proteases or esterases. Polymers can be engineered with peptide cross-linkers that are exclusively recognized and cleaved by these local enzymes, providing an additional layer of targeted payload release.
Polymer nanoparticles (PNPs) represent the most ubiquitous structural format for solid-state mRNA delivery. Unlike self-assembled vesicular systems, PNPs often consist of a dense, entangled polymeric matrix where the mRNA transcript is tightly complexed within the core (forming a polyplex) or physically entrapped via controlled nanoprecipitation. These solid-state architectures provide exceptionally robust steric shielding against extracellular ribonucleases. PNPs are highly tunable from an engineering standpoint; their hydrodynamic diameter (typically optimized between 50–150 nm) and polydispersity index (PDI) can be strictly controlled via microfluidic mixing parameters, ensuring highly reproducible endocytotic uptake and favorable in vivo biodistribution profiles.
For indications requiring localized and sustained mRNA expression—such as regional tumor immunotherapy or tissue engineering—polymeric hydrogels serve as highly effective macroscopic delivery depots. These three-dimensional, water-swollen networks are formulated using cross-linked hydrophilic polymers (e.g., chemically modified hyaluronic acid, alginate, or PEGylated derivatives). Advanced formulations utilize shear-thinning or stimuli-responsive in situ gelation mechanisms (e.g., Schiff base formation, click chemistry, or Michael-type addition) that allow for minimally invasive injection followed by rapid solidification at the target physiological site. The specific mesh size and hydrolytic degradation kinetics of the hydrogel matrix govern the spatiotemporal release profile of the encapsulated mRNA polyplexes, maximizing local transfection while minimizing systemic off-target toxicity.
Polymeric micelles are thermodynamically stable, core-shell nanostructures driven by the spontaneous self-assembly of amphiphilic block copolymers. In the context of mRNA therapeutics, polyion complex (PIC) micelles are specifically engineered to accommodate highly anionic payloads. These advanced structures feature a cationic, mRNA-binding inner segment that neutralizes and condenses the transcript, surrounded by a hydrophilic, non-ionic outer segment (typically a PEG corona). This highly ordered architecture shields the mRNA within a dense core while the dense steric corona provides superior colloidal stability and prevents opsonization by serum proteins. Crucially, their remarkably low critical micelle concentration (CMC) ensures structural integrity even upon extreme dilution in the systemic circulation.
Looking for Custom Polymers for mRNA Delivery?
Whether you need cationic polymers, biodegradable carriers, or stimuli-responsive platforms, our experts provide tailored synthesis and functionalization solutions to support your research and therapeutic development.
How Polymer Improve mRNA Stability and Delivery Efficiency?
The mechanism of action for polymer-based delivery involves a sequence of orchestrated events. Advanced polymer designs actively participate in overcoming biological barriers rather than serving merely as passive carriers.
- Protection Against RNase Degradation and Payload Damage: Through electrostatic complexation, cationic polymers condense mRNA into compact nanoparticles (polyplexes). This condensation sterically hinders RNases from accessing the mRNA backbone, effectively shielding the genetic code from enzymatic degradation in the bloodstream.
- Enhanced Colloidal Stability, Shelf-Life, and Formulation Robustness: Polymers can be engineered to prevent particle aggregation through steric stabilization or electrostatic repulsion. This results in formulations that maintain their size and polydispersity over time. Certain polymer formulations involve lyophilization (freeze-drying) protocols that allow mRNA therapeutics to be stored at higher temperatures compared to LNPs, simplifying the cold chain.
- Improved Cellular Uptake and Membrane Interaction: Cationic polymers interact with anionic proteoglycans on the cell membrane, triggering adsorptive endocytosis. Furthermore, the surface chemistry of polymer nanoparticles can be modified to exploit specific uptake pathways, such as caveolae-mediated or clathrin-mediated endocytosis, optimizing the internalization route.
- Endosomal Escape Mechanisms Enabled by Smart Polymer Design: This is the most critical function of the polymer carrier. Many polymers, particularly those with secondary and tertiary amines (like PEI and PBAEs), exhibit a high buffering capacity. This leads to the proton sponge effect, where the influx of protons into the endosome causes an influx of chloride ions and water, leading to osmotic swelling and endosomal rupture, thereby releasing the mRNA into the cytosol.
- Targeted Delivery Through Ligand-Modified Polymer Carriers: Polymers offer convenient functional handles for the conjugation of targeting ligands such as antibodies, peptides, or small molecules (e.g., folate or GalNAc). This active targeting ensures that the mRNA payload is delivered specifically to the disease site—such as tumor cells or hepatocytes—reducing off-target effects.
- Controlled Release, Reduced Off-Target Effects, and Lower Systemic Toxicity: By tuning the degradation rate of the polymer backbone (e.g., using hydrolysis-sensitive ester bonds), researchers can control the kinetics of mRNA release. This allows for sustained protein expression profiles and prevents the burst release often associated with toxicity.
Applications of Polymer-Based mRNA Delivery Systems
The clinical validation of mRNA-based SARS-CoV-2 vaccines unequivocally proved the immense therapeutic potential of transient gene expression. However, expanding the utility of mRNA beyond prophylactic vaccines into chronic diseases, oncology, and precision genomic medicine demands delivery vectors with highly specialized pharmacokinetic and pharmacodynamic profiles. Polymer-based systems, with their virtually limitless combinatorial design space, are uniquely positioned to address the distinct physiological barriers associated with these diverse clinical applications. By modulating the polymer architecture, researchers can engineer vectors optimized for systemic circulation, localized retention, or active cellular targeting.
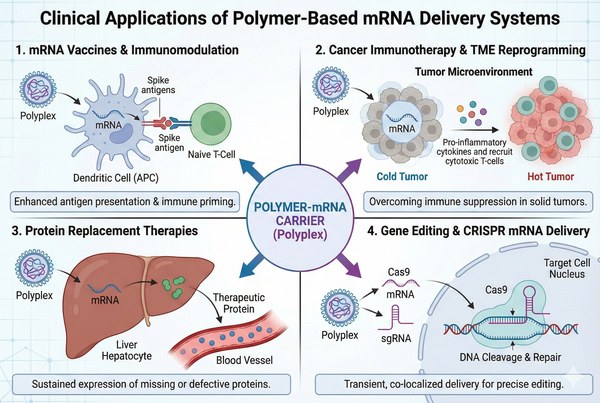 Fig. 3. Clinical applications of polymer-based mRNA delivery across four therapeutic areas (BOC Sciences Authorized).
Fig. 3. Clinical applications of polymer-based mRNA delivery across four therapeutic areas (BOC Sciences Authorized).
mRNA Vaccines for Infectious Diseases
While lipid nanoparticles currently dominate the commercial vaccine landscape, polymer systems are rapidly advancing to solve the lingering challenges of cold-chain dependence and suboptimal immunogenicity.
- Thermostability via Lyophilization: A major advantage of polymer-mRNA polyplexes is their structural resilience during freeze-drying processes. Polymers with high glass transition temperatures can stabilize the mRNA secondary structure, potentially allowing for the formulation of vaccines that remain stable at ambient temperatures, drastically simplifying global distribution logistics.
- Intrinsic Adjuvanticity: Unlike lipids, which require the addition of specific ionizable lipids to stimulate an immune response, certain cationic polymers possess intrinsic immunomodulatory properties. By subtly activating pattern recognition receptors (PRRs) such as Toll-like receptors (TLRs) or the cGAS-STING pathway, the polymer carrier acts as a self-adjuvant. This synergistic stimulation enhances the maturation of dendritic cells and promotes a robust Th1-biased immune response, often allowing for dose-sparing of the mRNA payload.
- Targeted Uptake by Antigen-Presenting Cells (APCs): Polymers can be readily functionalized with specific ligands, such as mannose or specific peptides, to actively target the C-type lectin receptors heavily expressed on the surface of macrophages and dendritic cells in the lymphatic system.
Cancer Immunotherapy and Tumor Microenvironment Modulation
In the realm of oncology, mRNA is being leveraged to break immune tolerance and mount a systemic attack against malignant cells. Polymers offer sophisticated mechanisms for navigating the complex and immunosuppressive tumor microenvironment (TME).
- Therapeutic Cancer Vaccines: Polymers are utilized to deliver mRNA encoding tumor-associated antigens (TAAs) or patient-specific neoantigens directly to secondary lymphoid organs. This approach primes cytotoxic CD8+ T cells to recognize and eliminate disseminated tumor cells.
- In Vivo CAR-T Cell Engineering: Emerging research is exploring the use of targeted polymer vectors to deliver mRNA encoding chimeric antigen receptors (CARs) directly to circulating T cells in vivo, potentially bypassing the arduous and expensive ex vivo cell manufacturing processes currently required for CAR-T therapy.
Protein Replacement Therapies
Treating monogenic disorders, such as Cystic Fibrosis, hemophilia, or lysosomal storage diseases, requires the delivery of mRNA to instruct patient cells to synthesize a missing or defective protein. This application presents the most stringent safety requirements for delivery vectors.
- The Necessity of Immune Stealth: Unlike vaccines, which rely on immune activation, protein replacement therapy requires absolute immune stealth to prevent systemic inflammation and the formation of anti-drug antibodies (ADAs). PEGylated, neutrally charged polymer platforms are critical here to ensure the vector remains invisible to the immune system.
- Biodegradability for Chronic Dosing: Because genetic diseases require lifelong, repeated administration of the therapeutic, the delivery vehicle must not accumulate in tissues. Rapidly biodegradable polymers, such as PBAEs or degradable polyesters, are essential. They undergo predictable hydrolysis into biologically inert metabolites that are easily cleared via renal filtration or hepatic processing.
- Extrahepatic Targeting: While delivering payloads to the liver (for conditions like hemophilia) is relatively straightforward due to natural venous drainage and fenestrated endothelium, targeting other organs is challenging. Optimized polymer libraries have demonstrated the unique ability to bypass hepatic clearance and deliver mRNA specifically to the pulmonary endothelium or spleen, opening new avenues for respiratory and systemic metabolic disorders.
Gene Editing and CRISPR mRNA Delivery
The therapeutic realization of CRISPR-Cas nucleases and base/prime editors relies entirely on the safe, transient, and co-localized delivery of the editing machinery. mRNA is the preferred format for the Cas endonuclease, as transient expression minimizes the risk of off-target DNA cleavage associated with constitutive expression from viral vectors.
- Packaging Large Payloads: The Cas9 mRNA transcript is exceptionally large (~4.5 kb). Complexing such large, highly anionic molecules into stable nanoparticles is challenging for traditional lipid systems. Polymers, however, exhibit highly cooperative electrostatic binding, allowing them to efficiently condense very large mRNA transcripts into tight, uniformly sized polyplexes.
- Co-delivery of mRNA and sgRNA: Effective gene editing requires the simultaneous presence of the Cas mRNA and the single guide RNA (sgRNA) within the same cell. Polymer systems excel at the co-encapsulation of diverse nucleic acids. The flexibility of the polymer chain allows it to enmesh both the long mRNA and the short sgRNA within a single nanocarrier, ensuring stoichiometric delivery to the target cytosol.
- Ribonucleoprotein (RNP) Delivery: In addition to pure nucleic acid delivery, certain amphiphilic and charge-dense polymers are being engineered to deliver pre-assembled Cas9/sgRNA Ribonucleoprotein (RNP) complexes, demonstrating the profound versatility of polymer materials in the gene editing landscape.
Design Considerations for Effective Polymer mRNA Carriers
The transition of a polymer-based mRNA therapeutic from in vitro proof-of-concept to in vivo clinical efficacy is governed by rigorous materials engineering. Designing an effective polymer carrier is not a monolithic task; rather, it is a complex, multivariate optimization problem. Formulators must exquisitely balance competing physicochemical parameters to satisfy the conflicting requirements of the delivery cascade: the vector must be highly stable in the extracellular environment yet structurally labile within the target cell. Understanding the structure-property relationships of the polymer is paramount to dictating the pharmacokinetic and pharmacodynamic fate of the mRNA payload.
Polymer Molecular Weight and Architecture
The spatial and dimensional properties of a polymer chain fundamentally dictate its interaction with the large, polyanionic mRNA transcript.
- The Molecular Weight Paradox: The molecular weight (Mw) of a polycation directly correlates with its condensing capacity. High-Mw polymers exhibit highly cooperative binding, forming highly stable, tightly packed polyplexes that offer superior protection against RNase degradation. However, this thermodynamic stability comes at a biological cost: high-Mw chains are notoriously cytotoxic, slow to degrade, and difficult for the cell to unpack, often leading to restricted translational efficiency. Conversely, low-Mw polymers are biocompatible but struggle to form stable nanoparticles under physiological ionic strengths.
- Architectural Influence: Beyond linear chain length, the topological architecture—whether branched, hyperbranched, star-shaped, or dendritic—profoundly influences the formulation. For example, dendrimers offer a highly controlled, monodisperse 3D structure with an exact number of surface functional groups. This enables highly reproducible mRNA complexation and predictable pharmacokinetics compared to linear polymers, which often suffer from higher polydispersity indices (PDI) and batch-to-batch structural variations.
Charge Density and mRNA Binding Strength
Electrostatic complexation is the driving force behind polyplex formation. The precise calibration of the cationic charge is arguably the most critical variable in formulation design.
- Optimizing the N/P Ratio: The formulation is mathematically guided by the Nitrogen-to-Phosphate (N/P) ratio, representing the molar ratio of amine (nitrogen) groups on the polymer to the phosphate groups on the mRNA backbone. An optimal N/P ratio (typically >1) ensures a net positive surface charge (Zeta potential), which promotes colloidal stability via electrostatic repulsion and facilitates binding to anionic cellular membranes.
- Tuning the pKa: The pKa of the ionizable groups must be carefully selected. Secondary and tertiary amines with a pKa between 5.5 and 6.5 are highly desirable; they remain largely unprotonated (and thus less toxic) at physiological pH (7.4) but undergo rapid protonation in the acidic endosome (pH 5.0–6.0). This targeted protonation acts as the chemical trigger for both the proton sponge mediated endosomal rupture and the intracellular dissociation of the polyplex. Too high of a persistent charge density leads to irreversible binding, trapping the mRNA within the polymer matrix and preventing ribosomal engagement.
Biodegradation Rate and Clearance Pathways
For applications requiring repeated administration, such as protein replacement therapies, the pharmacokinetic clearance of the delivery vehicle is as critical as the delivery of the payload itself. Bioaccumulation of high-molecular-weight synthetic polymers can lead to severe long-term organ toxicity.
- Cleavable Linkages: Modern polymer design mandates the incorporation of bio-labile linkages within the polymer backbone or cross-links. Polyesters, poly(amino acid)s, and poly(disulfide)s are engineered to undergo predictable degradation. The kinetics of this degradation—whether hydrolytic (e.g., ester bonds in PBAEs) or reductive (e.g., disulfide bonds in the presence of cytosolic glutathione)—must be tightly synchronized with the required duration of mRNA translation.
- Renal Filtration Limits: The ultimate design goal is for the polymer to disassemble in vivo into oligomeric or monomeric metabolic byproducts that fall well below the renal filtration threshold (approximately 30 to 50 kDa). This ensures rapid urinary excretion and minimizes hepatic or splenic accumulation, preserving the long-term safety profile of the therapeutic.
Formulation Optimization and Quality Control
Synthesizing the ideal polymer is only half the battle; the phase behavior of the polymer-mRNA interaction must be tightly controlled during formulation to meet GMP standards.
- Buffer and Mixing Kinetics: Polyplex self-assembly is highly sensitive to the formulation environment. The pH and ionic strength of the buffer directly impact the degree of polymer ionization and mRNA folding. Furthermore, the kinetics of mixing—transitioning from uncontrolled bulk mixing (vortexing) to highly controlled microfluidic mixing—dictates the CQAs of the nanoparticles.
- Analytical Characterization: Rigorous quality control relies on a suite of biophysical assays. Dynamic Light Scattering (DLS) and Electrophoretic Light Scattering (ELS) are standard for quantifying hydrodynamic diameter, PDI, and Zeta potential. More critically, fluorometric assays (such as the RiboGreen assay) and gel retardation assays are required to quantify the Encapsulation Efficiency (EE%) and verify that the mRNA remains structurally intact and fully protected following the thermodynamic stress of complexation.
Services
Custom Polymer Development for mRNA Delivery at BOC Sciences
At BOC Sciences, we offer comprehensive polymer-based solutions to support the development of efficient and reliable mRNA delivery systems. Our services cover polymer materials supply, custom polymer design and synthesis, mRNA carrier construction, formulation and encapsulation optimization, intracellular delivery enhancement, and full characterization and scale-up support. By integrating expertise in polymer chemistry and nucleic acid delivery technologies, we help our partners tailor key parameters such as stability, transfection efficiency, and intracellular release performance. From early-stage research to translational development, our solutions are designed to accelerate the advancement of polymer-based mRNA delivery platforms for vaccines, therapeutics, and gene editing applications.

Custom Polymer Synthesis for mRNA Therapeutics
- Provide PBAEs, PEI derivatives, PEGylated polymers, and biodegradable cationic polymers
- Block, branched, or functionalized polymer architectures can be customized on demand
- pKa, hydrophilic–hydrophobic balance, and mRNA binding affinity are optimized
- PEG chains, targeting ligands, or stimulus-responsive moieties can be introduced

Polymer-Based mRNA Carrier Development
- Construction of polyplex nanoparticles and polymeric micelle systems
- Optimization of mRNA complexation efficiency and carrier stability
- Precise control of key parameters such as particle size, PDI, and zeta potential
- Suitable for both in vitro transfection and in vivo delivery studies

Formulation & Encapsulation Optimization
- Enhancement of mRNA encapsulation efficiency and cargo stability
- Optimization of buffer systems, excipients, and processing conditions
- Support for self-assembly and microfluidic formulation strategies
- Improvement of storage stability and batch-to-batch reproducibility

Characterization & Scale-Up Support
- Characterization of polymer molecular weight, dispersity, and chemical structure
- Measurement of nanoparticle size, PDI, and surface potential
- Support for polymer synthesis scale-up from gram to kilogram scale
- Provision of COA, MSDS, and batch consistency assurance
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
Why use polymer carriers for mRNA delivery?
Polymer carriers offer unmatched structural versatility and chemical tunability compared to traditional lipid nanoparticles. Their synthetic flexibility allows formulation scientists to precisely control molecular weight, charge density, and degradation kinetics, enabling the development of highly stable, targeted, and safer mRNA delivery systems tailored for specific therapeutic applications.
How do polymers protect mRNA from degradation?
Cationic polymers protect mRNA through electrostatic complexation. Positively charged amine groups bind tightly to the negatively charged mRNA phosphate backbone, condensing it into dense, nano-sized polyplexes. This compact three-dimensional structure physically shields the fragile mRNA transcript from ubiquitous ribonucleases (RNases) and hydrolytic degradation in extracellular environments.
Can polymer carriers improve mRNA uptake and expression?
Yes. The net positive charge of polymer nanoparticles facilitates electrostatic interactions with anionic cell membranes, promoting robust cellular uptake via endocytosis. Furthermore, smart polymers possessing high buffering capacity trigger the proton sponge effect, disrupting the endosomal membrane and ensuring the mRNA successfully reaches the cytoplasm for ribosomal translation.
How are polymers selected for specific mRNA therapeutics?
Polymer selection depends heavily on the target tissue, required expression duration, and administration route. For instance, rapidly biodegradable poly(β-amino esters) are ideal for systemic lung delivery and chronic dosing, while ligand-functionalized polymers are chosen for active cellular targeting in cancer immunotherapy. The physicochemical profile must perfectly match the clinical objective.
How do polymer nanocarriers compare to lipid nanoparticles?
While lipid nanoparticles rely on the hydrophobic self-assembly of ionizable lipids and excel in vaccines, synthetic polymers offer superior covalent versatility. Polymers can be engineered with precise structural architectures, programmable degradation kinetics, and advanced targeting ligands, making them highly adaptable for chronic protein replacement therapies and complex extrahepatic targeting.
What are the primary toxicity concerns associated with cationic polymers?
High-molecular-weight polycations possessing excessive positive charge density can destabilize and disrupt anionic cellular membranes, causing acute cytotoxicity. Furthermore, non-degradable polymers risk long-term bioaccumulation. Modern formulations mitigate these risks by utilizing low-molecular-weight subunits interconnected via bio-labile linkages, ensuring rapid intracellular hydrolysis and efficient renal clearance.
How is the stability of polymer-mRNA formulations maintained during storage?
Unlike many lipid-based systems requiring stringent ultra-cold chain logistics, specific polymer-mRNA polyplexes exhibit remarkable thermodynamic resilience. By incorporating suitable cryoprotectants, these formulations can often be lyophilized (freeze-dried) into stable powders. This process preserves the fragile mRNA secondary structure, significantly extending shelf-life at conventional refrigeration or even ambient temperatures.

 Fig. 1. Polymer encapsulation overcomes enzymatic degradation and cellular uptake biological barriers (BOC Sciences Authorized).
Fig. 1. Polymer encapsulation overcomes enzymatic degradation and cellular uptake biological barriers (BOC Sciences Authorized). Fig. 2. Structural comparison of four core polymer architectures for mRNA delivery (BOC Sciences Authorized).
Fig. 2. Structural comparison of four core polymer architectures for mRNA delivery (BOC Sciences Authorized). Fig. 3. Clinical applications of polymer-based mRNA delivery across four therapeutic areas (BOC Sciences Authorized).
Fig. 3. Clinical applications of polymer-based mRNA delivery across four therapeutic areas (BOC Sciences Authorized).