How Polymer Microspheres Work in Drug Delivery: Mechanisms, Design, and Applications
Polymer microsphere drug delivery systems represent one of the most versatile and scientifically advanced platforms in controlled release technology. By engineering polymeric materials into microscale spherical carriers, researchers can precisely regulate drug encapsulation, release kinetics, stability, and targeting behavior. These systems are widely explored in pharmaceutical formulation development, biomaterials research, and advanced drug delivery design. Their performance depends on polymer chemistry, physicochemical interactions, structural morphology, and degradation behavior, all of which can be tailored through rational material selection and formulation strategies.
Resources
What Are Polymer Microspheres?
Polymer microspheres are highly engineered, spherical particulate systems typically ranging in diameter from 1 to 1000 micrometers. Structurally, they consist of a macromolecular network where active pharmaceutical ingredients are either dissolved, entrapped, or covalently attached. Depending on the specific fabrication method, these particles can take the form of microcapsules, featuring a distinct core-shell architecture, or micromatrices, where the therapeutic agent is uniformly dispersed throughout the continuous polymeric network. Their functionality is rooted in controlled release science, where drug diffusion, polymer erosion, or environmental triggers govern the liberation of active compounds. These systems enhance formulation stability, improve pharmacokinetic control, and enable sustained or programmed delivery profiles.
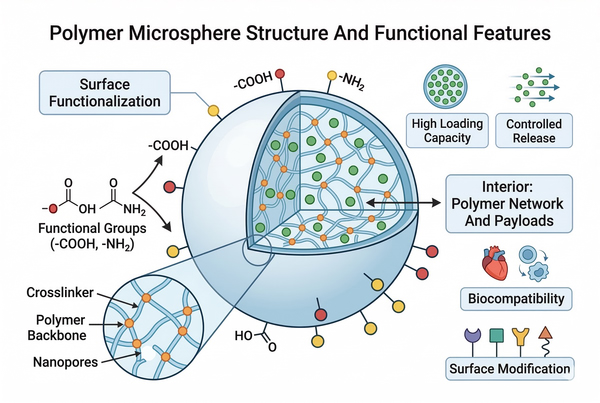 Fig. 1. Schematic illustration of polymer microsphere structure and functional features (BOC Sciences Authorized).
Fig. 1. Schematic illustration of polymer microsphere structure and functional features (BOC Sciences Authorized).
Core Principles of Microsphere-Based Drug Delivery
The foundational principle of this technology lies in the isolation of the active agent from the external biological environment until release is desired. This isolation mitigates premature metabolic degradation and rapid systemic clearance. By manipulating the physicochemical properties of the polymer shell or matrix, scientists can dictate the precise rate at which the active agent is liberated into the surrounding medium. This controlled liberation depends heavily on the complex interplay between polymer hydration, matrix swelling, and the thermodynamic driving forces of diffusion.
Why Polymer Materials Are Used in Controlled Release Systems?
Polymers are the material of choice for controlled release due to their immense structural diversity and tunable physicochemical properties. Synthetic chemistry allows for the precise control of molecular weight, polydispersity index, monomer ratios, and stereochemistry. These parameters directly dictate the glass transition temperature, crystallinity, and hydrophobicity of the final matrix. Consequently, formulation scientists can custom-design the macromolecular architecture to achieve an exact degradation timeline, ranging from a few days to several months, while maintaining excellent biocompatibility.
Advantages Over Conventional Drug Formulations
Conventional formulations often result in rapid peaks and troughs in systemic concentration, leading to potential toxicity or sub-therapeutic windows. Microsphere systems eliminate this volatility by providing a sustained, zero-order or pseudo-zero-order release profile. This mechanism drastically reduces dosing frequency, thereby enhancing formulation efficacy and stability. Furthermore, enclosing sensitive compounds within a polymeric matrix provides robust protection against enzymatic cleavage, oxidative stress, and hydrolytic degradation during storage and post-administration.
Types of Polymer Microspheres in Drug Delivery
The classification of polymer microspheres is largely dictated by their degradation behavior, origin, and responsiveness to environmental stimuli. Selecting the appropriate classification is the first and most critical step in formulation design, as it fundamentally determines the ultimate fate of both the carrier and its payload.
Biodegradable Polymer Microspheres
Biodegradable polymers represent the vanguard of advanced formulation science due to their transient nature in biological systems. These microspheres are constructed from polymers that undergo gradual chain scission, primarily via the hydrolysis of ester, amide, or anhydride linkages within their macromolecular backbone. As water penetrates the porous matrix, it initiates a cascade of hydrolytic cleavage, progressively reducing the polymer into lower molecular weight oligomers and ultimately into biocompatible monomeric units. Aliphatic polyesters are the paradigm of this class, offering precisely tunable degradation rates dictated by their crystalline-to-amorphous ratio and inherent molecular hydrophobicity. The transient structural nature of these microspheres is highly advantageous for injectable depot formulations, as the delivery vehicle safely dissolves and clears through standard metabolic pathways, negating any requirement for subsequent mechanical extraction.
Non-Biodegradable Polymer Microspheres
In stark contrast to their degradable counterparts, non-biodegradable polymer microspheres are engineered for absolute structural permanence. Fabricated from highly robust, chemically inert macromolecules such as poly(methyl methacrylate) or heavily crosslinked silicone elastomers, these matrices resist enzymatic degradation and hydrolytic cleavage indefinitely. Because the polymer backbone remains intact, the liberation of the active pharmaceutical ingredient is governed entirely by Fickian diffusion through the preexisting and static pore network of the matrix. This permanent architecture is exceptionally valuable for specialized localized applications, including long-term chemoembolization platforms where the simultaneous mechanical occlusion of vasculature and the protracted, steady-state diffusion of an active agent are required simultaneously.
Natural vs Synthetic Polymer-Based Microspheres
The dichotomy between natural and synthetic polymers fundamentally shapes the entire formulation strategy, with each category presenting distinct thermodynamic and processing advantages.
- Natural Polymers: Derived from biological sources such as complex polysaccharides or proteins, materials like alginate and chitosan inherently possess exceptionally high biocompatibility and abundant hydrophilic functional groups. These groups facilitate extremely mild, aqueous-based crosslinking mechanisms, making them ideal matrices for encapsulating highly sensitive biological macromolecules that would otherwise denature upon exposure to organic solvents. However, they frequently exhibit significant batch-to-batch variability in molecular weight and purity.
- Synthetic Polymers: Conversely, synthetic polymers offer unparalleled precision in structural engineering. Through rigorously controlled polymerization techniques, chemists can tightly regulate the molecular weight, polydispersity index, and stereoisomer ratios of the resulting macromolecules. This synthetic rigor translates to highly reproducible degradation kinetics, absolute material purity, and consistent performance across manufacturing scales.
| Feature | Natural Polymers | Synthetic Polymers |
|---|
| Origin | Biological extraction (polysaccharides, proteins) | Chemical synthesis (polyesters, polyamides) |
| Biocompatibility | Exceptionally high, intrinsic recognition | High, but depends on monomer breakdown products |
| Batch Consistency | Variable, highly dependent on source material | Exceptionally consistent and highly reproducible |
| Processing Conditions | Mild, often entirely aqueous-based | Often requires specific organic solvents |
| Degradation Control | Difficult to tune precisely | Highly tunable via molecular weight and co-monomer ratios |
Table 1. Comparison of natural and synthetic polymers.
Functionalized and Surface-Modified Microspheres
The interfacial properties of a microsphere dictate its immediate interaction with the surrounding biological fluid. Unmodified, strongly hydrophobic microspheres are highly susceptible to opsonization and rapid clearance by the reticuloendothelial system. To engineer a prolonged circulation half-life and prevent unwanted protein corona formation, the microsphere surface is rigorously modified. A prevalent strategy involves the covalent grafting of hydrophilic chains to create a dense steric barrier that repels protein adsorption. Beyond simple stealth properties, advanced surface functionalization allows for the introduction of reactive moieties, such as maleimides or activated esters. These functional groups serve as stable, orthogonal anchoring points for the precise covalent conjugation of targeting ligands, effectively transforming a passive particulate depot into a highly selective, actively homing delivery vector.
Stimuli-Responsive Polymer Microspheres
Stimuli-responsive, or smart, polymer microspheres represent the apex of customized controlled release technology. These advanced matrices are synthesized from complex macromolecules that undergo abrupt physical or chemical phase transitions in response to subtle fluctuations in their immediate microenvironment. By incorporating specific ionizable groups or thermo-responsive block segments into the primary polymer architecture, the overall matrix can be engineered to react dynamically to localized physiological triggers such as steep pH gradients, specific enzymatic concentrations, or elevated levels of reactive oxygen species. Upon encountering the designated stimulus, the microsphere may undergo rapid volumetric swelling, accelerated phase separation, or complete structural collapse. This dynamic behavior ensures that the encapsulated payload remains securely sequestered during systemic circulation and is liberated exclusively upon reaching the precise spatial coordinates of the targeted microenvironment.
Looking for Custom Polymer Microspheres?
Whether you need biodegradable microspheres, controlled-release systems, or surface-functionalized carriers, our experts provide tailored synthesis, formulation, and optimization solutions to support your research and product development.
Mechanism of Drug Release from Polymer Microspheres
Understanding the exact physical and chemical pathways by which an active ingredient transitions from the interior of a polymer matrix into the surrounding medium is essential for predicting and controlling the release profile. Release is rarely governed by a single pathway; rather, it is a synergistic combination of multiple kinetic phenomena.
- Drug Encapsulation Techniques: The mechanism of release is intrinsically tied to how the drug was originally encapsulated. Techniques such as single or double emulsion-solvent evaporation create specific internal morphologies. Spray drying often yields solid dispersions with high drug loading, while coacervation produces distinct core-shell structures. The distribution of the active pharmaceutical ingredient—whether as solid particulates, a molecular dispersion, or concentrated within an aqueous core—dictates the initial diffusion pathways and subsequent release kinetics.
- Diffusion-Controlled Release Mechanism: Diffusion is the primary driving force in the early stages of release. In reservoir systems, the active agent diffuses through a rate-limiting polymeric membrane. In matrix systems, the agent diffuses through the tortuous pore network of the polymer itself. This process is mathematically described by Fickian diffusion principles, where the rate is proportional to the concentration gradient, the diffusion coefficient of the molecule, and the porosity of the macromolecular network.
- Polymer Degradation and Erosion Mechanism: For biodegradable systems, release is intimately linked to the breakdown of the polymer backbone. Bulk erosion occurs when water penetrates the entire matrix, causing uniform degradation and a sudden, massive release of the remaining payload once a critical molecular weight threshold is breached. Surface erosion, characteristic of highly hydrophobic polymers, involves degradation strictly at the polymer-water interface, leading to a highly predictable, constant release rate that perfectly correlates with the loss of polymer mass.
- Swelling and Matrix Relaxation Mechanism: When hydrophilic polymer microspheres are exposed to aqueous environments, they absorb water and swell. This hydration process lowers the glass transition temperature of the polymer, causing the rigid macromolecular chains to relax into a rubbery state. This relaxation significantly increases the free volume within the matrix, thereby increasing the diffusion coefficient of the entrapped drug and initiating release.
- Combination and Multi-Phase Release Mechanisms: Most advanced formulations exhibit a multi-phase release profile. A common phenomenon is the initial burst release, where surface-associated or poorly entrapped drug diffuses rapidly upon administration. This is frequently followed by a lag phase with minimal release, and finally, a secondary sustained release phase driven by bulk polymer degradation. Formulators continuously strive to engineer the polymer structure to flatten these phases into a seamless, continuous profile.
- Targeted Delivery and Cellular Uptake: Beyond extracellular release, microspheres can be engineered for direct cellular interaction. Depending on their size and surface charge, specific microspheres undergo endocytosis or phagocytosis by target cells. Once internalized, the acidic environment of the endosome or the presence of intracellular enzymes can trigger the final degradation of the polymer matrix, delivering the payload directly into the cytosol and bypassing membrane-impermeability issues.
Materials Used in Polymer Microsphere Formulation
The architectural foundation of any controlled-release vehicle is the selection of its monomeric precursors and the resulting macromolecular framework. In polymer microsphere engineering, the choice of material is not merely a matter of structural containment; it is a strategic decision that dictates the thermodynamic stability of the payload, the mechanical integrity of the particle, and the precise kinetic profile of the release phase. Advanced delivery systems utilize a spectrum of aliphatic polyesters and specialized copolymers, each offering a distinct set of physicochemical parameters that can be precisely calibrated to meet the rigorous demands of modern therapeutics.
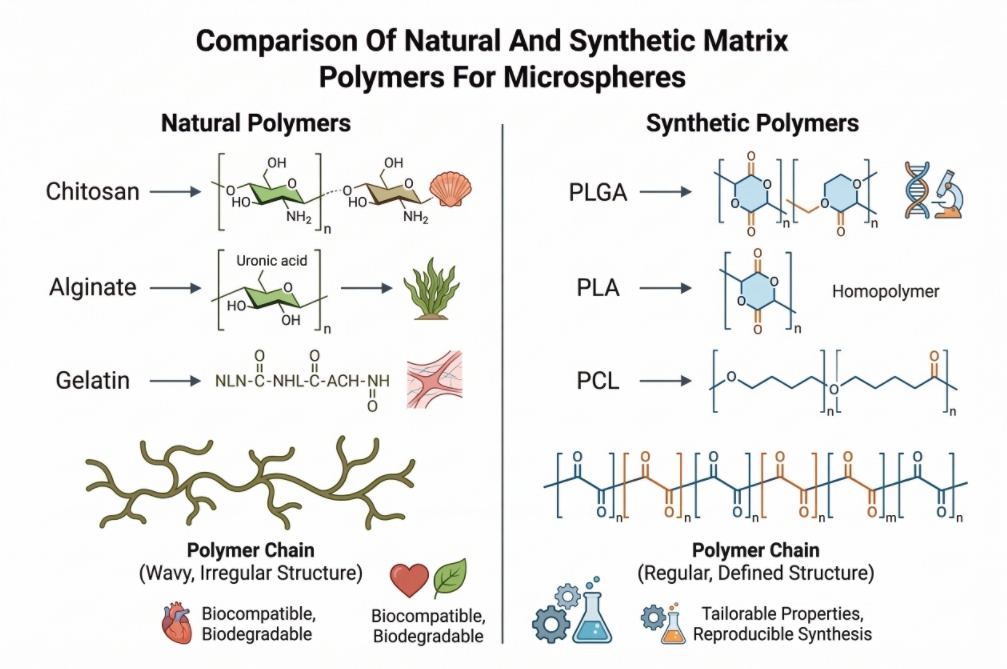 Fig. 2. Structural comparison of natural and synthetic polymers for microspheres (BOC Sciences Authorized).
Fig. 2. Structural comparison of natural and synthetic polymers for microspheres (BOC Sciences Authorized).
Poly(lactic acid) (PLA) and Poly(lactic-co-glycolic acid) (PLGA)
Poly(lactic-co-glycolic acid) (PLGA) and its homopolymer Poly(lactic acid) (PLA) are the most extensively utilized materials in the field of degradable microspheres. PLGA is a linear amorphous copolymer synthesized through the ring-opening polymerization of lactide and glycolide monomers. The primary advantage of PLGA lies in its highly tunable degradation rate, which is controlled by the molar ratio of its constituent monomers. Lactic acid contains a methyl group that renders the polymer more hydrophobic, impeding water penetration and slowing the rate of ester bond hydrolysis. Conversely, glycolic acid lacks this side chain, making the polymer more hydrophilic and susceptible to rapid hydration. By adjusting the lactide-to-glycolide ratio—common configurations include 50:50, 65:35, and 75:25—formulators can engineer microspheres that degrade over timescales ranging from two weeks to six months. Furthermore, the inherent biocompatibility of these materials is exceptional, as they eventually break down into endogenous metabolites that are cleared via the tricarboxylic acid cycle.
Polycaprolactone (PCL)
Polycaprolactone (PCL) is a semi-crystalline aliphatic polyester known for its high solubility in organic solvents and its remarkably slow degradation kinetics. Unlike the relatively rapid hydrolysis seen in PLGA, PCL possesses a high degree of crystallinity and significant hydrophobicity, which together create a formidable barrier to aqueous infiltration. In biological environments, PCL microspheres may take several years to fully erode. This makes PCL the premier material choice for ultra-long-acting depot formulations or for the delivery of active agents that require a very low, constant release rate over an extended duration. Additionally, PCL exhibits a low glass transition temperature and high permeability to many small-molecule drugs, allowing for the design of matrix-type systems where release is governed by steady-state diffusion rather than polymer erosion.
Polylactic Acid Derivatives and Copolymers
To address the limitations of conventional linear polyesters—such as the acidic microenvironment created during bulk erosion or poor encapsulation of hydrophilic payloads—advanced derivatives and block copolymers are frequently employed.
- PEGylated Copolymers (PLGA-PEG): The incorporation of Poly(ethylene glycol) (PEG) blocks into the polyester backbone creates amphiphilic copolymers. These materials self-assemble or facilitate the formation of microspheres with a hydrophilic corona, significantly reducing protein adsorption and enhancing the stability of the particles in physiological buffers.
- Branched and Star-Shaped Polymers: By utilizing multi-functional initiators during polymerization, star-shaped PLA or PLGA can be synthesized. These branched architectures possess different entanglement densities and crystalline behaviors compared to linear chains, offering unique mechanical properties and altered degradation profiles that can eliminate the common burst release effect.
- Poly(lactic acid-co-lysine): The introduction of functional amino acids into the polymer backbone provides pendant reactive groups, such as primary amines, which are essential for the subsequent covalent attachment of targeting moieties or for modifying the internal charge of the microsphere matrix.
Functionalized and Modified Polymers for Targeting
Standard structural polymers often lack the necessary chemical handles for the high-affinity binding required in active targeting. Consequently, the synthesis of functionalized polymers has become a critical focus in formulation design. This involves the strategic placement of reactive end-groups—such as maleimide, N-hydroxysuccinimide (NHS) esters, or alkyne groups for click chemistry—at the termini of the polymer chains. These activated polymers are then blended with standard materials during the microsphere fabrication process. Once the particles are formed, these surface-exposed functional groups allow for the site-specific, orientation-controlled conjugation of antibodies, peptides, or aptamers. This level of molecular engineering transforms the microsphere from a simple protective carrier into an intelligent, ligand-directed delivery system capable of recognizing specific cellular markers or navigating complex biological barriers.
Design and Formulation Strategies of Microsphere
Developing a polymer microsphere formulation is a rigorous exercise in chemical engineering and physical chemistry. It requires the delicate balancing of numerous process parameters to achieve optimal loading, stability, and release characteristics.
- Selection of Appropriate Polymer Materials: The initial design phase requires a deep understanding of the Flory-Huggins interaction parameters between the polymer and the active pharmaceutical ingredient. Highly hydrophobic drugs pair well with high-lactide PLGA or PCL, maximizing encapsulation efficiency through strong hydrophobic interactions. Conversely, highly water-soluble agents require modified hydrophilic blocks or complex double-emulsion techniques to prevent rapid partitioning into the continuous phase during fabrication.
- Surface Modification and Functionalization Strategies: Controlling the surface properties is essential for dictating the microsphere interactions with biological fluids. Adjusting the zeta potential through the selection of specific surfactants or polymer end-groups prevents aggregation and improves colloidal stability. Functionalization strategies often involve carbodiimide chemistry to covalently link stabilizing agents or stealth polymers directly to the microsphere surface, ensuring long-term stability post-administration.
- Targeted Delivery and Ligand Conjugation: When localized accumulation is required, ligand conjugation is employed. The stoichiometry and spatial orientation of the conjugated ligands are critical parameters. Too few ligands result in insufficient avidity, while excessive conjugation can alter the fundamental physicochemical properties of the microsphere or cause steric hindrance that blocks receptor binding. Optimization requires precise control over the conjugation chemistry and rigorous analytical quantification.
- Polymer–Drug Compatibility and Loading Optimization: Maximizing drug loading without compromising the structural integrity of the microsphere is a primary formulation challenge. High loading can induce phase separation within the matrix, leading to uncontrolled burst release. Formulation engineers utilize solid-state characterization techniques, such as differential scanning calorimetry and X-ray powder diffraction, to ensure the active ingredient remains in the desired amorphous or crystalline state within the polymer matrix.
- Controlling Release Kinetics and Duration: Release kinetics are finely tuned by manipulating the geometric and structural properties of the microspheres. Increasing the molecular weight of the polymer slows degradation, while increasing the internal porosity accelerates diffusion. Furthermore, controlling the particle size distribution through precise homogenization or microfluidic techniques ensures a uniform surface-area-to-volume ratio, which is critical for achieving reproducible batch-to-batch release profiles.
- Stability, Sterility, and Storage Considerations: Polymer microspheres are highly susceptible to hydrolytic degradation during storage. Therefore, lyophilization is frequently employed to remove residual moisture. However, the freezing and drying stresses can collapse the porous structure. Cryoprotectants are essential to preserve the microarchitecture. Additionally, terminal sterilization via gamma irradiation must be carefully calibrated, as excessive radiation can induce polymer chain scission and drastically alter the intended release kinetics.
- In Vitro and In Vivo Evaluation of Microsphere Systems: Rigorous analytical evaluation bridges the gap between synthesis and application. In vitro release testing utilizes specialized flow-through or sample-and-separate methods to map the kinetic release profile. Preclinical in vivo evaluation focuses on pharmacokinetic mapping in animal models to determine the area under the curve, peak concentration, and systemic half-life, ensuring the formulation meets the exact theoretical design specifications before further advancement.
Applications of Polymer Microsphere in Drug Delivery
The versatility of polymer microspheres has led to their broad adoption across a wide spectrum of therapeutic classes. By solving specific solubility, stability, and targeting challenges, these matrices enable the delivery of highly complex molecules that would otherwise be non-viable in a clinical setting. The ability to manipulate the polymer-to-drug ratio and the internal porosity of the matrix allows for the customization of delivery platforms for everything from systemic oncology treatments to localized vaccine depots.
Small Molecule Drugs
For small molecule APIs, especially those falling into the more challenging categories of the Biopharmaceutics Classification System (BCS), polymer microspheres offer a sophisticated solution to severe insolubility and rapid systemic clearance. By molecularly dispersing these hydrophobic molecules within an amorphous polymer matrix, formulation engineers can prevent drug crystallization and significantly enhance the apparent solubility. This entrapment effectively masks the drug’s inherent physicochemical limitations, extending the circulation half-life and maintaining consistent systemic exposure over prolonged periods. This is particularly beneficial for therapeutics with narrow window of efficacy, where maintaining a steady-state concentration is critical to avoiding toxicity.
Protein and Peptide Therapeutics
Large biologics, such as hormones, enzymes, and growth factors, are notoriously fragile and susceptible to rapid proteolytic cleavage or denaturation. Encapsulating proteins and peptides within a dense polymer matrix provides a robust physical barrier against enzymatic degradation in the biological environment. The primary design challenge in this scenario is maintaining the secondary and tertiary structures of the biologic during the encapsulation process. Advanced formulation techniques, such as solid-in-oil-in-water (S/O/W) or double emulsion methods, are utilized to shield the protein from harsh organic solvents, ensuring that the liberated therapeutic remains fully functional and bioactive upon release from the eroding microsphere.
Nucleic Acid Delivery (DNA, RNA, mRNA)
The delivery of negatively charged, highly degradable nucleic acids requires specialized polycationic or surface-modified polymer matrices. These polymers form electrostatic complexes with the nucleic acids, condensing them into stable polyplexes that are then sequestered within the microsphere. This structure protects the genetic material from pervasive nucleases in the bloodstream. Furthermore, these microspheres can be engineered with endosomolytic properties—often through the inclusion of polymers with a strong buffering capacity—to facilitate endosomal escape. This ensures the nucleic acid payload successfully reaches the cytosol or nucleus, enabling efficient gene silencing or protein expression.
Cancer Therapy and Targeted Oncology Delivery
In oncology, polymer microspheres are utilized to leverage the enhanced permeability and retention (EPR) effect or to serve as localized depots placed directly at the tumor site. By providing a continuous, high-concentration localized release of cytotoxic agents, these systems maximize the exposure of the malignant tissue to the active agent while drastically minimizing systemic distribution. This localized approach is particularly effective in chemoembolization, where microspheres physically block the blood supply to a tumor while simultaneously releasing a steady dose of chemotherapy. Advanced functionalization also allows these particles to be decorated with ligands that recognize tumor-specific receptors, further increasing the precision of the delivery.
Vaccine Delivery Systems
Polymer microspheres are exceptional platforms for advanced vaccine formulations, acting as both a physical reservoir for the sustained release of antigens and an inherent adjuvant. The particulate nature of the microspheres, typically engineered to be between 1 and 10 micrometers, mimics the physical dimensions of pathogens. This strongly promotes uptake by antigen-presenting cells (APCs) such as dendritic cells and macrophages. By controlling the polymer degradation rate, a single injection can provide a pulsed release of antigens over several weeks, effectively mimicking the effect of multiple booster shots and driving a more robust, long-lasting cellular and humoral immune response.
Long-Acting Injectable Formulations
The most commercially successful application of this technology is the development of long-acting injectable (LAI) suspensions for intramuscular or subcutaneous administration. These formulations are designed to slowly erode over weeks or even months, providing a highly reliable, zero-order release of the active agent. This approach is highly valued in the treatment of chronic conditions, such as psychiatric disorders or endocrine imbalances, where patient compliance is a significant factor in therapeutic success. By replacing daily oral dosing with a single monthly injection, polymer microsphere LAIs ensure that the drug concentration remains within the therapeutic window without the volatility associated with traditional dosage forms.
Services
Polymer Microsphere Development Services from BOC Sciences
BOC Sciences provides comprehensive polymer microsphere development services for pharmaceutical research, biomedical applications, and advanced material innovation. Leveraging expertise in monomer design, controlled polymer synthesis, biodegradable polymer engineering, and formulation development, we support projects from early-stage material design to process optimization and scale-up production. Our capabilities cover biodegradable microspheres, drug-loaded microspheres, controlled release systems, and functional polymer microspheres, enabling customized solutions tailored to specific application requirements. With advanced synthesis platforms and analytical technologies, BOC Sciences helps clients develop microsphere systems with precise particle size control, high stability, and optimized performance for research and industrial applications.

Custom Polymer Design and Synthesis for Microspheres
- Custom design and synthesis of polymers for microsphere fabrication, including biodegradable and functional materials.
- Structural development of PLGA, PLA, PCL, and other controlled-release polymers.
- Precise molecular weight and composition control to regulate degradation rate and performance.
- Development of functionalized polymers for enhanced application versatility.

Polymer Microsphere Fabrication and Process Development
- Development of microsphere preparation methods, including emulsion, solvent evaporation, and phase separation techniques.
- Optimization of particle size distribution and uniformity for specific delivery routes.
- Support for drug encapsulation microsphere systems with improved loading efficiency.
- Process optimization to ensure reproducibility and formulation stability.

Functionalization and Advanced Application Development
- Surface modification services for enhanced functionality and targeted delivery.
- Conjugation of peptides, antibodies, or bioactive molecules onto microsphere surfaces.
- Design of controlled and sustained release microsphere systems.
- Development of multi-functional microsphere platforms for drug delivery and diagnostic applications.

Characterization, Quality Control, and Scale-Up Production
- Particle size and morphological analysis to evaluate structural properties.
- Assessment of drug loading efficiency and release kinetics.
- Evaluation of stability and biodegradation performance.
- Support for batch consistency control and scale-up production from laboratory to larger quantities.
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
How do polymer microspheres control drug release?
They control release through a combination of physical and chemical mechanisms. Initially, release is governed by the diffusion of the active agent through the porous polymer network. Over time, as water penetrates the matrix, the polymer backbone begins to hydrolyze and degrade, leading to bulk or surface erosion which further releases the entrapped molecules in a sustained, highly predictable manner.
-
What types of drugs can be delivered using polymer microspheres?
These versatile systems can deliver an extensive range of therapeutic agents. They are highly effective for hydrophobic small molecules, fragile large biologics like proteins and peptides, and sensitive nucleic acids (DNA/RNA). The choice of polymer and fabrication technique can be adapted to accommodate the specific molecular weight, charge, and solubility profile of virtually any active pharmaceutical ingredient.
-
How are polymer microspheres customized for specific therapeutics?
Customization occurs at the chemical synthesis level. Scientists manipulate the molecular weight, hydrophobicity, and co-monomer ratios (such as the lactide-to-glycolide ratio in PLGA) to tune the degradation rate. Furthermore, the surface of the microspheres can be chemically modified with PEG for stealth properties or conjugated with specific ligands to achieve targeted accumulation at specific cellular sites.
-
What are the advantages of polymer microspheres over other delivery systems?
Compared to traditional immediate-release formulations or simple suspensions, polymer microspheres provide unparalleled control over release kinetics, enabling zero-order sustained release that drastically reduces dosing frequency. Compared to liposomes, solid polymer matrices offer significantly higher mechanical stability, superior protection against premature enzymatic degradation of the payload, and more highly predictable, longer-term degradation profiles.

 Fig. 1. Schematic illustration of polymer microsphere structure and functional features (BOC Sciences Authorized).
Fig. 1. Schematic illustration of polymer microsphere structure and functional features (BOC Sciences Authorized). Fig. 2. Structural comparison of natural and synthetic polymers for microspheres (BOC Sciences Authorized).
Fig. 2. Structural comparison of natural and synthetic polymers for microspheres (BOC Sciences Authorized).