How Synthetic Polymers Enable Targeted Vaccine Antigen Delivery?
The paradigm of vaccinology is rapidly shifting from traditional whole-pathogen formulations toward molecularly defined antigens, including recombinant subunits, synthetic peptides, and emerging nucleic acid (mRNA/DNA) technologies. While these next-generation antigens offer significant advantages in specificity and safety, their clinical efficacy is often limited by intrinsic physicochemical instability, short in vivo half-life, and poor membrane permeability. Exposed antigens are highly susceptible to nuclease or protease degradation in the complex physiological environment and are inefficiently taken up by antigen-presenting cells (APCs), resulting in weak immune responses. In this context, synthetic polymers, with their exceptional chemical versatility and controllable structures, have emerged as key tools to overcome these delivery bottlenecks. Compared with viral or lipid-based systems, synthetic polymers allow precise molecular engineering of molecular weight, topology, and surface functionalities, enabling the creation of intelligent delivery platforms that mimic pathogen features, protect fragile cargo, and achieve targeted lymph node delivery.
Resources
Introduction to Synthetic Polymers for Vaccine and Antigen Delivery
Synthetic polymers have emerged as versatile tools in modern vaccine and antigen delivery, offering precise control over size, composition, and functionalization. By enabling targeted delivery and sustained release, these polymers enhance immune responses while minimizing side effects. Their adaptability makes them a critical component in developing next-generation vaccines and immunotherapies.
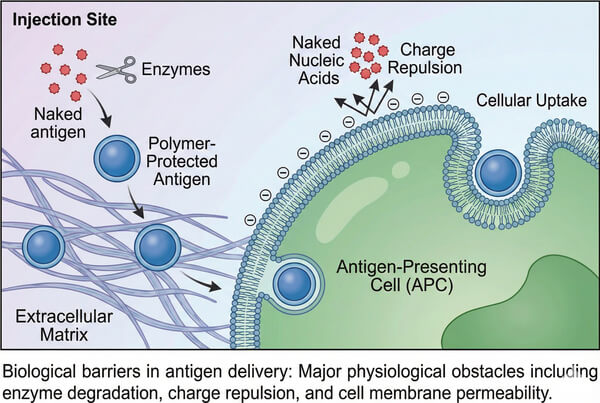 Fig. 1. Polymer encapsulation protects antigens from enzyme degradation and facilitates uptake by antigen-presenting cells (BOC Sciences Authorized).
Fig. 1. Polymer encapsulation protects antigens from enzyme degradation and facilitates uptake by antigen-presenting cells (BOC Sciences Authorized).
Challenges in Vaccine and Antigen Delivery
The core of vaccine development lies in delivering antigens intact to specific sites within the immune system and inducing effective humoral or cellular immune responses. This process, however, faces multiple biological barriers:
- Physicochemical instability: Exposed antigens, particularly RNA and peptides, are prone to aggregation, denaturation, or degradation in the complex physiological environment (e.g., pH changes, shear forces in bodily fluids).
- Enzymatic degradation: Nucleases and proteases commonly present in serum and tissue fluids rapidly eliminate exogenous antigens, leading to loss of activity before reaching target cells.
- Low cellular uptake: Antigen uptake by APCs, such as dendritic cells (DCs), is crucial for initiating immune responses. Most antigens are negatively charged or hydrophilic, hindering their penetration through the lipid bilayer of cell membranes.
- Lack of targeting: Non-specific systemic distribution not only reduces the effective concentration in lymphoid organs but may also cause systemic side effects.
Why Synthetic Polymers Are Used as Vaccine Carriers?
Compared with viral or lipid carriers, synthetic polymers offer unique advantages in vaccine delivery:
- Structural tunability: Polymerization reactions can be precisely controlled to adjust molecular weight (MW), polydispersity index (PDI), and topology (linear, star-shaped, dendritic).
- Functional modification: Polymers often contain abundant reactive side-chain groups (e.g., amino, carboxyl, hydroxyl) for conjugating targeting ligands, adjuvants, or PEGylation to prolong circulation.
- Environmental responsiveness: Polymers can be designed to respond to pH, temperature, redox potential, or enzymatic triggers, enabling antigen release in specific intracellular environments (e.g., endosomes or cytosol).
- Evasion of Pre-existing Immunity: Unlike viral vectors, synthetic polymers typically do not trigger anti-vector neutralizing antibodies, permitting effective repeated dosing and booster regimens.
- Manufacturing Scalability: Synthetic chemistry enables robust, cost-effective, and large-scale production with high batch-to-batch consistency, avoiding the complexity of biological manufacturing systems.
Functional Roles of Synthetic Polymers in Antigen Delivery
Synthetic polymers are not inert carriers—they actively modulate the pharmacokinetics and immunological properties of antigens. Through precise molecular design, polymer carriers can mimic pathogen characteristics and enhance immune recognition and processing.
Antigen Encapsulation and Structural Protection
Preserving the primary structure and conformational epitopes of antigens is critical for inducing neutralizing antibodies. Synthetic polymers form microspheres, nanocapsules, or complexes that provide a physical barrier against nuclease and protease attacks. For example, hydrophilic PEG shells create steric hindrance that prevents non-specific plasma protein adsorption (opsonization), reducing clearance by the reticuloendothelial system (RES) and maintaining antigen stability in circulation.
Controlled Antigen Release and Depot Effect
Traditional vaccination often produces a "pulsed" antigen concentration that rapidly declines. Polymer carriers enable sustained antigen release, mimicking prolonged exposure seen in natural infections.
- Depot effect: Polymeric microspheres form a reservoir at the injection site, slowly degrading and releasing antigen to continuously stimulate the immune system, potentially reducing booster requirements.
- Release kinetics: By tuning polymer degradation rates (e.g., adjusting lactic acid to glycolic acid ratios in PLGA), antigen release can be precisely controlled, either via diffusion from the matrix or polymer scaffold erosion.
Enhancement of Antigen Uptake by Antigen-Presenting Cells
DC maturation and antigen presentation are key to immune activation. Cationic polymers adsorb onto anionic cell membranes through electrostatic interactions, promoting adsorptive endocytosis. Some polymers are designed as pathogen-mimicking particles with surface modifications such as mannose or anti-DEC-205 antibodies, specifically targeting APC receptors and significantly enhancing uptake.
Modulation of Immune Responses through Polymer Design
Polymers themselves can act as immunostimulatory adjuvants:
- Proton sponge effect: Polymers with abundant buffering groups (e.g., PEI, PAMAM) absorb protons in acidic endosomes, causing chloride and water influx, endosomal swelling, and rupture, facilitating antigen escape to the cytosol. This is critical for CD8⁺ T cell-mediated cytotoxic immune responses.
- TLR activation: Certain synthetic polymers directly activate Toll-like receptors (TLRs), inducing cytokine secretion and modulating Th1/Th2 immune balance.
Types of Synthetic Polymers Used for Vaccine and Antigen Delivery
The diversity of synthetic polymers offers broad opportunities for vaccine design. Different types of polymers, based on their chemical structure, charge properties, and degradation mechanisms, are suitable for various antigens (such as nucleic acids, proteins, and peptides) and administration routes.
Polysaccharide-Based Synthetic Polymers for Antigen Encapsulation
Although polysaccharides (e.g., chitosan, glucans, hyaluronic acid) are naturally derived, precise synthetic modifications such as carboxymethylation, quaternization, or thiolation are often required to overcome poor solubility or inconsistent immunogenicity. These chemical modifications not only significantly improve material stability and antigen loading capacity but also impart unique mucoadhesive properties, enabling transient opening of epithelial tight junctions—particularly suitable for nasal or oral vaccine delivery. In addition, certain modified polysaccharides can specifically recognize and bind lectin receptors on antigen-presenting cells (APCs), exerting intrinsic adjuvant effects and enhancing immune responses without the need for conventional adjuvants.
Poly(amino acids) for Biodegradable Vaccine Carriers
Poly(amino acids) such as poly(glutamic acid) (PGA), poly(lysine) (PLL), and poly(aspartic acid) (PASP) are synthetic polymers that mimic natural protein structures. Their key advantage lies in being fully degradable in vivo by proteases into non-toxic endogenous amino acids, addressing safety concerns associated with polymer accumulation. Rich functional groups on their side chains (e.g., amino, carboxyl) allow convenient hydrophobic side-chain branching or covalent antigen conjugation to form amphiphilic core-shell nanomicelles. This architecture not only effectively protects antigens from enzymatic degradation but also allows tuning of micelle size and surface charge to promote efficient dendritic cell uptake.
Polyamines for Efficient Nucleic Acid Delivery
Polyamines, exemplified by polyethyleneimine (PEI) and its derivatives, possess high-density positive charges and are considered the "gold standard" among non-viral carriers for compacting and delivering mRNA or DNA vaccines. They electrostatically condense negatively charged nucleic acids into nanoscale polyplexes and induce endosomal rupture through the "proton sponge effect," enabling nucleic acids to escape into the cytoplasm for translation. To balance high-molecular-weight PEI's transfection efficiency with potential cytotoxicity, current research focuses on developing biodegradable crosslinked low-molecular-weight PEI or PEGylated copolymers for safer clinical applications.
Polyesters for Controlled Antigen Release
Aliphatic polyesters, particularly poly(lactic-co-glycolic acid) (PLGA) and polycaprolactone (PCL), are among the few synthetic polymers approved by the FDA for human drug delivery and have extensive safety data. By precisely tuning the monomer ratios (e.g., lactic acid to glycolic acid) and polymer molecular weight, the degradation kinetics of microspheres or nanoparticles can be controlled, achieving sustained antigen release over weeks to months (Depot Effect). This long-term release mimics continuous antigen exposure seen in natural infections and is ideal for subunit vaccines that require prolonged humoral immune memory, reducing booster doses and improving patient compliance.
Poly(amidoamines) (PAMAM) for Targeted Immune Delivery
PAMAM dendrimers feature highly ordered, radially branched architectures and extremely low polydispersity, allowing highly predictable pharmacokinetics and precise drug design. Their abundant surface functional groups enable multivalent modifications, allowing simultaneous conjugation of antigens, targeting ligands (e.g., mannose), and immunomodulators on a single carrier for specific immune cell targeting. The hydrophobic cavities inside PAMAM can encapsulate small-molecule adjuvants, while their nanoscale size facilitates lymphatic drainage and direct targeting of lymph nodes, maximizing T-cell activation at the source of immune initiation.
Looking for Custom Polymers for Vaccine Delivery?
Whether you need cationic polymers, biodegradable carriers, or stimuli-responsive platforms, our experts provide tailored synthesis and functionalization solutions to support your research and therapeutic development.
Synthetic Polymer-Based Antigen Delivery Technologies
Depending on antigen properties and the desired immune response, synthetic polymers can be engineered into various sophisticated physical forms. These technological platforms not only modulate antigen pharmacokinetics but also optimize immune recognition and processing through unique structural effects.
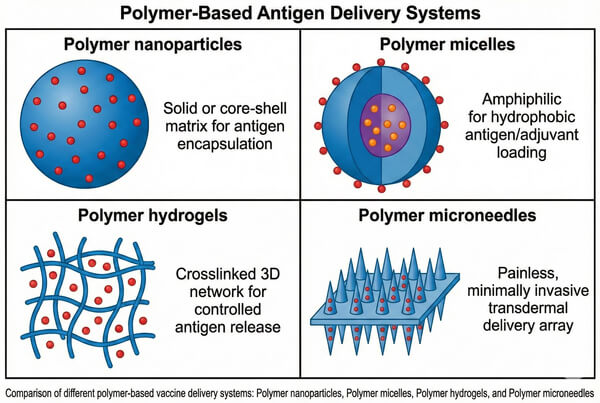 Fig. 2. Structural comparison of common polymer delivery vehicles: nanoparticles, micelles, hydrogels and microneedles (BOC Sciences Authorized).
Fig. 2. Structural comparison of common polymer delivery vehicles: nanoparticles, micelles, hydrogels and microneedles (BOC Sciences Authorized).
Polymer Nanoparticles as Vaccine Delivery Platforms
Polymer nanoparticles (PNPs) typically exist as solid nanospheres or core-shell nanocapsules. Their submicron size (usually<200 nm) enables passive lymphatic targeting to lymph nodes and direct interaction with resident immune cells. PNPs protect antigens from enzymatic degradation in peripheral environments and allow surface functionalization (e.g., PEGylation or ligand conjugation) to tune circulation time and cellular uptake, enabling precise intracellular antigen delivery. This platform is particularly suitable for antigens requiring intracellular processing, as it promotes endosomal escape and enhances cross-presentation to effectively activate CD8⁺ cytotoxic T lymphocyte (CTL) responses.
Polymer Micelles and Complexes for Antigen Delivery
Polymer micelles, formed by the self-assembly of amphiphilic block copolymers in aqueous solution, create core-shell structures with hydrophobic cores that encapsulate hydrophobic immunomodulators (e.g., TLR agonists) and hydrophilic shells that physically adsorb or chemically conjugate hydrophilic antigens, enabling co-delivery of antigens and adjuvants. Polyelectrolyte complexes (PECs) leverage electrostatic interactions between positively charged polymers and negatively charged nucleic acids or protein antigens. Their mild preparation conditions preserve the native bioactivity of macromolecules. These carriers offer high structural flexibility and can be designed to be pH- or redox-sensitive for smart intracellular antigen release.
Polymer Microspheres for Antigen and Adjuvant Delivery
Polymer microspheres typically range from 1–100 μm. Their larger size prevents rapid phagocytic clearance, allowing them to act as "antigen depots" at the injection site. By tuning the polymer matrix (e.g., PLGA) molecular weight and degradation rate, microspheres can achieve sustained or programmed pulsed antigen release, mimicking continuous exposure during natural infections. This controlled release maintains long-term antibody titers and may enable single-shot vaccines, addressing compliance challenges associated with multiple injections.
Polymer Hydrogels for Controlled Antigen Release
Polymer hydrogels are three-dimensional hydrophilic networks formed by crosslinked polymer chains. Their high-water content closely mimics the natural extracellular matrix, preserving protein antigen conformation and stability. Injectable in situ-forming hydrogels undergo sol-gel transitions under physiological conditions, creating local scaffolds that not only provide sustained antigen release but also recruit dendritic cells via chemokine gradients, forming "immune incubators." This local immunomodulation significantly enhances antigen uptake and presentation, particularly suitable for vaccines requiring strong localized immune responses, such as cancer vaccines.
Polymer Microneedles for Minimally Invasive Vaccine Administration
Polymer microneedle arrays, typically fabricated from soluble or biodegradable polymers (e.g., PVP, hyaluronic acid), painlessly penetrate the stratum corneum to deliver antigens directly to Langerhans cell-rich epidermal and dermal layers, eliciting robust mucosal and systemic immunity. Compared with conventional liquid formulations, incorporating antigens into the polymer matrix improves stability of thermosensitive antigens, potentially enabling cold-chain-independent storage. Microneedle technology also enables minimally invasive and convenient vaccination, enhancing accessibility and patient compliance for large-scale immunization.
Application Scenarios of Synthetic Polymers in Vaccine and Antigen Delivery
As vaccine technologies diversify, synthetic polymers provide highly customizable physical and chemical properties to enable targeted delivery strategies for various antigen types, from conventional proteins to advanced nucleic acids.
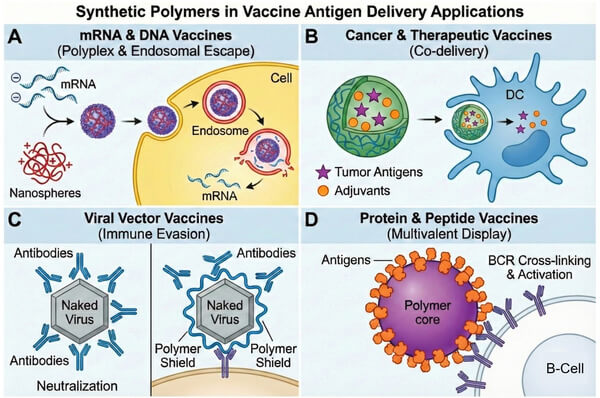 Fig. 3. Key strategies: mRNA polyplexes, cancer co-delivery, stealth viral vectors, and multivalent antigen display (BOC Sciences Authorized).
Fig. 3. Key strategies: mRNA polyplexes, cancer co-delivery, stealth viral vectors, and multivalent antigen display (BOC Sciences Authorized).
Synthetic Polymers for Protein and Subunit Vaccines
Subunit protein vaccines are safe but often poorly immunogenic due to the lack of pathogen-associated molecular patterns (PAMPs). Synthetic polymer particles can display protein antigens at high density on their surface using multivalent display, mimicking viral particle structures to enhance B-cell receptor cross-linking and induce stronger humoral immunity. Additionally, polymer carriers can co-encapsulate TLR agonists (e.g., CpG ODN) with protein antigens, ensuring spatiotemporal synchronization within the same APC, avoiding systemic toxicity from free adjuvants and significantly enhancing antibody titers and affinity.
Synthetic Polymers for Peptide Vaccines
Peptide antigens are small and prone to enzymatic degradation, resulting in very short in vivo half-life and weak immune memory when injected directly. Covalent conjugation of peptides to high-molecular-weight synthetic polymer backbones or self-assembly of amphiphilic polymers into nanofibers can significantly increase hydrodynamic radius, preventing rapid renal clearance. pH-sensitive polymer carriers can exploit the "proton sponge effect" to promote endosomal escape of peptides into the cytosol, enabling processing via MHC-I pathways, which is critical for inducing CD8⁺ cytotoxic T lymphocyte (CTL) responses against intracellular pathogens or tumor cells.
Synthetic Polymers for mRNA and DNA Vaccines
The efficacy of nucleic acid vaccines relies entirely on delivery systems that protect their integrity and ensure efficient intracellular transfection. Cationic synthetic polymers (e.g., poly(β-amino esters) [PBAEs] or modified PEI) can electrostatically condense mRNA or DNA into stable polyplexes, preventing nuclease degradation and promoting cellular uptake. Compared to lipid nanoparticles (LNPs), polymer carriers typically offer superior lyophilization stability and chemical modification flexibility. Current research focuses on developing polymers with ionizable and biodegradable properties to reduce cytotoxicity and improve in vivo transfection efficiency.
Synthetic Polymers for Cancer and Therapeutic Vaccines
Therapeutic cancer vaccines face the challenge of overcoming tumor-induced immunosuppressive microenvironments. Smart synthetic polymers are designed to co-deliver tumor-associated antigens (TAAs/TSAs) with immune checkpoint inhibitors (e.g., anti-PD-1 antibodies) or immunomodulators (e.g., STING agonists) to tumor-draining lymph nodes, synergistically activating effector T cells. Polymers responsive to the acidic tumor microenvironment enable site-specific release, enhancing tumor-infiltrating lymphocyte (TIL) activity and reversing immunosuppression to achieve an "in situ vaccine" effect.
Synthetic Polymers for Viral Vector Vaccines
Although viral vectors are inherently efficient at transduction, pre-existing neutralizing antibodies in humans often limit delivery efficiency and repeated dosing. Surface chemical modification or physical coating of viral vectors (e.g., adenovirus) with "stealth" polymers such as PEG or zwitterionic polymers can shield viral capsid epitopes, helping the vector evade immune recognition and clearance. This polymer-virus hybrid strategy prolongs circulation time and enables functional ligand attachment to alter viral tropism, achieving targeted transduction of specific tissues or cell types.
Design and Optimization Strategies for Polymer-Based Antigen Delivery
Developing clinically viable vaccine formulations requires systematic optimization of polymer structures and administration strategies, necessitating a deep understanding of materials chemistry and immunology.
Key Design Criteria for Antigen Delivery Polymers
- Biocompatibility and biodegradability: Carrier materials and their degradation products must be non-toxic, non-immunogenic (unless serving as adjuvants), and metabolizable for safe elimination.
- Loading capacity: Polymers must achieve high antigen loading through electrostatic adsorption, hydrophobic interactions, or covalent conjugation.
- Size and surface charge: Nanoscale dimensions (typically 20–200 nm) favor lymphatic drainage; moderate positive charges facilitate interaction with negatively charged cell membranes, while excessive charge may induce cytotoxicity.
Optimization of Polymer Structure
- Molecular Weight Optimization: Polymer molecular weight directly influences degradation rate and renal clearance. For carriers requiring prolonged circulation, polymers above the renal filtration threshold (~30–50 kDa) are typically chosen; for rapidly cleared carriers, molecular weight is controlled or cleavable bonds are introduced.
- Charge Density Optimization: Zeta potential affects colloidal stability and cellular interactions. Adjusting the proportion of cationic monomers helps balance transfection efficiency and cytotoxicity, with an ideal Zeta potential usually between +10 and +30 mV.
- Hydrophilicity/Hydrophobicity Optimization (HLB value): Tuning the hydrophilic-lipophilic balance of amphiphilic polymers determines micelle morphology (spherical, rod-like, vesicular) and critical micelle concentration (CMC), affecting antigen loading stability and release behavior.
- Adjuvant Properties of Polymers: Incorporating specific functional groups (e.g., mannose, imidazole) into the polymer backbone imparts intrinsic immune-stimulatory capacity, enabling integrated "carrier–adjuvant" design.
Optimizing Administration Routes
- Intradermal Administration: Polymer microneedles or nanoparticles precisely target the epidermis and dermis, rich in Langerhans cells and dermal dendritic cells, significantly enhancing antigen presentation efficiency and enabling dose sparing.
- Subcutaneous Administration: Suitable for polymer microspheres or in situ hydrogels, this route forms antigen depots in subcutaneous tissue, slowly draining to lymph nodes and sustaining germinal center stimulation.
- Intramuscular Injection: The most conventional route; large polymer particles form local depots for sustained antigen release, while cationic polymer nanocarriers protect unstable antigens (e.g., mRNA) for efficient uptake and expression by muscle cells.
- Intravenous Injection: Rarely used for prophylactic vaccines, intravenous polymer nanovaccines achieve systemic distribution and are preferentially captured by marginal zone B cells and dendritic cells in the spleen, often employed for therapeutic cancer vaccines to induce systemic T-cell responses.
- Mucosal Administration: Polymers with mucoadhesive or permeation-enhancing modifications (e.g., chitosan derivatives) overcome mucosal barriers and induce antigen-specific secretory IgA (sIgA), establishing a first line of defense on respiratory or gastrointestinal mucosa.
- Intranodal Injection: Using ultrasound guidance, ultrapure polymer nanovaccines are injected directly into lymph nodes, bypassing peripheral losses and non-specific clearance, maximizing exposure to naive T cells and triggering rapid, potent immune responses.
Services
Polymer Development Platform for Vaccine and Antigen Delivery at BOC Sciences
BOC Sciences provides a one-stop solution from polymer material screening and functional modification to vaccine/antigen delivery system development. Our platform supports custom polymer design to improve vaccine stability, delivery efficiency, and immunogenicity, offering reliable support for both research and industrial applications.

Custom Polymer Screening for Antigen Delivery Applications
- Select optimal polymer carriers based on antigen size, charge, and stability.
- Assess polymer biocompatibility and immune activation potential.
- High-throughput screening across multiple polymer types (PLGA, PEG-PLGA, PEI, PBAE, etc.).
- Optimize polymer–antigen binding efficiency and complex stability.

Polymer Modification and Functionalization for Vaccine Systems
- PEGylation, glycosylation, or peptide modification to extend circulation time and reduce immune clearance.
- Introduction of pH-, temperature-, or enzyme-responsive units for controlled intracellular release.
- Surface modifications to enhance self-assembly stability and nanoparticle formation.
- Conjugation of targeting ligands to improve delivery to specific cells or tissues.

Polymer–Antigen Compatibility and Formulation Optimization
- Evaluate polymer–antigen complex stability.
- Measure loading efficiency, protective performance, and immunological activity.
- Optimize particle size, charge, and distribution.
- Optimize formulation storage conditions and verify long-term stability.

Polymer-Based Drug Delivery System Development
- Construct polymer–antigen nanoparticles, micelles, and complex systems.
- Support mRNA, protein, and peptide vaccines.
- Optimize delivery efficiency, endosomal escape, and targeting.
- Evaluate immunogenicity and expression levels both in vitro and in vivo.
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
What are synthetic polymers used for in antigen delivery?
Synthetic polymers serve dual roles in vaccine development as multifunctional carriers and immune adjuvants. They act as physical scaffolds to efficiently load proteins, peptides, or nucleic acids, protecting them from degradation in vivo. Surface chemical modifications enable targeted delivery to lymph nodes or antigen-presenting cells (APCs). Additionally, some functionalized polymers possess intrinsic immune-stimulatory properties, synergistically activating the innate immune system and enhancing overall vaccine immunogenicity and translation efficiency.
How do polymers improve antigen stability in vaccines?
Synthetic polymers form physical structures such as microspheres, nanocapsules, or dense complexes to shield unstable antigens, protecting them from nuclease and protease degradation in serum. Polymer matrices provide localized buffering to maintain pH stability and prevent thermal aggregation or conformational denaturation. This comprehensive protection is critical for maintaining the bioactivity of labile antigens, such as mRNA, during manufacturing, storage, and circulation in vivo.
Are synthetic polymer vaccine carriers safe?
Safety is a core consideration for clinical translation. Widely used polymers such as PLGA, PLA, PCL, and PEG exhibit excellent biocompatibility, with several materials FDA-approved. These biodegradable polymers are ultimately metabolized into non-toxic endogenous small molecules like lactic acid and glycolic acid, eliminating long-term accumulation risk. For potentially cytotoxic cationic polymers, strategies such as PEGylation or introducing cleavable bonds are used to maintain delivery efficiency while significantly improving biocompatibility.
Can polymer delivery systems enhance immune response?
Yes, polymers can enhance immune response by providing controlled release, targeted delivery, and adjuvant effects. By designing polymer properties such as charge, hydrophobicity, or particle size, you can influence antigen uptake by dendritic cells and macrophages. This results in stronger humoral and cellular immune responses, supporting more effective vaccination strategies.
What types of polymers are suitable for vaccine delivery?
You can use a variety of polymers, including biodegradable options like PLGA, PLA, PCL, and PEGylated polymers, as well as natural polymers like chitosan and alginate. Selection depends on the antigen type, delivery route, and desired release kinetics. Your customization allows control over particle size, degradation rate, and surface functionalization to meet specific vaccine requirements.

 Fig. 1. Polymer encapsulation protects antigens from enzyme degradation and facilitates uptake by antigen-presenting cells (BOC Sciences Authorized).
Fig. 1. Polymer encapsulation protects antigens from enzyme degradation and facilitates uptake by antigen-presenting cells (BOC Sciences Authorized). Fig. 2. Structural comparison of common polymer delivery vehicles: nanoparticles, micelles, hydrogels and microneedles (BOC Sciences Authorized).
Fig. 2. Structural comparison of common polymer delivery vehicles: nanoparticles, micelles, hydrogels and microneedles (BOC Sciences Authorized). Fig. 3. Key strategies: mRNA polyplexes, cancer co-delivery, stealth viral vectors, and multivalent antigen display (BOC Sciences Authorized).
Fig. 3. Key strategies: mRNA polyplexes, cancer co-delivery, stealth viral vectors, and multivalent antigen display (BOC Sciences Authorized).