Conventional Micelles vs. Polymeric Micelles for Drug Delivery: Choosing the Right Carrier
Micellar nanostructures have become important tools for improving the formulation of hydrophobic or poorly soluble therapeutic molecules. By exploiting the self-assembly of amphiphilic molecules in aqueous environments, micelles create nanoscale compartments capable of solubilizing compounds that otherwise exhibit limited bioavailability in water-based systems. Among the various micellar systems studied in pharmaceutical research, conventional surfactant micelles and polymeric micelles represent two widely investigated carrier platforms. Although both systems rely on amphiphilicity-driven self-assembly, their physicochemical characteristics, stability, and drug delivery performance differ substantially. Understanding these differences is essential when selecting an appropriate carrier for specific drug molecules and formulation objectives.
Resources
Introduction to Micellar Drug Delivery Systems
Micellar drug delivery systems are nanoscale assemblies formed by amphiphilic molecules in aqueous environments. Their ability to encapsulate hydrophobic compounds within a core region while maintaining colloidal stability in aqueous media has made them attractive platforms in pharmaceutical formulation and nanomedicine research. Advances in polymer chemistry, nanotechnology, and molecular engineering have significantly expanded the structural diversity and functionality of micellar systems.
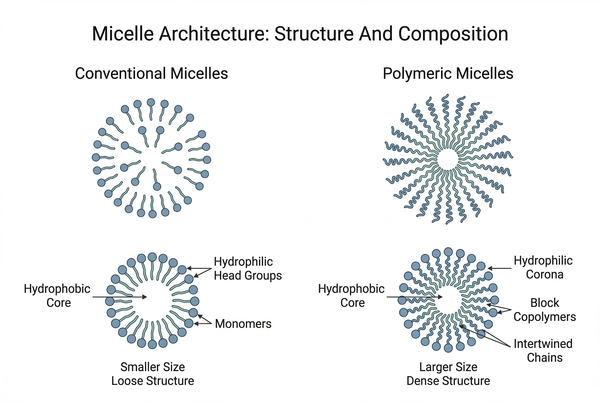 Fig. 1. Structural comparison between conventional micelles and polymeric micelle architectures (BOC Sciences Authorized).
Fig. 1. Structural comparison between conventional micelles and polymeric micelle architectures (BOC Sciences Authorized).
What Are Micelles in Drug Delivery?
Micelles are self-assembled colloidal structures typically formed when amphiphilic molecules are dispersed in water above a threshold concentration. These molecules contain hydrophilic segments that interact with water and hydrophobic segments that avoid aqueous environments. When the concentration of amphiphiles reaches a specific level, hydrophobic interactions drive the aggregation of the hydrophobic domains into a core, while hydrophilic chains extend outward toward the surrounding solvent. In drug delivery research, micelles function as nanoscale carriers that encapsulate poorly water-soluble molecules within their hydrophobic core. The hydrophilic shell stabilizes the structure in aqueous media, enabling dispersion, transport, and controlled interaction with biological environments.
Why Micelles Are Used for Poorly Soluble Drugs?
A large proportion of small-molecule drug candidates exhibit low aqueous solubility, which can significantly limit their formulation options and bioavailability. Micellar systems address this limitation by providing a hydrophobic microenvironment capable of solubilizing such compounds. Encapsulation within micelles enhances apparent solubility, protects labile molecules from degradation, and can influence drug distribution behavior. In addition, micelles may enable the incorporation of multiple functional elements, such as targeting ligands or stimuli-responsive components, which further improve delivery performance.
Key Factors Influencing Micelle-Based Drug Delivery Performance
The effectiveness of micelle-based drug delivery systems depends on a range of physicochemical and structural parameters that influence micelle formation, stability, drug loading, and release behavior. These factors determine whether a micellar carrier can maintain structural integrity while efficiently transporting hydrophobic molecules in aqueous environments. Key factors include the following:
- Critical Micelle Concentration (CMC): The critical micelle concentration defines the minimum concentration of amphiphilic molecules required for micelle formation. Systems with lower CMC values generally exhibit improved structural stability because micelles are less likely to dissociate upon dilution. Polymeric micelles typically possess significantly lower CMC values than conventional surfactant micelles, which contributes to enhanced stability in dilute environments.
- Amphiphilic Molecular Architecture: The structural design of the amphiphilic molecule strongly affects micelle formation and stability. Parameters such as block length ratio, molecular weight, and hydrophilic–hydrophobic balance determine the size, morphology, and stability of the resulting micelles. Carefully engineered amphiphilic block copolymers can produce micelles with controlled core dimensions and enhanced drug compatibility.
- Hydrophobic Core Compatibility with Drug Molecules: Efficient drug encapsulation depends on the chemical compatibility between the hydrophobic core of the micelle and the drug molecule. Strong hydrophobic interactions, van der Waals forces, and specific molecular affinities improve drug loading capacity and reduce premature leakage.
- Micelle Size and Particle Size Distribution: The nanoscale dimensions of micelles influence their dispersion behavior and interaction with biological systems. Particle size distribution also affects colloidal stability, diffusion properties, and overall delivery performance. Uniform and well-controlled particle sizes are typically preferred for reproducible formulation outcomes.
- Drug Loading Capacity and Encapsulation Efficiency: Drug loading capacity refers to the amount of drug that can be incorporated into the micelle relative to the carrier material, while encapsulation efficiency measures the fraction of drug successfully retained within the micelle. These parameters depend on polymer composition, micelle preparation methods, and drug–polymer interactions.
- Thermodynamic and Kinetic Stability: Micelles exist in dynamic equilibrium with individual amphiphilic molecules in solution. Thermodynamic stability determines the ability of micelles to remain assembled under equilibrium conditions, whereas kinetic stability relates to how rapidly the structure dissociates. Polymeric micelles often demonstrate enhanced kinetic stability due to stronger intermolecular interactions.
- Drug Release Mechanisms: The mechanism by which drugs are released from micelles plays a critical role in delivery performance. Release may occur through diffusion from the core, micelle disassembly, polymer degradation, or environmental stimuli. Controlled release behavior is typically achieved through careful selection of polymer composition and core properties.
- Surface Properties and Hydrophilic Corona Composition: The hydrophilic outer layer of micelles stabilizes the structure in aqueous media and influences interactions with surrounding biological components. Surface properties such as charge, steric stabilization, and hydration layers affect micelle aggregation behavior and dispersion stability.
- Environmental Conditions: External factors such as pH, ionic strength, temperature, and the presence of biomolecules can influence micelle stability and drug retention. Designing micellar systems that remain stable under varying environmental conditions is essential for reliable drug delivery performance.
Conventional Micelles: Structure, Formation, and Characteristics
Conventional micelles are nanoscale aggregates formed by low–molecular weight surfactants in aqueous environments. These systems represent one of the earliest and most widely studied colloidal carriers for improving the solubility and dispersion of hydrophobic compounds. In aqueous media, amphiphilic surfactant molecules spontaneously self-assemble into micellar structures once the concentration exceeds a specific threshold. This process is driven primarily by hydrophobic interactions, which minimize unfavorable contact between nonpolar molecular segments and water molecules. From a formulation perspective, conventional micelles provide a relatively simple approach for solubilizing poorly water-soluble molecules. Their formation requires minimal processing and occurs under mild conditions without the need for complex polymer synthesis. As a result, surfactant-based micellar systems have long been explored in pharmaceutical, cosmetic, and biochemical research as solubilizing carriers and dispersion stabilizers.
Molecular Structure of Conventional Surfactant Micelles
The molecular architecture of conventional micelles is determined by the amphiphilic nature of surfactant molecules. Each surfactant molecule contains a hydrophilic head group and a hydrophobic tail, typically composed of a hydrocarbon chain. When dispersed in water, the hydrophobic segments tend to avoid contact with the surrounding solvent, while the hydrophilic head groups interact favorably with water molecules. As the surfactant concentration increases, these molecules organize into spherical aggregates in which the hydrophobic tails orient inward to form a nonpolar core, while the hydrophilic head groups face outward toward the aqueous environment. This arrangement reduces the overall free energy of the system by shielding hydrophobic segments from water.
Although spherical micelles are the most common structure observed in dilute solutions, surfactant assemblies can also adopt cylindrical, rod-like, or lamellar structures depending on factors such as surfactant concentration, temperature, ionic strength, and molecular packing constraints. The shape and size of micelles are often described by the packing parameter, which relates the volume and length of the hydrophobic tail to the effective surface area of the hydrophilic head group.
Critical Micelle Concentration (CMC) and Micelle Stability
A defining characteristic of conventional micelles is the critical micelle concentration. This parameter represents the minimum surfactant concentration required for spontaneous micelle formation in solution. Below the CMC, surfactant molecules exist primarily as individual monomers dispersed in the solvent. Once the concentration exceeds this threshold, additional surfactant molecules preferentially assemble into micelles rather than remaining as free monomers. The CMC is influenced by several factors, including the length of the hydrophobic tail, the structure of the hydrophilic head group, temperature, and the presence of electrolytes in solution. Surfactants with longer hydrophobic chains generally exhibit lower CMC values because stronger hydrophobic interactions promote aggregation.
Conventional surfactant micelles typically have relatively high CMC values compared with polymeric micelles. As a result, dilution of the solution can cause micelles to dissociate back into monomers. This dynamic equilibrium between monomers and assembled micelles can lead to structural instability under conditions where the surfactant concentration falls below the CMC.
Common Surfactants Used in Conventional Micelles
A variety of surfactants have been utilized to form conventional micellar systems, and they are generally categorized according to the charge of their hydrophilic head groups. Nonionic surfactants are widely used due to their relatively low toxicity and good compatibility with many drug molecules. Examples include polyoxyethylene-based surfactants and polysorbates. These surfactants form micelles through hydrophobic interactions while maintaining neutral surface charge. Anionic surfactants, such as sulfate or carboxylate derivatives, carry a negatively charged head group. These molecules can generate micelles with strong electrostatic interactions, although their compatibility with certain biological systems may be limited.
Cationic surfactants contain positively charged head groups and are often used in specialized applications involving electrostatic interactions with negatively charged molecules. Zwitterionic surfactants contain both positive and negative charges within the same molecule, offering unique interfacial properties and improved stability in certain environments. The selection of an appropriate surfactant depends on the chemical characteristics of the drug molecule, desired micelle size, solubilization efficiency, and compatibility with the intended formulation environment.
Advantages and Limitations of Conventional Micellar Systems
Conventional micelles offer several practical advantages for drug formulation research. Their self-assembly occurs rapidly and spontaneously under suitable conditions, eliminating the need for complex synthesis procedures. Surfactant-based micelles are also capable of significantly improving the apparent solubility of hydrophobic compounds by incorporating them into the hydrophobic core. Another advantage is the relatively small size of micelles, typically ranging from a few nanometers to several tens of nanometers. This nanoscale dimension enables efficient dispersion in aqueous environments and can facilitate interactions with biological systems.
Despite these advantages, conventional micellar systems also present notable limitations. Their relatively high critical micelle concentrations mean that micelles may dissociate upon dilution, which can lead to instability and premature release of encapsulated molecules. In addition, the small size of the hydrophobic core can limit drug loading capacity for larger or highly hydrophobic compounds. Furthermore, the dynamic equilibrium between micelles and free surfactant molecules can affect formulation stability and reproducibility. These limitations have motivated the development of alternative systems such as polymeric micelles, which offer enhanced stability and greater structural tunability for advanced drug delivery applications.
Polymeric Micelles: Design and Functional Architecture
Polymeric micelles represent an advanced class of nanostructured carriers formed through the self-assembly of amphiphilic block copolymers in aqueous environments. Compared with conventional surfactant micelles, polymeric micelles exhibit significantly improved structural stability, lower critical micelle concentrations, and greater flexibility in molecular design. These characteristics have made them an important platform in modern drug delivery research, particularly for solubilizing hydrophobic compounds and constructing multifunctional nanocarriers.
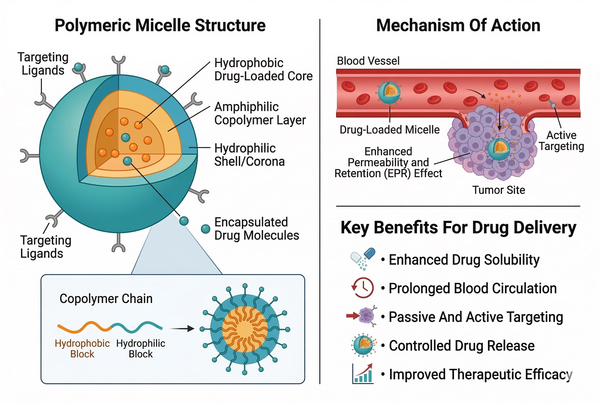 Fig. 2. Overview of polymeric micelle structure, targeting mechanism, and delivery benefits (BOC Sciences Authorized).
Fig. 2. Overview of polymeric micelle structure, targeting mechanism, and delivery benefits (BOC Sciences Authorized).
Amphiphilic Block Copolymers and Self-Assembly Mechanisms
Polymeric micelles are formed from amphiphilic block copolymers that contain chemically distinct hydrophilic and hydrophobic segments. When these copolymers are dispersed in water above a certain concentration, the hydrophobic blocks aggregate to minimize unfavorable interactions with water molecules. At the same time, the hydrophilic blocks remain solvated in the surrounding aqueous environment. This process leads to the spontaneous formation of nanoscale aggregates in which the hydrophobic polymer segments form the interior core and the hydrophilic chains extend outward to form a protective corona. The self-assembly process is governed by thermodynamic factors, including hydrophobic interactions, polymer chain mobility, and entropic contributions associated with solvent interactions. By carefully adjusting these parameters, researchers can control micelle size, morphology, and stability. Typical polymeric micelles have diameters ranging from approximately 10 to 100 nanometers, which allows them to remain well dispersed in aqueous environments.
Core–Shell Structure and Drug Encapsulation Mechanisms
A defining structural feature of polymeric micelles is the core–shell architecture. The hydrophobic core forms through the aggregation of nonpolar polymer segments and provides a microenvironment capable of incorporating hydrophobic molecules. The hydrophilic corona, often composed of water-soluble polymer chains, stabilizes the nanostructure through steric repulsion and hydration. Drug encapsulation within polymeric micelles generally occurs through noncovalent interactions. Hydrophobic interactions between the drug molecule and the core-forming polymer segments represent the primary driving force for incorporation. Additional interactions such as van der Waals forces, hydrogen bonding, and π–π stacking may further stabilize the drug within the micellar core depending on the chemical structure of the encapsulated compound. The strength of these interactions influences both drug loading capacity and retention stability. Polymers with core segments that exhibit strong chemical compatibility with the drug molecule often demonstrate higher loading efficiency and reduced premature release. As a result, selecting appropriate core-forming polymers is a critical step in micelle design.
Common Polymers Used for Polymeric Micelle Formation
A wide variety of amphiphilic block copolymers have been explored for polymeric micelle formation. In many systems, the hydrophilic segment is composed of polyethylene glycol due to its excellent water solubility and ability to form highly hydrated surface layers. The presence of polyethylene glycol chains in the corona also contributes to steric stabilization, which helps prevent aggregation in aqueous media. The hydrophobic segments of the copolymer typically consist of biodegradable or hydrophobic polymers capable of forming a stable core. Common examples include polycaprolactone, polylactic acid, polylactic-co-glycolic acid, polyaspartate derivatives, and polyesters. These polymers provide hydrophobic environments suitable for encapsulating poorly water-soluble compounds while also allowing for tunable degradation properties. Other polymer systems may incorporate polyamino acids, polycarbonates, or polyethers as core-forming segments. By combining different polymer blocks, researchers can generate micelles with diverse physicochemical characteristics tailored for specific formulation requirements.
Stimuli-Responsive and Functional Polymeric Micelles
One of the major advantages of polymeric micelles is the ability to introduce functional elements that enable responsive or targeted behavior. Modern polymer synthesis techniques allow the incorporation of responsive linkages or functional groups into polymer backbones, enabling micelles to respond to environmental triggers. Stimuli-responsive micelles may undergo structural changes in response to factors such as pH variation, temperature shifts, redox conditions, or enzymatic activity. These environmental triggers can induce changes in polymer solubility, micelle stability, or core structure, which in turn influence drug release behavior. Functional polymeric micelles may also include targeting ligands, imaging probes, or reactive groups on the micelle surface. Surface modification strategies enable interactions with specific biological molecules or cellular structures, thereby expanding the versatility of micellar nanocarriers in advanced drug delivery research. Through careful molecular design, polymeric micelles can therefore be engineered not only as solubilizing carriers but also as multifunctional nanoplatforms capable of controlled drug transport, environmental responsiveness, and molecular targeting.
Looking for Custom Polymer Micelles?
From amphiphilic block copolymers to stimuli-responsive systems, we offer tailored synthesis and formulation solutions for drug delivery research.
Conventional Micelles vs. Polymeric Micelles: Key Differences
Although conventional micelles and polymeric micelles are both formed through the self-assembly of amphiphilic molecules in aqueous environments, their structural organization, physicochemical stability, and functional capabilities differ substantially. These differences arise primarily from the molecular scale of the building blocks used to form the micelles. Understanding the fundamental distinctions between these two systems is essential when selecting an appropriate carrier platform for drug solubilization and nanocarrier development. The following comparison highlights several key parameters that differentiate conventional surfactant micelles from polymeric micelles.
| Comparison Aspect | Conventional Micelles | Polymeric Micelles |
|---|
| Building Blocks | Formed from low–molecular weight surfactants with distinct hydrophilic head groups and hydrophobic tails. | Constructed from amphiphilic block copolymers composed of hydrophilic and hydrophobic polymer segments. |
| Molecular Weight of Amphiphiles | Typically low molecular weight molecules with relatively small hydrophobic domains. | High molecular weight polymers with extended chain structures that enable more complex assembly behavior. |
| Self-Assembly Driving Forces | Primarily driven by hydrophobic interactions among surfactant tails. | Driven by hydrophobic interactions along with additional stabilization from polymer chain entanglement and steric effects. |
| Critical Micelle Concentration | Generally high critical micelle concentrations, making micelles more sensitive to dilution. | Significantly lower critical micelle concentrations, resulting in improved structural stability even under diluted conditions. |
| Micelle Stability | Dynamic equilibrium between micelles and surfactant monomers may lead to dissociation. | Enhanced kinetic and thermodynamic stability due to stronger polymer–polymer interactions. |
| Core Size and Drug Loading Capacity | Hydrophobic core is relatively small, which may limit the loading of large or highly hydrophobic molecules. | Larger hydrophobic cores allow higher drug loading capacity and improved encapsulation efficiency. |
| Structural Tunability | Limited ability to modify structure beyond surfactant type and concentration. | Highly tunable through polymer design, including block length, architecture, and functional group incorporation. |
| Drug Release Behavior | Drug release may occur rapidly due to micelle dissociation or diffusion from a small core. | Release behavior can be tuned through polymer composition, core structure, and responsive linkages. |
| Surface Functionalization | Difficult to incorporate targeting ligands or functional groups due to the small molecular structure. | Surface modification is feasible through polymer chemistry, enabling ligand attachment and functional design. |
| Particle Stability in Complex Environments | Stability may decrease in the presence of salts, proteins, or dilution. | Polymer corona provides steric stabilization that helps maintain dispersion stability. |
| Formulation Complexity | Relatively simple formulation and rapid self-assembly in aqueous solution. | Requires controlled polymer synthesis and careful formulation optimization. |
Applications in Pharmaceutical and Biomedical Research
Micellar nanocarriers have attracted substantial attention in pharmaceutical and biomedical research due to their ability to solubilize hydrophobic compounds, protect sensitive molecules, and enable tunable nanoscale delivery platforms. Both conventional and polymeric micelles have been explored for a variety of research applications, particularly in situations where the physicochemical properties of a molecule limit its direct formulation in aqueous environments. With advances in polymer chemistry and nanomaterials engineering, polymeric micelles have become particularly important in modern drug delivery research. Their tunable molecular architecture allows researchers to control particle size, surface properties, and encapsulation behavior, enabling the development of multifunctional nanocarriers for a wide range of biomedical applications.
Polymeric Micelles for Cancer Therapy
Polymeric micelles have been widely investigated as nanocarriers for anticancer compounds, especially hydrophobic chemotherapeutic molecules that exhibit poor aqueous solubility. Many anticancer agents contain aromatic or lipophilic structural elements that limit their ability to dissolve in physiological media. Encapsulation within micellar cores can significantly improve their apparent solubility and facilitate dispersion in aqueous systems. In research environments, polymeric micelles are often designed to carry cytotoxic molecules within hydrophobic polymer domains, thereby protecting the drug during transport and enabling controlled release under specific conditions. The nanoscale size of polymeric micelles also enables them to remain well dispersed in aqueous formulations while maintaining a relatively uniform particle distribution. Another important advantage is the ability to introduce functional components into the micelle structure. Surface-modified polymeric micelles can incorporate ligands, imaging probes, or responsive polymer segments, enabling more sophisticated carrier systems for oncology-related drug delivery research.
Micelle-Based Delivery of Peptides and Proteins
Peptides and proteins represent an important class of therapeutic molecules, but their formulation can be challenging due to limited stability, aggregation tendencies, or susceptibility to degradation. Micellar carriers have been explored as protective environments that can stabilize certain biomolecules and improve their dispersion in aqueous formulations. Polymeric micelles may provide a microenvironment that reduces aggregation and protects sensitive peptide structures from unfavorable interactions with the surrounding medium. In some cases, amphiphilic polymers can be engineered to interact with peptide molecules through electrostatic interactions, hydrogen bonding, or hydrophobic interactions. Additionally, micellar systems can help modulate the release behavior of peptide-based molecules by controlling the diffusion of encapsulated molecules from the micelle core. This controlled release capability can be useful in research settings where sustained or gradual delivery of peptide therapeutics is desired.
Delivery of Poorly Soluble Small-Molecule Drugs
A large proportion of small-molecule drug candidates identified during drug discovery exhibit limited aqueous solubility. This characteristic presents a major challenge during early formulation development, as poor solubility can restrict bioavailability and complicate experimental evaluation. Micellar carriers provide a practical strategy for addressing this challenge by incorporating hydrophobic compounds into the micelle core. The hydrophobic environment of the core allows these molecules to remain solubilized within an otherwise aqueous formulation. This capability is particularly valuable during preclinical formulation studies, where researchers often need to evaluate compounds that are not easily dissolved in conventional solvents. Polymeric micelles, in particular, offer improved solubilization capacity compared with conventional surfactant micelles due to their larger core volume and stronger hydrophobic interactions. These properties make them useful tools for screening and delivering poorly soluble drug candidates in pharmaceutical research.
Emerging Applications in Gene and Nucleic Acid Delivery
In addition to small-molecule drugs and peptides, micellar nanocarriers have also been explored for the delivery of nucleic acids such as DNA, messenger RNA, and small interfering RNA. These molecules present unique formulation challenges because they are highly charged, relatively large, and sensitive to degradation in biological environments. To address these challenges, specialized polymeric micelles have been developed that incorporate cationic or ionizable polymer segments capable of interacting with negatively charged nucleic acids. These electrostatic interactions allow the formation of polymer–nucleic acid complexes that can be stabilized within micellar structures. In some cases, hybrid micelle systems combine hydrophobic drug encapsulation with nucleic acid complexation, enabling co-delivery strategies for advanced therapeutic research. Ongoing developments in polymer chemistry and nanomaterial design continue to expand the potential applications of micellar carriers in nucleic acid delivery and related areas of biomedical research.
Design Considerations When Choosing the Right Micellar Carrier
Selecting an appropriate micellar carrier requires a systematic evaluation of both the physicochemical properties of the drug molecule and the structural characteristics of the carrier system. While micelles offer significant advantages for solubilizing hydrophobic compounds and stabilizing sensitive molecules, their performance is strongly influenced by the compatibility between the drug and the micelle-forming materials. In addition, factors such as carrier stability, release behavior, and formulation scalability must be carefully considered during the design phase.
Drug Physicochemical Properties and Compatibility
The physicochemical properties of the drug molecule represent one of the most important factors when designing micellar delivery systems. Parameters such as molecular weight, hydrophobicity, polarity, and the presence of functional groups determine how effectively a drug can interact with the micelle core. Highly hydrophobic compounds tend to exhibit strong affinity for the nonpolar core region of micelles, which facilitates efficient encapsulation. In contrast, molecules with moderate hydrophobicity may require careful selection of core-forming polymers or surfactants to achieve stable incorporation. Structural features such as aromatic rings, long hydrocarbon chains, or lipophilic substituents often enhance compatibility with hydrophobic polymer segments. Other considerations include drug stability in aqueous environments and potential interactions with the micelle-forming materials. Hydrogen bonding, π–π interactions, and electrostatic interactions can contribute to improved drug retention within the micelle core. Evaluating these molecular interactions is essential for achieving high drug loading capacity and stable encapsulation.
Selection of Surfactants vs. Amphiphilic Block Copolymers
Another critical design consideration is the choice of amphiphilic molecules used to construct the micellar carrier. Conventional micelles formed from low–molecular weight surfactants offer the advantage of simple preparation and rapid self-assembly in aqueous solutions. These systems can be effective for basic solubilization tasks and may be suitable for certain small hydrophobic molecules. However, surfactant micelles often exhibit relatively high critical micelle concentrations, which can result in structural instability when the formulation is diluted. In contrast, polymeric micelles formed from amphiphilic block copolymers generally possess significantly lower critical micelle concentrations and enhanced resistance to dissociation. Amphiphilic block copolymers also provide greater flexibility for structural design. By modifying polymer block lengths, molecular weight, and chemical composition, researchers can tailor the micelle size, core properties, and surface characteristics. This tunability makes polymeric micelles particularly attractive for advanced drug delivery research and nanocarrier engineering.
Stability Requirements for In Vivo Applications
Micellar carriers must maintain structural integrity under conditions that mimic physiological environments. Stability considerations include resistance to dilution, protection against aggregation, and the ability to retain encapsulated drug molecules during circulation. Dilution stability is particularly important because micellar systems may encounter highly diluted conditions after administration. Systems with high critical micelle concentrations may dissociate under such conditions, leading to premature drug release. Polymeric micelles with low critical micelle concentrations generally demonstrate improved stability under dilution. Additional stability factors include interactions with proteins, salts, and other biomolecules present in biological fluids. Surface characteristics of the micelle corona, including hydrophilicity and steric stabilization, play an important role in maintaining dispersion stability and minimizing unwanted interactions.
Scale-Up, Manufacturing, and Regulatory Considerations
Beyond molecular design and formulation performance, practical considerations related to manufacturing and scalability must also be taken into account when selecting a micellar carrier system. Laboratory-scale preparation methods must be adaptable to larger-scale production while maintaining consistent particle size distribution and drug loading efficiency. Polymer synthesis processes must also be reproducible and capable of producing materials with controlled molecular weight and narrow polydispersity. Variations in polymer composition or structure can significantly influence micelle formation and stability, making quality control an important aspect of carrier development. Additionally, the choice of raw materials and excipients should consider safety, purity, and regulatory acceptance. Surfactants and polymers used in micellar formulations must meet appropriate quality standards and demonstrate compatibility with pharmaceutical formulation requirements. Careful consideration of these factors during the design phase helps ensure that micellar carrier systems can be reliably produced and applied in advanced drug delivery research.
Services
Custom Polymer and Micelle Development Services from BOC Sciences
BOC Sciences has extensive expertise in functional monomer development, controlled polymerization, polymer material design, and nanocarrier research, providing professional custom polymer and micelle development services for academic institutions, biotechnology companies, and pharmaceutical organizations. Supported by advanced polymer synthesis platforms, nanocarrier formulation capabilities, and comprehensive analytical characterization facilities, we offer integrated solutions covering amphiphilic block copolymer design, polymeric micelle construction, drug loading optimization, micelle characterization, and process scale-up. By precisely controlling polymer structure, molecular weight, and hydrophilic–hydrophobic balance, BOC Sciences helps clients develop polymeric micelle nanocarriers with excellent stability, high drug loading capacity, and controlled release performance, accelerating the development of advanced drug delivery systems.

Custom Synthesis of Amphiphilic Block Copolymers
- Custom design and synthesis of amphiphilic block copolymers for polymeric micelle nanocarriers.
- Structural design and molecular weight control of PEG, PLA, PCL, and PLGA polymers.
- Support for controlled polymerization techniques, including RAFT, ATRP, and ring-opening polymerization (ROP).
- Development of functionalized polymers and multiblock copolymers.

Polymeric Micelle Formulation and Drug Loading Optimization
- Development of polymeric micelle preparation and self-assembly systems.
- Support for encapsulation of hydrophobic small-molecule drugs to enhance solubility.
- Optimization of drug loading capacity and encapsulation efficiency.
- Development of single-drug and combination drug loading systems.

Analytical Characterization of Micellar Nanocarriers
- Particle size distribution and nanostructure analysis.
- Determination of critical micelle concentration (CMC).
- Morphological characterization using techniques such as TEM and SEM.
- Evaluation of drug release behavior and formulation stability.

Scale-Up Production and Specialized Polymer Development
- Scale-up production of polymer materials from milligram to kilogram quantities.
- Custom production of high-purity functional polymers and monomers.
- Batch consistency control and quality analysis.
- Development of specialized polymers for drug delivery applications.
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What are the main differences between conventional and polymeric micelles?
Conventional micelles are formed from low–molecular weight surfactants and generally have relatively high critical micelle concentrations, which can make them sensitive to dilution. Polymeric micelles are assembled from amphiphilic block copolymers with much larger molecular structures. This difference results in improved stability, larger hydrophobic cores, and greater flexibility for structural design in polymeric micelles.
-
Which micelle system provides better stability and drug release control?
Polymeric micelles generally provide better structural stability and more controllable drug release behavior. Their amphiphilic block copolymer structure produces very low critical micelle concentrations and stronger intermolecular interactions within the micelle core. These features help maintain micelle integrity in diluted environments and allow the release profile to be tuned through polymer composition and architecture.
-
Can polymeric micelles deliver proteins, peptides, or nucleic acids?
Polymeric micelles can be engineered to accommodate a variety of biomolecules, including peptides, proteins, and nucleic acids. This is achieved by incorporating functional polymer segments that enable electrostatic interactions, hydrogen bonding, or hydrophobic interactions with the biomolecule. Such modifications allow the formation of stable complexes that help protect sensitive biomolecules in aqueous environments.
-
How to choose the right micelle system for specific drug therapeutics?
Selecting a suitable micelle system depends on the physicochemical properties of the drug molecule, including hydrophobicity, molecular size, and functional groups. Conventional micelles may be appropriate for simple solubilization tasks, while polymeric micelles are often preferred when higher stability, tunable drug release, or advanced carrier functionality is required.

 Fig. 1. Structural comparison between conventional micelles and polymeric micelle architectures (BOC Sciences Authorized).
Fig. 1. Structural comparison between conventional micelles and polymeric micelle architectures (BOC Sciences Authorized). Fig. 2. Overview of polymeric micelle structure, targeting mechanism, and delivery benefits (BOC Sciences Authorized).
Fig. 2. Overview of polymeric micelle structure, targeting mechanism, and delivery benefits (BOC Sciences Authorized).