Biodegradable Polymers for Drug Delivery: Materials, Design, and Controlled Release Strategies
As pharmaceutical innovation increasingly focuses on complex and poorly soluble active compounds, biodegradable polymers have emerged as essential materials for advanced drug delivery system (DDS) design. Rather than acting as passive carriers, these polymers actively regulate drug release, stability, and distribution by undergoing controlled degradation in physiological environments. Their ability to break down into non-toxic byproducts makes them particularly attractive for designing safe and tunable delivery platforms. By precisely engineering polymer composition, molecular weight, and architecture, researchers can tailor degradation kinetics and release profiles to match specific formulation requirements. Biodegradable polymers enable the development of delivery systems that improve solubility, protect sensitive molecules, and achieve sustained or stimuli-responsive release. As a result, they play a central role in modern formulation strategies for small molecules, peptides, proteins, and nucleic acids.
Resources
Overview of Biodegradable Polymer Systems for Drug Delivery
Biodegradable polymer systems for drug delivery are advanced material platforms designed to transport therapeutic agents in a controlled, safe, and efficient manner while naturally degrading into biocompatible byproducts within the body. These systems are typically constructed from polymers such as polylactic acid (PLA), polyglycolic acid (PGA), and their copolymers (PLGA), which have been extensively studied and validated for pharmaceutical applications. By enabling tunable degradation rates, drug release kinetics, and targeted delivery profiles, biodegradable polymers significantly improve drug bioavailability, reduce systemic toxicity, and enhance patient compliance. From an engineering and pharmaceutical perspective, biodegradable polymer-based carriers—including nanoparticles, microspheres, hydrogels, and implants—can be precisely tailored to meet specific therapeutic requirements. Factors such as polymer composition, molecular weight, crystallinity, and surface functionality play critical roles in dictating drug encapsulation efficiency and release behavior. Importantly, these systems eliminate the need for surgical removal after drug depletion, making them particularly valuable for long-term and implantable therapies.
Biodegradable polymers used in drug delivery can be broadly categorized based on their chemical structure, degradation mechanism, and origin. Each class offers distinct advantages in terms of release control, biocompatibility, and formulation flexibility. Selecting the appropriate polymer system is critical for achieving optimal performance in delivery design.
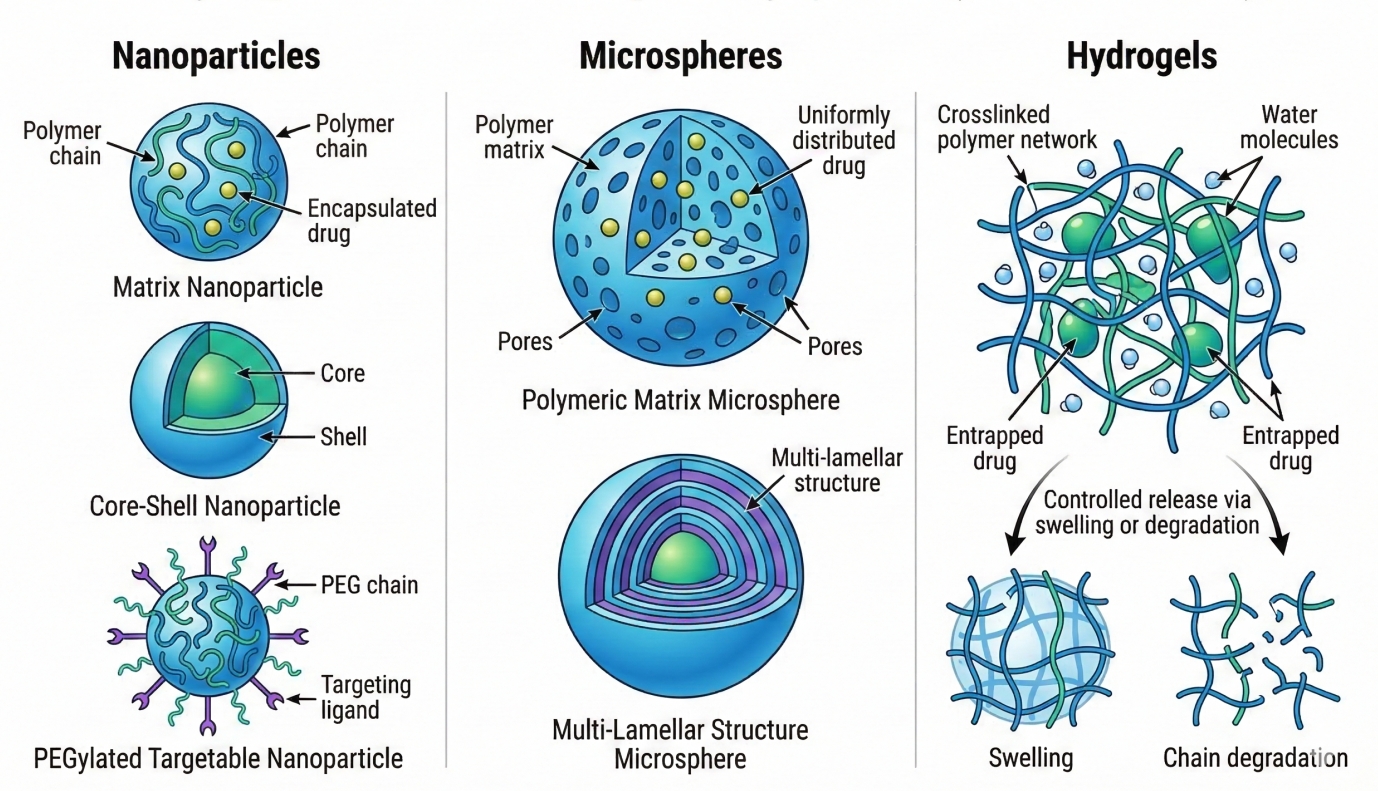 Fig. 1. Structural illustration of biodegradable polymer nanoparticles, microspheres, and hydrogels used in controlled drug delivery systems (BOC Sciences Authorized).
Fig. 1. Structural illustration of biodegradable polymer nanoparticles, microspheres, and hydrogels used in controlled drug delivery systems (BOC Sciences Authorized).
- Aliphatic Polyesters: This class includes widely used materials such as PLA, PLGA, and PCL. These polymers degrade primarily through hydrolysis of ester bonds, producing biocompatible metabolites. Their degradation rates can be finely tuned by adjusting monomer ratios, crystallinity, and molecular weight, making them highly versatile for sustained release formulations.
- Poly(ethylene glycol)-Based Copolymers: PEG is often incorporated into biodegradable systems to improve hydrophilicity and reduce protein adsorption. PEG-based block copolymers form self-assembled nanostructures such as micelles, enhancing solubility and enabling controlled release of hydrophobic drugs.
- Natural Biodegradable Polymers: Materials such as chitosan, alginate, and hyaluronic acid offer inherent biocompatibility and biofunctionality. Their enzymatic degradability and ability to interact with biological interfaces make them suitable for responsive and mucoadhesive delivery systems.
Major Challenges in Biodegradable Polymer-Based Delivery Systems
Despite their advantages, biodegradable polymers must overcome several formulation and material-related challenges to ensure reliable performance and reproducibility:
- Controlled Degradation Kinetics: Achieving predictable degradation profiles remains complex, as factors such as polymer crystallinity, molecular weight, and environmental pH significantly influence hydrolysis rates.
- Drug–Polymer Compatibility: Interactions between the polymer matrix and the active compound can affect drug stability, loading efficiency, and release behavior.
- Initial Burst Release: Rapid release of surface-associated drug molecules can compromise controlled delivery performance and requires careful formulation optimization.
- Scalability and Reproducibility: Manufacturing biodegradable polymer systems with consistent particle size, morphology, and drug loading remains a key challenge in industrial production.
- Environmental Sensitivity: Degradation behavior can vary depending on temperature, pH, and enzymatic conditions, necessitating robust design strategies.
Why Biodegradable Polymers Are Essential in Modern Drug Delivery Design
The use of biodegradable polymers is driven by their unique ability to combine material safety with precise control over drug release. These systems offer multiple functional advantages that are critical for advanced formulation development:
- Tunable Release Profiles: By adjusting polymer composition and structure, it is possible to design delivery systems with sustained, delayed, or stimuli-responsive release characteristics.
- Improved Drug Stability: Encapsulation within biodegradable matrices protects sensitive molecules from degradation and environmental stress.
- Enhanced Solubility: Amphiphilic polymer systems improve the dispersion of poorly water-soluble compounds, increasing formulation efficiency.
- Reduced Long-Term Accumulation: Complete degradation into non-toxic byproducts eliminates concerns related to material accumulation in biological systems.
Overall, biodegradable polymers provide a powerful platform for designing advanced drug delivery systems that balance performance, safety, and formulation flexibility.
Advanced Design Principles and Functional Mechanisms of Delivery Systems
Biodegradable polymer-based drug delivery systems represent a cornerstone of modern formulation science, offering unparalleled control over drug release kinetics, material degradation, and structural performance. Unlike inert carriers, these systems are inherently dynamic—undergoing predictable chemical or enzymatic breakdown that directly governs drug release behavior. This unique property enables the design of delivery platforms that integrate material degradation with controlled therapeutic exposure. The performance of biodegradable polymer systems is dictated by a combination of molecular-level parameters, including polymer composition, chain architecture, crystallinity, and molecular weight distribution. By precisely tuning these variables, researchers can engineer delivery systems with highly specific degradation timelines, diffusion characteristics, and mechanical stability. This level of control is critical for optimizing formulation performance across a wide range of drug modalities.
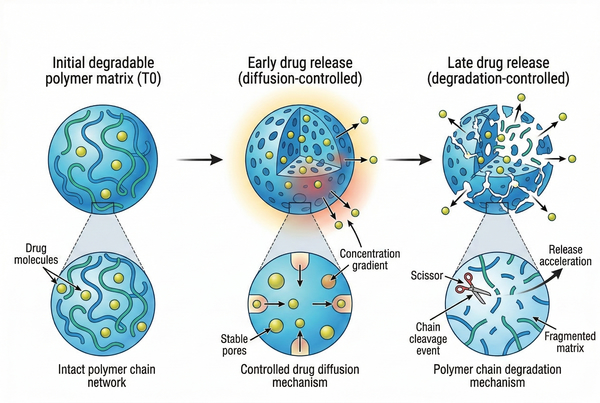 Fig. 2. Schematic representation of polymer chain degradation and diffusion-controlled drug release behavior (BOC Sciences Authorized).
Fig. 2. Schematic representation of polymer chain degradation and diffusion-controlled drug release behavior (BOC Sciences Authorized).
Functional Roles of Biodegradable Polymers in Drug Delivery Systems
In advanced drug delivery design, biodegradable polymers serve as multifunctional materials that actively regulate encapsulation, protection, transport, and release of active compounds. Their role extends far beyond structural support, influencing both physicochemical stability and kinetic behavior:
- Programmable Degradation-Driven Release: The hydrolysis of ester or amide bonds within polymers such as PLGA, PLA, and PCL leads to time-dependent chain scission. This degradation process progressively increases matrix porosity and facilitates controlled drug diffusion, enabling sustained and predictable release profiles.
- Microenvironment Regulation: Degradation byproducts (e.g., lactic acid, glycolic acid) can locally modulate pH within the polymer matrix. This internal microenvironment influences drug stability, solubility, and release kinetics, particularly for pH-sensitive compounds.
- Encapsulation Efficiency and Drug Protection: Biodegradable polymers provide a physical barrier that shields active molecules from hydrolytic, oxidative, or enzymatic degradation during formulation and storage.
- Interfacial and Surface Engineering: Functional groups along polymer backbones enable chemical modification, allowing the introduction of hydrophilic segments, targeting ligands, or stimuli-responsive moieties to further optimize delivery performance.
Classes of Biodegradable Polymers and Structure–Property Relationships
The selection of biodegradable polymers is fundamentally governed by their chemical structure and resulting physicochemical properties. Understanding structure–property relationships is essential for rational material design:
Aliphatic Polyesters (Hydrolytically Degradable):
- PLA (Polylactic Acid): Exhibits relatively high crystallinity and hydrophobicity, resulting in slower water penetration and prolonged degradation profiles.
- PLGA (Poly(lactic-co-glycolic acid)): The copolymer composition controls hydrophilicity and degradation rate; higher glycolide content accelerates hydrolysis due to increased water uptake.
- PCL (Polycaprolactone): Characterized by semi-crystalline structure and low glass transition temperature, leading to very slow degradation and long-term release capability.
Amphiphilic and PEG-Based Copolymers:
- Incorporation of PEG segments increases hydrophilicity, reduces protein adsorption, and promotes self-assembly into micellar or nanoparticulate systems.
- Block copolymer design enables phase separation between hydrophobic and hydrophilic domains, controlling drug loading and release pathways.
Natural Biopolymers (Enzymatically Degradable):
- Chitosan: Cationic nature enhances electrostatic interaction with negatively charged molecules and improves mucoadhesion.
- Alginate: Forms ionically crosslinked hydrogels, with release governed by swelling and diffusion mechanisms.
- Hyaluronic Acid: Degraded by hyaluronidase, enabling enzyme-responsive release behavior.
Advanced Architectures:
- Block Copolymers: Enable self-assembly into core–shell nanostructures with tunable drug partitioning.
- Crosslinked Networks (Hydrogels): Provide structural stability and allow diffusion-controlled release through mesh size regulation.
- Dendritic Polymers: Highly branched structures offering high loading capacity and precise functionalization.
Degradation Pathways and Release Kinetics Modeling
Drug release from biodegradable polymer systems is governed by the interplay between polymer degradation, water diffusion, and drug transport. A mechanistic understanding of these processes is essential for predictive formulation design:
- Hydrolytic Chain Scission: Ester bond cleavage reduces molecular weight over time, transitioning the polymer from a solid matrix to soluble oligomers.
- Autocatalytic Degradation: Accumulation of acidic degradation products can accelerate internal hydrolysis, leading to heterogeneous degradation behavior.
- Diffusion-Controlled Release (Higuchi Model): Early-stage drug release is dominated by diffusion through the polymer matrix.
- Degradation-Controlled Release: At later stages, polymer erosion governs drug release as structural integrity is lost.
- Swelling-Controlled Systems: In hydrogels, water uptake expands the network, facilitating drug diffusion.
- Stimuli-Responsive Systems: Polymers designed with pH-, redox-, or enzyme-sensitive linkages enable environment-triggered release.
Formulation Engineering Strategies and Processing Techniques
The translation of biodegradable polymers into functional drug delivery systems requires precise control over formulation parameters and processing methods:
- Emulsion-Based Techniques: Single and double emulsion methods are widely used for encapsulating hydrophobic and hydrophilic drugs into polymeric microspheres.
- Nanoprecipitation: Enables the formation of uniform nanoparticles with narrow size distribution through solvent displacement.
- Spray Drying: Produces dry polymeric particles suitable for scalable manufacturing and improved stability.
- Microfluidic Processing: Provides precise control over particle size and reproducibility for advanced nanocarrier fabrication.
- Crosslinking Strategies: Chemical or physical crosslinking controls hydrogel network density and release kinetics.
Looking for High-Performance Biodegradable Polymers?
At BOC Sciences, we provide tailored biodegradable polymer materials and formulation support to meet diverse drug delivery requirements. From polymer design to advanced delivery system development, our team delivers reliable and scalable solutions for your research and production needs.
Formulation Platforms Enabled by Biodegradable Polymers for Drug Delivery Systems
Biodegradable polymers enable the development of a wide range of formulation platforms by providing tunable structural, physicochemical, and degradation properties. Through rational material selection and processing techniques, these polymers can be engineered into delivery systems with precise control over particle size, morphology, drug loading, and release kinetics. Each formulation platform offers distinct advantages depending on the drug characteristics and desired release profile. Understanding the structural features and functional mechanisms of these systems is essential for selecting the most suitable delivery strategy.
Polymeric nanoparticles are among the most widely used biodegradable delivery systems due to their small size, high surface area, and tunable surface properties. These systems are typically fabricated using methods such as nanoprecipitation or emulsion-based techniques, allowing precise control over particle size distribution and morphology.
- Controlled Release Behavior: Drug release is governed by a combination of diffusion and polymer degradation, with smaller particle sizes favoring faster release kinetics.
- Surface Functionalization: Surface modification enables tuning of hydrophilicity, charge, and interaction with surrounding environments.
- Versatile Drug Loading: Suitable for encapsulating both hydrophobic and hydrophilic compounds through matrix or core–shell structures.
Microspheres and microcapsules provide larger-scale polymer matrices that are particularly suitable for long-term sustained release applications. These systems are commonly prepared via solvent evaporation or spray drying techniques, enabling high drug loading and controlled morphology.
- Extended Release Profiles: Larger particle size and reduced surface area slow drug diffusion, supporting prolonged release durations.
- High Encapsulation Efficiency: Particularly effective for hydrophobic drugs and macromolecules.
- Core–Shell Structures: Microcapsules with distinct core and shell layers allow more precise control over release kinetics.
Polymeric micelles are self-assembled nanostructures formed from amphiphilic block copolymers. They consist of a hydrophobic core and a hydrophilic shell, enabling efficient encapsulation of poorly soluble drugs.
- Enhanced Solubilization: Hydrophobic drugs are incorporated into the core, significantly improving apparent solubility.
- Dynamic Stability: Micelle formation depends on critical micelle concentration (CMC), which influences stability under dilution conditions.
- Stimuli-Responsive Behavior: Block copolymer design enables responsiveness to pH, temperature, or redox conditions.
Hydrogel-Based Delivery Systems
Polymeric Hydrogels are three-dimensional crosslinked polymer networks capable of retaining large amounts of water. Their highly hydrated structure makes them suitable for encapsulating sensitive biomolecules and enabling diffusion-controlled release.
- Diffusion-Controlled Release: Drug transport is governed by network mesh size and swelling behavior.
- Tunable Mechanical Properties: Crosslinking density and polymer composition control stiffness and degradation rate.
- Responsive Hydrogels: Systems can be designed to respond to environmental stimuli, enabling controlled release under specific conditions.
Nanofibers and Porous Scaffolds
Biodegradable polymers can also be processed into nanofibrous or porous structures using techniques such as electrospinning or phase separation. These architectures provide high surface area and interconnected porosity, enabling controlled drug loading and release.
- High Surface Area: Facilitates rapid drug diffusion and interaction with the surrounding environment.
- Structural Versatility: Fiber diameter, porosity, and alignment can be tuned to control release behavior.
- Multi-Drug Loading Capability: Enables incorporation of multiple agents within different structural domains.
Hybrid and Multi-Component Systems
Advanced formulation strategies increasingly involve hybrid systems that combine biodegradable polymers with other materials to enhance functionality and performance.
- Polymer–Lipid Hybrid Nanoparticles: Integrate polymer stability with lipid biocompatibility.
- Polymer–Inorganic Composites: Combine controlled degradation with structural robustness and functional properties.
- Layered or Core–Shell Architectures: Enable spatial separation of drugs and controlled sequential release.
Comparative Analysis of Biodegradable Polymer-Based Formulation Platforms
Biodegradable polymers can be engineered into a variety of formulation platforms, each offering distinct advantages in terms of drug loading, release kinetics, and structural properties. Selecting the appropriate formulation type is essential for achieving optimal delivery performance and aligning with specific drug characteristics. The following comparison highlights the key differences between major biodegradable polymer-based formulation systems, providing guidance for rational design and platform selection.
| Feature / Metric | Polymeric Nanoparticles | Microspheres / Microcapsules | Polymeric Micelles | Hydrogels |
|---|
| Typical Size Range | 50–300 nm | 1–100 μm | 10–100 nm | Bulk networks (macro- to microscale) |
| Drug Loading Mechanism | Matrix encapsulation or core–shell structures | Matrix dispersion or reservoir-type encapsulation | Hydrophobic core solubilization | Physical entrapment within crosslinked network |
| Release Mechanism | Diffusion + polymer degradation | Predominantly degradation-controlled with diffusion contribution | Diffusion and micelle dissociation | Diffusion through swollen network + degradation |
| Release Duration | Short to medium term (hours to weeks) | Medium to long term (days to months) | Rapid to short term (hours to days) | Tunable (hours to weeks depending on crosslinking density) |
| Suitable Drug Types | Small molecules, proteins, nucleic acids | Small molecules, peptides, proteins | Hydrophobic small molecules | Hydrophilic drugs, proteins, macromolecules |
| Encapsulation Efficiency | Moderate to high (depends on formulation method) | High, especially for hydrophobic compounds | Moderate, limited by core capacity | Moderate, dependent on network structure |
| Structural Tunability | High (size, surface properties, composition) | Moderate (size and polymer composition) | High at molecular level (block copolymer design) | Very high (crosslink density, mesh size, composition) |
| Stability Considerations | Sensitive to aggregation and surface interactions | High physical stability | Dependent on critical micelle concentration (CMC) | Sensitive to swelling and environmental conditions |
| Manufacturing Complexity | Moderate to high (nanoprecipitation, emulsification) | Moderate (emulsion solvent evaporation, spray drying) | Moderate (self-assembly processes) | Variable (crosslinking and gelation control required) |
| Key Advantages | Versatile platform with tunable size and release | Long-term sustained release and high drug loading | Excellent solubilization of hydrophobic drugs | Suitable for sensitive biomolecules and controlled diffusion |
| Key Limitations | Potential burst release and stability challenges | Larger size may limit certain applications | Limited stability upon dilution | Mechanical weakness and complex tuning requirements |
Key Considerations for Selecting Biodegradable Polymers in Drug Delivery Design
The selection of biodegradable polymers is a critical step in designing efficient and reliable drug delivery systems. A well-matched polymer–drug combination ensures optimal encapsulation, stability, and release performance. This process requires a systematic evaluation of both material properties and formulation requirements. The following factors play a decisive role in determining the suitability of a biodegradable polymer for a given application.
Physicochemical Properties of the Drug
- Solubility Profile: Hydrophobic drugs benefit from polymeric micelles or nanoparticles with hydrophobic cores, while hydrophilic drugs require hydrogel or matrix-based systems. In practice, solubility mismatch between drug and polymer strongly influences encapsulation efficiency and phase distribution; parameters such as partition coefficient (logP), Hansen solubility parameters, and drug–polymer interaction energy should be considered to optimize loading and minimize premature leakage.
- Molecular Size: Large biomolecules require polymers that preserve structural integrity and prevent denaturation. Macromolecular drugs (e.g., proteins, nucleic acids) are particularly sensitive to shear stress, interfacial exposure, and dehydration during formulation; therefore, polymer systems with mild processing conditions, low interfacial tension, and protective hydration environments are preferred to maintain conformational stability.
- Chemical Stability: Sensitive compounds require protective polymer matrices to prevent degradation during formulation and release. Factors such as susceptibility to hydrolysis, oxidation, or pH-induced degradation must be evaluated, and polymer selection should consider barrier properties, microenvironmental pH evolution, and potential catalytic effects of degradation byproducts.
Polymer Degradation and Release Requirements
- Degradation Rate: Must align with the desired release timeline, ranging from rapid release to long-term sustained delivery. This rate is influenced by molecular weight, copolymer ratio (e.g., lactide:glycolide), crystallinity, and hydrophilicity; fine-tuning these parameters enables precise control over degradation half-life and release duration.
- Degradation Mechanism: Bulk vs surface erosion significantly impacts release kinetics. Bulk-degrading polymers often exhibit autocatalytic effects and heterogeneous internal degradation, while surface-eroding systems enable more linear and predictable release profiles; understanding water diffusion relative to degradation rate is essential for mechanism selection.
- Byproduct Safety: Degradation products should be non-toxic and easily eliminated. In addition to toxicity, accumulation of acidic or reactive byproducts may alter local microenvironment conditions, potentially affecting drug stability and release kinetics; buffering strategies or polymer modifications may be required to mitigate these effects.
Formulation Strategy and Delivery Format
- Nanoparticles vs Microspheres: Particle size influences release rate, distribution, and stability. Smaller particles provide higher surface area-to-volume ratios, leading to faster diffusion-driven release, whereas larger microspheres favor prolonged degradation-controlled release; size distribution uniformity is also critical for reproducible performance.
- Hydrogel Systems: Suitable for sustained release and high water content environments. Release behavior is governed by mesh size, crosslinking density, and swelling kinetics, which can be engineered to control diffusion coefficients and responsiveness to environmental stimuli.
- Surface Functionalization: Enables further tuning of interaction and release behavior. Surface chemistry can be modified to alter hydrophilicity, charge density, and binding affinity, influencing protein adsorption, cellular interaction, and overall delivery efficiency.
Manufacturing and Scalability Considerations
- Process Reproducibility: Consistent particle size and drug loading are essential. Variability in emulsification energy, solvent removal rate, and mixing conditions can significantly affect final product characteristics, requiring tight process control and robust optimization.
- Material Availability: Reliable sourcing of polymers with consistent quality is critical. Batch-to-batch variations in molecular weight distribution, end-group chemistry, and residual monomers can impact degradation behavior and release performance.
- Cost Efficiency: Polymer selection impacts overall formulation cost and scalability. Considerations include raw material cost, process complexity, yield, and scalability of synthesis and formulation methods, all of which influence feasibility in large-scale production.
Application Scope of Biodegradable Polymers Across Different Drug Modalities
Biodegradable polymers demonstrate broad applicability across diverse drug modalities due to their tunable physicochemical properties and adaptable formulation capabilities. By tailoring polymer composition, architecture, and degradation behavior, these materials can be optimized to accommodate drugs with varying molecular sizes, solubility profiles, and stability requirements. A modality-driven design approach is essential to ensure compatibility between the drug and polymer matrix, enabling efficient encapsulation, protection, and controlled release. The following sections outline how biodegradable polymers are strategically applied across major drug classes.
Small Molecule Drug Delivery
Small molecule drugs, particularly those with poor aqueous solubility, benefit significantly from biodegradable polymer-based delivery systems. Hydrophobic polymers such as PLA, PLGA, and PCL provide compatible matrices that enhance drug loading efficiency and minimize premature diffusion.
- Solubility Enhancement: Amphiphilic block copolymers form micellar structures with hydrophobic cores, improving dispersion and apparent solubility of poorly soluble compounds.
- Controlled Release Profiles: Drug release can be tuned through polymer degradation rate, crystallinity, and matrix density.
- Stability Improvement: Encapsulation reduces exposure to environmental degradation factors such as hydrolysis and oxidation.
Peptides and Protein Delivery
Peptides and proteins present unique formulation challenges due to their structural complexity and sensitivity to environmental conditions. Biodegradable polymers provide protective microenvironments that preserve molecular integrity while enabling sustained release.
- Structural Stabilization: Polymer matrices reduce denaturation caused by interfacial stress, temperature fluctuations, and dehydration.
- Controlled Microenvironment: Formulation strategies must address pH shifts caused by polymer degradation, which can affect protein stability.
- Encapsulation Strategies: Double emulsion and mild processing techniques are commonly employed to maintain bioactivity.
Nucleic Acid Delivery Systems
Nucleic acids such as DNA and RNA require specialized delivery systems due to their large size, negative charge, and susceptibility to enzymatic degradation. Biodegradable cationic polymers enable complex formation and provide protection during delivery.
- Electrostatic Complexation: Cationic polymers (e.g., poly(beta-amino esters)) form stable complexes with negatively charged nucleic acids, facilitating encapsulation.
- Protection from Degradation: Polymer matrices shield nucleic acids from nuclease activity and environmental stress.
- Stimuli-Responsive Release: Polymers can be engineered with pH- or redox-sensitive linkages to enable triggered release under specific conditions.
Hydrophilic Macromolecules and Biologics
Hydrophilic macromolecules require delivery systems that maintain high water content and minimize aggregation or denaturation. Biodegradable hydrogels and polymer networks are particularly suitable for these applications.
- Hydrated Polymer Networks: Hydrogels provide aqueous environments that preserve structural integrity of sensitive macromolecules.
- Diffusion-Controlled Release: Release behavior is governed by mesh size and swelling properties of the polymer network.
- Reduced Aggregation Risk: Encapsulation within hydrated matrices minimizes intermolecular interactions that lead to aggregation.
Multi-Component and Combination Systems
Increasing formulation complexity has driven the development of systems capable of delivering multiple active components simultaneously. Biodegradable polymers provide structural flexibility for designing such multi-functional platforms.
- Co-Encapsulation Strategies: Different drug types can be incorporated within distinct domains of polymer matrices or core–shell structures.
- Sequential Release Design: Layered or multi-phase polymer systems enable staged release of multiple agents.
- Hybrid Delivery Platforms: Integration with lipids or inorganic materials further enhances loading capacity and functional diversity.
Services
Custom Biodegradable Polymer Solutions for Drug Delivery
BOC Sciences is a specialized provider of monomer and polymer chemical products, offering integrated solutions for biodegradable polymer-based drug delivery system development. With extensive expertise in polymer chemistry, material engineering, and formulation science, we support both research and industrial clients in designing high-performance delivery platforms tailored to specific application needs. Our capabilities span from monomer selection and polymer synthesis to advanced functionalization and formulation development. By combining precise control over polymer structure, degradation behavior, and physicochemical properties, we enable the development of delivery systems with optimized drug loading, stability, and release profiles. Our scalable production and customization services ensure consistent quality from early-stage research to large-scale applications.

Biodegradable Polymer Material Supply
- Extensive portfolio of biodegradable polymers including PLA, PLGA, PCL, PEG-based copolymers, and natural polymers.
- Customizable specifications such as molecular weight, polydispersity, end-group functionality, and composition ratios.
- High-purity materials with strict quality control and batch consistency.
- Flexible supply scale from milligram quantities for research to kilogram-level production.

Custom Polymer Design and Synthesis
- Tailored synthesis of biodegradable polymers with controlled architecture, including block, graft, and random copolymers.
- Design of polymers with tunable degradation rates and targeted physicochemical properties.
- Development of functionalized polymers with reactive groups for further modification.
- Comprehensive analytical characterization including molecular weight distribution, composition, and structural validation.

Drug–Polymer Conjugation and Functionalization
- Advanced conjugation strategies for small molecules, peptides, proteins, and nucleic acids.
- Design and synthesis of cleavable linkers for controlled and stimuli-responsive drug release.
- Surface modification techniques for tuning hydrophilicity, charge, and interaction properties.
- Integration of targeting ligands or responsive moieties to enhance delivery performance.

Formulation Development and Optimization
- Development of biodegradable polymer-based nanoparticles, microspheres, micelles, and hydrogel systems.
- Optimization of drug loading efficiency, encapsulation stability, and release kinetics.
- Process development including emulsification, nanoprecipitation, spray drying, and microfluidics.
- Support for scale-up, reproducibility, and process consistency in formulation manufacturing.
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What are the most commonly used biodegradable polymers in drug delivery?
The most commonly used biodegradable polymers include PLA, PLGA, and PCL due to their well-characterized degradation behavior and tunable physicochemical properties. These materials allow precise control over degradation rate, mechanical strength, and drug release kinetics. Additionally, PEG-based copolymers are widely used to improve hydrophilicity and enhance formulation flexibility in complex delivery systems.
-
How does polymer degradation affect drug release?
Polymer degradation plays a central role in controlling drug release by gradually breaking down the polymer matrix through hydrolysis or enzymatic processes. As degradation progresses, increased porosity and reduced molecular weight facilitate drug diffusion. The balance between degradation rate and diffusion determines whether release is primarily diffusion-controlled, degradation-controlled, or a combination of both mechanisms.
-
How can burst release be minimized in biodegradable systems?
Burst release can be minimized by optimizing formulation parameters such as increasing polymer molecular weight, improving drug encapsulation efficiency, and reducing surface-associated drug content. Additional strategies include modifying polymer hydrophobicity, using core–shell structures, and controlling particle size distribution to limit rapid initial diffusion from the outer regions of the delivery system.
-
What is the difference between bulk and surface degradation?
Bulk degradation occurs uniformly throughout the polymer matrix as water penetrates the entire structure, often leading to rapid molecular weight reduction and internal autocatalysis. In contrast, surface degradation happens layer by layer at the material interface, maintaining structural integrity while enabling more predictable and linear drug release profiles over time.
-
How do you select the right biodegradable polymer?
Selecting the appropriate biodegradable polymer requires evaluating drug properties, desired release profile, and processing conditions. Key considerations include polymer composition, molecular weight, degradation rate, and compatibility with the drug. A systematic approach ensures optimal encapsulation efficiency, stability, and controlled release performance for the intended formulation strategy.
-
Are biodegradable polymers suitable for all drug types?
Biodegradable polymers are highly versatile and can be adapted for a wide range of drug types, including small molecules, peptides, proteins, and nucleic acids. However, formulation strategies must be carefully optimized to address specific challenges such as solubility, stability, and molecular size, ensuring compatibility between the drug and the polymer matrix.
-
What formulation types use biodegradable polymers?
Biodegradable polymers are used in various formulation platforms, including nanoparticles, microspheres, micelles, and hydrogels. Each system offers distinct advantages in terms of drug loading, release control, and structural properties. The choice of formulation depends on drug characteristics, desired release kinetics, and processing feasibility for scalable manufacturing.

 Fig. 1. Structural illustration of biodegradable polymer nanoparticles, microspheres, and hydrogels used in controlled drug delivery systems (BOC Sciences Authorized).
Fig. 1. Structural illustration of biodegradable polymer nanoparticles, microspheres, and hydrogels used in controlled drug delivery systems (BOC Sciences Authorized). Fig. 2. Schematic representation of polymer chain degradation and diffusion-controlled drug release behavior (BOC Sciences Authorized).
Fig. 2. Schematic representation of polymer chain degradation and diffusion-controlled drug release behavior (BOC Sciences Authorized).