Controlled & Sustained Release Polymer Systems for Precision Drug Delivery
Controlled and sustained release polymer systems have become essential technologies in modern pharmaceutical science, enabling more precise control over drug pharmacokinetics and therapeutic performance. By incorporating active pharmaceutical ingredients into polymer matrices, nanoparticles, hydrogels, or implantable devices, these systems can regulate the rate, duration, and location of drug release within the body. Compared with conventional formulations, polymer-based delivery platforms help maintain stable drug concentrations, reduce dosing frequency, and minimize systemic side effects. Biodegradable polymers, functionalized synthetic polymers, and natural polymers derived biomaterials can be carefully designed to achieve predictable degradation profiles, tunable drug diffusion, and responsive release behaviors under specific physiological conditions. These capabilities make polymer systems particularly valuable for the delivery of small molecules, peptides, proteins, and nucleic acid therapeutics.
Resources
Introduction to Controlled and Sustained Release Polymer Systems
The evolution of precision drug delivery has been fundamentally driven by advancements in materials science, specifically the development of engineered macromolecular frameworks. Controlled and sustained release polymer systems represent a sophisticated approach to modulating the pharmacokinetics and biodistribution of active pharmaceutical ingredients. By utilizing specific monomers and carefully controlled polymerization techniques, scientists can create carriers that release therapeutics at predetermined rates over extended periods. This paradigm shift moves the pharmaceutical industry away from conventional immediate-release dosage forms toward highly optimized, patient-centric therapeutic profiles.
What Are Controlled Release Polymer Drug Delivery Systems?
At their core, controlled release polymer drug delivery systems are engineered composite platforms where an active therapeutic agent is thermodynamically dispersed, physically encapsulated, or covalently tethered within a macromolecular network. These systems function by establishing a structural, rate-limiting barrier between the highly concentrated therapeutic payload and the surrounding aqueous biological fluids. The release of the active agent is strictly mediated by the physicochemical properties of the selected polymer, such as its glass transition temperature, degree of crystallinity, and susceptibility to hydrolytic or enzymatic cleavage. Consequently, the polymer matrix orchestrates a precise, programmable discharge of the therapeutic agent, ensuring the systemic concentration remains within a highly specific, predefined therapeutic window.
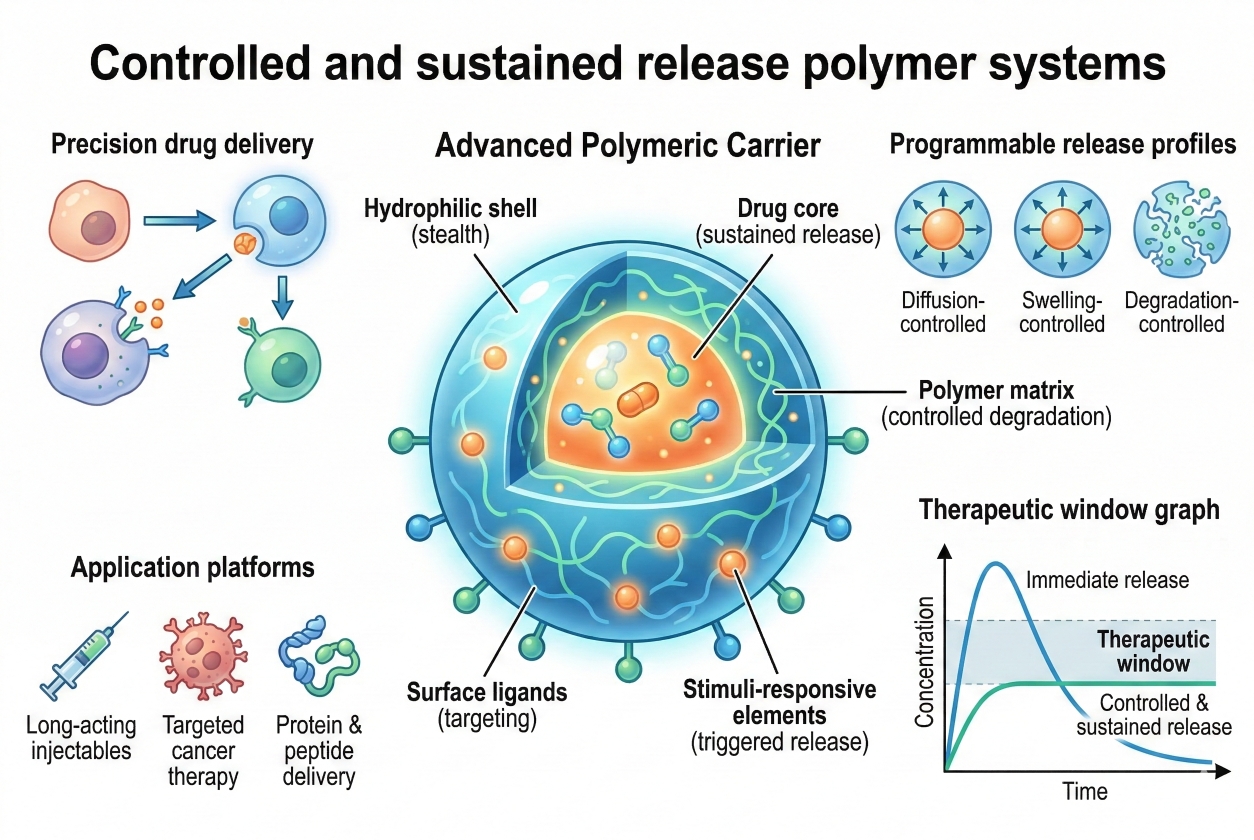 Fig. 1. Mechanisms and applications of controlled and sustained release polymers (BOC Sciences Authorized).
Fig. 1. Mechanisms and applications of controlled and sustained release polymers (BOC Sciences Authorized).
Differences Between Controlled Release and Sustained Release
While frequently conflated in general formulation literature, sustained release and controlled release delineate entirely distinct pharmacokinetic and physicochemical profiles. Sustained release systems are designed to prolong the dissolution and absorption phases over an extended timeframe. This typically results in first-order release kinetics, where the rate of drug diffusion is directly proportional to the declining concentration gradient within the matrix. As the internal concentration depletes, the release rate proportionally decelerates.
Conversely, true controlled release systems are engineered to achieve zero-order release kinetics without relying on concentration gradients. In this advanced release model, the delivery rate remains absolutely constant and independent of the initial or remaining active ingredient concentration. This steady, non-declining release is achieved by designing highly specialized polymer architectures—such as precisely tailored osmotic platforms or strictly surface-eroding polyanhydrides. In these sophisticated systems, the structural mechanics, swelling behavior, and degradation profile of the polymer matrix function as the sole rate-determining steps. This ensures a highly accurate, flat release profile over the entire predefined duration, establishing strict control over the temporal delivery of the therapeutic payload.
Why Precision Drug Delivery Requires Polymer-Based Carriers?
The vast majority of newly discovered active pharmaceutical ingredients exhibit severe biopharmaceutical limitations, most notably extreme hydrophobicity, rapid enzymatic degradation, and exceedingly short systemic half-lives. Precision drug delivery mandates the use of engineered polymer carriers to systematically overcome these thermodynamic and physiological barriers. By encapsulating a lipophilic agent within an amphiphilic block copolymer micelle, scientists can drastically alter the apparent aqueous solubility of the payload. Furthermore, the immense spatial conformation of high-molecular-weight polymers provides intense steric hindrance. This structural shielding protects fragile molecules from premature metabolic clearance, prevents non-specific protein adsorption, and allows the delivery system to navigate complex biological fluids with remarkable stability.
Advantages of Controlled Release Polymer Drug Delivery
The integration of polymers into drug delivery formulations offers a multitude of physicochemical and biopharmaceutical advantages that optimize therapeutic outcomes.
- Improved Drug Bioavailability and Therapeutic Efficacy: By protecting the active pharmaceutical ingredient from premature degradation and modulating its release, polymer matrices significantly enhance the fraction of the drug that reaches systemic circulation. This optimized bioavailability ensures that the therapeutic agent remains active and available at the target site, thereby maximizing its pharmacological efficacy.
- Reduced Dosing Frequency and Enhanced Patient Compliance: A direct consequence of improved bioavailability and prolonged circulation is the ability to drastically reduce dosing frequency. Transitioning from multiple daily doses to once-daily, weekly, or even monthly administrations simplifies medication regimens, thereby significantly enhancing overall adherence to the therapeutic protocol.
- Minimized Side Effects and Toxicity: Conventional dosage forms often lead to a rapid spike in plasma drug concentration, potentially crossing the toxic threshold before rapidly metabolizing. Polymer systems smooth out this pharmacokinetic curve, eliminating the initial burst effect and maintaining concentrations safely below toxic levels, which minimizes off-target side effects.
- Enhanced Stability of Sensitive Therapeutics: Complex therapeutics, including peptides, proteins, and nucleic acids, are notoriously unstable in physiological environments. Encapsulation within a dense polymeric network provides robust steric shielding against nucleases, proteases, and extreme pH environments, preserving the structural integrity of the payload until it reaches the intracellular target.
- Flexibility in Polymer Design and Functionalization: The vast chemical space of monomer selection allows for infinite tunability. Scientists can adjust the ratio of hydrophilic to hydrophobic blocks, alter the degree of crosslinking, and modify end-groups to achieve precise degradation rates and mechanical properties. This architectural flexibility makes polymers universally adaptable to almost any class of therapeutic agent.
Key Mechanisms Behind Controlled and Sustained Drug Release
The temporal profile of drug release from a polymeric matrix is dictated by complex thermodynamic and kinetic mechanisms. Understanding these physical pathways is essential for rationally designing systems that meet specific pharmacokinetic requirements.
Diffusion-Controlled Polymer Drug Release
Diffusion is the most fundamental mechanism governing release. In reservoir systems, an inner core of the drug is surrounded by an insoluble polymeric membrane, and the drug diffuses through this rate-limiting barrier following Fickian principles. In matrix systems, the drug is uniformly dispersed throughout a solid polymer network. As biological fluids penetrate the matrix, the drug dissolves and diffuses out through the porous channels created by the polymer chains.
Polymer Degradation and Erosion-Based Release
For biodegradable polymers, release is heavily dependent on the chemical breakdown of the polymer backbone. This can occur via surface erosion, where the polymer degrades strictly at the material-fluid interface, leading to highly predictable release kinetics. Alternatively, bulk degradation occurs when water penetrates the entire polymer matrix, initiating random chain scission throughout the volume, which eventually leads to a sudden structural collapse and mass release of the remaining drug.
Swelling-Controlled Drug Delivery Systems
Swelling-controlled systems rely on the hydration dynamics of hydrophilic polymers. Initially, the polymer exists in a glassy, dehydrated state, restricting drug mobility. Upon exposure to aqueous environments, water molecules penetrate the matrix, lowering the glass transition temperature of the polymer. This causes the polymer chains to relax and expand, transitioning into a rubbery state. The drug then diffuses through the expanded macromolecular network.
Stimuli-Responsive Polymer Release Mechanisms
Advanced formulations utilize polymers that undergo abrupt physical or chemical transitions in response to specific environmental triggers. These triggers can be endogenous, such as localized pH variations, specific enzymatic activity, or temperature gradients, or exogenous, such as applied magnetic fields or light. The stimuli induce conformational changes, cleavage of functional groups, or rapid swelling, prompting a spatially and temporally controlled burst of drug release.
Types of Polymers Used in Controlled Drug Delivery Systems
The architectural foundation of any advanced pharmacokinetic modulation platform lies in the precise selection of its macromolecular constituents. The physicochemical identity, stereochemistry, and molecular weight distribution of the polymer directly dictate the thermodynamic interactions with the active pharmaceutical ingredient and the surrounding aqueous medium. By categorizing these materials based on their origin, degradation profiles, and environmental responsiveness, formulation scientists can rationally design matrices that achieve exact release kinetics—ranging from hours to several months—while maintaining strict biocompatibility and structural integrity.
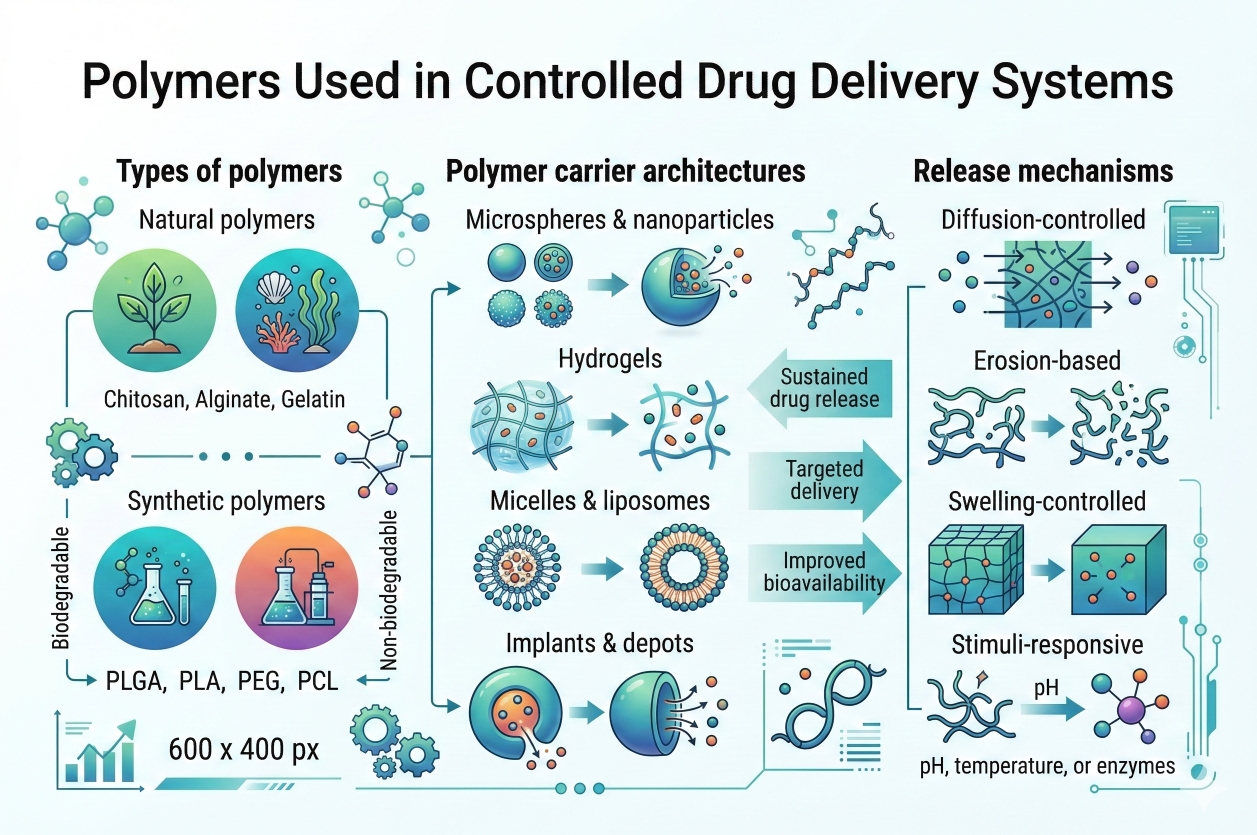 Fig. 2. Classification and mechanisms of polymers in controlled drug delivery (BOC Sciences Authorized).
Fig. 2. Classification and mechanisms of polymers in controlled drug delivery (BOC Sciences Authorized).
Biodegradable Polymers for Sustained Drug Release
Biodegradable macromolecules are synthesized to undergo controlled chemical scission upon exposure to aqueous conditions, eliminating the requirement for subsequent extraction of the carrier matrix. The degradation kinetics are primarily governed by the susceptibility of backbone linkages, such as esters, orthoesters, or anhydrides, to hydrolytic cleavage. Aliphatic polyesters represent a cornerstone in this category. By modulating the stoichiometric ratio of distinct monomeric units during copolymerization, scientists can precisely tune the hydrophobicity, glass transition temperature, and crystallinity of the resulting matrix. This structural tunability allows for the exact programming of degradation timelines, shifting the release profile from rapid bulk erosion to highly predictable, long-term surface erosion. Polyanhydrides, possessing highly water-labile linkages but extremely hydrophobic backbones, are particularly valuable for achieving steady zero-order release through strict surface-degradation mechanisms.
Synthetic Polymers in Precision Drug Delivery
Synthetic polymers provide unmatched batch-to-batch reproducibility, high chemical purity, and the ability to engineer highly complex molecular architectures, including multiblock copolymers, star-shaped macromolecules, and dendrimers. These materials are broadly classified into non-degradable and water-soluble categories. Non-degradable variants, featuring robust carbon-carbon or siloxane backbones, are heavily utilized in the fabrication of highly durable reservoir devices and diffusion-controlled, rate-limiting membranes. Conversely, water-soluble synthetic polymers are frequently deployed as stealth steric stabilizers. Through covalent conjugation to therapeutic agents or nanocarrier surfaces, these highly hydrated chains create a dense conformational cloud that minimizes non-specific protein adsorption and significantly extends the systemic circulation half-life of the active payload.
Natural Polymers for Controlled Drug Delivery
Polymers derived from botanical, marine, or microbial sources offer exceptional biomimetic properties and inherent biocompatibility. These complex macromolecules, primarily encompassing polysaccharides and proteinaceous materials, are characterized by a high density of reactive functional groups distributed along their backbones, including primary amines, hydroxyls, and carboxylic acids. This structural richness facilitates straightforward chemical functionalization and extensive crosslinking capabilities without requiring harsh organic solvents. Furthermore, many natural polyions possess the unique ability to undergo spontaneous ionic gelation upon interaction with oppositely charged crosslinkers. This enables the mild, aqueous encapsulation of highly sensitive biotherapeutics, preserving their complex secondary and tertiary structures that would otherwise denature under conventional synthetic processing conditions.
Smart and Functional Polymers for Programmable Release
Stimuli-responsive, or smart, polymers represent the vanguard of programmable delivery systems, designed to undergo rapid, reversible, and non-linear physicochemical transitions in response to precise microenvironmental triggers. Thermoresponsive polymers are engineered with specific phase-transition parameters, shifting abruptly from a highly soluble, extended state to a collapsed, hydrophobic state upon crossing a critical temperature threshold. pH-responsive variants exploit the protonation or deprotonation of weakly acidic or basic pendant groups, driving massive hydration and volumetric swelling in targeted ionic environments. Advanced formulations also incorporate redox-sensitive linkages, such as disulfide bonds, which remain highly stable in general circulation but undergo rapid reductive cleavage upon entering specific intracellular compartments, triggering an immediate, localized discharge of the therapeutic agent.
Looking for Custom Polymers for Drug Delivery?
Whether you need cationic polymers, biodegradable carriers, or stimuli-responsive platforms, our experts provide tailored synthesis and functionalization solutions to support your research and therapeutic development.
Major Polymer-Based Controlled Release Drug Delivery Platforms
The architectural formulation of macromolecular carriers dictates the spatial distribution, payload capacity, and administration route of the final therapeutic system. Transitioning from raw polymer chains to highly engineered delivery platforms requires a profound understanding of interfacial phenomena, thermodynamic stability, and precise fabrication methodologies. By manipulating the physical chemistry of the polymer-solvent-active pharmaceutical ingredient system, formulation scientists can construct diverse microscale and nanoscale architectures tailored to specific pharmacokinetic requirements and targeting strategies.
Polymer Microspheres and Microcapsules
The fabrication of polymer microspheres and microcapsules relies heavily on complex multiphase fluid dynamics and precise control over interfacial tension. Typically formulated via water-in-oil-in-water double emulsion or coacervation techniques, these microparticulate systems require the careful selection of surfactants and organic solvents to stabilize the emulsion droplets before solidification. During the solvent evaporation phase, the polymer precipitates at the solvent-water interface, forming a dense, continuous shell around a liquid core in microcapsules, or a monolithic matrix with dispersed therapeutic agents in microspheres. The rate of solvent extraction, the shear force applied during emulsification, and the polymer concentration directly influence the final particle size distribution, surface morphology, and internal porosity, which collectively govern the subsequent release kinetics.
Polymer Nanoparticles and Nanocarriers
At the nanoscale, polymer carrier formation is driven by rapid macromolecular self-assembly and nanoprecipitation processes. When a dissolved polymer and active pharmaceutical ingredient are introduced into an antisolvent under high shear or via microfluidic mixing, spontaneous supersaturation occurs. This thermodynamic instability forces the polymer chains to undergo rapid nucleation and growth, encapsulating the payload within a sub-micron matrix. For amphiphilic block copolymers, structural formation is governed by the critical micelle concentration. Above this threshold, the hydrophobic segments spontaneously associate to minimize contact with the aqueous phase, creating a hydrophobic core capable of solubilizing lipophilic agents. This core is surrounded by a hydrophilic corona that provides steric stabilization and prevents rapid physiological clearance.
Polymeric Hydrogels for Sustained Drug Delivery
Polymeric hydrogels are three-dimensional, highly hydrated networks structurally maintained through either covalent crosslinking or physical entanglements. The formation of chemical hydrogels involves the free-radical polymerization of specific hydrophilic monomers alongside multifunctional crosslinkers, creating a permanent, irreversible macroscopic lattice. Conversely, physical hydrogels rely on transient molecular interactions, including hydrogen bonding, ionic complexation, or hydrophobic associations, which allow for shear-thinning and injectability. The sustained release capability of these platforms is dictated by the thermodynamic balance between the osmotic pressure driving fluid into the network and the elastic restorative force of the polymer chains resisting expansion. By precisely tuning the crosslinking density and the polymer volume fraction, formulation scientists can establish specific mesh sizes that strictly control the diffusion rate of encapsulated macromolecules.
Polymer Implants and Long-Acting Drug Depots
Polymeric implants and macroscopic depots represent the pinnacle of long-acting, localized delivery, requiring advanced solid-state engineering and precise rheological control. Solid implants are typically fabricated through hot-melt extrusion or injection molding, processes that subject the polymer and active pharmaceutical ingredient to elevated thermal and mechanical stresses. The formulation must be thermodynamically optimized to ensure the therapeutic agent remains uniformly dispersed within the molten polymer matrix without undergoing degradation. Alternatively, in situ forming depots utilize solutions of biodegradable polymers dissolved in biocompatible organic solvents. Upon injection, the solvent rapidly diffuses into the surrounding aqueous environment, prompting a solvent-exchange phase inversion. This thermodynamic phase separation solidifies the polymer locally, trapping the therapeutic payload within a tortuous, porous structure that provides a highly predictable release profile over several months.
Design Strategies for Controlled and Sustained Release Systems
Translating a basic polymer-drug mixture into a highly optimized, predictable delivery vehicle requires rigorous physicochemical engineering. Formulation scientists must bridge the gap between materials science and pharmacokinetics by manipulating molecular architectures, thermodynamic mixing parameters, and surface chemistry. A successful design strategy relies heavily on mathematical modeling to predict release profiles and precise chemical functionalization to dictate the spatial and temporal fate of the therapeutic payload in complex biological environments.
Polymer Molecular Weight and Drug Release Kinetics
The molecular weight and the polydispersity index of a polymer are fundamental parameters that dictate the mechanical integrity, porosity, and degradation timeline of the delivery matrix. High molecular weight chains exhibit extensive physical entanglement, which elevates the glass transition temperature and creates a dense macromolecular network. This high entanglement density severely restricts the diffusivity of both water entering the matrix and the therapeutic agent escaping it. To precisely engineer and predict this release, formulation scientists rely on mathematical modeling, most notably the Korsmeyer-Peppas equation. By manipulating the polymer's molecular weight and crosslinking density, scientists can tune the exponent n to shift the release mechanism from Fickian diffusion to anomalous transport or perfect zero-order release, ensuring the pharmacokinetic profile matches the required therapeutic window.
Drug–Polymer Interactions and Encapsulation Efficiency
Achieving high encapsulation efficiency while preventing premature burst release requires a deep understanding of the thermodynamic compatibility between the active pharmaceutical ingredient and the polymer matrix. This compatibility is often evaluated using the Flory-Huggins solution theory, where the interaction parameter (χ) quantifies the energy of mixing. A low or negative Flory-Huggins parameter indicates high thermodynamic miscibility, allowing the drug to remain uniformly dispersed in an amorphous solid solution. When the drug and polymer share complementary functional groups, powerful intermolecular forces—such as hydrogen bonding, dipole-dipole interactions, and electrostatic complexation—lock the active payload within the matrix. Maximizing these non-covalent interactions restricts the mobility of the drug molecules, preventing them from migrating to the surface of the particle during fabrication. This thermodynamic stabilization is critical for suppressing the initial burst release and maintaining structural homogeneity throughout the shelf life of the product.
Surface Functionalization for Targeted and Controlled Delivery
The surface chemistry of a polymeric carrier strictly governs its interfacial interactions with biological fluids and cellular membranes. Bare hydrophobic polymer surfaces rapidly adsorb circulating proteins via hydrophobic interactions, leading to rapid opsonization and clearance by the mononuclear phagocyte system. To circumvent this, the surface is covalently grafted with highly hydrophilic, flexible polymer chains. This functionalization creates a dense, steric hydration layer that physically repels opsonins, drastically prolonging the systemic circulation time of the carrier. Beyond passive shielding, the terminal ends of these surface polymers can be chemically activated—using techniques like carbodiimide crosslinking—to attach specific targeting ligands, including aptamers, peptides, or monoclonal antibodies. This active targeting strategy drastically alters the biodistribution profile, shifting the carrier's accumulation from generic clearance organs directly to the specific cellular receptors of the target tissue, thereby locally amplifying the therapeutic concentration.
Engineering Multi-Stage and Dual-Release Systems
Modern therapeutic protocols often require the simultaneous or sequential delivery of multiple active agents with vastly different physicochemical properties. To accommodate this, scientists engineer complex, multi-compartmental polymeric architectures, such as core-shell microparticles or multilayered thin films. Fabricated via advanced techniques like coaxial electrohydrodynamic spraying or precision microfluidics, these systems feature distinct spatial domains. The outer shell can be formulated with a rapidly dissolving, low-molecular-weight polymer to deliver an immediate, high-dose pulse of a sensitizing agent. Simultaneously, the inner core is formulated from a high-molecular-weight, slowly degrading matrix, providing a sustained, zero-order release of a second therapeutic agent over several weeks. This structural compartmentalization prevents the different drugs from interacting prematurely and allows for precise, independent tuning of their respective release kinetics.
Applications of Controlled & Sustained Release Polymer Systems
The ultimate objective of engineering macromolecular architectures is to resolve the severe biopharmaceutical limitations of raw active pharmaceutical ingredients. By applying the rigorous thermodynamic and kinetic design strategies previously discussed, formulation scientists can develop highly specialized delivery platforms tailored to specific physiological environments. These advanced polymer systems transcend traditional formulation boundaries, enabling the precise spatial and temporal control required for modern, complex therapeutic modalities.
Cancer Therapy and Targeted Chemotherapy Delivery
Formulating antineoplastic agents presents a massive physicochemical challenge due to their extreme hydrophobicity and high indiscriminate cytotoxicity. Polymer science addresses this by utilizing amphiphilic block copolymers that spontaneously self-assemble into nanoscopic micelles, effectively solubilizing lipophilic drugs within their hydrophobic cores. Once injected, these nanocarriers exploit the distinct pathophysiological architecture of tumor microenvironments. Tumors typically exhibit defective, highly porous vasculature and impaired lymphatic drainage. Polymeric nanoparticles engineered to a specific hydrodynamic radius—typically between 20 and 150 nanometers—can passively extravasate through these vascular fenestrations and accumulate preferentially in the target tissue, a phenomenon known as the enhanced permeability and retention effect. To further optimize this, the outer hydrophilic corona of the polymer is functionalized with targeting ligands that bind exclusively to overexpressed surface receptors on the target cells, driving receptor-mediated endocytosis and highly localized intracellular release.
Long-Acting Injectable Formulations
For therapeutic regimens requiring strict, continuous pharmacological intervention, long-acting injectable formulations provide a transformative pharmacokinetic advantage. These systems primarily utilize biodegradable aliphatic polyesters formulated into precisely sized microspheres or in situ precipitating depots. The primary biopharmaceutical goal is to flatten the plasma concentration-time curve. Conventional injectables produce rapid systemic peaks that border on toxicity, followed by rapid clearance and sub-therapeutic troughs. By dispersing the active pharmaceutical ingredient within a dense, slowly degrading polymer matrix, the release is strictly governed by hydrolytic chain scission and steady diffusion. This mechanism provides a continuous, zero-order release profile that maintains optimal systemic concentrations for weeks or months following a single administration, eliminating the massive peak-to-trough fluctuations and significantly improving the overall pharmacokinetic stability.
Protein, Peptide, and Nucleic Acid Delivery
Biologics, including highly complex proteins, therapeutic peptides, and nucleic acids, are notoriously fragile. Their tertiary structures are highly susceptible to denaturation from shear stress, temperature fluctuations, and enzymatic degradation by ubiquitous proteases and nucleases in biological fluids. Polymer systems offer indispensable steric shielding for these macromolecules. For nucleic acid delivery, cationic polymers are specifically synthesized to possess a high density of protonatable amines. These polycations spontaneously interact with the negatively charged phosphate backbones of genetic material, condensing them into stable, nanoscale polyplexes. Upon cellular uptake via endocytosis, these specific polymers buffer the acidic environment of the endosome—a mechanism known as the proton sponge effect. This buffering causes massive osmotic swelling and subsequent rupture of the endosomal membrane, safely releasing the intact nucleic acid payload directly into the cytosol before lysosomal degradation can occur.
Implantable Drug Delivery Systems for Chronic Diseases
When systemic delivery is fundamentally inefficient or when potent therapeutics must be strictly confined to a localized tissue compartment, macroscopic polymer implants become the formulation of choice. These systems are geometrically engineered as solid rods, flexible rings, or flat wafers, utilizing either non-degradable siloxanes for diffusion-controlled release or specialized polyanhydrides for strict surface-erosion release. Implantable systems are surgically or minimally invasively placed directly at the required site of action. Because the polymer matrix isolates the payload from immediate systemic dissolution, these platforms can safely house massive quantities of highly potent active agents. As the matrix dictates a slow, constant release, it establishes a high, sustained drug concentration exclusively in the surrounding local tissue while keeping systemic circulation levels remarkably low, thereby circumventing widespread physiological clearance mechanisms and minimizing off-target exposure.
Services
Polymer Materials and Custom Development Services for Drug Delivery
Controlled and sustained release polymer systems play a critical role in modern drug development by enabling prolonged therapeutic effects, reduced dosing frequency, and improved patient compliance. Leveraging extensive expertise in polymer chemistry, material engineering, and drug delivery system design, BOC Sciences provides specialized support for the development of controlled and sustained release formulations. Based on the physicochemical properties of drug molecules and specific application requirements, our team offers services including polymer material selection, carrier system design, drug encapsulation, and release behavior studies. These capabilities help researchers develop stable polymer delivery platforms with predictable release profiles, supporting the development and optimization of advanced pharmaceutical formulations.

Controlled Release Polymer Design and Synthesis
- Custom design and polymer synthesis of functional polymers for controlled drug delivery systems.
- Structural design and molecular weight control of PLGA, PLA, PCL, and PEG-based polymers.
- Support for controlled polymerization techniques, including RAFT, ATRP, and ring-opening polymerization (ROP).
- Development of biodegradable and functionalized polymer materials.

Construction of Controlled Release Delivery Systems
- Development of polymer nanoparticles, polymeric micelles, and microsphere systems.
- Design of sustained and long-acting drug delivery platforms.
- Development of systems compatible with various administration routes.
- Optimization of carrier structure and formulation performance.

Drug Encapsulation and Formulation Development
- Development of encapsulation systems for hydrophobic and hydrophilic drugs.
- Optimization of drug loading capacity and encapsulation efficiency.
- Development of single-drug and combination drug delivery systems.
- Optimization of formulation stability and release performance.

Characterization and Performance Evaluation
- Polymer structure and molecular weight analysis.
- Particle size distribution and morphological characterization.
- Evaluation of drug release kinetics and polymer degradation behavior.
- Comprehensive performance evaluation and system optimization.
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
How do controlled and sustained release polymers improve drug therapy?
Controlled and sustained release polymers improve drug therapy by regulating the rate and duration of drug release within the body. These systems maintain stable therapeutic concentrations over extended periods, reduce dosing frequency, minimize side effects, and enhance patient compliance. Polymer-based carriers can also protect sensitive drugs and improve overall treatment efficacy.
-
Which polymers are commonly used in controlled drug release systems?
Common polymers used in controlled and sustained drug delivery include biodegradable materials such as poly(lactic acid) (PLA), poly(lactic-co-glycolic acid) (PLGA), and polycaprolactone (PCL), as well as polyethylene glycol (PEG) and chitosan. These polymers provide tunable degradation rates, biocompatibility, and controllable drug release profiles.
-
What factors influence drug release kinetics in polymer-based delivery systems?
Drug release kinetics in polymer systems depend on several factors, including polymer composition, molecular weight, degradation rate, drug–polymer interactions, particle size, and system architecture. Environmental conditions such as pH, temperature, and biological enzymes can also affect release behavior and overall therapeutic performance.
-
Why are biodegradable polymers widely used in controlled drug delivery?
Biodegradable polymers are widely used because they can gradually degrade into biocompatible byproducts within the body, eliminating the need for surgical removal. Materials such as PLA, PLGA, and PCL allow predictable degradation rates, controlled drug release profiles, and improved safety, making them ideal for long-acting and implantable drug delivery systems.
-
What are the advantages of sustained release polymer drug delivery systems?
Sustained release polymer systems provide several therapeutic benefits, including prolonged drug activity, reduced dosing frequency, improved patient compliance, and more stable drug concentrations. These systems can also enhance drug stability, minimize systemic toxicity, and enable the delivery of sensitive molecules such as peptides, proteins, and nucleic acids.
-
Which drug types benefit most from controlled release polymer systems?
Controlled release polymer systems are particularly beneficial for drugs that require long-term administration or stable plasma concentrations. These include anticancer drugs, hormones, peptides, proteins, vaccines, and anti-inflammatory agents. Polymer carriers help improve bioavailability, reduce side effects, and support precision drug delivery.

 Fig. 1. Mechanisms and applications of controlled and sustained release polymers (BOC Sciences Authorized).
Fig. 1. Mechanisms and applications of controlled and sustained release polymers (BOC Sciences Authorized). Fig. 2. Classification and mechanisms of polymers in controlled drug delivery (BOC Sciences Authorized).
Fig. 2. Classification and mechanisms of polymers in controlled drug delivery (BOC Sciences Authorized).