Polymer Nanoparticles for Drug Delivery: Formulation Strategies and Applications
The pharmaceutical landscape is increasingly shifting away from conventional systemic administration toward sophisticated, localized, and smart delivery systems. Among the most versatile tools in this domain are polymer nanoparticles (PNPs). Unlike pre-formed solid implants or simple solutions, PNPs represent a class of colloidal drug delivery systems that offer precise control over pharmacokinetics and biodistribution. These systems are engineered to overcome historic challenges such as poor bioavailability, rapid renal clearance, and off-target toxicity, serving as dynamic reservoirs that modulate the therapeutic index of encapsulated payloads.
Resources
Defining Polymer Nanoparticles
Polymer nanoparticles are solid, colloidal particles ranging in size from 10 to 1000 nm. Fundamentally, they act as carriers where the active pharmaceutical ingredient (API) is dissolved, entrapped, encapsulated, or chemically attached to a polymer matrix. Distinct from hydrogels which are water-swollen networks, PNPs are typically defined by a compact structure that can be formulated as nanospheres (matrix systems where the drug is physically dispersed) or nanocapsules (reservoir systems consisting of a polymeric shell surrounding an oily or aqueous core). This structural versatility allows them to encapsulate a wide variety of therapeutics, from hydrophobic small molecules to hydrophilic biologics.
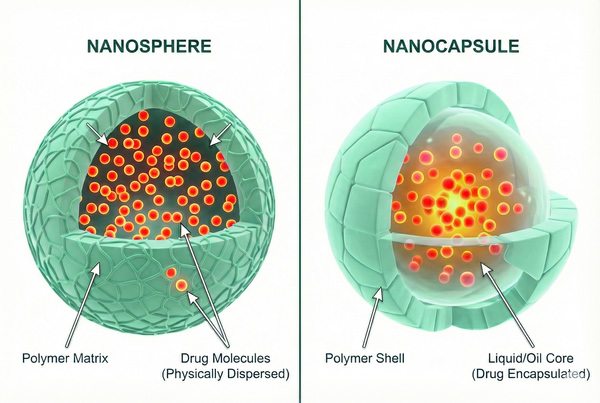 Fig. 1. 3D cross-sectional comparison illustrating differences between polymer nanospheres and nanocapsules (BOC Sciences Authorized).
Fig. 1. 3D cross-sectional comparison illustrating differences between polymer nanospheres and nanocapsules (BOC Sciences Authorized).
Key Mechanisms of Action
The efficacy of PNPs relies heavily on their ability to reach the target site. Passive targeting exploits the enhanced permeability and retention (EPR) effect, particularly in oncology. Tumor blood vessels are often leaky with wide fenestrations, allowing nanoparticles to accumulate in the tumor tissue, while lymphatic drainage is impaired, aiding retention. Active targeting, conversely, involves surface engineering of the nanoparticle with ligands (such as antibodies or peptides) that bind specifically to receptors overexpressed on diseased cells (e.g., CD44 receptors). This dual approach maximizes efficacy at the target while minimizing systemic exposure.
Key Advantages of Polymer Nanoparticles for Drug Delivery
- Protection of Drugs from Degradation: The polymer matrix serves as a physical barrier, shielding sensitive cargo such as proteins, peptides, and nucleic acids from enzymatic degradation (proteases and nucleases) in the physiological environment.
- Controlled and Sustained Release: Release kinetics are governed by diffusion through the polymer matrix and erosion of the matrix itself, allowing for zero-order release profiles that maintain plasma concentrations within the therapeutic range for extended periods.
- Enhanced Solubility and Bioavailability of Therapeutics: Hydrophobic drugs can be partitioned into the hydrophobic domains of the nanoparticle (e.g., the core of a micelle), significantly enhancing their apparent solubility and retention.
- Targeted Delivery and Cellular Uptake Enhancement: Through size optimization and surface functionalization, PNPs can improve cellular internalization via endocytosis and facilitate intracellular trafficking.
Classification of Polymers Used in Drug Delivery Systems
The design of an effective delivery system begins with the selection of the appropriate polymer backbone, as this dictates the degradation kinetics, mechanical stiffness, and biological interaction of the carrier. Polymers are generally categorized by their origin and biodegradability, with the choice often depending on the required therapeutic window and the route of administration.
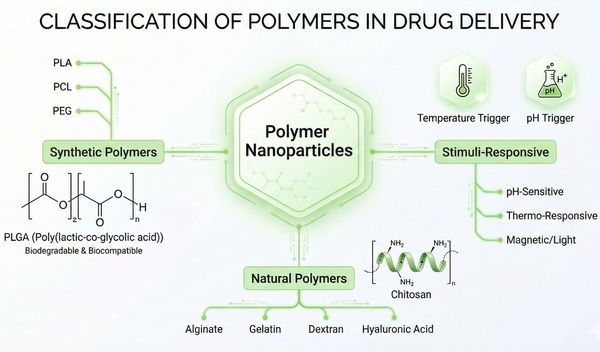 Fig. 2. Mind map classifying synthetic, natural, and stimuli-responsive polymers for drug delivery (BOC Sciences Authorized).
Fig. 2. Mind map classifying synthetic, natural, and stimuli-responsive polymers for drug delivery (BOC Sciences Authorized).
Synthetic biodegradable polymers represent the frontier of controlled delivery technologies, heavily favored for their batch-to-batch consistency and the ability to precisely tune their physicochemical properties through chemical synthesis. Engineering synthetic polymers offers superior control over molecular weight, polydispersity, and reproducible mechanical properties, making them the preferred choice for strictly controlled drug delivery systems requiring long-term stability.
PLGA (Poly(lactic-co-glycolic acid)) and PLA (Polylactic acid) are extensively utilized. These synthetic polyesters degrade via the hydrolytic cleavage of ester bonds, producing lactic acid and glycolic acid. These degradation products subsequently enter the Krebs cycle and are safely eliminated as CO2 and water, ensuring a definitive advantage in clearing the body without toxic accumulation in the liver or spleen. A distinct formulator advantage of PLGA is the tunability of its degradation and release kinetics by modulating the Lactide to Glycolide (LA/GA) ratio. A higher glycolide content generally increases hydrophilicity and accelerates hydrolysis rates, whereas a higher lactide content prolongs the degradation profile, allowing for customized therapeutic windows. Furthermore, PCL (Polycaprolactone) serves as another critical synthetic option. PCL exhibits a significantly slower degradation rate and high lipophilicity, making it an optimal backbone for highly permeable, ultra-long-term delivery systems.
Natural polymers are derived from biological sources and possess intrinsic biocompatibility, often mimicking the mammalian extracellular matrix (ECM) and offering unique cell-signaling capabilities. While they may require rigorous quality control to mitigate batch-to-batch variability and rapid enzymatic degradation, their natural origin ensures exceptional tolerability.
Chitosan, derived from the deacetylation of chitin, is a cationic linear polysaccharide featuring amine groups with a pKa of approximately 6.5. It is particularly noted for its mucoadhesive properties, making it an ideal candidate for transmucosal drug delivery systems. Alginate, an anionic polysaccharide, undergoes a rapid, non-covalent ionotropic gelation process in the presence of divalent cations like Ca2+ or Mg2+. This exceptionally mild crosslinking environment is highly friendly to sensitive biologics and structural proteins. Protein-based biopolymers, such as Gelatin and Albumin, bridge the gap with natural bioactivity. Gelatin intrinsically contains RGD sequences for cell attachment, providing versatile platforms for bioactive encapsulation. Albumin nanoparticles are highly valued for their endogenous transport pathways and abundant functional surface groups that facilitate high drug loading and targeted surface modifications.
Non-Biodegradable Polymers
While biodegradable systems are universally preferred for continuous systemic administration to ensure polymer residues fall below the renal filtration threshold and avoid accumulation, non-biodegradable polymers occupy highly specialized functional niches. Polymers such as PMMA (Polymethyl methacrylate) and Polystyrene are not broken down into non-toxic metabolites via hydrolysis or enzymatic action.
Instead, they provide extreme structural rigidity and absolute resistance to physiological degradation. In the realm of nanoparticulate systems, these materials are predominantly leveraged for ex vivo diagnostics, calibration standards in flow cytometry, and highly specialized localized delivery devices where the matrix is either superficially applied or physically retrieved post-treatment. Their unyielding structural integrity ensures that particle size, morphology, and surface charge remain completely static during the assay or localized application, providing predictable performance where biodegradation would otherwise introduce unwanted variables.
Functionalized and Stimuli-Responsive Polymers
The evolution of smart polymeric systems enables spatiotemporal control over drug release, as these materials undergo abrupt physiochemical changes in response to specific environmental triggers. pH-sensitive polymers are engineered with ionizable pendant groups, such as carboxylic acids or amines. A transition from formulation pH to physiological or microenvironmental pH triggers protonation or deprotonation, leading to electrostatic repulsion, changes in hydrodynamic volume, and subsequent payload release. This mechanism is highly critical for targeting the acidic tumor microenvironment or protecting drugs through the gastrointestinal tract.
Temperature-sensitive polymers typically rely on a lower critical solution temperature (LCST) mechanism. Below the LCST, hydrogen bonding with water molecules dominates, maintaining a highly solvated solution state; however, as the temperature rises to body temperature, hydrophobic interactions prevail, driving the dehydration of hydrophobic blocks and causing rapid structural aggregation. Additionally, enzyme-responsive and reactive oxygen species (ROS)-responsive polymers are designed with specific cleavable peptide sequences or functional bonds. These highly targeted networks remain structurally intact in systemic circulation but rapidly degrade to release their therapeutic cargo only when encountering the elevated concentrations of specific enzymes present at the localized disease site.
Looking for Custom Polymer Nanoparticles?
Whether you need biodegradable nanoparticles, surface-functionalized carriers, or stimuli-responsive platforms, our experts provide tailored synthesis and optimization solutions to support your research and therapeutic development.
Key Formulation Strategies for Polymer Nanoparticles
The transition from a promising polymer candidate to a functional drug delivery system requires a rigorous formulation strategy. This phase demands a delicate balance between thermodynamic stability, rheological performance, and pharmacokinetic targets. Successful design hinges on optimizing the interplay between the physicochemical properties of the drug payload and the network architecture of the polymer. Formulation scientists must select appropriate manufacturing techniques based on the hydrophobicity, molecular weight, and required release profile of the therapeutic agent.
Solvent Evaporation and Solvent Diffusion Methods
Achieving high encapsulation efficiency and minimizing the burst release effect are the primary challenges in formulation. The loading strategy must be tailored to the drug hydrophobicity and molecular weight. For hydrophobic small molecules, solvent evaporation and diffusion methods are highly effective. These techniques allow the therapeutic agent to partition into the hydrophobic domains of the polymer matrix, which significantly enhances drug solubility and retention. By carefully controlling the evaporation or diffusion rate of the organic solvent, formulators can dictate the final mesh size of the resulting polymer network, acting as a molecular sieve that governs the diffusion rate of the encapsulated therapeutics.
Nanoprecipitation (Interfacial Deposition)
Nanoprecipitation is a highly reproducible method designed for rapid, single-step nanoparticle formation. It leverages the instantaneous precipitation of a polymer when an organic solution is introduced into an aqueous anti-solvent. This method is highly advantageous for scaling up production while maintaining batch-to-batch consistency, a crucial factor for synthetic biodegradable block copolymers. The rapid interfacial deposition results in a dense polymer matrix that can physically entrap hydrophilic macromolecules within its structure. Adjusting parameters such as polymer concentration during this process allows formulators to modify the particle size and mitigate premature diffusion.
Emulsification-Polymerization
Instead of utilizing pre-formed polymer chains, emulsification-polymerization constructs the nanoparticle directly from monomeric precursors in situ. This strategy often relies on chemical crosslinking, formed through permanent covalent bonds via specific chemical reactions. Chemical crosslinking allows for precise control over the structural mesh size and swelling ratio, offering robust stability. To further prevent premature leakage during the polymerization process, drugs can be covalently tethered to the polymer backbone via cleavable linkers such as ester, hydrazone, or disulfide bonds. This converts the release mechanism from simple diffusion to a highly controlled hydrolysis process.
Self-Assembly and Micellar Formulations
The self-assembly of amphiphilic triblock copolymers, such as PLGA-PEG-PLGA, represents a sophisticated approach to nanoparticle formulation. In aqueous environments, the hydrophilic blocks, such as PEG, remain solvated and stabilize the exterior. Simultaneously, thermodynamic forces drive the hydrophobic blocks, such as PLGA, to undergo dehydration and associate into dense micellar cores. At critical concentrations, these individual micelles aggregate to form stable, supramolecular nanostructures. This core-shell micelle structure is exceptionally efficient for encapsulating hydrophobic drug molecules within the core while utilizing the hydrophilic shell to ensure systemic stability.
Surface Modification and Functionalization Techniques
The foundational polymer matrix provides the structural architecture for drug encapsulation, but the external surface properties ultimately dictate the fate of the nanoparticle within a biological environment. Surface modification and functionalization are critical engineering steps designed to optimize the interface between the delivery system and surrounding tissues. By meticulously altering the exterior chemistry, formulation scientists can prevent rapid clearance, direct the payload to specific cellular addresses, and ensure the structural integrity of the carrier remains intact until it reaches the target site.
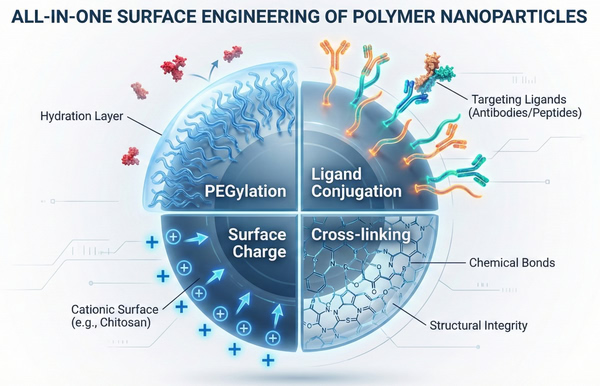 Fig. 3. 3D visualization of four multifunctional surface engineering strategies for polymer nanoparticles (BOC Sciences Authorized).
Fig. 3. 3D visualization of four multifunctional surface engineering strategies for polymer nanoparticles (BOC Sciences Authorized).
PEGylation Strategies
The physiological environment actively identifies and clears foreign materials. To combat this, the nanoparticle surface must be engineered to resist non-specific protein adsorption, which serves as the primary precursor to immune system recognition and subsequent clearance. PEGylation, the process of conjugating polyethylene glycol (PEG) chains to the polymer exterior, is the most widely adopted strategy to overcome this hurdle. The hydrophilic PEG blocks dominate the surface interface, creating a dense hydration layer that provides significant steric hindrance. This modification effectively shields the nanoparticle from opsonization, thereby delaying phagocytosis by the reticuloendothelial system and drastically extending the systemic circulation half-life of the therapeutic payload.
Ligand Conjugation for Active Targeting
While extended circulation allows nanoparticles to accumulate in target tissues passively, active targeting strategies utilize surface-bound ligands to achieve precise cellular interactions. Advanced formulation strategies involve incorporating specific affinity ligands into the polymer network. By conjugating targeting moieties such as monoclonal antibodies, aptamers, or specific peptides to the nanoparticle exterior, the carrier can bind directly to receptors overexpressed on diseased cells. For instance, modifying the surface with hyaluronic acid leverages its natural affinity for CD44 receptors, allowing for the highly efficient active targeting of CD44-overexpressing tumor cells. This receptor-mediated interaction significantly enhances cellular internalization via endocytosis.
Modulating Surface Charge (Zeta Potential)
The Zeta potential of a nanoparticle reflects its surface charge, a critical parameter that influences both the physical stability of the colloidal dispersion and its biological behavior. Modulating this charge is often achieved by selecting polymers with specific ionizable groups. Cationic modifications are particularly valuable; polymers with cationic functional groups, such as the amine groups found in chitosan, readily interact electrostatically with negatively charged cell membranes to promote cellular uptake. Furthermore, this positive surface charge is absolutely essential for nucleic acid delivery, as the cationic groups interact electrostatically with the negatively charged phosphate backbone of DNA or RNA to form stable polyplexes.
Cross-linking Strategies
Cross-linking techniques are employed to reinforce the physical stability of the polymer nanoparticle and fine-tune its release kinetics. This can be achieved through physically crosslinked networks formed via non-covalent interactions, such as ionic interactions or hydrophobic associations. For more robust stability, chemically crosslinked networks are formed through permanent covalent bonds utilizing reactions such as Michael-type addition, Schiff base formation, or photopolymerization. Chemical crosslinking allows for highly precise control over the structural mesh size. Because the mesh size of the resulting network acts as a molecular sieve that dictates the diffusion rate of encapsulated therapeutics , tuning the cross-linking density allows formulators to tightly control release kinetics and achieve highly desirable zero-order release profiles.
Critical Characterization Techniques for Quality Control
Comprehensive characterization is essential to predict in vivo performance and ensure the safety of polymer-based delivery systems. Translating a formulation from the bench to functional applications requires addressing critical stability and regulatory hurdles. For formulation scientists, rigorous quality control validates that the nanoparticle dispersion meets strict specifications for size, morphology, surface charge, and payload retention before proceeding to downstream applications.
Particle Size and Morphology Analysis
Dynamic light scattering (DLS) is employed to accurately determine the hydrodynamic diameter and polydispersity index (PDI) of nanoparticles suspended in aqueous solutions. A low PDI indicates a narrow, uniform size distribution, which is critical for maintaining predictable degradation profiles and ensuring consistent cellular interaction. High-resolution imaging techniques, specifically transmission electron microscopy (TEM) and scanning electron microscopy (SEM), are utilized to visualize the solid-state architecture. These electron microscopy methods reveal the precise surface texture and structural morphology, confirming whether the particles form uniform spheres, porous networks, or specific core-shell micelle structures.
Surface Charge and Stability Assessment
Zeta potential measurements are critical for evaluating the electrostatic repulsion between particles, serving as a primary indicator of colloidal stability in suspension. High absolute Zeta potential values (either highly cationic or highly anionic) prevent particle aggregation through strong electrostatic repulsion, ensuring a stable dispersion over time. Long-term stability studies monitor changes in particle size, molecular weight, and drug retention under varied storage conditions to establish a reliable shelf-life. To ensure long-term stability and prevent hydrolysis during storage, aqueous polymer solutions often undergo lyophilization, or freeze-drying. This lyophilization process requires the addition of specific cryoprotectants, such as trehalose or sucrose, to prevent polymer aggregation and maintain the structural integrity upon reconstitution.
Encapsulation Efficiency and Drug Loading Capacity
Encapsulation efficiency quantifies the precise percentage of the initial therapeutic payload that is successfully entrapped within the polymer matrix. Drug loading capacity measures the mass ratio of the encapsulated active ingredient relative to the total mass of the final nanoparticle formulation. Achieving high encapsulation efficiency and minimizing the burst release effect are the primary challenges in formulation design. Optimization involves systematically adjusting parameters such as the polymer concentration, the molecular weight, and the specific loading strategy, selecting between physical entrapment and chemical conjugation based on the payload properties.
In Vitro Drug Release Profiling
Standard dissolution methods, such as dialysis bag techniques or USP Apparatus 4, are utilized to accurately quantify drug release rates over a specified timeframe. These in vitro assays must strictly simulate physiological conditions and frequently require sink conditions to accurately mimic in vivo clearance mechanisms. The release kinetics are governed by two concurrent mechanisms: the diffusion of the drug through the polymer matrix and the continuous erosion of the matrix itself. Experimental data is fitted to mathematical kinetic models, such as the Higuchi model for diffusion-controlled release or the Korsmeyer-Peppas model to evaluate complex mechanisms involving both diffusion and polymer relaxation. By analyzing these release profiles, formulators can perfectly synchronize the degradation rate of the polymer with the desired therapeutic window.
Applications of Polymer Nanoparticles in Drug Delivery
The versatility of polymer matrices has catalyzed their adoption across a broad spectrum of therapeutic areas. By acting as dynamic and programmable reservoirs, these colloidal systems provide an effective solution to overcome critical pharmaceutical challenges, including poor bioavailability, rapid systemic clearance, and off-target toxicity. They offer a robust platform for the precise placement and sustained delivery of diverse therapeutic agents, maximizing efficacy at the target while minimizing systemic exposure.
Small Molecule Drug Delivery
For hydrophobic small molecule drugs, inherent poor solubility and rapid clearance from the target site are persistent hurdles. Polymer nanoparticles, particularly those formulated from amphiphilic block copolymers, address these issues by incorporating hydrophobic drugs directly into their dense micellar cores. This encapsulation strategy significantly enhances apparent solubility. Furthermore, the release is strictly governed by the diffusion of the drug through the polymer matrix and the simultaneous erosion of the polymer backbone. This dual mechanism ensures a sustained, linear release profile, effectively mitigating the burst effect that is typical of simple pharmaceutical suspensions.
Protein and Peptide Therapeutics
Biologics, such as proteins, peptides, and monoclonal antibodies, are notoriously unstable and highly prone to denaturation, aggregation, and enzymatic hydrolysis in physiological environments. Unlike traditional formulations that may expose these sensitive cargo molecules to harsh conditions, specialized polymer nanoparticles provide a gentle, protective encapsulation environment. The tailored internal architecture of hydrophilic or amphiphilic nanoparticles helps maintain the protein's native tertiary structure. This supreme preservation of bioactivity is critical for delivering growth hormones or targeted therapies safely to their intended site of action.
Nucleic Acid (DNA, RNA, mRNA) Delivery
The revolutionary rise of gene therapy and mRNA therapeutics has created an absolute demand for carriers that can protect highly fragile nucleic acids from ubiquitous nucleases. Cationic polymer nanoparticle systems—utilizing materials such as Chitosan or specific polyamine derivatives—interact electrostatically with the negatively charged phosphate backbone of DNA or RNA. This interaction forms highly stable polyplexes within the nanoparticle matrix. Upon reaching the target cell, the polymer architecture facilitates cellular uptake via endocytosis and actively promotes endosomal escape—a highly critical step for ensuring the high transfection efficiency of non-viral gene vectors.
Oncology and Cancer Immunotherapy
Systemic chemotherapy is often heavily limited by severe dose-limiting toxicities, such as cardiotoxicity or neutropenia. Polymer nanoparticles address this by enabling highly localized or actively targeted administration, a strategy that maximizes the therapeutic index. By engineering nanoparticles to accumulate via the EPR effect or active receptor targeting, a high local concentration gradient of chemotherapeutics is created within the tumor mass. This ensures sustained cytotoxicity to cancer cells while keeping systemic plasma concentrations negligible. Additionally, nanoparticles are increasingly used to retain immunomodulators within the tumor microenvironment, potentiating the immune response while preventing severe systemic autoimmune adverse events often associated with immunotherapy.
Vaccine Delivery and Adjuvant Systems
Advanced nucleic acid vaccines and recombinant protein antigens rely on sophisticated delivery carriers to ensure structural integrity and robust immune activation. Polymer nanoparticles act as highly efficient dual-function platforms; they physically protect the fragile antigen payload from premature degradation while simultaneously controlling its presentation to the immune system. The highly tunable physicochemical properties of the polymer backbone allow these nanocarriers to mimic the size and morphology of natural pathogens. This strategic structural mimicry significantly enhances targeted uptake by antigen-presenting cells, making them exceptionally versatile tools for modulating specific cellular pathways and boosting targeted immune responses.
Services
Partnering with BOC Sciences for Polymer Nanoparticle Development
We deliver streamlined polymeric nanoparticle solutions for advanced drug and bioactive delivery. From custom polymer design and nanoparticle formulation to loading optimization, surface functionalization, and performance evaluation, our integrated services help you achieve better stability, higher efficiency, and reliable reproducibility—accelerating your project from concept to application-ready systems.

Polymer Materials Supply for Nanoparticles
- Supply of PEG, PLGA, PLA, PCL, PBAE, and other functionalized polymers
- Support for controlled customization of molecular weight, end groups, and functional moieties
- Custom block, copolymer, branched, or biodegradable polymer architectures
- Introduction of PEG, targeting ligands, or stimuli-responsive groups (pH/redox/enzyme, etc.)

Polymeric Nanoparticle Formulation Development
- Construction of nanospheres, nanocapsules, polymeric micelles, and related systems
- Support for self-assembly, emulsion–solvent evaporation, nanoprecipitation, and other preparation strategies
- Precise control of key parameters such as particle size, PDI, and surface charge
- Optimization of system stability and batch-to-batch reproducibility

Drug/Bioactive Loading & Encapsulation
- Suitable for small molecules, peptides, proteins, and nucleic acids
- Optimization of encapsulation efficiency, drug loading, and distribution uniformity
- Design of controlled-release or triggered-release strategies
- Enhanced stability and delivery efficiency of active ingredients

Characterization & Performance Evaluation
- Particle size, PDI, zeta potential, and morphology analysis (DLS, electron microscopy, etc.)
- Structural characterization of polymers and carriers (NMR, GPC, FTIR)
- Evaluation of drug loading, encapsulation efficiency, and release behavior
- Support for stability studies and in vitro functional performance testing
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
What are polymer nanoparticles used for in drug delivery?
Polymer nanoparticles are primarily utilized to create sophisticated, targeted, and sustained-release drug delivery systems. They serve to overcome historic pharmaceutical challenges such as poor bioavailability, rapid systemic clearance, and off-target toxicity. By acting as dynamic colloidal reservoirs, they allow for the precise delivery of chemotherapeutics, biological proteins, and anti-inflammatory agents directly to specific cellular targets or tissues. This approach significantly improves the therapeutic index of the encapsulated payload and offers a highly versatile alternative to traditional systemic administration or macroscopic surgical implants.
How do polymer nanoparticles enhance drug stability and bioavailability?
These nanocarriers enhance stability by serving as a robust physical barrier that shields sensitive cargo molecules from proteases, nucleases, and harsh physiological environments. For hydrophobic small molecules, polymer nanoparticles significantly enhance bioavailability by partitioning the active ingredients into the hydrophobic domains of the polymer matrix (such as a PLGA core), thereby overcoming inherent solubility issues. Furthermore, surface engineering techniques, such as PEGylation, create a hydration layer that prevents non-specific protein adsorption, delaying immune clearance and extending the systemic circulation half-life of the drug.
Can polymer nanoparticles deliver proteins, peptides, or nucleic acids?
Yes, polymer nanoparticles are exceptionally well-suited for the delivery of complex biologics. Unlike rigid systems that may require harsh organic solvents, properly formulated hydrophilic or amphiphilic polymer matrices provide a protective encapsulation environment that prevents protein denaturation and aggregation. For gene therapy applications, polymers with cationic functional groups (such as chitosan or specific polypeptides) interact electrostatically with the negatively charged phosphate backbone of DNA or RNA. This interaction forms highly stable polyplexes that protect the nucleic acids from enzymatic degradation and actively facilitate cellular uptake and endosomal escape.
How are polymer nanoparticles customized for specific therapeutic applications?
Formulation scientists customize these delivery systems by meticulously adjusting foundational polymer parameters, including molecular weight, polymer concentration, and the specific ratio of hydrophobic to hydrophilic blocks (such as the Lactide to Glycolide ratio in PLGA). These structural modifications precisely tune the degradation rate, the mechanical integrity of the particle, and the payload release kinetics to match the specific biological requirements of the target tissue. Additionally, the surface can be functionalized with specific targeting ligands to enable precise receptor-mediated cellular interactions.
What drug types are best suited for polymer nanoparticles?
The architectural versatility of polymer nanoparticles makes them suitable for a remarkably broad spectrum of therapeutic agents. Hydrophobic small molecules are highly suitable, as they can be efficiently partitioned into the hydrophobic micellar cores. Hydrophilic macromolecules, including proteins and therapeutic antibodies, are also excellent candidates and are typically incorporated via physical entrapment within the aqueous or hydrophilic phases of the polymer network. Finally, negatively charged molecules like mRNA and siRNA are ideally suited for complexation with cationic polymer systems.
How do polymer nanoparticles compare to lipid nanoparticles?
While both are premier colloidal delivery systems, they offer distinct formulation advantages. Lipid nanoparticles excel in rapid cell membrane fusion and are the current industry standard for specific mRNA vaccine applications. However, polymer nanoparticles generally offer superior physical stability, a much higher degree of structural rigidity, and more highly predictable, tunable degradation profiles. The diverse chemistry of synthetic and natural polymers allows for highly complex covalent surface modifications and cross-linking strategies, making polymer nanoparticles the preferred choice when ultra-long-term sustained release or highly specific structural functionalization is required.
What factors influence in vivo performance and safety?
In vivo performance and safety are fundamentally dictated by the biocompatibility and biodegradability of the selected polymer. The polymer material must be chemically inert and non-immunogenic. A critical safety factor is ensuring that the polymer degradation products degrade into non-toxic metabolites and eventually fall below the renal filtration threshold (typically less than 40-60 kDa) to guarantee clearance from the body without toxic accumulation in the liver or spleen. Additionally, particle size, surface charge, and the presence of stealth coatings directly influence the circulation time, cellular uptake efficiency, and the potential for triggering a foreign body response.

 Fig. 1. 3D cross-sectional comparison illustrating differences between polymer nanospheres and nanocapsules (BOC Sciences Authorized).
Fig. 1. 3D cross-sectional comparison illustrating differences between polymer nanospheres and nanocapsules (BOC Sciences Authorized). Fig. 2. Mind map classifying synthetic, natural, and stimuli-responsive polymers for drug delivery (BOC Sciences Authorized).
Fig. 2. Mind map classifying synthetic, natural, and stimuli-responsive polymers for drug delivery (BOC Sciences Authorized). Fig. 3. 3D visualization of four multifunctional surface engineering strategies for polymer nanoparticles (BOC Sciences Authorized).
Fig. 3. 3D visualization of four multifunctional surface engineering strategies for polymer nanoparticles (BOC Sciences Authorized).