Targeted Polymer Drug Delivery Systems: Site-Specific Therapy and Controlled Release
Targeted polymer drug delivery systems represent a transformative approach in modern therapeutics, enabling precise site-specific therapy and highly controlled drug release. By leveraging advanced polymer chemistry, functional design strategies, and smart material engineering, these systems can improve drug stability, enhance bioavailability, and minimize systemic side effects. From cancer treatment and gene delivery to inflammatory and chronic disease management, polymer-based delivery platforms are reshaping how active pharmaceutical ingredients are transported and released in the body. With increasing demand for precision medicine and optimized therapeutic performance, targeted polymer technologies are becoming a core focus in pharmaceutical research and development, driving innovation in drug formulation, nanomedicine, and next-generation controlled release systems.
Resources
Introduction to Targeted Polymer Drug Delivery Systems
Polymer-based therapeutics represent a transformative frontier in pharmaceutical and materials science, bridging the gap between active pharmaceutical ingredients and their precise biological targets. By engineering macromolecular carriers, researchers can manipulate the pharmacokinetics and biodistribution of therapeutic agents, mitigating systemic toxicity while maximizing local efficacy. This sophisticated approach relies on the programmable nature of synthetic and natural polymers to navigate complex biological barriers, ensuring that the payload reaches the intended site of action with exceptional accuracy.
What Are Targeted Polymer Drug Delivery Systems?
Targeted polymer drug delivery systems are specialized macromolecular constructs designed to transport and release therapeutic compounds exclusively at desired pathological sites. These platforms typically consist of a polymeric backbone that physically encapsulates or chemically conjugates the drug. By tailoring the molecular weight, hydrophobicity, and architectural geometry of the polymer, scientists create nanocarriers that protect the drug from premature degradation in the bloodstream while directing it toward specific cellular or tissue microenvironments.
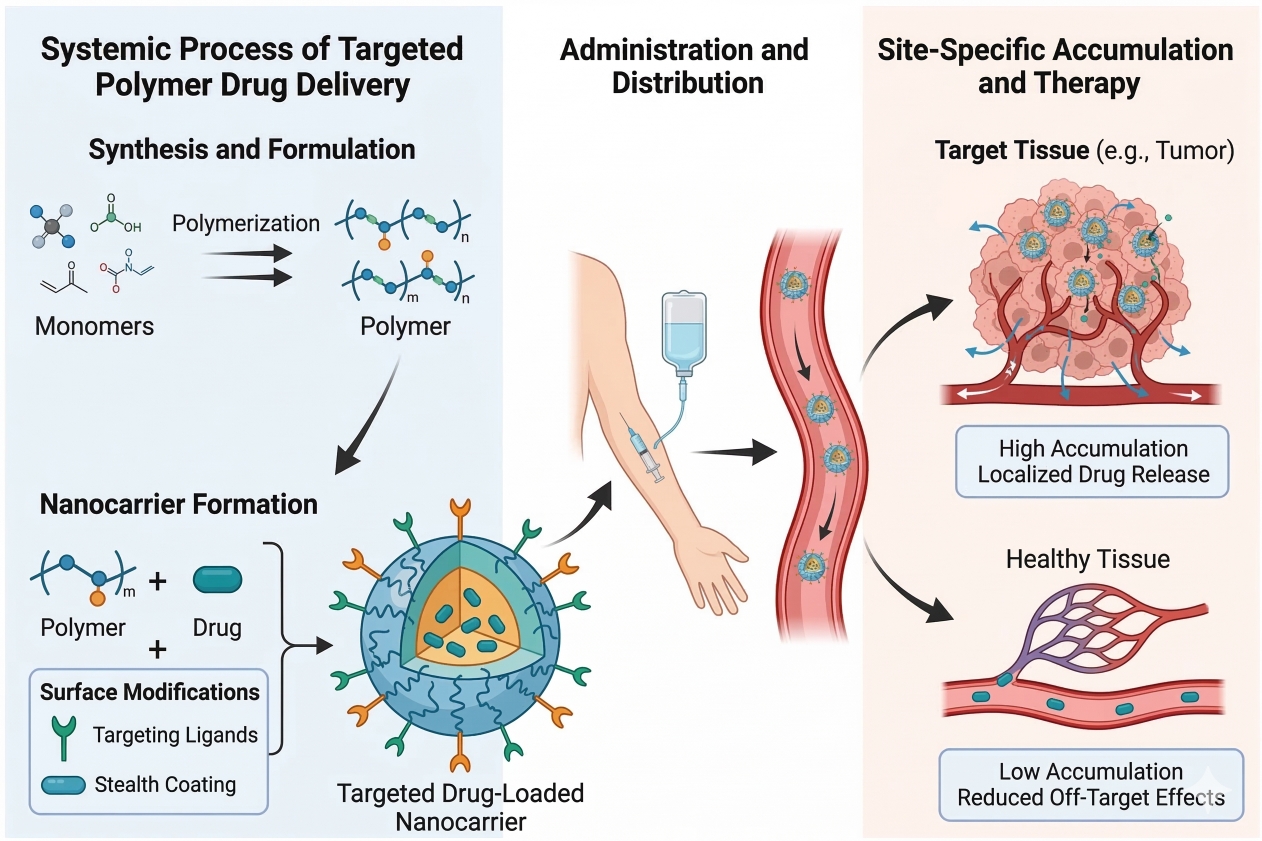 Fig. 1. Schematic of synthesis, administration, and targeted accumulation of polymeric nanocarriers (BOC Sciences Authorized).
Fig. 1. Schematic of synthesis, administration, and targeted accumulation of polymeric nanocarriers (BOC Sciences Authorized).
Why Site-Specific Drug Delivery Matters in Modern Medicine?
Standard pharmacological interventions often suffer from unfavorable biodistribution, where only a microscopic fraction of the administered dose reaches the intended tissue, leaving the remainder to interact indiscriminately with healthy organs. Site-specific delivery fundamentally alters this dynamic. By restricting the pharmacological action to a precise anatomical or cellular locus, researchers can vastly improve the therapeutic index of highly potent molecules, significantly reducing collateral damage to healthy tissues and addressing complex physiological challenges at a molecular level.
Advantages of Polymer-Based Drug Carriers Over Conventional Delivery
Macromolecular carriers offer distinct physicochemical advantages over free drug administration. Polymers vastly improve the aqueous solubility of hydrophobic drugs, stabilize fragile biomolecules against enzymatic cleavage, and prolong circulation half-life by evading rapid renal clearance and reticuloendothelial system recognition. Furthermore, their highly functionalizable surfaces allow for the attachment of targeting moieties and stealth coatings, providing a degree of modularity and precision impossible to achieve with small-molecule formulations alone.
Key Components of a Targeted Polymer Drug Delivery Platform
A sophisticated polymeric delivery vehicle is generally composed of three integral elements. The core is the active therapeutic payload, ranging from small molecules to complex nucleic acids. The carrier itself is the polymeric scaffold, which determines the structural integrity, loading capacity, and release kinetics. Finally, the surface often features targeting ligands, such as antibodies, peptides, or aptamers, alongside stabilizing hydrophilic chains that prevent protein aggregation and subsequent immune clearance.
Mechanisms of Targeted Drug Delivery Using Polymers
Achieving precise spatial localization requires a deep understanding of both human pathophysiology and nanocarrier transport physics. Polymers can be engineered to exploit unique biological phenomena or actively seek out specific cellular signatures, thereby accumulating selectively in diseased tissues rather than healthy ones.
Passive Targeting Through the Enhanced Permeability and Retention (EPR) Effect
Many rapidly growing pathological tissues, particularly solid tumors, exhibit uniquely defective vascular architecture characterized by large endothelial fenestrations and impaired lymphatic drainage. Polymeric nanocarriers within a specific size range generally between 10 and 200 nanometers can exploit this anomaly by extravasating through the leaky vasculature and accumulating in the interstitial space. This phenomenon, known as the enhanced permeability and retention effect, serves as the foundational mechanism for the passive accumulation of macromolecular therapeutics.
Active Targeting via Ligand–Receptor Interactions
To achieve a higher degree of specificity beyond passive accumulation, polymeric surfaces can be decorated with specific affinity molecules. Active targeting involves the conjugation of ligands that recognize and bind to overexpressed receptors on the surface of target cells. Once the nanocarrier binds to the complementary receptor, it frequently triggers receptor-mediated endocytosis, facilitating the direct cellular internalization of the macromolecular complex and drastically increasing the intracellular concentration of the active payload.
Stimuli-Responsive Targeting Strategies for Controlled Drug Release
Advanced polymeric carriers can be synthesized to remain structurally inert until they encounter specific biochemical triggers inherent to the target microenvironment. Diseased tissues often exhibit unique physiological hallmarks, such as localized hypoxia, elevated reactive oxygen species, or distinct enzymatic profiles. By incorporating chemical linkages that cleave or change conformation exclusively in the presence of these specific local stimuli, the polymer ensures that payload release is highly restricted to the targeted area.
Intracellular Targeting and Organelle-Specific Delivery
Reaching the correct cell is often only the first hurdle; many therapeutics, particularly biologics and genetic materials, must reach specific subcellular compartments like the nucleus or mitochondria to exert their effects. Polymeric systems can be engineered with endosomolytic properties, allowing them to rupture endosomal membranes after internalization and escape into the cytosol. Further functionalization with organelle-targeting signaling peptides ensures the ultimate delivery of the payload to its precise subcellular destination.
Controlled Drug Release Mechanisms in Polymer Systems
The temporal control of an active pharmaceutical ingredient is just as critical as its spatial targeting. Polymeric networks manage the release profile through a variety of thermodynamic, kinetic, and chemical processes, ensuring the payload is deployed at a therapeutic rate over a desired timeframe.
Diffusion-Controlled Drug Release
Diffusion is the most fundamental mechanism governing the elution of drugs from a polymeric matrix or reservoir. In these systems, the release rate is determined by the concentration gradient and the physical constraints of the polymer network. The drug molecules must navigate through the tortuous pores or the macromolecular mesh of the polymer. By adjusting the cross-linking density and hydration characteristics of the polymer, scientists can precisely tune the diffusion coefficient, resulting in predictable, sustained release profiles.
Polymer Degradation and Erosion Mechanisms
Many therapeutic applications utilize biodegradable polymers that physically break down to release their cargo. This can occur through surface erosion, where the polymer matrix degrades uniformly from the exterior inwards, offering zero-order release kinetics. Alternatively, bulk degradation involves the simultaneous scission of polymer chains throughout the entire matrix, leading to a sudden burst release once a critical molecular weight threshold is breached. Selecting the appropriate erosion mechanism is essential for matching the release timeline to the required therapeutic window.
pH-Responsive and Enzyme-Responsive Drug Release
Chemical strategies often utilize the body's natural gradients to trigger payload release. pH-responsive polymers take advantage of the acidic microenvironments found in endosomes, lysosomes, or specific pathological tissues. These polymers may contain acid-labile bonds that quickly degrade at lower pH levels, or ionizable groups that induce rapid swelling and payload discharge. Similarly, enzyme-responsive systems incorporate specific peptide sequences that are exclusively recognized and cleaved by proteases locally overexpressed at the target site.
External Stimuli-Triggered Drug Delivery Systems
Beyond utilizing inherent biological triggers, polymers can be engineered to respond to exogenous physical stimuli applied by a practitioner. Materials integrating magnetic nanoparticles, photo-responsive chromophores, or thermo-sensitive blocks can change their physical state in response to external magnetic fields, near-infrared light, or focused ultrasound. This approach offers unparalleled temporal control, allowing for on-demand pulsatile release precisely when the external stimulus is applied.
Types of Polymers Used in Targeted Drug Delivery Systems
Selecting the appropriate macromolecular backbone is the most critical decision in designing a targeted delivery vehicle. By carefully modulating molecular weight, polydispersity indices, and monomeric composition, material scientists can engineer sophisticated polymeric matrices that perfectly align with the precise biophysical and spatial requirements of the intended cellular target.
Biodegradable Polymers for Drug Delivery Applications
Biodegradable polymers are foundational to modern controlled-release architectures because they are designed to safely undergo chemical breakdown within physiological environments, eliminating the need for subsequent surgical removal. Aliphatic polyesters, such as polylactic acid, polyglycolic acid, and their copolymeric derivatives, are extensively utilized due to their highly predictable ester bond hydrolysis. By adjusting the stoichiometric ratio of crystalline to amorphous monomeric units, formulation scientists can precisely tune the rate of water penetration and subsequent polymer chain scission. Furthermore, materials like polyanhydrides are specifically selected when strict surface-erosion is required, allowing for a steady, zero-order release of the payload as the polymer degrades uniformly from the exterior interface inward, contrasting with the bulk erosion typical of many polyesters.
Synthetic Polymers for Precision Drug Transport
Synthetic polymers offer an unparalleled degree of structural and chemical control, enabled by advanced macromolecular engineering techniques such as reversible deactivation radical polymerization. Methods including atom transfer radical polymerization (ATRP) and reversible addition-fragmentation chain-transfer polymerization (RAFT) allow chemists to synthesize chains with extremely narrow molecular weight distributions and complex, predetermined architectures. Polyethylene glycol remains the gold standard for creating a hydrophilic steric barrier around nanocarriers, a stealth corona that effectively suppresses opsonization and evades rapid clearance by the mononuclear phagocyte system. Beyond steric shielding, synthetic backbones like polyacrylamides and polyoxazolines provide robust, highly functionalizable scaffolds that can be modified with exact stoichiometric precision for complex drug conjugation and ligand attachment.
Natural Polymers and Their Biomedical Advantages
Polysaccharides and protein-based biopolymers derived from natural sources provide exceptional inherent biocompatibility coupled with unique biological recognition capabilities. Polyanionic natural polymers, such as hyaluronic acid, possess an intrinsic binding affinity for specific cellular receptors overexpressed in numerous pathological states, simultaneously acting as both the structural carrier and the active targeting moiety. Conversely, polycationic natural polymers like chitosan offer a high density of primary amines, facilitating strong electrostatic interactions with negatively charged cellular membranes and mucosal surfaces to significantly enhance tissue permeation. The utility of natural polymers is further amplified by their susceptibility to site-specific enzymatic degradation, ensuring that the carrier matrix is dismantled precisely when it encounters the distinct enzymatic profiles localized at the target site.
Polymeric Micelles, Nanoparticles, and Dendrimers
The geometric configuration and supramolecular assembly of polymers drastically alter their pharmacokinetic behavior and payload capacity. Amphiphilic block copolymers spontaneously self-assemble in aqueous media above their critical micelle concentration, forming polymeric micelles that feature a dense, hydrophobic core ideal for solubilizing highly lipophilic compounds, surrounded by a stabilizing hydrophilic shell. Polymeric nanoparticles provide a more rigid, solid-state matrix that can encapsulate both hydrophilic and hydrophobic entities within a protective polymeric mesh. Dendrimers represent a distinct class of highly branched, architecturally perfect macromolecules synthesized via rigorous, step-growth iterative processes. This step-wise synthesis generates a perfectly monodisperse structure with a precisely controlled number of surface functional groups, offering exceptional multivalency for dense targeting ligand presentation and internal voids for secure payload sequestration.
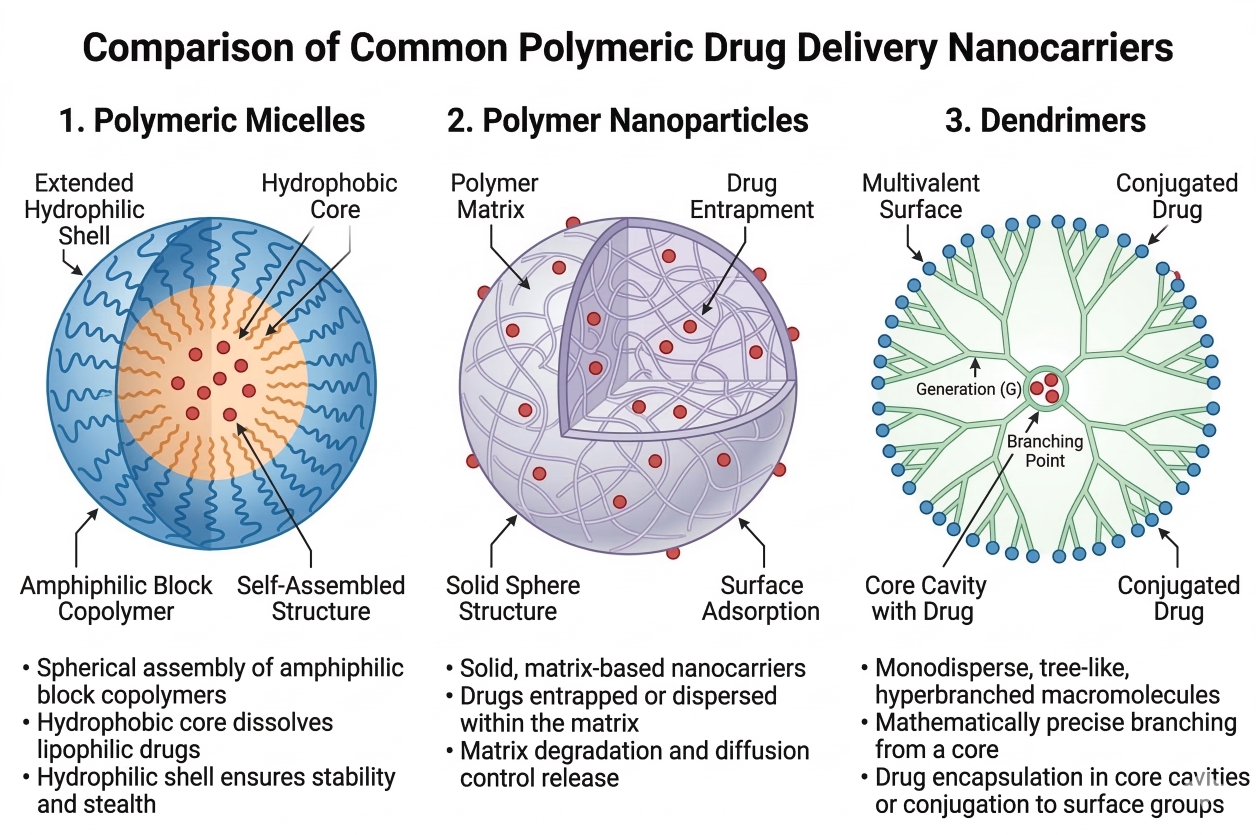 Fig. 2. Comparative overview of polymeric micelles, nanoparticles, and dendrimers (BOC Sciences Authorized).
Fig. 2. Comparative overview of polymeric micelles, nanoparticles, and dendrimers (BOC Sciences Authorized).
Stimuli-Responsive Polymers for Smart Drug Release
Smart polymeric systems are engineered to undergo rapid, reversible, and sharp physicochemical transitions in response to microscopic fluctuations in their immediate microenvironment. Thermoresponsive polymers are designed with a specific lower critical solution temperature carefully calibrated to physiological thresholds; upon encountering localized temperature variations, these polymer chains undergo a dramatic coil-to-globule phase transition, effectively squeezing out the encapsulated payload. Similarly, pH-responsive systems incorporate ionizable functional groups, such as carboxylic acids or tertiary amines, that undergo rapid protonation or deprotonation across physiological pH gradients. This ionization alters the internal electrostatic repulsion within the polymer network, triggering massive structural swelling or complete matrix dissolution precisely upon entry into acidic subcellular compartments like endosomes and lysosomes.
Looking for Targeted Polymer Drug Delivery Solutions?
Whether you need ligand-conjugated polymers, tumor-targeting carriers, or precision-engineered delivery systems, our experts provide tailored synthesis and functionalization solutions to support your research and therapeutic development.
Design Strategies for Site-Specific Polymer Drug Delivery
Translating raw macromolecular materials into sophisticated, site-specific delivery vehicles requires a highly meticulous approach that bridges synthetic chemistry and biophysics. By systematically engineering the overarching polymer topology, meticulously tuning the surface chemistry, and precisely controlling the internal payload distribution, formulation scientists can construct highly autonomous nanocarriers. These engineered systems are capable of navigating systemic circulation, evading premature immune clearance, and localizing their chemical payload precisely at the intended microscopic locus with exceptional reliability.
Polymer Architecture: Linear, Branched, and Dendritic Structures
The topological arrangement of the polymer chain exerts a profound influence on the hydrodynamic volume, rheological behavior, and steric profile of the resulting nanocarrier. Linear polymers, characterized by a single continuous backbone, offer straightforward synthetic pathways and predictable folding dynamics, making them highly suitable for fundamental steric shielding and basic matrix formation. Branched architectures, encompassing star-shaped and comb-like topologies, introduce a higher degree of steric hindrance and alter the solution viscosity, providing multiple terminal functional groups for subsequent chemical modification. Dendritic structures represent the pinnacle of precise architectural control. Synthesized through rigorous, iterative step-growth processes, dendrimers are perfectly monodisperse, highly branched macromolecules. They offer a mathematically defined number of peripheral functional groups, enabling ultra-dense, multivalent presentation of targeting ligands, while their internal hydrophobic cavities provide highly uniform compartments for precise payload sequestration.
Surface Functionalization for Targeting Ligands
The exterior interface of a polymeric vehicle serves as its primary communication module with the biological environment, dictating both its stealth capabilities and its active targeting avidity. Surface functionalization involves the covalent attachment of specific biological recognition moieties, such as peptides, aptamers, or engineered proteins, to the polymer corona. To ensure the targeting ligand retains its native, active conformation, chemists employ highly efficient and bioorthogonal conjugation techniques, including azide-alkyne cycloaddition, maleimide-thiol coupling, and N-hydroxysuccinimide ester reactions. A critical design parameter is optimizing the ligand density on the carrier surface. Insufficient functionalization leads to weak receptor binding, whereas excessive ligand crowding can drastically compromise the protective stealth layer, inadvertently triggering rapid opsonization and subsequent recognition by the mononuclear phagocyte system.
Polymer–Drug Conjugation Approaches
In scenarios where physical encapsulation yields insufficient payload stability or premature leaching, formulation scientists employ polymer-drug conjugation strategies. This approach involves covalently tethering the active pharmaceutical ingredient directly to the polymeric backbone, transforming the entire construct into a macromolecular prodrug. The success of this strategy hinges entirely on the rational design of the intermediary spacer or linker molecule. This chemical bridge must possess absolute thermodynamic stability during systemic transit to prevent off-target toxicity, yet it must be highly labile upon reaching the target microenvironment. Common strategies involve integrating stimuli-responsive linkers, such as hydrazone bonds that cleave rapidly in acidic subcellular compartments, or disulfide bridges that undergo rapid reduction in the presence of elevated intracellular glutathione concentrations, ensuring the therapeutic agent is liberated in its fully active, unmodified form.
Nanostructure Engineering for Controlled Release Profiles
Beyond molecular-level chemistry, the macroscopic physical engineering of the polymer matrix determines the precise temporal deployment of the therapeutic payload. Advanced nanostructure engineering allows for the fabrication of multi-compartmental architectures, such as core-shell nanoparticles, where distinct therapeutic agents can be localized in separate spatial domains for sequential release. By meticulously calibrating the cross-linking density, matrix porosity, and the stoichiometric ratio of hydrophobic to hydrophilic segments, researchers can dictate the rate of aqueous diffusion into the matrix and the subsequent outward migration of the drug. Furthermore, techniques such as layer-by-layer assembly permit the creation of polymeric shells with highly tunable thicknesses, enabling the design of complex kinetic profiles, including delayed-onset release or sustained zero-order elution that precisely matches the required pharmacological concentration curve.
Polymer-Based Drug Delivery Platforms and Nanocarriers
The physical manifestation of a macromolecular delivery strategy relies entirely on the successful formulation of the synthesized polymer into a distinct three-dimensional architecture. These platforms serve as the structural chassis for therapeutic transport, dictating the spatial distribution, physical stability, and ultimate release kinetics of the active payload. By manipulating the supramolecular assembly, phase behavior, or chemical cross-linking of specific polymer chains, formulation scientists can engineer a diverse array of delivery vehicles. Each platform possesses unique biophysical properties meticulously tailored to overcome specific biopharmaceutical barriers and optimize the spatial and temporal delivery profile of complex molecules.
Polymeric Micelles for Hydrophobic Drug Delivery
The formulation of highly lipophilic active ingredients presents a persistent biophysical challenge in pharmaceutical materials science. Polymeric micelles address this limitation by utilizing amphiphilic block copolymers that undergo spontaneous, thermodynamic self-assembly in aqueous environments once their concentration exceeds a specific threshold known as the critical micelle concentration. This supramolecular arrangement yields a highly ordered core-shell architecture. The inner hydrophobic core acts as a robust solubilization reservoir, physically sequestering water-insoluble compounds through intense hydrophobic interactions and intermolecular forces. Concurrently, the hydrophilic corona provides a dense steric barrier, preventing macroscopic aggregation and minimizing non-specific interactions during transit.
Polymer Nanoparticles and Nanospheres
Solid polymeric nanoparticles represent a highly versatile class of colloidal carriers characterized by a dense, kinetically stable matrix. Unlike the dynamic equilibrium that governs micellar structures, nanospheres provide a rigid framework where the therapeutic payload is either physically entrapped within the polymer mesh or homogeneously dispersed throughout the particle volume. Engineered via highly controlled techniques such as nanoprecipitation or complex emulsion solvent evaporation, these structures offer exceptional structural integrity. The elution of the entrapped agent is governed by a predictable combination of aqueous diffusion through the porous matrix and the gradual hydrolytic degradation of the polymer backbone, enabling highly tunable, multiphasic release profiles over extended temporal windows.
Polymer–Drug Conjugates for Targeted Therapy
Shifting away from physical encapsulation, polymer-drug conjugates utilize precise synthetic chemistry to covalently tether the active compound directly to a macromolecular scaffold. This approach essentially creates a high-molecular-weight prodrug, fundamentally altering the solubility, circulation half-life, and biodistribution of the parent molecule. The core design principle relies on the incorporation of highly specific, stimulus-responsive spacer linkages between the polymer backbone and the payload. These chemical bridges are engineered to remain entirely stable in systemic circulation while undergoing rapid, selective cleavage via specific intracellular enzymes or localized reducing environments only upon reaching the targeted subcellular compartment, ensuring strict spatial control over payload activation.
Hydrogels and Injectable Polymer Delivery Systems
For applications requiring highly localized, long-term sustained release, macroscopic three-dimensional polymer networks offer an optimal biophysical solution. Polymeric hydrogels are characterized by their extensive chemical or physical cross-linking, allowing them to absorb and retain massive volumes of aqueous fluids while maintaining distinct structural cohesiveness. Injectable, in situ-forming hydrogels represent a significant advancement in this domain. These systems are formulated as low-viscosity polymeric solutions that undergo a rapid phase transition in response to physiological triggers, such as temperature shifts or ionic variations upon injection. Once solidified at the target anatomical site, the porous macroscopic matrix acts as a substantial therapeutic depot, providing sustained, diffusion-controlled elution of the payload over highly prolonged durations.
Therapeutic Applications of Targeted Polymer Drug Delivery
The continuous evolution of macromolecular engineering has enabled localized intervention across a broad spectrum of complex and highly localized pathologies, radically changing the approach to therapeutic targeting.
Targeted Cancer Therapy and Tumor-Specific Drug Delivery
Solid tumors present a primary target for polymeric delivery platforms. By leveraging both the enhanced permeability and retention effect for passive accumulation and specific overexpressed receptors for active binding, polymers can deliver highly cytotoxic chemotherapeutic agents directly to malignant cells. This site-specific delivery minimizes exposure to highly vulnerable tissues such as bone marrow and cardiac muscle, fundamentally improving the safety and tolerance profiles of rigorous oncological interventions.
Polymer Drug Delivery in Gene and Nucleic Acid Therapies
Nucleic acids, including short interfering RNA and messenger RNA, are highly unstable and require robust protection to prevent rapid degradation by extracellular nucleases. Cationic polymers effectively condense these negatively charged genetic materials into stable polyplexes. These polymeric vectors shield the nucleic acids during transit, facilitate cellular uptake via charge-based interactions with cell membranes, and promote crucial endosomal escape, enabling the successful regulation of target gene expression.
Targeted Delivery for Inflammatory and Autoimmune Diseases
Chronic inflammation involves highly localized sites of immune overactivation. Polymeric nanocarriers can be engineered to target specific inflammatory markers or reactive oxygen species present exclusively within these inflamed joint tissues or gastrointestinal tracts. By delivering potent immunosuppressive agents or non-steroidal anti-inflammatory drugs directly to the site of cellular infiltration, systemic immune suppression is avoided, preserving the body's overall defensive capabilities while treating the local pathology.
Central Nervous System Drug Delivery Strategies
The blood-brain barrier is one of the most formidable obstacles in therapeutic science, effectively preventing the vast majority of therapeutic molecules from entering the central nervous system. Polymeric systems are actively designed to traverse this barrier through receptor-mediated transcytosis, utilizing surface ligands that bind to transport receptors expressed on brain endothelial cells. This specialized targeting facilitates the non-invasive transport of neurotrophic factors and complex therapeutics for the management of neurodegenerative conditions.
Services
Custom Polymer Development and Support Services for Targeted Drug Delivery
BOC Sciences has extensive expertise in functional monomer development, polymer material design, targeting modification technologies, and nanocarrier construction, providing comprehensive support for the development of targeted polymer drug delivery systems. To enhance drug accumulation in specific tissues, cells, or tumor microenvironments, we offer integrated solutions covering targeted polymer design, ligand conjugation, nanocarrier formulation, and performance optimization. By precisely controlling polymer structure, surface functionality, and conjugation strategies, BOC Sciences helps clients develop polymer-based delivery systems with high targeting efficiency, improved in vivo stability, and controlled release properties, supporting advanced drug delivery research and translational development.

Targeted Functional Polymer Design and Synthesis
- Custom design and polymer synthesis of functional polymers for targeted delivery applications.
- Development of amphiphilic block copolymers and biodegradable polymers.
- Precise control of molecular weight, composition, and hydrophilic–hydrophobic balance.
- Supply of reactive functional polymers for subsequent targeting modification.

Targeting Ligand Conjugation and Surface Functionalization
- Targeting ligand conjugation services to enhance active targeting capability.
- Conjugation of folic acid, peptides, antibody fragments, and aptamers.
- Support for click chemistry and bioorthogonal conjugation strategies.
- Polymer surface functionalization and PEGylation to improve stability and circulation time.

Construction of Targeted Nanocarrier Systems
- Development of targeted polymer nanoparticles and micelle systems.
- Support for hydrophobic drug loading and targeted delivery optimization.
- Design of multifunctional delivery platforms for precision therapeutics.
- Development of systems compatible with various administration routes.

Performance Evaluation and System Optimization
- Particle size, morphology, and structural characterization.
- Support for targeting efficiency and cellular uptake studies.
- Evaluation of drug release behavior and in vivo stability.
- Comprehensive system optimization and technical support.
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What are targeted polymer drug delivery systems?
Targeted polymer drug delivery systems are advanced therapeutic platforms that use specially designed polymers to deliver drugs directly to specific tissues, cells, or disease sites. These systems improve treatment precision by enhancing site-specific accumulation, reducing off-target effects, and enabling controlled drug release. They are widely used in cancer therapy, gene delivery, and chronic disease management.
-
How do polymer-based drug delivery systems achieve site-specific targeting?
Polymer-based delivery systems achieve site-specific targeting through mechanisms such as passive targeting (e.g., enhanced permeability and retention effect), active targeting using ligands that bind to specific receptors, and stimuli-responsive release triggered by pH, enzymes, temperature, or other biological signals. These strategies ensure that the therapeutic payload is released primarily at the desired location.
-
How do targeted polymers improve drug delivery efficiency?
Targeted polymers improve drug delivery efficiency by enhancing site-specific accumulation, reducing systemic toxicity, and enabling controlled and sustained drug release. Through mechanisms such as active ligand-mediated targeting, passive tumor accumulation, and stimuli-responsive release, polymer-based systems increase drug concentration at the disease site while minimizing off-target exposure. This leads to improved therapeutic outcomes, optimized dosing, and better patient compliance.
-
What polymers are commonly used in targeted delivery?
Commonly used polymers in targeted drug delivery include biodegradable polymers such as poly(lactic acid) (PLA), poly(lactic-co-glycolic acid) (PLGA), and polycaprolactone (PCL), as well as synthetic and functional polymers like polyethylene glycol (PEG), polyethylenimine (PEI), and stimuli-responsive polymers. These materials are selected based on their biocompatibility, degradation profile, surface modification potential, and ability to support drug conjugation or nanoparticle formation.
-
What are the advantages of using polymers for controlled drug release?
Polymers offer multiple advantages for controlled drug release, including tunable degradation rates, chemical versatility, biocompatibility, and structural flexibility. They allow precise control over drug release kinetics, improve drug stability, enhance bioavailability, and support long-term or sustained therapy. Additionally, functionalized polymers can be engineered for smart, stimuli-responsive delivery systems.

 Fig. 1. Schematic of synthesis, administration, and targeted accumulation of polymeric nanocarriers (BOC Sciences Authorized).
Fig. 1. Schematic of synthesis, administration, and targeted accumulation of polymeric nanocarriers (BOC Sciences Authorized). Fig. 2. Comparative overview of polymeric micelles, nanoparticles, and dendrimers (BOC Sciences Authorized).
Fig. 2. Comparative overview of polymeric micelles, nanoparticles, and dendrimers (BOC Sciences Authorized).