Injectable Polymer Hydrogels for Long-Acting Drug Delivery: Design Principles and Material Selection
Injectable polymer hydrogels are versatile long-acting drug delivery platforms that can be administered in a flowable state and converted into localized depots after injection through physical association, ionic interaction, solvent exchange, or chemical crosslinking. Unlike rigid implants or discrete particulate systems, hydrogel depots provide a water-rich three-dimensional polymer network that can retain payloads, regulate transport, and adjust release behavior through mesh size, swelling, degradation, and drug–matrix interactions. Because these systems can be tuned for injectability, gelation rate, mechanical integrity, and residence time, they are widely explored for localized and depot-style delivery of small molecules, peptides, proteins, and selected nucleic acid-related payloads. For formulation design, injectable hydrogels are especially useful when the goal is to combine minimally invasive administration with controllable matrix-based long-acting release.
Resources
Injectable Polymer Hydrogels for Long-Acting Drug Delivery
Injectable hydrogel systems occupy a distinct place in advanced drug delivery because they bridge formulation science, polymer chemistry, and depot engineering. A well-designed hydrogel can be injected through a practical device, form a stable reservoir after administration, and then regulate release through a combination of diffusion resistance, swelling, matrix relaxation, and controlled degradation. This platform is especially attractive when long-acting performance must be achieved without relying on preformed implants or dense hydrophobic particles. Compared with polymer nanoparticles, injectable hydrogels generally offer a larger local reservoir and slower matrix evolution. Compared with compact polyester depots, they provide a more hydrated environment that can be advantageous for sensitive payloads and localized retention.
What Defines a Hydrogel Drug Delivery System?
A hydrogel drug delivery system is built from a crosslinked or physically associated polymer network that can absorb and retain substantial amounts of water while maintaining enough structural integrity to act as a functional matrix. In drug delivery, the hydrogel is not merely a swollen excipient. It acts as a transport-regulating environment whose mesh size, charge distribution, water content, and degradation behavior influence how quickly an incorporated payload diffuses, partitions, or is retained. When the network is engineered for long-acting delivery, the hydrogel becomes a depot rather than a simple gel carrier, and its material design directly shapes both retention and release.
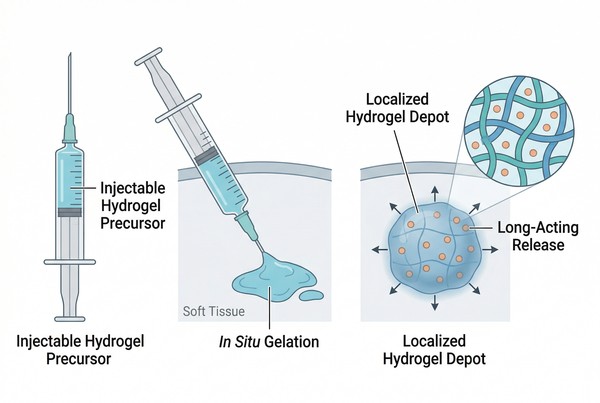 Fig. 1. Injectable hydrogels transition from flowable systems to localized depots after administration (BOC Sciences Authorized).
Fig. 1. Injectable hydrogels transition from flowable systems to localized depots after administration (BOC Sciences Authorized).
What Makes a Hydrogel Injectable?
Injectability depends on whether the formulation can flow through a syringe or related delivery device before or during administration without unacceptable force, premature gel blocking, or destructive shear effects. A material may be an excellent hydrogel after formation but still fail as an injectable system if viscosity is too high before injection or if gelation begins too early. The best injectable hydrogels balance pre-injection flow with post-injection structure formation. In practical terms, this means the formulation must remain processable long enough to be administered, yet convert into a depot quickly enough to avoid leakage, dilution, or uncontrolled burst release.
Why Injectable Hydrogels Are Useful for Long-Acting Release?
Injectable hydrogels are useful for long-acting release because they create a localized polymer network that slows transport and can be designed to evolve over time. The depot can restrict drug diffusion, swell gradually, degrade in a controlled way, or interact with the payload through ionic, hydrophobic, or hydrogen-bonding effects. This enables release profiles that are often more adjustable than simple solution dosing and less mechanically invasive than preformed implants. The platform is particularly attractive when a project requires local retention, reduced dosing frequency, or a matrix that can accommodate water-sensitive or structurally delicate molecules more gently than some solvent-driven or rigid particulate systems.
How Injectable Hydrogels Differ from Particles and Implants
Injectable hydrogels differ from particles and implants in both structure and release logic. Particulate systems, including polymer nanoparticles and microspheres, depend heavily on surface area, particle morphology, and matrix density. Implants rely more on geometry, dense structure, and long-path diffusion. Hydrogels, by contrast, are dominated by water-rich networks, mesh-size effects, swelling, and network relaxation. This makes them especially useful when drug mobility must be managed in a hydrated environment or when in situ depot formation is preferred over premanufactured solid dosage forms. When platform choice is still open, it is often helpful to compare hydrogel design against broader carrier trade-offs discussed in choosing the right drug delivery system.
What Polymer Materials Are Used in Injectable Hydrogels?
Material selection is the core design decision in injectable hydrogel development. The polymer controls not only whether the system gels, but how it gels, how much water it absorbs, how it interacts with the payload, how strong the final depot becomes, and how the network evolves after administration. Because hydrogel behavior depends on both polymer chemistry and network architecture, formulators should select materials based on the intended gelation trigger, payload properties, target release window, and handling constraints rather than choosing a polymer solely because it is widely cited.
Natural and Semi-Synthetic Hydrogel Polymers
Natural and semi-synthetic polymers are common starting points when a hydrated, biologically compatible matrix is needed. Materials from the broader class of natural polymers often provide strong water affinity, mild processing conditions, and useful functional groups for ionization or derivatization. Examples such as chitosan, dextran, alginate-type systems, and poly-γ-glutamic acid derivatives are especially relevant because they can support swelling-driven matrices, ionic interactions, and water-rich depots. These polymers are often useful when gentle gel formation or higher aqueous compatibility is more important than rigid structure.
Synthetic Polymers for Injectable Hydrogel Systems
Synthetic polymers offer more direct control over chain architecture, substitution pattern, reactive functionality, and gelation response. This can be important when injectability, reproducibility, and defined network behavior matter more than the intrinsic biofunctionality of a natural polymer. Materials drawn from broader families of polymer materials can be adapted for hydrogel formation through pendant reactive groups, amphiphilic balance, ionizable segments, or crosslinkable termini. In practical development, synthetic systems are often selected when the project requires tighter control over gelation kinetics, mechanical properties, or structure–property consistency.
Thermosensitive Hydrogel-Forming Polymers
Thermosensitive hydrogels are designed to remain more fluid during preparation or injection and become more structured as temperature changes. These systems are attractive because they can support simple administration without requiring a separate external crosslinking step at the time of injection. Their performance depends on the balance between hydrophilic and hydrophobic segments, association strength, and the temperature range over which chain interactions intensify. For long-acting delivery, thermosensitive systems must be tuned carefully: a transition that is too weak can lead to depot instability, while one that is too aggressive can compromise injectability or create heterogeneous gel formation.
Ionic and Chemically Crosslinkable Polymers
Ionic and chemically crosslinkable polymers are useful when stronger post-injection structure is required. Ionic systems form networks through charge-driven interactions, while chemically crosslinkable systems create covalent links after administration or during controlled processing. These materials are often chosen when the formulation must support longer residence time, tighter diffusion control, or improved mechanical strength. Crosslinkable polymers must be matched carefully to the gelation mechanism, because the same reactivity that strengthens the network can also alter payload stability, pre-injection viscosity, or storage behavior if not properly controlled.
How Polymer Class Affects Depot Behavior?
Polymer class changes how the depot behaves after administration. Highly hydrophilic polymers tend to promote water uptake and swelling, which can support gentle release but may also increase burst risk. More associative or crosslinked systems can improve retention, but may reduce syringeability or create slower network equilibration. Charge-bearing polymers may enhance retention of oppositely charged payloads but complicate release if binding becomes too strong. Degradable hydrogel materials can gradually relax or erode, especially when they are built from or combined with biodegradable polymers. The best material class is therefore the one whose network behavior aligns with both payload needs and formulation constraints.
| Polymer Class | Typical Examples | Main Gelation Mode | Key Advantage | Main Limitation | Best-Fit Use |
|---|
| Natural and semi-synthetic polymers | Chitosan, dextran derivatives, poly-γ-glutamic acid sodium salt | Ionic association, physical gelation, derivatized crosslinking | Mild aqueous processing and high water compatibility | Batch variability and less predictable network structure | Localized depots and sensitive payload environments |
| Synthetic hydrophilic polymers | PEG-based systems, poly(N-vinylacetamide), poly(vinylamine) derivatives | Chemical crosslinking or designed associative gelation | Better control over structure and reproducibility | May need additional design for strong payload retention | Defined injectability and tunable release studies |
| Thermosensitive polymers | Temperature-responsive copolymer systems | Temperature-triggered physical gelation | Simple in situ depot formation | Narrow process window between flow and gelation | Minimally invasive depot formation |
| Ionic or covalently crosslinkable systems | Functionalized natural or synthetic polymers | Ionic gelation or post-injection crosslinking | Improved mechanical strength and retention | Greater complexity in gelation control | Longer-lasting depots with stronger matrices |
Need a Custom Injectable Hydrogel Design Strategy?
From polymer screening and hydrogel synthesis to gelation design and release optimization, we support injectable hydrogel development for long-acting and depot-style drug delivery.
How Injectable Hydrogels Form After Administration?
The success of an injectable hydrogel depends not only on the final network, but on how that network forms. Post-injection gelation determines whether the material remains localized, whether the payload is retained effectively, and whether the final structure is uniform enough to support reproducible release. A formulation that gels too slowly may leak or dilute before depot formation, while one that gels too quickly may be difficult to inject or may form a heterogeneous matrix. Understanding gelation pathways is therefore essential to both performance and manufacturability.
Physical Gelation in Injectable Hydrogel Systems
Physical gelation relies on noncovalent interactions such as hydrophobic association, hydrogen bonding, chain entanglement, crystallite formation, or temperature-triggered assembly. These systems can be attractive because they avoid potentially harsh crosslinking chemistry and often support simpler processing. However, they are also sensitive to concentration, ionic environment, temperature, and dilution. In long-acting delivery, physically formed hydrogels must be engineered so that the resulting network is strong enough to remain localized while still allowing appropriate transport of water and payload through the matrix.
Chemical Crosslinking After Injection
Chemical crosslinking creates covalent bonds after or during administration and can provide stronger and more persistent network structure than many purely physical systems. This is useful when longer residence time, higher gel strength, or tighter diffusion control is needed. The challenge is that reactive formulations must remain sufficiently stable before use and form the network in a controlled way after delivery. Crosslink density, reaction rate, and the distribution of reactive groups all influence injectability, final network homogeneity, and the release behavior of the depot.
In Situ Forming Hydrogels Versus Preformed Gels
In situ forming hydrogels are low-viscosity or moderately viscous systems that undergo gelation after administration, whereas preformed gels are already structured before injection and rely more on shear-thinning or deformation during delivery. In situ systems can provide better site-localized formation and more precise post-injection structure building, but they depend heavily on timing and local conditions. Preformed gels can simplify certain aspects of network definition, yet they may require greater injection force and may show different structural recovery after administration. The choice depends on whether the project prioritizes easy flow, immediate localization, or tighter pre-injection control.
How Gelation Rate Affects Formulation Performance?
Gelation rate is one of the most practical formulation variables in injectable hydrogels. A slow rate may allow payload diffusion or convective loss before the depot stabilizes, increasing burst release. An excessively fast rate may raise injection force, shorten handling time, or create local heterogeneity because the system begins to structure before uniform placement is complete. The ideal gelation rate is therefore not simply "fast" or "slow," but matched to the administration process, payload sensitivity, and desired early release behavior.
How Injectable Hydrogels Control Drug Release?
Release from injectable hydrogels is controlled by a dynamic combination of network structure, water transport, matrix evolution, and payload interaction. Unlike dense polyester depots where erosion can dominate later stages, hydrogel release often begins with a highly hydrated system whose internal transport pathways are already partially open. This makes release control more dependent on mesh size, swelling behavior, binding interactions, and network relaxation. The main development task is to identify which of these mechanisms is dominant for the payload and then tune the matrix so the overall profile matches the intended long-acting release goal.
Mesh Size and Diffusion-Controlled Release
Mesh size determines how easily a solute moves through the hydrated polymer network. When the mesh is substantially larger than the payload, diffusion can be relatively rapid and long-acting release may be weak. When the mesh is comparable to or smaller than the effective hydrodynamic size of the payload, transport slows and release becomes more controlled. For small molecules, mesh restriction alone may not be enough unless the network is dense or interacting strongly with the drug. For larger biomolecules, modest changes in crosslink density can substantially alter mobility and retention.
Swelling and Water Transport in Hydrogel Depots
Swelling changes both the physical dimensions and the transport environment of the depot. As water enters the network, the hydrogel may expand, dilute local polymer concentration, and widen effective transport pathways. This can accelerate release if the payload is not otherwise retained. Swelling can also alter local mechanical properties and create time-dependent changes in mesh size. For long-acting systems, it is not enough to know the initial gel state; one must understand how water uptake evolves over the intended release period.
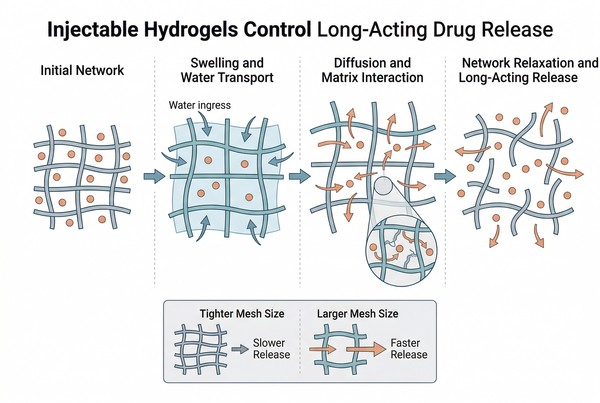 Fig. 2. Hydrogel release is shaped by mesh size, swelling, degradation, and drug–matrix interactions (BOC Sciences Authorized).
Fig. 2. Hydrogel release is shaped by mesh size, swelling, degradation, and drug–matrix interactions (BOC Sciences Authorized).
Polymer Degradation and Network Relaxation
In degradable or labile hydrogel systems, the network does not remain static. Bond cleavage, chain relaxation, and mass loss can enlarge transport pathways and alter release from a primarily diffusion-controlled process to a relaxation- or degradation-assisted one. This is especially relevant when the hydrogel is constructed from functionalized natural polymers, degradable linkages, or hybrid systems containing segments from biodegradable polymers. The timing of network change matters as much as the initial structure, because a depot that looks adequately retentive on day one may become much more permeable later.
Drug–Matrix Interactions in Hydrogel Release
Many hydrogel systems depend on more than size exclusion. Charged polymers, hydrogen-bonding motifs, hydrophobic domains, and specific functional groups can all slow release by temporarily associating with the payload. For example, cationic or amphoteric matrices may retain anionic molecules more effectively, while hydrophobic pockets may help limit the mobility of less polar solutes. These interactions can be useful, but they must be balanced carefully. If retention is too weak, burst release occurs; if it is too strong, release may become incomplete or highly environment-dependent.
How to Select Hydrogel Materials for Different Payloads?
Payload type is one of the most important variables in injectable hydrogel design because it determines which release mechanisms are realistic, which polymer interactions are useful, and which formulation risks are most likely to dominate development. A hydrogel that works well for a peptide may release a small molecule too quickly, while a matrix designed for local retention of a hydrophobic compound may be poorly suited for a charged macromolecule. For this reason, hydrogel selection should begin with payload behavior rather than polymer familiarity. In practical formulation work, the most useful selection variables are aqueous solubility, molecular size, conformational sensitivity, charge state, diffusion tendency, and target release duration. These properties should then be matched to network density, gelation mechanism, swelling profile, degradability, and polymer–payload interaction potential. The goal is not simply to form a gel, but to build a depot whose structure and microenvironment are compatible with both the drug and the intended long-acting release profile.
Hydrogel Selection for Small Molecules
Small molecules are often difficult to retain in injectable hydrogels because their diffusion through hydrated networks can be very fast, especially when the drug is water-soluble and the gel swells quickly after administration. In these systems, mesh restriction alone is often insufficient for long-acting release. Effective material selection therefore focuses on whether the hydrogel can provide an additional retention mechanism, such as hydrophobic association, hydrogen bonding, ionic interaction, or a denser post-gelation network that slows transport more meaningfully.
The best hydrogel materials for small molecules are usually those that balance injectability with controlled network tightening after administration. If polymer concentration is increased too far to slow diffusion, syringeability may become impractical; if the network is too open, burst release remains high. A useful screening strategy is to compare drug partitioning, early release, swelling behavior, and polymer–drug affinity together. When the small molecule remains too mobile even in optimized hydrogels, it may indicate that a different long-acting platform is more suitable than a purely hydrogel-based depot.
Hydrogel Selection for Peptides and Proteins
Peptides and proteins often fit hydrogel systems better than highly mobile small molecules because the water-rich environment can reduce exposure to harsh organic processing and support a gentler depot microenvironment. However, hydrogel selection for these payloads must consider more than release duration. Protein conformation, aggregation tendency, adsorption to polymer domains, local pH, ionic strength, and crosslinking chemistry can all affect stability during gelation, storage, and release. A hydrogel that retains a protein well but destabilizes it is not a useful formulation choice.
For proteins and larger peptides, suitable materials typically combine mild gelation conditions with a network dense enough to slow transport without creating excessive binding or structural stress. Polymer matrices that support buffered aqueous environments and avoid overly reactive or strongly denaturing interactions are usually preferred. In screening, release amount should always be interpreted alongside payload integrity, because incomplete recovery, aggregation, or structural change may reveal that the apparent retention advantage of a hydrogel is actually a compatibility problem rather than true long-acting control.
Hydrogel Selection for Nucleic Acids and Sensitive Macromolecules
Nucleic acids and similarly sensitive macromolecules require a different material-selection strategy because successful delivery depends on both depot behavior and molecular protection. A neutral hydrogel may provide physical entrapment, but it is often insufficient for highly charged, enzymatically vulnerable payloads unless the system also offers electrostatic association, protective complexation, or a hybrid carrier architecture. This makes polymer charge density, gelation mildness, and compatibility with auxiliary carrier components especially important selection criteria.
In many cases, the hydrogel is most effective not as the sole delivery material, but as a secondary depot around a preformed nucleic acid carrier. Materials should therefore be selected based on whether they preserve carrier integrity during gel formation and whether they allow gradual release without excessive binding or rapid disassembly. If the matrix binds too weakly, release is too fast; if it binds too strongly, release becomes incomplete or condition-dependent. For these payloads, hydrogel selection should always distinguish between retaining the molecule and preserving its functional delivery state.
Matching Polymer Properties to Payload Behavior
The most reliable selection strategy is to convert payload properties directly into hydrogel design requirements. A fast-diffusing hydrophilic drug generally needs stronger interaction or a tighter network, while a structurally sensitive biomolecule may require milder gelation and a more stable aqueous microenvironment. Charged payloads may benefit from complementary ionic motifs, but only when reversible release remains possible. In this way, polymer hydrophilicity, charge, crosslinking mode, swelling tendency, and degradability become practical levers for matching the matrix to the therapeutic.
A useful decision framework is to evaluate five variables together: payload mobility, payload sensitivity, target release duration, need for local depot retention, and tolerance to the gelation process. When these are mapped against polymer class and network behavior, material choice becomes much more rational. The right hydrogel is therefore not the one with the strongest mechanics or the most complex chemistry, but the one whose structure solves the dominant release and stability problem of the payload with the least added formulation risk.
Common Challenges in Injectable Hydrogel Formulation Development
Injectable hydrogels can appear conceptually simple, yet they present recurring development difficulties because administration, depot formation, and release all happen within a coupled material system. Problems that look similar at the release stage can arise from very different causes, including poor injectability, incomplete gelation, excessive swelling, weak payload retention, or instability during storage. The most efficient development programs therefore treat these issues as connected variables rather than isolated formulation defects.
- Burst release from weak drug retention: Burst release usually appears when the hydrogel forms too slowly, the mesh is too open, or the drug has little affinity for the matrix. In highly hydrated systems, payload loss can begin immediately if the network does not create an effective transport barrier. This is especially common for small hydrophilic drugs and for systems where gelation occurs after significant dilution. Solving the problem may require stronger interaction motifs, faster in situ structuring, or reformulation of the matrix rather than simple concentration changes.
- Poor syringeability and injection force: A hydrogel formulation can fail before depot formation if it cannot be administered reproducibly. High pre-injection viscosity, premature structuring, strong associative interactions, or poor shear recovery can all increase injection force. This challenge becomes more severe as formulators try to increase polymer concentration to improve retention. The right balance is to build enough structure after delivery without making the pre-injection state impractical for handling or dosing.
- Weak gel strength after administration: Weak gel strength leads to poor localization, faster relaxation, and less predictable release. The issue may arise from insufficient crosslink density, low polymer concentration, weak association strength, or adverse interactions between the payload and the forming matrix. A depot that appears to form may still lack the structural persistence needed for long-acting release. For long-acting use, gel strength should be evaluated as a functional property connected to residence time and release control, not just as a material descriptor.
- Inconsistent gelation under practical conditions: Gelation that works well in one controlled setup may behave differently when concentration, ionic environment, temperature, or administration timing changes. This inconsistency can produce large differences in depot structure and early release. In situ systems are particularly sensitive because small shifts in local conditions can alter network formation rate or homogeneity. Development should therefore test gelation under realistic preparation and administration conditions instead of relying on idealized benchtop observations alone.
- Storage stability and formulation compatibility issues: Some injectable hydrogel systems are unstable before use because reactive groups, associative segments, or suspended payload domains change during storage. Viscosity drift, premature structuring, phase separation, or loss of payload quality can all compromise performance. Storage compatibility must therefore be considered early, especially when the hydrogel contains sensitive biomolecules, labile linkers, or multicomponent precursor systems that are designed to gel after administration.
| Development Issue | Likely Cause | What to Evaluate | Practical Optimization Direction |
|---|
| Burst release | Weak retention, slow gelation, open network | Early release profile, gelation timing, mesh-related indicators | Increase interaction strength or improve post-injection structuring |
| Poor syringeability | High viscosity, premature association, weak shear recovery | Injection force, viscosity profile, handling window | Adjust concentration, associative balance, or precursor design |
| Weak gel strength | Low network density or insufficient crosslinking | Mechanical properties, gel recovery, depot persistence | Refine polymer architecture or crosslink density |
| Inconsistent gelation | Sensitivity to temperature, ions, or mixing history | Gelation time under practical conditions | Broaden process tolerance and simplify gel trigger |
| Storage instability | Reactive drift, aggregation, phase separation | Viscosity change, appearance, payload integrity | Redesign precursor stability and storage format |
How to Evaluate and Optimize Injectable Hydrogel Performance?
Hydrogel optimization should connect pre-injection behavior, post-injection structure, and release performance into one decision framework. It is not enough to know that a gel forms or that a release curve exists. A useful evaluation strategy should explain how rheology influences injectability, how gelation determines depot quality, how swelling and degradation change transport, and how those changes affect the payload over time. This structure–process–performance approach allows formulators to improve hydrogel design rationally instead of treating each experiment as an isolated trial.
- Rheology and injectability testing: Rheology is essential because it predicts whether the formulation can be handled, filled, and delivered reproducibly. Shear-thinning behavior, viscosity under relevant shear conditions, structural recovery, and injection force all help define whether the material is realistically injectable. These measurements are especially important for systems that depend on associative interactions or partially structured precursors before administration.
- Gelation time and mechanical property assessment: Gelation time should be measured under conditions that reflect actual preparation and administration, not only under simplified laboratory triggers. Mechanical assessment should then determine whether the formed depot has sufficient strength and resilience to remain localized while supporting the intended release period. Together, these tests show whether the hydrogel transitions into a usable depot rather than merely forming a weak gel in principle.
- Swelling, degradation, and release studies: Swelling and degradation studies reveal how the hydrogel evolves after formation, while release studies show how that evolution affects payload escape. These datasets should be interpreted together. A system that swells rapidly may still be useful if payload interactions remain strong, while a degrading system may support long-acting release acceptably if network relaxation is gradual and controlled. The goal is to understand the sequence of structural change rather than just to report isolated endpoints.
- How to link hydrogel properties to application goals: Every measured property should connect back to an application need. If local retention is the goal, gelation speed and post-injection mechanics may matter most. If the payload is a sensitive biomolecule, mild gel formation and matrix compatibility may dominate. If the project needs extended release of a mobile small molecule, drug–matrix interaction and network density become central. Optimization works best when the target product logic is defined early and testing is selected to answer that logic directly.
- When an injectable hydrogel is the right platform choice: An injectable hydrogel is the right platform when a project benefits from minimally invasive depot formation, localized matrix retention, and release control in a hydrated environment. It is especially attractive when the payload would benefit from gentler aqueous processing or when a preformed implant is not desirable. However, if the system cannot retain a small mobile drug or cannot form a consistent depot under realistic conditions, another platform such as polymer nanoparticles or a denser long-acting matrix may be more appropriate.
Services
Advanced Injectable Hydrogel Design and Synthesis Services
BOC Sciences provides integrated support for injectable hydrogel development, from polymer selection and functional modification to hydrogel synthesis, gelation design, and release-focused evaluation. Our capabilities are built around the practical variables that control whether a hydrogel becomes a reliable long-acting depot, including precursor composition, crosslinking strategy, injectability, network architecture, payload compatibility, and formulation stability. By linking polymer chemistry to depot behavior, we help teams develop hydrogel systems that are better aligned with localized and long-acting drug delivery goals.

Polymer Selection & Hydrogel Design
- Screening of natural, semi-synthetic, and synthetic hydrogel-forming polymers.
- Selection of gelation triggers for in situ and injectable depot systems.
- Matching polymer class to payload behavior and release targets.
- Support for localized and long-acting formulation logic.

Injectable Hydrogel Synthesis
- Custom polymer hydrogel synthesis for injectable and in situ forming systems.
- Functionalization for thermosensitive, ionic, and crosslinkable networks.
- Optimization of precursor composition and gelation pathways.
- Development of tailored water-rich depot materials.

Injectability and Release Evaluation
- Rheology, injectability, and gelation time testing.
- Mechanical characterization of post-injection depots.
- Swelling, degradation, and release-profile assessment.
- Correlation of hydrogel structure with long-acting release behavior.

Formulation Optimization Support
- Improvement of burst control, retention, and matrix stability.
- Adjustment of crosslink density and payload compatibility.
- Evaluation of storage stability and precursor robustness.
- Integrated support from material design to depot performance.
Need Support with Injectable Hydrogel Development?
Discuss hydrogel polymer selection, in situ gelation, injectability, and long-acting release design with a team focused on advanced polymer-based drug delivery systems.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What makes an injectable hydrogel suitable for long-acting drug delivery?
An injectable hydrogel is suitable when it can be administered as a flowable system and then form a stable depot after injection. Its value depends on controllable gelation, appropriate mesh size, predictable swelling or degradation, and enough drug retention within the network to avoid rapid early loss.
-
How do injectable hydrogels control drug release?
Injectable hydrogels control release through diffusion restriction, water uptake, network relaxation, matrix degradation, and drug–polymer interactions. The dominant mechanism depends on polymer chemistry, crosslink density, payload size, charge, and how strongly the incorporated drug partitions into or binds within the formed hydrogel depot.
-
Why do some injectable hydrogels show burst release?
Burst release usually occurs when the drug is weakly retained, the network forms too slowly, or the final mesh is too open to slow transport effectively. It can also result from rapid swelling, poor drug–matrix compatibility, or insufficient post-injection structuring under realistic administration conditions.
-
What polymers are commonly used in injectable hydrogel systems?
Common materials include natural or semi-synthetic polymers such as chitosan, dextran derivatives, and poly-γ-glutamic acid systems, as well as synthetic hydrogel-forming polymers with thermosensitive, ionic, or chemically crosslinkable behavior. The right choice depends on gelation method, payload sensitivity, and target release duration.
-
How should injectable hydrogels be evaluated during formulation development?
Evaluation should include rheology, injectability, gelation time, mechanical integrity, swelling behavior, degradation pattern, and in vitro release. Together, these tests show whether the system can be administered reliably, form a useful depot after injection, and support long-acting release that matches the intended payload and application.

 Fig. 1. Injectable hydrogels transition from flowable systems to localized depots after administration (BOC Sciences Authorized).
Fig. 1. Injectable hydrogels transition from flowable systems to localized depots after administration (BOC Sciences Authorized). Fig. 2. Hydrogel release is shaped by mesh size, swelling, degradation, and drug–matrix interactions (BOC Sciences Authorized).
Fig. 2. Hydrogel release is shaped by mesh size, swelling, degradation, and drug–matrix interactions (BOC Sciences Authorized).