Polymeric Transdermal Patches for Long-Acting Drug Delivery: Matrix Design and Release Control
Polymer-based transdermal patches remain one of the most practical long-acting drug delivery formats because they combine controlled release with noninvasive administration, scalable manufacturing, and flexible design options. Unlike simple topical films or rapidly depleted surface formulations, polymeric patch systems are engineered to regulate how a drug is stored, how it moves through the matrix, how it contacts the skin, and how long it can maintain a useful input profile. In long-acting delivery, that balance is especially important because successful patch performance depends on more than drug release alone. The formulation must also support stable skin contact, predictable transport across the stratum corneum, resistance to crystallization or phase separation, and sufficient physical stability during wear and storage.
Resources
What Are Polymeric Transdermal Patches?
Polymeric transdermal patches are structured drug delivery systems designed to maintain contact with the skin and deliver an active ingredient over an extended period through a controlled-release matrix, reservoir, or adhesive-based layer. Their long-acting value comes from the fact that the polymer does not merely hold the drug; it also governs diffusion, adhesion, flexibility, occlusion, and the physical integrity of the patch during wear. In practical formulation development, this means the patch is a transport-regulating system rather than a passive carrier. The patch architecture, the polymer class, and the interaction between the drug and the matrix all work together to determine whether delivery is short-lived, sustained, or truly long-acting.
Definition and Basic Patch Architecture
A transdermal patch typically contains one or more functional layers, including a backing layer, a drug-containing region, a skin-contact layer, and in some cases a distinct rate-controlling membrane. In drug-in-adhesive systems, the adhesive itself serves as the drug reservoir. In matrix patches, the drug is distributed through a continuous polymer phase that contacts the skin either directly or through an adhesive interface. In reservoir systems, the drug-rich core is separated from the skin by a membrane that modulates release. Although these structures differ, they all rely on polymer design to manage drug distribution, structural stability, and transport to the skin surface.
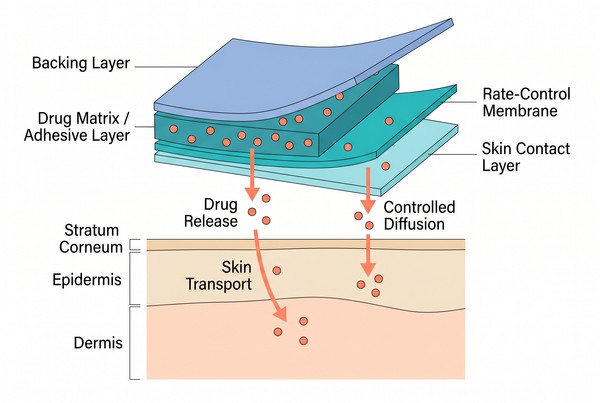 Fig. 1. Typical layer structure of polymeric transdermal patch systems (BOC Sciences Authorized).
Fig. 1. Typical layer structure of polymeric transdermal patch systems (BOC Sciences Authorized).
How Transdermal Patches Differ from Topical Films and Microneedles?
Transdermal patches differ from topical films because their objective is not simply local coverage or superficial deposition, but controlled input through the skin over time. They also differ from microneedle systems because they generally rely on diffusion through intact skin rather than mechanical disruption of the barrier. This distinction is important for long-acting delivery. A patch must work within the limits of the skin barrier, so material selection and release control become central. By contrast, microneedles shift more of the challenge toward barrier bypass. Patches therefore remain especially relevant when the payload and target delivery rate are compatible with a noninvasive diffusion-based strategy.
Why Patches Remain Relevant in Long-Acting Delivery?
Patches remain relevant because they provide a strong balance of usability, formulation control, and wear-based delivery. They can support prolonged exposure without the complexity of injectable depots or the device burden of larger implants. They are also highly adaptable in terms of structure, allowing developers to choose among adhesive-based, matrix-based, membrane-controlled, and multilayer formats. When the payload is appropriate for transdermal input, patches offer a practical route to long-acting delivery with clear advantages in dose continuity, convenience, and platform scalability.
How Polymeric Transdermal Patches Achieve Controlled Release?
Controlled release in a transdermal patch is created by limiting how quickly the drug can leave its internal environment, reach the skin surface, and then partition into or across the skin. This makes polymer design central to release performance. The matrix must be open enough to allow sustained movement, but restrictive enough to prevent rapid depletion. At the same time, the patch must remain physically stable throughout the wear period, because structural changes in the matrix often translate directly into changes in release. For long-acting delivery, the goal is not merely slower release, but a reproducible balance between early availability and prolonged transport.
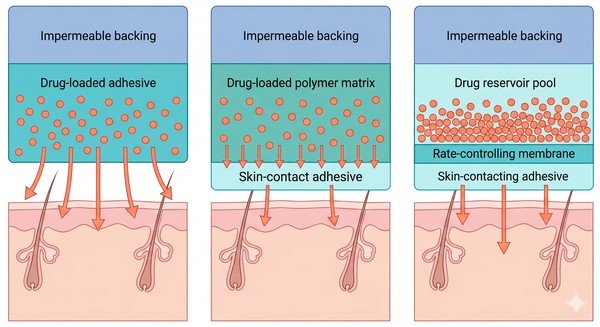 Fig. 2. How polymeric patch structures control skin transport and sustained release (BOC Sciences Authorized).
Fig. 2. How polymeric patch structures control skin transport and sustained release (BOC Sciences Authorized).
Matrix Diffusion Through Thin Polymer Structures
In matrix patches, the drug is dispersed or dissolved throughout a polymer network and leaves the system by diffusing through that structure toward the skin-contact side. The effective release rate depends on polymer mobility, free volume, internal drug state, and the degree of compatibility between the drug and the surrounding matrix. Thin structures can still provide long-acting performance when diffusion is sufficiently restricted and the drug remains evenly distributed. The key challenge is ensuring that the matrix does not release too quickly at the start or become transport-limited to the point that meaningful delivery cannot be maintained.
Surface-Area Effects on Burst and Sustained Phases
Patch geometry and exposed surface area influence how much of the loaded drug is available near the skin-contact interface during the early phase of wear. If too much drug is concentrated near the surface, the system may show a stronger initial burst. If the drug is distributed more uniformly or restrained by an additional barrier, the sustained phase can become more dominant. This balance matters in long-acting patch design because users often need an initial onset without sacrificing duration. Surface-area effects are therefore not only a geometric issue; they are a release-design parameter that interacts with polymer thickness, internal drug distribution, and membrane resistance.
Erosion and Degradation in Biodegradable Patch Systems
Although many transdermal patches rely primarily on diffusion rather than erosion, biodegradable polymers can become relevant in advanced or hybrid patch concepts where controlled structural change supports release. In these systems, gradual hydration, matrix softening, or degradation can alter transport pathways over time. The challenge is to ensure that such changes improve release control rather than destabilize it. For long-acting patches, degradable behavior is useful only when it contributes to predictable delivery and remains compatible with the wear period, storage conditions, and desired mechanical performance.
Polymer Materials Used in Long-Acting Patch Systems
Material choice defines the operating window of a patch. The polymer determines whether the matrix can hold the drug uniformly, whether the adhesive layer can maintain skin contact, whether the backing can tolerate wear, and whether the system remains stable under heat and humidity. This is why patch development is fundamentally a materials problem as much as a dosage-form problem. A useful long-acting patch usually requires more than one functional material, and the best results come from selecting those materials according to role rather than by using a single familiar polymer everywhere in the structure.
Pressure-Sensitive Adhesive Polymers
Pressure-sensitive adhesive polymers are central in many patch formats because they must secure the patch to the skin while remaining compatible with the drug and, in some cases, serving as the primary drug-containing phase. The adhesive must balance tack, peel behavior, cohesion, and comfort over the intended wear period. If the adhesive is too soft, edge lift and cold flow may appear. If it is too rigid, skin conformity and comfort may suffer. In long-acting systems, adhesive performance is directly tied to delivery consistency because unstable contact often means unstable input.
Matrix-Forming Polymers for Sustained Release
Matrix-forming materials govern how the drug is retained and released within the patch body. Hydrophilic components such as Polyethylene glycol can influence matrix hydration and internal mobility, while more hydrophobic structures can slow drug movement and extend release. A matrix polymer must therefore be chosen according to how it affects both drug distribution and the rate of transport through the film. The best material is not simply the one that loads the most drug; it is the one that supports the intended release profile without causing phase instability or compromising patch handling.
Membrane-Forming and Backing-Layer Polymers
In membrane-controlled or multilayer designs, the membrane and backing layers play distinct but equally important roles. The membrane regulates how quickly the drug can move from the reservoir toward the skin, while the backing protects the patch from environmental exposure and helps maintain structural integrity during wear. Materials associated with Polyurethane (PU), polyethers, or selected barrier-forming systems may be relevant when flexibility, toughness, and permeability control are required together. These layers are not passive supports; they are part of the release-control system.
Smart and Hydrogel-Forming Polymers for Advanced Patch Designs
Advanced patch systems sometimes incorporate moisture-responsive, thermoresponsive, or gel-forming materials to refine skin contact and release behavior. In some designs, the patch may benefit from logic similar to Polymer hydrogels, especially where hydration-dependent transport or soft interfacial contact is useful. Other systems may rely on specialty materials including Silicones when flexibility and barrier properties need to be combined. These advanced materials are most valuable when they solve a clear delivery or wear problem rather than being added only for novelty.
Need Support for Long-Acting Patch Design?
From polymer selection and matrix screening to skin transport and release-control strategy, we support the development of polymeric transdermal patches for sustained and long-acting delivery.
Patch Design Formats and Their Trade-Offs
Patch architecture determines how the polymer layers work together and what trade-offs define the system. Two patches carrying the same drug can behave very differently depending on whether the drug sits directly in the adhesive, in a matrix body, or in a separate reservoir. For long-acting delivery, these structural decisions affect release duration, dose capacity, manufacturing complexity, and wear reliability. Selecting the right architecture therefore depends on aligning the payload and target performance with the strengths and weaknesses of each format.
Drug-in-Adhesive Systems
Drug-in-adhesive patches are structurally simple because the adhesive layer holds both the drug and the skin-contact function. This can make them attractive for scalable manufacturing and relatively straightforward wear behavior. The main limitation is that the adhesive must satisfy multiple demands at once: it must retain the drug, control release, maintain adhesion, and resist crystallization or phase separation. For long-acting delivery, this format works best when the payload is compatible with the adhesive environment and does not overload the system.
Matrix Patches
Matrix patches separate the sustained-release function more clearly by embedding the drug within a dedicated polymer structure. This provides more flexibility in controlling diffusion and matrix composition than a pure drug-in-adhesive design. However, the matrix must still remain physically stable and must deliver the drug uniformly throughout wear. Matrix patches are often a strong choice when long-acting release needs to be tuned through polymer composition without moving into the higher complexity of reservoir systems.
Reservoir Patches
Reservoir patches use a drug-rich compartment separated from the skin by a membrane or regulating layer. This can provide stronger control over release when the system is engineered well, because the transport barrier is more explicit. The trade-off is greater design and manufacturing complexity. Seal integrity, membrane consistency, and reservoir stability become critical. Reservoir patches are most useful when release precision is worth the added structural demands.
Multilayer and Wearable Patch Designs
Multilayer and wearable designs extend patch architecture by assigning different functions to different layers, such as adhesion, comfort, reservoir support, moisture management, or directional transport. These systems are attractive when the product concept needs more than one performance characteristic to be optimized simultaneously. Their challenge lies in integration: each added layer can introduce new interfacial stability issues, manufacturing variables, and wear-related failure modes. Still, for advanced long-acting applications, multilayer architecture can be the most flexible design route.
| Patch Format | Main Release Mechanism | Typical Polymer Role | Key Strength | Main Limitation | Best-Fit Scenario |
|---|
| Drug-in-Adhesive | Diffusion from adhesive layer | Adhesion and drug hosting in one phase | Simple structure and efficient manufacturing | Loading and crystallization can be limiting | Moderate-dose patches with simpler release needs |
| Matrix Patch | Diffusion through polymer matrix | Drug retention and sustained release | Flexible control through matrix composition | Uniformity and burst control require careful design | Sustained release with balanced structural simplicity |
| Reservoir Patch | Membrane-controlled transport | Reservoir containment and rate control | More defined release regulation | Higher structural and manufacturing complexity | Products needing tighter control over flux |
| Multilayer / Wearable Patch | Combined diffusion and layer-specific control | Role-specific layers for support, release, and contact | High design flexibility | Integration and stability complexity | Advanced long-acting or user-specific formats |
Formulation Strategy for Skin Transport and Stability
Transdermal performance is shaped by what happens inside the patch before the drug even reaches the skin. A patch that seems acceptable structurally may fail because the drug crystallizes, partitions unevenly, or does not remain soluble within the matrix over time. Likewise, a system that releases well in a model experiment may perform poorly if adhesion drops or if the matrix evolves under heat and humidity. Long-acting patch development therefore requires formulation decisions that link internal stability with external transport.
Drug Solubility in Polymer Matrices
The degree to which the drug remains dissolved or molecularly dispersed in the patch matrix strongly affects release uniformity and long-term stability. If solubility is insufficient, the drug may crystallize, migrate, or become unevenly distributed, all of which can change release behavior during storage and wear. Matrix selection must therefore account for how the drug behaves in the polymer phase at relevant concentrations rather than only under ideal preparation conditions.
Crystallization Control and Uniform Drug Distribution
Crystallization is one of the most common patch-development problems because it reduces available dissolved drug and can destabilize the release profile. Uniform distribution is also important because local enrichment can cause variable input and patch-to-patch inconsistency. Managing these issues often requires careful balance among polymer polarity, drying conditions, residual solvent management, and the use of compatible modifiers. A patch cannot behave as a reliable long-acting system if its internal drug state changes significantly during storage.
Use of Plasticizers, Permeation Enhancers, and Modifiers
Plasticizers and related modifiers can improve flexibility, reduce brittleness, and alter internal mobility in the matrix. Permeation enhancers may improve flux through the skin, but their effect must be weighed against stability, skin comfort, and wear consistency. The useful formulation strategy is not to maximize enhancement, but to create a controlled and reproducible transport environment. In long-acting patches, excessive enhancement can shorten duration just as insufficient enhancement can make delivery ineffective.
Balancing Adhesion, Comfort, and Sustained Delivery Performance
A long-acting patch must stay in place, feel acceptable during wear, and keep releasing at the intended rate. These goals are often in tension. Stronger adhesion can improve wear stability but may reduce comfort or complicate removal. A softer matrix can improve feel but may increase drug mobility or weaken structural integrity. The best formulation strategy is therefore one that treats adhesion and release as interconnected performance variables rather than as separate optimization tasks.
| Polymer Property | Effect on Adhesion | Effect on Drug Loading | Effect on Release Control | Effect on Skin Comfort | Main Formulation Trade-Off |
|---|
| High Tack / Soft Adhesive Behavior | Improves initial skin contact | May limit stable high loading | Can increase early release if diffusion is less restricted | Usually improves conformity | Better contact versus structural stability |
| Dense Matrix Structure | May reduce flexibility if excessive | Can support more stable drug state in some systems | Slows diffusion and can extend duration | May feel less comfortable if too rigid | Longer release versus comfort |
| High Hydrophilicity | Can alter adhesive performance under moisture | May favor some drug classes but destabilize others | Often accelerates matrix mobility and release | Can improve soft feel but raise sensitivity to humidity | Transport support versus storage stability |
| Barrier-Forming / Low-Permeability Behavior | Usually neutral to moderate effect | May constrain useful loading if drug mobility is too low | Improves release restraint and flux regulation | Depends on flexibility of the full layer system | Better control versus slower input |
Evaluation of Patch Quality and Delivery Performance
Evaluating a long-acting patch means assessing more than the amount of drug released. The system must maintain adhesion, preserve drug distribution, support predictable transport, and remain stable under wear and storage conditions. Because these properties influence one another, a useful test strategy links patch mechanics to release and release to skin transport. This helps reveal whether the formulation is robust enough to function as a practical long-acting system rather than only a promising laboratory film.
- Adhesion, Tack, and Peel Properties: Adhesion testing is critical because loss of skin contact often leads directly to reduced or inconsistent delivery. Tack influences initial fixation, peel affects removal and edge-lift resistance, and cohesive strength helps determine whether the adhesive maintains integrity during wear. These properties should always be interpreted alongside intended wear duration and matrix composition.
- Drug Content Uniformity and Release Testing: Uniform drug content is essential for reproducible dose delivery, especially in long-acting systems where small differences in internal distribution may become more significant over time. Release testing should capture both early and sustained phases, since acceptable total release can still hide an undesirable front-loaded profile.
- Skin Permeation and In Vitro Diffusion Studies: A patch may release drug effectively from its own matrix and still fail as a transdermal system if transport across skin remains insufficient. Permeation testing helps determine whether the architecture produces a transport profile that actually matches the intended long-acting delivery objective.
- Stability Under Heat, Humidity, and Packaging Stress: Heat and humidity can change polymer mobility, adhesive behavior, and drug crystallization risk, making packaging part of the performance system rather than only a container. A robust long-acting patch should preserve both mechanical properties and release behavior through storage without major shifts in structure or drug state.
When Polymeric Patches Are the Right Choice?
Polymeric patches are the right long-acting platform when the delivery objective can be achieved through controlled skin transport without the need to mechanically bypass the barrier or implant a larger sustained-release device. Their strongest value lies in combining noninvasive wear, tunable release, and practical manufacturability. However, they are not universally suitable. The platform works best when the payload, target flux, and skin compatibility all fit the limits of patch-based delivery.
- Best-Fit Payload and Product Scenarios: Polymeric patches are especially useful for drugs that can be delivered through the skin at meaningful rates and benefit from prolonged, relatively even input over time. They are also attractive when the product concept values user convenience, defined wear periods, and scalable manufacturing with well-controlled matrix or reservoir architecture.
- Main Limitations of Patch-Based Long-Acting Delivery: The largest limitations are often skin permeability, drug loading limits, crystallization risk, and the challenge of maintaining stable adhesion while also controlling release. A patch may be elegantly designed yet still fail if the payload is fundamentally unsuited to passive or enhancer-assisted skin transport.
- When Microneedles, Hydrogels, or Implants May Be Better: Microneedles may be more appropriate when the main challenge is bypassing the stratum corneum rather than fine-tuning passive transport. Hydrogel-oriented systems may be preferable when hydration-mediated interfacial behavior dominates the design logic. Implants or depot systems may offer better control when much longer duration or larger payload capacity is required than a wearable patch can realistically support.
Research Applications of Polymeric Patches in Drug Delivery
Polymeric transdermal patches support a wide range of research applications because they combine controlled release, wearable administration, and material tunability within a relatively mature dosage platform. In research and formulation development, their value is especially clear when the objective is to maintain more stable input over time, reduce rapid exposure fluctuation, or create a noninvasive delivery route for molecules that are difficult to manage with conventional dosage forms. The specific application value of a patch depends on the interaction among payload potency, skin permeability, matrix compatibility, and the ability of the polymer system to maintain both adhesion and release consistency during wear.
Small-Molecule Research for Sustained Transdermal Input
Polymeric patches are most widely studied for potent small molecules that can cross the skin barrier at a practical rate while remaining stable within adhesive or matrix-based systems. In this area, researchers typically focus on drugs that benefit from prolonged and relatively even exposure rather than rapid peak concentration. The main formulation questions usually involve whether the molecule remains molecularly dispersed in the patch, whether it crystallizes during storage, and whether the selected polymer matrix can support both controlled release and acceptable skin contact. This is why small-molecule patch research often centers as much on matrix thermodynamics and phase behavior as on skin permeation itself.
Hormones and Other Low-Dose Molecules Requiring Stable Exposure
Polymeric patches are also highly relevant in research on hormones and other low-dose bioactive molecules where exposure stability is a major design goal. For these systems, the scientific challenge is not simply delivering the molecule through the skin, but maintaining a reproducible release rate over the intended wear period while minimizing variability caused by adhesive changes, drug migration, or matrix aging. Literature in this area frequently uses polymer patches as model systems for studying long-duration input, because even small changes in matrix composition or membrane resistance can significantly alter the delivery profile of low-dose molecules.
Analgesic, Neuromodulatory, and CNS-Active Molecule Delivery
Polymeric patches are often investigated for analgesic and neuromodulatory molecules when sustained transdermal input is preferred over repeated short-interval administration. In these studies, the patch platform is valued for its ability to smooth exposure over time and reduce the dependence on rapid absorption cycles. Research examples in this space commonly focus on how polymer composition affects release uniformity, how skin transport can be sustained without excessive burst, and how long-wear adhesion influences dose consistency. As a result, this area often serves as a model for studying the relationship between matrix design and prolonged pharmacokinetic input.
Molecules with Limited Oral or Conventional Delivery Suitability
Another important application area involves molecules that are less suitable for conventional oral delivery because of stability limitations, variable absorption, or the need for more controlled systemic exposure. In these cases, polymeric patches are studied as an alternative platform that can bypass some formulation constraints associated with gastrointestinal processing while introducing a different set of challenges related to skin permeability and matrix compatibility. Research in this category often emphasizes permeation enhancement, drug solubilization within the polymer phase, and methods to prevent crystallization or phase separation during storage.
Functional and Responsive Patch Systems for Advanced Delivery Research
Beyond conventional patch formats, polymeric systems are increasingly used in advanced delivery research involving responsive matrices, hydrogel-assisted interfaces, multilayer wearable systems, and hybrid transdermal structures. These studies typically examine how smart polymers, hydration-sensitive materials, or layered transport barriers can improve residence, comfort, or release control for more demanding payloads. In the literature, such systems are often explored not because every design is immediately practical, but because they help clarify how polymer structure, water uptake, and interfacial transport can be engineered together in next-generation transdermal platforms.
Disease-Oriented Research Areas Commonly Linked to Patch Development
From a research perspective, polymeric patches are most often associated with long-term delivery concepts relevant to pain-related disorders, hormone-regulated conditions, cardiovascular support, smoking-cessation research, and selected central nervous system applications. The common thread across these fields is not the disease label itself, but the need for sustained input, manageable dosing frequency, and a platform capable of maintaining relatively stable delivery over time. For this reason, patch research is often organized around exposure control requirements rather than around one narrowly defined molecular class.
Services
Advanced Polymer Synthesis and Transdermal Patch Development Services
At BOC Sciences, polymeric transdermal patch development is treated as an integrated material-and-transport design problem, where matrix chemistry, adhesive behavior, skin contact, and long-acting release must be aligned from the outset. Our team supports patch programs through polymer synthesis, material screening, formulation strategy development, and performance-oriented characterization, helping clients translate polymer selection into practical patch architecture and predictable skin delivery. By connecting polymer function with wear behavior, drug distribution, and transport control, BOC Sciences provides technical support for transdermal systems designed to achieve sustained and long-acting performance.

Custom Polymer & Patch Material Design
- Selection of adhesive, matrix, membrane, and backing-support materials.
- Polymer matching based on loading, adhesion, and release-control targets.
- Support for hydrophilic, hydrophobic, and barrier-forming patch materials.
- Material strategies tailored to long-acting transdermal delivery goals.

Patch Architecture and Formulation Development
- Support for drug-in-adhesive, matrix, reservoir, and multilayer designs.
- Optimization of internal drug distribution and crystallization control.
- Guidance on release-barrier selection and structural integration.
- Formulation strategies that balance adhesion, comfort, and duration.

Skin Transport and Release Evaluation
- Assessment of release profiles from matrix and reservoir systems.
- Support for diffusion and permeation study design.
- Evaluation of polymer effects on transport consistency and wear behavior.
- Interpretation of structure–release relationships in long-acting patches.

Stability and Packaging Support
- Assessment of humidity, heat, and packaging effects on patch integrity.
- Adhesion and storage-risk evaluation for long-wear systems.
- Support for reproducibility across patch batches and matrix structures.
- Development workflows aimed at practical, robust patch performance.
Need a Consultation?
Talk with BOC Sciences about polymeric transdermal patch development, from material screening and architecture selection to skin transport and sustained-release optimization.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What polymers are commonly used in long-acting transdermal patches?
Long-acting transdermal patches commonly use pressure-sensitive adhesive polymers, matrix-forming polymers, membrane-forming materials, and flexible backing-layer polymers. The final choice depends on drug compatibility, required adhesion, target release duration, and whether the patch is designed as a drug-in-adhesive, matrix, or reservoir system.
-
What is the difference between matrix and reservoir transdermal patches?
Matrix patches distribute the drug throughout a continuous polymer phase, so release depends mainly on diffusion through the matrix. Reservoir patches keep the drug in a separate compartment and use a membrane or barrier layer to regulate transport, often allowing tighter control but with greater structural complexity.
-
How do polymeric patches control drug release through skin?
Polymeric patches control release by regulating how the drug is stored, dissolved, and transported within the patch before it reaches the skin surface. Polymer composition, matrix density, membrane resistance, and interfacial contact all influence whether the patch delivers a rapid burst, a sustained profile, or a more stable long-acting input.
-
What affects adhesion and release performance in transdermal patches?
Adhesion and release are influenced by adhesive composition, polymer mobility, drug loading, moisture sensitivity, and backing-layer design. A patch must maintain stable skin contact while also preserving internal drug distribution and release control. If adhesion changes during wear, delivery consistency can decline even when the formulation looks acceptable initially.
-
When are polymeric patches better than microneedles?
Polymeric patches are often the better choice when the drug can cross intact skin at a useful rate and the product concept favors noninvasive, wear-based delivery. They are especially suitable when long-acting performance can be achieved through controlled diffusion, without the added complexity of barrier disruption or microneedle device integration.

 Fig. 1. Typical layer structure of polymeric transdermal patch systems (BOC Sciences Authorized).
Fig. 1. Typical layer structure of polymeric transdermal patch systems (BOC Sciences Authorized). Fig. 2. How polymeric patch structures control skin transport and sustained release (BOC Sciences Authorized).
Fig. 2. How polymeric patch structures control skin transport and sustained release (BOC Sciences Authorized).