Polymeric Microneedles and Transdermal Systems for Long-Acting Drug Delivery
Polymer-based microneedles and transdermal systems are emerging as versatile long-acting drug delivery platforms because they combine skin-access enhancement with tunable release control. By integrating needle geometry, matrix design, reservoir coupling, and material selection, these systems can support more prolonged input than conventional topical formulations while avoiding the complexity of larger implantable devices. Depending on the platform design, long-acting behavior may come from rapid skin barrier penetration followed by sustained release from the needle matrix, or from a microneedle-enabled pathway that allows a connected patch or reservoir to maintain controlled delivery over an extended period.
Resources
Polymeric Microneedles and Transdermal Systems
Long-acting skin delivery platforms occupy a unique space in formulation development because they sit between conventional transdermal patches and more invasive depot or implant systems. Polymeric microneedles can create transient microchannels in the stratum corneum, deposit drug directly into the upper skin layers, or act as structural interfaces that connect the skin to a sustained-release matrix. At the same time, polymer-based transdermal systems can extend input through hydrated networks, backing-supported reservoirs, or patch-integrated release layers. The result is a broad design space in which material choice, device architecture, and release logic must be considered together rather than separately.
Definition of Polymeric Microneedles
Polymeric microneedles are microstructured projections fabricated from drug-compatible polymers and designed to penetrate the outer skin barrier without relying on conventional hypodermic injection geometry. Depending on the application, they may dissolve after insertion, swell while remaining structurally intact, or serve as coated or reservoir-coupled structures that facilitate delivery from a secondary drug source. Their role in long-acting delivery is not simply to puncture skin, but to create a controlled interface through which drug deposition, local retention, or sustained transdermal input can be engineered.
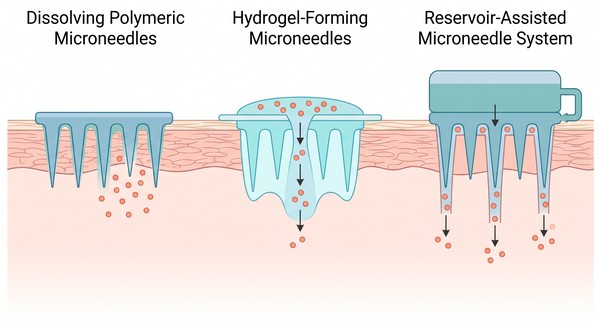 Fig. 1. Drug delivery pathways in long-acting microneedle-enabled transdermal systems (BOC Sciences Authorized).
Fig. 1. Drug delivery pathways in long-acting microneedle-enabled transdermal systems (BOC Sciences Authorized).
How Microneedles Differ from Conventional Transdermal Systems?
Conventional transdermal systems generally depend on passive diffusion across intact skin, which limits the range of payloads that can be delivered and constrains the achievable input rate. Microneedles differ because they actively reduce the barrier effect of the stratum corneum. This makes them especially valuable for molecules that would otherwise show poor skin permeation. In practice, polymeric microneedles are best viewed as an enabling structure rather than only a dosage form: they may deliver drug directly from the needle itself, or they may serve as the access component of a broader transdermal system that continues release after insertion.
Key Features of Long-Acting Skin Delivery Platforms
A skin-based platform becomes truly relevant to long-acting drug delivery when it does more than improve initial penetration. It must also support prolonged residence, delayed depletion, or controlled input over time. That performance is strongly influenced by the use of biodegradable polymers, swellable matrix networks, reservoir-coupled structures, and release-modulating backing layers. In other words, a long-acting microneedle system is defined not only by insertion, but by what happens after insertion.
Why Use Microneedles for Long-Acting Drug Delivery?
The main reason to use microneedles for long-acting delivery is that many payloads cannot achieve meaningful or reliable exposure through passive topical or transdermal routes alone. A conventional formulation may remain largely on the skin surface, diffuse too slowly, or deliver drug in a profile that is too short to be useful. Microneedles address this by improving barrier access, while polymer-based matrices and connected systems provide the structural basis for release that continues beyond the initial application step.
Limits of Conventional Topical and Transdermal Delivery
Traditional creams, gels, and standard patches often struggle to maintain prolonged input for hydrophilic molecules, large molecules, or drugs with unfavorable skin partition behavior. Even when passive permeation is possible, the resulting delivery rate may be too variable or too low for a long-acting product concept. The skin barrier therefore becomes the central limitation. Without an active access strategy, the formulation may be forced into a narrow range of payload classes and flux profiles.
How Microneedles Improve Skin Barrier Penetration?
Microneedles improve delivery by creating controlled micro-scale entry points through the outer barrier layer. This reduces the dependence on passive diffusion through intact skin and expands the design space for both payload selection and release engineering. In dissolving systems, the drug can be placed directly into the skin as the microneedle matrix dissolves. In hydrogel-forming systems, the needles may remain as hydrated conduits that connect a patch or reservoir to the skin. In both cases, the initial insertion event is only part of the platform logic; the real development question is how to sustain input afterward.
Why Polymer-Controlled Release Matters?
Polymer-controlled release matters because barrier access alone does not guarantee long-acting performance. A platform may insert successfully and still fail to provide sustained delivery if the matrix dissolves too quickly, the reservoir depletes too rapidly, or the skin-device interface lacks consistency. Release control depends on polymer hydration, swelling, dissolution rate, drug diffusion path length, and structural integrity under use conditions. These variables determine whether the system behaves like a short-lived penetration aid or a true long-acting transdermal platform.
Types of Polymeric Microneedles for Long-Acting Delivery
Polymeric microneedles are not a single platform class. Their performance depends heavily on how the needle itself behaves during and after application. Some systems disappear by dissolution, some remain as hydrated interfaces, and others rely on coatings or coupled reservoirs rather than a fully drug-loaded needle matrix. Understanding these distinctions is essential for selecting a platform that truly supports long-acting delivery rather than only short-term enhanced penetration.
Dissolving Microneedles
Dissolving microneedles are fabricated from water-soluble or rapidly hydrating polymer matrices that soften or dissolve after insertion. Their main value lies in depositing a dose into the skin without leaving behind a rigid structure. For long-acting delivery, their usefulness depends on whether the inserted dose itself is sustained by local matrix behavior, secondary depot formation, or controlled diffusion from the skin deposition zone. If dissolution is too rapid and the payload diffuses away quickly, the system behaves more like an enhanced immediate-delivery platform than a long-acting one.
Hydrogel-Forming Microneedles
Hydrogel-forming microneedles are especially relevant to long-acting delivery because they can remain in place as hydrated interfaces rather than dissolving completely. After insertion, they swell and create a transport pathway between the skin and an attached drug reservoir or patch layer. This allows the platform to separate the penetration function from the bulk of the drug load. In many development programs, this architecture provides a more flexible route to sustained input than fully dissolving needles, particularly when the product concept requires longer application times or tighter control over delivery kinetics through polymer hydrogels.
Coated, Hollow, and Reservoir-Based Systems
Other microneedle-enabled systems include coated needles, hollow microstructures, and reservoir-based devices. Coated systems typically carry drug on the needle surface and may be more suitable for rapid deposition than for extended release unless combined with a secondary sustained component. Hollow systems create fluid pathways for active transport, while reservoir-based systems allow the needle array to function as an interface between the skin and a larger controlled-release matrix. Among these, reservoir-assisted concepts often show the strongest alignment with long-acting transdermal objectives because they can decouple insertion performance from total drug load and release duration.
Drug Release After Skin Insertion
Once the microneedles enter the skin, release may proceed through several distinct pathways. A dissolving matrix may liberate drug directly within hydrated skin tissue. A swelling interface may allow transport from a backing reservoir through the swollen polymer. A coated system may rely on rapid surface release followed by subsequent diffusion from the deposited zone. Because these mechanisms are not equivalent, long-acting performance must be defined in terms of the full system pathway, not just the needle format.
Polymer Selection for Microneedles and Transdermal Systems
Material selection is one of the most important determinants of long-acting behavior in microneedle-enabled delivery. The polymer must support enough mechanical strength for insertion, remain compatible with the payload, and provide the intended hydration or dissolution profile once applied. For true long-acting performance, the material also needs to support consistent post-insertion behavior, whether that means rapid dissolution followed by delayed tissue release, or prolonged swelling as a controlled conduit. This is why material selection is best approached as a structure–function problem rather than a simple list of acceptable excipients.
Water-Soluble Polymers for Dissolving Microneedles
Water-soluble polymers are commonly used when the goal is to form dissolving microneedles that release their payload after insertion. Materials such as Polyvinyl Alcohol (PVA), Polyethylene glycol, and selected polysaccharide or protein-derived matrices are attractive because they can be cast into needle structures and later hydrate efficiently in skin tissue. The challenge is that the same hydrophilicity that supports fast dissolution may also shorten the effective release window if the rest of the system does not provide a secondary sustained mechanism.
Hydrogel-Forming and Swellable Polymers
Swellable materials are central to hydrogel-forming microneedles and other long-acting transdermal interfaces. In these systems, the matrix should absorb fluid and expand without dissolving too quickly or collapsing structurally. Materials and blends based on chitosan, Alginate, Gelatin, and related hydrophilic networks may be considered when bioadhesion, swelling control, and interfacial transport are important. The correct choice depends on how much hydration is needed, how stable the swollen structure must remain, and whether the matrix is acting as the drug carrier, the conduit, or both.
Mechanical Strength and Dissolution Trade-Offs
One of the central material trade-offs in microneedle development is balancing insertion strength against post-application transformation. A matrix that dissolves very quickly may not be mechanically robust enough to penetrate reliably. A matrix that is extremely strong may dissolve or swell too slowly, reducing delivery efficiency. This trade-off becomes even more important in long-acting systems because the polymer must often support both successful insertion and controlled behavior after insertion. Material design therefore requires more than choosing a strong polymer or a soluble polymer; it requires choosing one that changes in the right way at the right time.
How Polymer Choice Affects Loading and Release?
Polymer choice affects not only device mechanics but also where the drug can be loaded, how uniformly it is distributed, and how the release path develops during use. Hydrophilic polymers may favor certain payload classes and release routes, while slower-eroding materials such as Polycaprolactone (PCL) or biodegradable matrices based on Poly(lactic-co-glycolic acid) (PLGA) may be more suitable in reservoir-associated components or supporting layers where prolonged release is desired. In every case, the important question is how the chosen polymer contributes to the full long-acting pathway rather than to a single isolated property.
| Microneedle / System Type | Main Long-Acting Mechanism | Polymer Requirement | Key Strength | Main Limitation |
|---|
| Dissolving Microneedles | Rapid skin insertion followed by matrix dissolution and local drug deposition | Fast-hydrating, formable, drug-compatible matrix | Simple administration and direct dose placement | May not provide a sufficiently long release window without secondary control |
| Hydrogel-Forming Microneedles | Swollen interface enables prolonged transport from connected systems | High swelling capacity with retained structural integrity | Good fit for sustained transdermal input | System complexity is usually higher than dissolving formats |
| Coated / Hollow / Reservoir-Based Systems | Barrier access combined with secondary drug source or active transport | Mechanically reliable interface plus compatible reservoir design | Flexible separation of insertion and release functions | More demanding device integration and consistency control |
Looking for Polymers and Device Support for Long-Acting Skin Delivery?
From microneedle matrices and hydrogel interfaces to reservoir-linked transdermal systems, we provide polymer-focused support for long-acting skin delivery development.
Formulation and Device Design for Long-Acting Performance
In microneedle-enabled long-acting delivery, performance emerges from the whole device architecture rather than from the needle composition alone. Geometry controls insertion depth and reliability, drug loading strategy determines where the payload resides, and backing or reservoir elements often define the duration of delivery. For this reason, device design should be treated as part of formulation development, not as a separate engineering step that happens afterward.
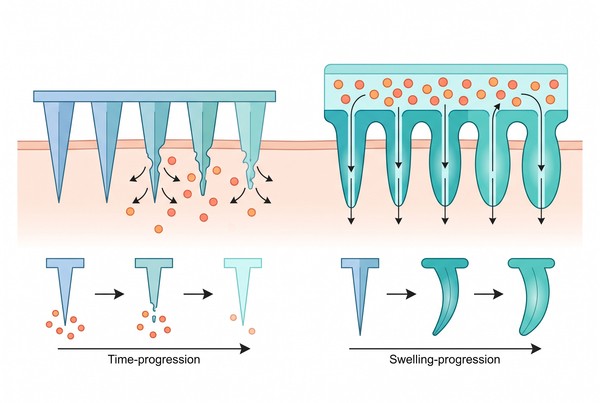 Fig. 2. Material-property trade-offs in dissolving and hydrogel-forming microneedles (BOC Sciences Authorized).
Fig. 2. Material-property trade-offs in dissolving and hydrogel-forming microneedles (BOC Sciences Authorized).
Needle Geometry and Insertion Depth
Needle height, base width, tip sharpness, aspect ratio, and array density all influence whether the microneedles insert consistently and reach the intended skin depth. Geometry must be compatible with the mechanical properties of the selected polymer, because even a well-chosen material can fail in use if the geometry is too aggressive or too fragile. For long-acting delivery, insertion depth also affects the local environment in which the drug is deposited or transported, which in turn influences release kinetics and reproducibility.
Drug Loading in Tips, Matrices, and Reservoirs
The placement of drug within the system is a major design variable. Tip loading can improve delivery efficiency when only a small quantity needs to be inserted into the skin quickly. Full matrix loading may simplify fabrication, but it can also couple total dose and mechanical behavior too tightly. Reservoir coupling is often preferable for long-acting systems because it allows the microneedle array to function mainly as a skin-access interface while the larger drug load resides in a connected transdermal compartment. The optimal strategy depends on dose, stability, required duration, and the desired balance between immediate and delayed release.
Backing Layers and Patch Integration
Backing layers and support structures are especially important when microneedles are integrated with transdermal systems rather than used as stand-alone dissolving arrays. These layers may provide occlusion, protect the device, serve as a drug reservoir, or maintain contact over extended wear periods. Flexible structural polymers such as Polyurethane (PU) may be relevant when the device requires both skin conformity and mechanical support. In practical formulation terms, the backing is not a passive accessory. It can determine wear stability, moisture management, and effective long-acting input.
Moisture Sensitivity and Packaging Design
Many polymeric microneedles are highly sensitive to environmental moisture because hydration directly alters mechanical strength, swelling behavior, and drug stability. This makes drying history and packaging design central to performance. A system that is nominally strong enough for insertion may soften during storage if humidity control is poor. Conversely, excessive drying may affect polymer brittleness or compromise payload stability. Long-acting systems place even greater demands on packaging because they rely on carefully tuned post-application behavior that must remain stable until use.
Long-Acting Release Strategies in Microneedle Systems
Long-acting release in microneedle-enabled systems can be achieved through more than one pathway. In some cases, the microneedles themselves carry the dose and release it over time after insertion. In other cases, the needles primarily create or maintain access while a connected reservoir or patch governs the prolonged input phase. Understanding this distinction is essential, because it determines which component truly controls duration, which material properties matter most, and where optimization effort should be concentrated.
Immediate Insertion with Sustained Post-Application Release
Some systems use the insertion step to establish rapid access and then rely on the behavior of the inserted or deposited matrix to extend delivery. This can occur when dissolving microneedles deposit drug-rich material in the skin or when layered formulations create a delayed diffusion pathway after insertion. These approaches can support long-acting behavior, but only when the deposited material or local release environment has enough structural persistence to prevent immediate depletion.
Hydrogel Microneedles Combined with Reservoir Systems
One of the most coherent long-acting strategies involves hydrogel-forming microneedles coupled to a transdermal reservoir or patch layer. In this architecture, the swollen needles act as a pathway rather than the primary drug store. The benefit is that release duration can be adjusted through the reservoir composition, patch design, and interfacial transport resistance without overloading the needle matrix itself. This is often more scalable for prolonged input than forcing all long-acting behavior into a dissolving needle body.
Microneedle-Assisted Transport vs Direct Payload Release
A useful way to classify microneedle systems is to ask whether the needles mainly release payload directly or mainly assist transport from another source. Direct-release systems are typically simpler in concept, but they may be constrained by how much drug can be loaded into the needles. Microneedle-assisted transport systems can support larger total payloads and longer application periods, but they require stronger integration between the needle array and the secondary transdermal structure. Neither approach is universally better; the right choice depends on dose, duration, payload type, and formulation stability.
Payload Fit by Molecule Type
Payload class strongly influences which long-acting transdermal strategy is realistic. Small molecules with sufficient potency and acceptable skin stability may fit either direct microneedle release or reservoir-coupled delivery. More hydrophilic or larger molecules may benefit more from microneedle-enabled pathways that bypass passive skin limitations. In some platform comparisons, alternative carriers such as polymer nanoparticles may be considered for different biodistribution goals, but microneedle-enabled systems remain particularly attractive when long-acting delivery must be achieved through the skin rather than through systemic carrier circulation.
Performance Evaluation and Platform Selection
Evaluation of long-acting microneedle systems must go beyond proving that the needles can pierce the skin. The real question is whether the system delivers the intended dose consistently over the intended period while preserving structural and formulation integrity. This requires a combined assessment of mechanics, transformation behavior, drug transport, and storage risk. It also requires an honest platform selection step, because not every long-acting delivery goal is best served by a microneedle-based system.
Mechanical Strength and Insertion Testing
Mechanical testing should confirm that the needles can tolerate handling and application without fracture, buckling, or incomplete insertion. However, strength alone is not enough. The system must also maintain consistent penetration across the array so that dose input remains reproducible. This is especially important when long-acting performance depends on sustained interface formation rather than only on initial deposition.
Dissolution, Swelling, and Release Behavior
After insertion, the matrix may dissolve, swell, or remain as a connected structure, and each pathway must be evaluated under realistic use conditions. The timing of these changes determines whether the product behaves as a rapid-release enhancer or as a platform capable of prolonged input. Release testing should therefore focus on transformation kinetics as well as total drug liberation, with attention to how the system behaves over the full intended wear or residence period.
Skin Permeation and Dose Consistency
Skin permeation studies are necessary to understand whether the created pathway produces stable transport or only short-lived enhancement. For long-acting systems, dose consistency over time is often more important than peak delivery rate. A system that inserts well but shows variable transport or premature depletion may not meet the product target even if its initial delivery is impressive. This is why permeation and dose-reproducibility testing should be interpreted together.
Stability, Risk, and Platform Choice
Long-acting microneedles are the right choice when the product concept benefits from skin-enabled prolonged input, moderate device complexity, and a polymer matrix that can be tuned for insertion and post-application behavior at the same time. They are especially attractive when a program needs a platform between standard patches and more invasive long-acting systems. However, they are not always the best option. Conventional patches may be sufficient when passive transdermal flux is already adequate. Hydrogel-heavy systems may be better when prolonged hydrated interfacing dominates the design logic. Depots or implants may be preferable when very long residence and highly defined release geometry are required. Careful platform selection therefore depends on aligning payload needs, duration goals, and realistic device constraints.
When Microneedles Are the Right Long-Acting Platform?
Polymeric microneedles are not automatically the best option for every long-acting skin-delivery goal, but they are highly valuable when a project requires both barrier bypass and controlled post-application release. Their strongest advantage lies in combining skin access with tunable polymer behavior, allowing developers to design platforms that balance insertion reliability, drug loading, and sustained input. At the same time, microneedle systems must be evaluated against other long-acting formats to ensure that the selected platform truly matches the payload, target duration, and overall formulation strategy.
Best-fit Scenarios for Long-Acting Microneedle-Enabled Delivery
Microneedle-enabled systems are particularly well suited when passive transdermal delivery is too limited to achieve the intended therapeutic input, yet a more invasive long-acting platform is not desirable. They are often a strong fit for product concepts that need controlled skin penetration, localized deposition, or sustained transport through a polymer-mediated interface. These systems are also attractive when the formulation benefits from separating the penetration function from the main drug reservoir, as seen in hydrogel-forming or reservoir-assisted architectures. In practical development terms, polymeric microneedles are most useful when long-acting delivery depends on both device structure and material-controlled release rather than on passive diffusion alone.
Key Limitations and Material-Design Constraints
Despite their versatility, polymeric microneedles impose several important material and design constraints. The selected polymer must provide enough mechanical integrity for reliable insertion while still delivering the intended dissolution, swelling, or interfacial transport behavior after application. This often creates trade-offs between strength and transformation rate, especially in systems designed for sustained delivery. Drug loading capacity may also be limited in fully matrix-based microneedles, making certain long-duration concepts more difficult unless a secondary reservoir is introduced. In addition, moisture sensitivity, storage stability, and reproducible skin contact can all affect whether a microneedle platform remains practical beyond early-stage feasibility.
When Patches, Hydrogels, Depots, or Implants May Be Better?
Other long-acting platforms may be more appropriate when the target profile depends less on skin barrier penetration and more on sustained bulk release, structural persistence, or very long-duration dosing. Conventional patches may be sufficient when passive transdermal flux is already adequate and no microneedle-enabled access is needed. Polymer hydrogels may be a better choice when prolonged hydrated interfacing or local matrix retention is the dominant requirement. Depots and implants may outperform microneedles when the product concept requires highly defined geometry, larger payload capacity, or much longer release duration than a wearable skin platform can reasonably maintain. For this reason, microneedles should be selected when their combined advantages in skin access, polymer tunability, and controlled transdermal delivery clearly align with the intended long-acting design.
Services
Advanced Polymer Synthesis and Microneedle Development Services
At BOC Sciences, we approach long-acting skin delivery from a polymer-engineering perspective, connecting material design with practical device and formulation performance. Our team supports projects involving dissolving microneedles, hydrogel-forming interfaces, reservoir-integrated transdermal systems, and related polymer-enabled delivery formats through polymer synthesis, material screening, formulation optimization, and performance-oriented characterization. By aligning polymer structure, mechanical behavior, drug compatibility, and release logic, BOC Sciences helps developers build microneedle and transdermal systems that are not only manufacturable, but also better matched to long-acting delivery goals.

Custom Polymer & Material Design
- Selection of drug-compatible polymers for dissolving and hydrogel-forming microneedles.
- Support for hydrophilic, swellable, and biodegradable matrix development.
- Optimization of mechanical behavior, hydration profile, and release function.
- Material design aligned with long-acting skin delivery targets.

Microneedle Formulation & Device Support
- Guidance on needle geometry, drug placement, and backing-layer integration.
- Support for dissolving, swellable, and reservoir-assisted platform selection.
- Optimization of matrix loading, tip loading, and patch-connected architectures.
- Evaluation of formulation variables that shape long-acting performance.

Release and Performance Evaluation
- Mechanical testing, insertion reliability, and transformation behavior assessment.
- Dissolution, swelling, and sustained-input evaluation under relevant conditions.
- Drug loading and compatibility analysis for polymer matrices and reservoirs.
- Support for long-acting release interpretation and platform comparison.

Transdermal System Integration
- Design support for microneedle-enabled transdermal patches and reservoirs.
- Material matching across needle arrays, backing layers, and connected delivery elements.
- Moisture, packaging, and storage-risk assessment for polymeric skin-delivery systems.
- Development workflows tailored to long-acting transdermal product concepts.
Need a Consultation?
BOC Sciences supports polymeric microneedle and transdermal system development with material selection, formulation design, and long-acting release strategy guidance.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What polymers are commonly used in polymeric microneedles for long-acting delivery?
Common choices include water-soluble matrix polymers for dissolving systems, hydrogel-forming polymers for swollen interface systems, and supportive structural polymers for backing or reservoir-integrated designs. The right choice depends on insertion strength, transformation behavior, and the intended release pathway.
-
How do dissolving microneedles differ from hydrogel-forming microneedles?
Dissolving microneedles usually release drug as the needle matrix hydrates and disappears, while hydrogel-forming microneedles typically swell and remain as transport interfaces that connect the skin to an attached reservoir or patch.
-
Can polymeric microneedles really provide long-acting drug delivery?
Yes, but long-acting performance depends on the full platform architecture. Some systems rely on sustained release from deposited material, while others rely on prolonged transport through a swollen or reservoir-coupled interface.
-
What controls insertion strength, dissolution speed, and release duration?
These properties are controlled by polymer choice, needle geometry, matrix density, hydration behavior, payload placement, and whether the system is integrated with a patch or reservoir.
-
When are microneedles better than conventional transdermal patches?
Microneedles are especially useful when passive skin permeation is insufficient, when the payload needs more direct skin access, or when long-acting delivery requires a controlled interface beyond what a standard patch can provide.

 Fig. 1. Drug delivery pathways in long-acting microneedle-enabled transdermal systems (BOC Sciences Authorized).
Fig. 1. Drug delivery pathways in long-acting microneedle-enabled transdermal systems (BOC Sciences Authorized). Fig. 2. Material-property trade-offs in dissolving and hydrogel-forming microneedles (BOC Sciences Authorized).
Fig. 2. Material-property trade-offs in dissolving and hydrogel-forming microneedles (BOC Sciences Authorized).