Polymeric Micelles for Poorly Soluble Drugs: Enhanced Solubility and Controlled Delivery
High hydrophobicity severely restricts systemic absorption, resulting in erratic bioavailability and rendering many fundamentally promising molecules pharmacologically ineffective. Traditional formulation approaches, which often rely on harsh organic co-solvents or low-molecular-weight surfactants, frequently introduce severe dose-limiting toxicities and thermodynamic instability upon systemic administration. Polymeric micelles have emerged as a highly sophisticated, supramolecular solution to these pervasive delivery challenges. Through the spontaneous self-assembly of amphiphilic block copolymers, these advanced nanocarriers form a thermodynamically stable core-shell architecture. The inner lipophilic core serves as an optimized nanoreservoir to encapsulate and seamlessly solubilize hydrophobic drugs, while the protective hydrophilic corona prevents aggregation and significantly prolongs systemic circulation.
Resources
Introduction to Challenges in Poorly Soluble Drug Delivery
The transition of newly discovered chemical entities from the laboratory bench to viable pharmaceutical formulations is frequently obstructed by a critical physicochemical barrier: severe hydrophobicity. In contemporary drug discovery, high-throughput screening and structure-based design heavily favor large, lipophilic molecules that bind tightly to target receptors. Consequently, a staggering majority of pipeline compounds fall into the Biopharmaceutics Classification System Class II and Class IV, characterized by extremely low aqueous solubility. This inherent insolubility prevents these molecules from seamlessly integrating into the highly aqueous environment of biological systems. Overcoming this thermodynamic barrier requires sophisticated formulation engineering that can bridge the gap between lipophilic molecular structures and the necessary aqueous delivery mediums without compromising molecular stability.
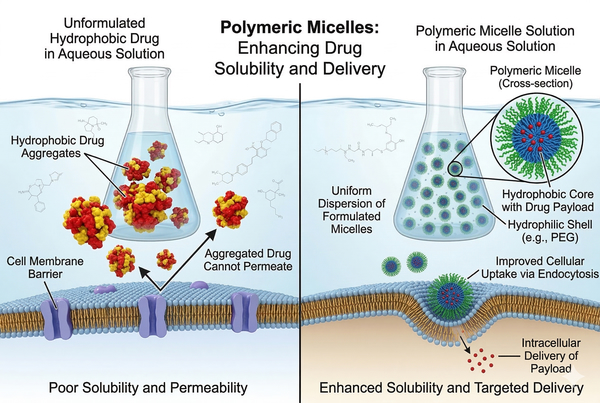 Fig. 1. Illustrating how polymeric micelles overcome drug solubility and delivery barriers (BOC Sciences Authorized).
Fig. 1. Illustrating how polymeric micelles overcome drug solubility and delivery barriers (BOC Sciences Authorized).
The Impact of Poor Aqueous Solubility on Bioavailability
Aqueous solubility is the fundamental rate-limiting step for systemic absorption. According to the principles governed by the Noyes-Whitney equation, the dissolution rate of a solid compound is directly proportional to its solubility in the surrounding medium. When an active pharmaceutical ingredient lacks sufficient solubility, it fails to dissolve adequately in gastrointestinal fluids or blood plasma. This failure creates a cascade of pharmacokinetic challenges. Un-dissolved drug particles cannot diffuse across the unstirred water layer surrounding cellular membranes, nor can they partition into the lipid bilayer for transcellular transport. Instead of being absorbed, hydrophobic molecules tend to agglomerate, leading to erratic absorption profiles, sub-therapeutic plasma concentrations, and extremely high intra-subject variability. Ultimately, this physicochemical limitation directly translates to dismal bioavailability, rendering the active ingredient virtually useless regardless of its intrinsic receptor affinity.
Limitations of Conventional Delivery Methods
Historically, formulation scientists have relied on brute-force techniques to force hydrophobic compounds into solution, but these conventional methods present severe limitations. Techniques such as physical micronization or nano-milling increase the surface area of the drug, but the resulting high surface energy often leads to rapid re-agglomeration or Ostwald ripening during storage. Liquid formulations traditionally utilize organic co-solvents like ethanol or propylene glycol, or low-molecular-weight surfactants. While these excipients can temporarily solubilize the drug, they suffer from a fatal flaw: upon injection and subsequent infinite dilution in the bloodstream, the solvent capacity drops precipitously, causing the immediate precipitation of the drug into hazardous crystalline emboli. Furthermore, traditional surfactants are notorious for interacting with cellular membranes, leading to dose-limiting toxicities, severe hemolysis, off-target nerve damage, and profound hypersensitivity reactions. These toxicological profiles drastically narrow the therapeutic index of the formulation.
Importance of Solubility Enhancement for Therapeutic Efficacy
Addressing the solubility barrier is not merely an exercise in formulation convenience; it is a strict prerequisite for achieving predictable and reproducible therapeutic efficacy. Proper solubilization ensures that the active pharmaceutical ingredient follows linear and predictable pharmacokinetics, establishing true dose proportionality. When a drug is fully dissolved, formulation scientists can map an accurate dose-response curve, which is impossible when absorption is erratically limited by dissolution rates. Moreover, effective solubility enhancement allows for a massive reduction in the total administered dose. By ensuring that a higher percentage of the drug actually enters systemic circulation, the requisite payload is minimized. This targeted efficiency significantly reduces the metabolic burden on clearance organs like the liver and kidneys, and minimizes localized gastrointestinal toxicity caused by unabsorbed drug irritating the mucosal lining.
Why Polymeric Micelles Offer a Superior Alternative for Formulation?
Polymeric micelles present a thermodynamically stable, structurally elegant, and highly tunable solution to these pervasive solubility challenges. Unlike harsh surfactants, these supramolecular assemblies are formed from amphiphilic block copolymers that spontaneously self-assemble in aqueous media driven by an entropy-favorable process. They organize into a distinct core-shell architecture: the inner hydrophobic core acts as a highly efficient nanoreservoir, sequestering poorly soluble drugs via hydrophobic interactions and physical entrapment. Simultaneously, the dense hydrophilic corona provides robust steric stabilization. This outer shell relies on entropic repulsion to prevent micellar aggregation and acts as a biological shield against rapid clearance mechanisms. Crucially, block copolymers possess a significantly lower critical micelle concentration compared to conventional surfactants. This grants polymeric micelles extraordinary thermodynamic and kinetic stability, ensuring they maintain their structural integrity and hold their payload securely even upon extreme dilution in systemic circulation. Their nanoscopic size and modular chemistry make them vastly superior to traditional delivery systems, offering a highly controlled and biocompatible pathway for hydrophobic drug delivery.
Key Advantages of Polymeric Micelles for Hydrophobic Drugs
The unique architectural features of polymeric micelles confer multiple physicochemical and biological advantages over traditional lipid-based or surfactant-based delivery systems. By carefully selecting the block copolymer constituents, scientists can engineer nanocarriers that independently optimize drug loading, systemic circulation, and site-specific release.
- Significant Enhancement of Aqueous Solubility and Dissolution Rates: The primary function of a polymeric micelle is to encapsulate lipophilic compounds within its hydrophobic core through hydrophobic interactions, hydrogen bonding, or π-π stacking. This encapsulation dramatically increases the apparent aqueous solubility of the active ingredient, often by several orders of magnitude. Consequently, the dissolution rate is accelerated, ensuring the drug remains in a bioavailable state upon administration.
- Controlled and Sustained Drug Release: Unlike simple surfactant micelles that may rapidly dissociate upon infinite dilution in the bloodstream, polymeric micelles boast a notably low critical micelle concentration. This thermodynamic stability ensures that the encapsulated payload is released in a controlled and sustained manner. Release kinetics can be modulated by altering the core-forming polymer length, crystallinity, and the strength of drug-polymer interactions.
- Improved Bioavailability and Targeted Cellular Uptake: The nanometer size range of polymeric micelles, typically between 10 and 100 nanometers, facilitates superior permeation across biological barriers. Their hydrophilic corona minimizes opsonization, extending circulation half-life and allowing the nanocarriers to accumulate in targeted tissues. Furthermore, micelles can be internalized by cells via endocytosis, efficiently delivering the hydrophobic payload directly into the intracellular compartment.
- Protecting Active Pharmaceutical Ingredients (APIs) from in vivo Degradation: Hydrophobic drugs, particularly complex natural products or easily hydrolyzable molecules, are highly susceptible to enzymatic degradation and rapid clearance. The dense hydrophilic shell of a polymeric micelle acts as a robust physical shield. It limits the diffusion of degrading enzymes and water molecules into the core, thereby preserving the structural integrity and pharmacological activity of the API during transit.
- Reduction of Systemic Toxicity and Off-Target Side Effects: By sequestering the active pharmaceutical ingredient within the core, polymeric micelles drastically reduce the interaction of the free drug with healthy, non-targeted tissues. This spatial confinement minimizes off-target toxicity. Additionally, replacing harsh traditional solubilizers with biocompatible block copolymers eliminates the severe side effects conventionally associated with excipient toxicity.
- Customizable Pharmacokinetics and Favorable Biodistribution: The pharmacokinetic profile of a micellar formulation can be fine-tuned by adjusting the molecular weight and ratio of the hydrophilic and hydrophobic blocks. A well-designed hydrophilic corona imparts a stealth effect, prolonging systemic circulation. This extended duration allows for a highly favorable biodistribution profile, ensuring a higher fraction of the administered dose reaches the desired localized microenvironment.
Block Copolymers Used in Polymeric Micelles
The functional performance, thermodynamic stability, and biological fate of a polymeric micelle are fundamentally dictated by the physicochemical properties of its constituent amphiphilic block copolymers. Designing an optimal micellar delivery system requires precise macromolecular engineering. Formulation scientists must meticulously match the structural characteristics of the polymer blocks to the specific properties of the hydrophobic payload. By manipulating variables such as molecular weight and the volume fraction of the segments, researchers exert exact control over the self-assembly process and the ultimate morphological architecture of the nanocarrier.
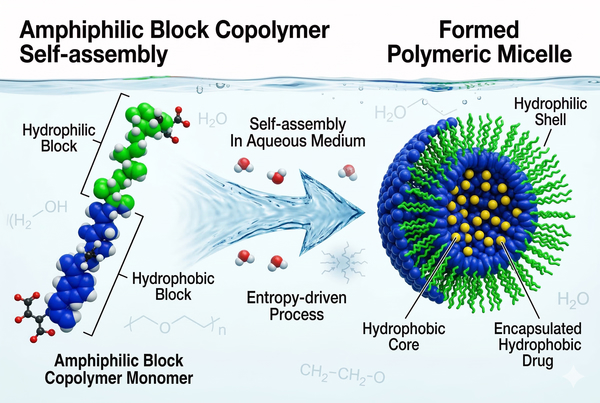 Fig. 2. Amphiphilic block copolymers self-assembling into a drug-loaded polymeric micelle (BOC Sciences Authorized).
Fig. 2. Amphiphilic block copolymers self-assembling into a drug-loaded polymeric micelle (BOC Sciences Authorized).
Hydrophilic Blocks in Copolymers
The hydrophilic block is responsible for forming the protective outer corona, which dictates the colloidal stability and systemic circulation of the micelle. Selecting the optimal hydrophilic polymer is critical for establishing a robust steric barrier against protein adsorption and opsonization.
- Polyethylene Glycol (PEG): Recognized as the industry standard due to its exceptional hydration capacity. Each ether oxygen in the polymer backbone coordinates with multiple water molecules, creating a dense, highly effective biological shield.
- Poly(2-oxazoline)s (POx): Advanced, next-generation alternatives that offer highly tunable hydrophilicity through side-chain variations. They possess lower innate viscosity, demonstrate remarkable chemical stability, and mitigate the accelerated blood clearance phenomenon associated with repeated polymer administration.
Hydrophilic Antifouling Polymers
Beyond synthetic polyethers, a diverse array of biomimetic and naturally derived polymers serves as exceptionally effective, multifunctional hydrophilic coronas. These macromolecules often provide distinct biological and physicochemical advantages over traditional synthetic blocks.
- Polyamino Acids: Materials such as poly(glutamic acid) or poly(aspartic acid) offer inherent enzymatic biodegradability alongside highly functional carboxyl groups, enabling straightforward secondary chemical modifications.
- Highly Hydrated Polysaccharides: Dextran, heparin, and chitosan confer massive hydration shells and extreme antifouling characteristics, significantly improving the thermodynamic stability of the micelle in complex biological fluids.
- Receptor-Targeting Polysaccharides: Hyaluronic acid and chondroitin sulfate possess intrinsic biological functionalities. For instance, hyaluronic acid naturally binds to CD44 receptors, transforming a simple stealth micelle into an actively targeted nanocarrier without the need for additional complex conjugation chemistry.
Hydrophobic Polymers in Block Copolymers
The hydrophobic core is the functional engine of the micelle, sequestering and stabilizing the poorly soluble active pharmaceutical ingredient. The selection of this core-forming block requires a profound thermodynamic affinity for the target drug to prevent premature leakage.
- Polyesters (PCL, PLA, PLGA): Extensively utilized due to their well-established hydrolytic degradation profiles, exceptional biocompatibility, and highly predictable clearance pathways.
- Polyethers (e.g., PPO): Provide temperature-dependent assembly characteristics, allowing for thermodynamic tuning of the core environment.
- Hydrophobic Polyamino Acids: Polymers like poly(L-leucine) or poly(L-phenylalanine) offer aromatic and hydrophobic side chains that facilitate strong π-π stacking and hydrogen bonding with complex lipophilic drugs, drastically enhancing loading capacity.
- Polyoxazolines and Polyoxazines: Deliver unprecedented synthetic modularity, allowing chemists to fine-tune the core environment to perfectly match the precise lipophilicity of almost any small molecule.
Stimuli-Responsive Block Copolymers
To achieve precise spatial and temporal control over drug release, block copolymers are engineered to undergo rapid architectural changes in response to specific localized microenvironmental triggers. This responsiveness ensures the payload is deployed exactly where it is needed.
- pH-Sensitive Architectures: Incorporate ionizable groups that protonate in acidic environments, such as endosomes. This protonation induces powerful electrostatic repulsion, destabilizing the micelle and facilitating rapid intracellular payload release.
- Thermo-Responsive Blocks: Utilize polymers with a highly specific lower critical solution temperature designed to undergo a sharp phase transition from hydrophilic to hydrophobic upon reaching a designated physiological temperature.
- Redox-Sensitive Backbones: Incorporate reversible disulfide bridges within the polymer structure. These bonds remain entirely stable in systemic circulation but are rapidly cleaved by the highly reducing environment created by elevated intracellular glutathione concentrations.
Drug Conjugates and Block Copolymer Complexes
Advancing beyond the physical encapsulation of payloads, contemporary formulation strategies often utilize precise chemical integration or robust non-covalent interactions to drastically improve the loading efficiency and stability of the micellar system.
- Polymer-Drug Conjugates (Prodrug Micelles): Covalently link the active pharmaceutical ingredient directly to the hydrophobic polymer block via stimulus-cleavable linkers, effectively eliminating the risk of premature burst release and achieving exceptionally high, reproducible loading.
- Block Copolymer Complexes (Polyion Complex Micelles): Rely on strong, non-covalent electrostatic coacervation between block ionomers and oppositely charged therapeutic molecules. They assemble spontaneously in purely aqueous environments, providing a robust, solvent-free formulation pathway for sensitive ionic compounds.
Unimolecular Micelles and Crosslinked Micelles
A fundamental limitation of traditional, self-assembled polymeric micelles is their dynamic thermodynamic nature; they risk premature dissociation if diluted below their critical micelle concentration. Formulation scientists engineer structurally locked nanocarriers to completely circumvent this vulnerability.
- Unimolecular Micelles: Utilize complex, highly branched macromolecular architectures—such as dendrimers or multi-arm star polymers—that inherently possess a core-shell topology within a single molecule, rendering them completely concentration-independent.
- Crosslinked Micelles: Introduce covalent crosslinks either within the hydrophobic core or throughout the hydrophilic shell post-assembly. This locks the once-dynamic structure into a rigid, static nanostructure, granting extraordinary mechanical stability and heavily prolonging circulation time.
Looking for Custom Polymer Micelles?
From amphiphilic block copolymers to stimuli-responsive systems, we offer tailored synthesis and formulation solutions for drug delivery research.
Formulation and Design Strategies for Polymeric Micelles
Translating precisely engineered block copolymers into viable, highly reproducible drug delivery systems requires rigorous, multi-faceted formulation strategies. The self-assembly process is governed by delicate thermodynamic balances, meaning formulation scientists must exert exact control over preparation conditions to ensure batch-to-batch consistency. A successful formulation strategy bridges the gap between raw polymer synthesis and a functional nanosuspension by optimizing thermodynamic compatibility, thoroughly characterizing physical morphology, and establishing comprehensive analytical profiles to predict biological behavior.
Drug Loading Capacity and Excipient-Derived Toxicity
Maximizing drug loading capacity while maintaining colloidal stability is the primary objective of micellar formulation. The theoretical loading limit is dictated by the Flory-Huggins interaction parameter (χ) between the specific drug molecule and the core-forming polymer block. Formulation scientists utilize techniques such as solvent evaporation, thin-film hydration, or rapid nanoprecipitation to kinetically trap the hydrophobic payload within the assembling core. By optimizing these parameters, researchers can achieve exceptionally high encapsulation efficiencies, significantly reducing the total mass of polymer required per administered dose. This optimization is critical because it fundamentally eliminates the need for massive quantities of traditional low-molecular-weight surfactants, thereby completely bypassing the severe, dose-limiting excipient toxicities conventionally associated with hydrophobic drug formulations.
Pharmacokinetic Analysis and Duration
Comprehensive pre-clinical pharmacokinetic profiling is absolute requisite to validate the systemic advantages of a micellar design. Analytical methodologies—often involving high-performance liquid chromatography coupled with mass spectrometry—must precisely differentiate between the total drug concentration, the unencapsulated free drug fraction, and the polymer-associated payload. The fundamental goal of micellar encapsulation is to drastically alter the intrinsic absorption, distribution, metabolism, and excretion profile of the bare molecule. A successfully formulated stealth micelle will exhibit a profoundly reduced systemic clearance rate (CL) and a significantly extended elimination half-life (t1/2). This prolonged circulation duration maximizes the area under the curve, ensuring the therapeutic payload remains available in the bloodstream long enough to accumulate selectively within targeted tissue microenvironments.
Hydrodynamic Size, Morphology, and Drug Distribution
The physicochemical signature of a polymeric micelle dictates its physiological fate, making advanced morphological characterization indispensable. Dynamic light scattering is strictly employed to measure the hydrodynamic radius (Rh) and evaluate the polydispersity index of the micellar suspension. A remarkably narrow size distribution is essential for predictable biodistribution and uniform cellular uptake. Beyond mere size, spatial morphology is analyzed using transmission electron microscopy, atomic force microscopy, or small-angle X-ray scattering. These techniques differentiate between simple spherical micelles, cylindrical architectures, and worm-like nanostructures. Furthermore, rigorous analytical techniques ensure that the drug molecules are homogeneously distributed within the lipophilic core rather than adsorbed onto the hydrophilic corona, which would result in uncontrolled burst release.
Combination Therapies and Their Impact on Drug Retention
Polymeric micelles offer an unparalleled platform for the simultaneous co-delivery of multiple therapeutic agents, a highly sophisticated strategy utilized to overcome multi-drug resistance pathways. Co-encapsulating a highly hydrophobic primary drug alongside a secondary chemosensitizer requires meticulous assessment of competitive core interactions. Because the finite volume of the hydrophobic core has specific spatial and thermodynamic limits, formulators must map the partition coefficients of each active agent. Careful optimization of the polymer architecture ensures that the retention kinetics of both molecules remain synchronized. This prevents the premature leakage of one component over the other, guaranteeing that the calculated synergistic ratio of the combination therapy is precisely maintained upon arrival at the target site.
In Vitro and In Vivo Evaluation of Polymeric Micelle Systems
Rigorous pre-clinical evaluation protocols serve as the vital bridge between physicochemical formulation and biological validation. In vitro release assays are typically conducted using tightly controlled dialysis setups maintained under perfect sink conditions to accurately map the release kinetics over time. These dissolution profiles are frequently fitted to mathematical models, such as the Korsmeyer-Peppas equation, to determine whether the release is governed by Fickian diffusion or polymer chain relaxation. Subsequent in vivo evaluation utilizes complex animal models to assess true circulation time, targeted tissue accumulation, and metabolic degradation pathways. Advanced imaging techniques employing fluorescently tagged polymers or radioactive tracers are utilized to independently track the spatial distribution of the nanocarrier versus its encapsulated payload, proving the systemic superiority of the intact micelle.
Stability, Sterility, and Storage Considerations
Maintaining the strict thermodynamic and kinetic stability of polymeric nanosuspensions over an extended shelf-life represents one of the most complex formulation challenges. Aqueous suspensions are continuously vulnerable to physical degradation pathways, including Ostwald ripening, micellar fusion, and uncontrolled aggregation, as well as the chemical hydrolysis of vulnerable polymer backbones. Consequently, advanced lyophilization techniques are standard practice. Freeze-drying protocols are meticulously designed with the addition of optimized cryoprotectants—such as trehalose or sucrose—to prevent the collapse of the core-shell architecture during the desiccation phase. This results in a highly stable, dry powder that can be instantaneously reconstituted into fully functional micelles. Finally, sterility is typically achieved through sterile filtration using 0.22 um membranes, avoiding the structural damage that high-heat autoclaving or intense gamma irradiation might inflict upon the polymer matrix.
Application Scenarios of Polymeric Micelles in Poorly Soluble Drugs
The highly modular nature of polymeric micelles makes them an exceptionally versatile platform in advanced drug delivery. By meticulously tailoring the core-shell architecture, polymer chemists and formulation scientists can adapt these supramolecular nanocarriers to solve complex delivery challenges across entirely different classes of therapeutic molecules.
Delivery of Hydrophobic Small Molecule Drugs
Small molecules with high lipophilicity often suffer from rapid precipitation and poor cellular penetrability in aqueous biological environments. Polymeric micelles excel in rescuing these compounds, particularly those categorized under the Biopharmaceutics Classification System Class II and IV. By embedding lipophilic compounds—such as complex polycyclic structures, poorly soluble kinase inhibitors, or highly hydrophobic natural product derivatives—into the lipophilic core, formulators completely eliminate the necessity for harsh organic co-solvents. The hydrophobic interactions within the core, characterized by a highly favorable free energy of encapsulation (ΔG < 0), enable safe intravenous administration, vastly improved tissue distribution, and the avoidance of excipient-induced hemolysis commonly associated with traditional small-molecule formulations.
Protein and Peptide Therapeutics with Low Solubility
Beyond small molecules, large biologics present unique delivery hurdles. Proteins and peptides frequently suffer from rapid enzymatic degradation, physical instability, and a high propensity for aggregation in non-ideal physiological environments. Specifically engineered block copolymers can encapsulate highly hydrophobic or amphipathic peptides, effectively shielding their complex three-dimensional conformations from premature unfolding. This spatial sequestration within the micellar core prevents aggregation-induced immunogenicity and protects delicate structural motifs from proteolytic enzymes. By modulating the degradation rate of the polymer matrix, scientists can facilitate highly controlled, sustained release profiles for biologics that would otherwise exhibit impractically short biological half-lives.
Nucleic Acid Delivery (DNA, RNA, mRNA) Using Polymeric Micelles
While nucleic acids are inherently highly water-soluble due to their phosphodiester backbones, their large molecular weight and dense negative charge prevent them from passively crossing lipophilic cellular membranes. To address this, specialized polyion complex micelles are formulated through the electrostatic coacervation of block catiomers and polyanionic nucleic acids. This spontaneous complexation creates a dense, electroneutral core that protects the fragile genetic payload from abundant serum nucleases. The surrounding hydrophilic PEGylated shell provides necessary colloidal stability in systemic circulation. This sophisticated supramolecular assembly enables efficient endocytosis and subsequent endosomal escape, facilitating robust intracellular delivery without the severe cellular toxicity typically associated with standard cationic lipid nanoparticles.
Targeted Oncology Therapies and Combination Treatments
In the realm of advanced targeted therapies, polymeric micelles elegantly leverage the enhanced permeability and retention effect characteristic of highly fenestrated pathological microenvironments. Their optimized nanoscopic size—typically ranging between 10 nm and 100 nm—allows the micelles to passively extravasate into targeted tissues while avoiding rapid renal clearance. Furthermore, the immense loading capacity of the hydrophobic core allows for sophisticated combination treatments. Formulators can seamlessly co-encapsulate a primary therapeutic agent alongside a secondary sensitizer or efflux pump inhibitor. This precise co-delivery ensures that both molecules reach the target cell simultaneously at an optimized synergistic ratio, a feat nearly impossible to achieve with separate administrations of free drugs due to their differing pharmacokinetic profiles.
Services
Polymeric Micelles for Poorly Soluble Drugs Support Services
BOC Sciences leverages strong capabilities in functional monomer development, controlled polymerization technologies, nanocarrier design, and advanced analytical characterization to support the development of polymeric micelle systems for poorly soluble drugs. To address challenges such as low aqueous solubility and limited bioavailability of hydrophobic small-molecule drugs, we provide integrated services covering amphiphilic polymer design, micelle construction, drug loading optimization, and physicochemical characterization. By precisely tuning polymer structures and hydrophilic–hydrophobic balance, BOC Sciences helps clients develop polymeric micelle systems with enhanced solubility, improved stability, and controlled drug release, supporting research and formulation development.

Amphiphilic Polymer Design and Synthesis
- Custom design and synthesis of amphiphilic block copolymers for polymeric micelle carriers.
- Structural design and molecular weight control of PEG, PLA, PCL, and PLGA polymers.
- Optimization of hydrophilic–hydrophobic block ratios to enhance solubility and micelle stability.
- Development of functionalized polymers and multiblock copolymers.

Poorly Soluble Drug Loading Development
- Development of polymeric micelle drug loading systems to improve hydrophobic drug solubility.
- Optimization of drug loading capacity and encapsulation efficiency.
- Development of single-drug and combination drug loading systems.
- Optimization of self-assembly and dialysis-based micelle preparation methods.

Custom Polymeric Micelle Synthesis and Development
- Custom design and synthesis of polymeric micelle carriers.
- Optimization of amphiphilic polymer structures and self-assembly behavior.
- Development of delivery systems for hydrophobic small molecules and anticancer drugs.
- Integrated development from material design to micelle formulation.

Polymeric Micelle Characterization and Analysis
- Particle size distribution and nanostructure analysis.
- Determination of critical micelle concentration (CMC).
- Morphological characterization using techniques such as TEM and SEM.
- Evaluation of drug release behavior and formulation stability.
Do You Need A Consultation?
BOC Sciences combines advanced polymer chemistry and drug delivery expertise to accelerate your research, providing tailored polymer platforms for small molecules, biologics, and nucleic acid therapeutics.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
How do polymeric micelles improve solubility of hydrophobic drugs?
Polymeric micelles dramatically enhance solubility by functioning as dispersible nanoscale reservoirs. Driven by thermodynamic self-assembly, amphiphilic block copolymers form a dense, lipophilic inner core where poorly soluble molecules partition via hydrophobic interactions. Simultaneously, the outer hydrophilic corona maintains the entire drug-loaded supramolecular complex evenly suspended and highly concentrated within aqueous physiological environments.
-
Can polymeric micelles provide controlled and sustained release?
Yes, these nanocarriers excel at providing controlled and sustained payload release. Because block copolymers possess an exceptionally low critical micelle concentration, they maintain structural integrity even upon massive systemic dilution. Release kinetics are primarily governed by drug diffusion outward from the solid polymer core and the gradual hydrolytic degradation of the constituent block copolymers.
-
Are polymeric micelles suitable for proteins, peptides, or nucleic acids?
Absolutely. Through precise macromolecular engineering, block copolymers can be adapted to encapsulate complex biologics. Hydrophobic peptides are sequestered to prevent structural unfolding and aggregation. For highly charged macromolecules like nucleic acids, oppositely charged block ionomers are utilized to form polyion complex micelles, which effectively neutralize the charge and shield delicate structures from enzymatic degradation.
-
How are polymeric micelles customized for specific poorly soluble drugs?
Customization is achieved by meticulously selecting and synthesizing specific copolymer blocks. Formulation scientists precisely match the thermodynamic properties of the hydrophobic core to the target molecule, maximizing the Flory-Huggins interaction parameter for optimal loading. Additionally, corona length is adjusted to dictate circulation time, while functional groups can be chemically grafted to enable stimuli-responsive payload release.

 Fig. 1. Illustrating how polymeric micelles overcome drug solubility and delivery barriers (BOC Sciences Authorized).
Fig. 1. Illustrating how polymeric micelles overcome drug solubility and delivery barriers (BOC Sciences Authorized). Fig. 2. Amphiphilic block copolymers self-assembling into a drug-loaded polymeric micelle (BOC Sciences Authorized).
Fig. 2. Amphiphilic block copolymers self-assembling into a drug-loaded polymeric micelle (BOC Sciences Authorized).