Polymer Microspheres for Long-Acting Drug Delivery: Materials, Release Control, and Formulation Strategy
Polymer microspheres are one of the most established application forms in polymeric long-acting drug delivery systems, offering a practical way to convert rapid drug exposure into sustained and more controlled release profiles. By selecting the right polymer chemistry, particle structure, loading approach, and manufacturing process, formulators can tailor microsphere systems for small molecules, peptides, proteins, and other challenging payloads. Compared with immediate-release formulations, polymer microspheres can reduce concentration fluctuation, extend dosing intervals, improve formulation robustness, and support both local and systemic delivery goals through tunable degradation and diffusion behavior.
Resources
Polymer Microspheres for Long-Acting Drug Delivery
Polymer microspheres occupy a central position in long-acting formulation development because they combine tunable material design with scalable particle engineering. In practical terms, they allow a formulator to embed an active ingredient inside a biodegradable polymer or slowly permeable polymer matrix, then regulate release through a combination of diffusion, hydration, pore evolution, and polymer degradation. This makes microspheres highly relevant when a project needs a more sustained exposure profile than an immediate-release dosage form can provide, but does not require the structural permanence of a bulk implant or the phase-transition behavior of an in situ forming depot. When designed well, microspheres can provide a useful balance among manufacturability, injectability, release control, and material flexibility.
Definition and Structural Characteristics of Polymer Microspheres
Polymer microspheres are discrete spherical particles, typically in the micrometer size range, composed of a polymeric matrix or shell structure that carries a drug payload. In matrix-type microspheres, the drug is dispersed throughout the polymer bulk, and release occurs as water penetrates the particle, dissolved molecules diffuse outward, and the polymer gradually erodes or relaxes. In more reservoir-like structures, the payload may be concentrated in internal domains or encapsulated in a core-like region, though true reservoir behavior is less common in classical biodegradable microsphere systems than in implants. Structural features such as particle diameter, porosity, surface smoothness, internal phase separation, and polymer density all influence release behavior. Because these structural variables can be intentionally tuned, microspheres are widely used as engineered carriers rather than simple particulate excipients.
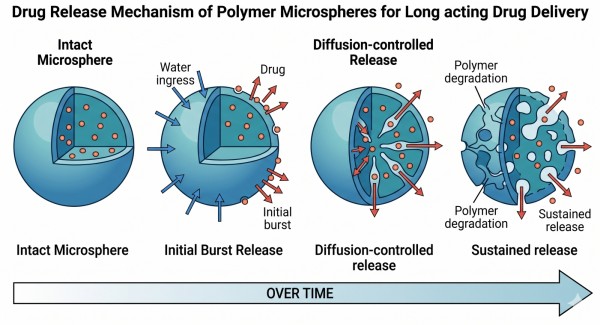 Fig. 1. Diffusion, degradation, and burst release behavior in polymer microspheres (BOC Sciences Authorized).
Fig. 1. Diffusion, degradation, and burst release behavior in polymer microspheres (BOC Sciences Authorized).
How Microspheres Differ from Nanoparticles, Depots, and Implants?
Microspheres differ from polymer nanoparticles primarily in size, release timescale, and usage logic. Nanoparticles are usually selected when systemic circulation behavior, tissue penetration, or intracellular uptake matters most, whereas microspheres are commonly chosen when the goal is longer release duration from a localized or injectable particulate depot. Compared with in situ depots, microspheres are preformed solid particles before administration, which can improve control over particle attributes such as size distribution and internal morphology. Compared with implants, microspheres offer a less device-like format and are often easier to suspend and administer, but they may provide less geometric precision over release than a carefully engineered implant. These distinctions matter because developers are rarely choosing "a polymer" in the abstract; they are choosing an application form with a defined set of trade-offs in manufacturability, release control, and administration behavior.
Why Microspheres Are a Core Platform in Long-Acting Drug Delivery?
Microspheres are considered a core long-acting platform because they translate polymer properties into adjustable release duration without requiring complex device assembly. Their usefulness comes from the fact that multiple levers can be tuned simultaneously: polymer composition can alter degradation rate, molecular weight can change matrix density and hydration behavior, particle size can regulate diffusion distance, and process conditions can determine porosity and drug distribution. This combination of formulation flexibility and material control makes microspheres a practical choice when developers need sustained release over days, weeks, or longer while still working with a particle-based dosage form. They are also relevant when a project needs to reduce rapid peak exposure associated with immediate-release systems and create a more extended release phase that aligns better with the intended product profile.
Why Polymer Microspheres Are Used Instead of Immediate-Release Formulations?
The practical reason microspheres remain so important is that many active ingredients do not perform well when delivered in a rapid, pulse-like manner. Immediate-release formats can create sharp concentration peaks followed by rapid decline, which is often undesirable for long-term disease management or for local dosing situations where prolonged residence is needed. Polymer microspheres are therefore not just a packaging option; they are a release-engineering solution that helps reshape exposure over time. Their value is clearest when the therapeutic objective requires steadier release, lower fluctuation, or reduced dosing frequency compared with conventional immediate-release approaches.
Problems Caused by Rapid Drug Release and Fluctuating Exposure
In an immediate-release system, the drug often dissolves and becomes available quickly, which may produce a high initial concentration followed by a short effective window. For some APIs this pattern is acceptable, but for others it leads to inefficient exposure, repeated dosing, or unnecessary variability in local or systemic levels. In local administration contexts, rapid release can expose the target tissue to a large early dose that dissipates before the desired duration is achieved. In systemic contexts, the same pattern may generate large peak-to-trough swings. These limitations are a major reason long-acting systems were developed in the first place. Microspheres address this by using the polymer matrix as a transport barrier and structural regulator rather than allowing the drug to immediately partition into the surrounding medium.
How Microspheres Support Prolonged and More Even Drug Release?
Microspheres extend release by slowing the sequence of events required for drug escape. Water must first penetrate the particle; the drug must then dissolve or mobilize inside the matrix; diffusion pathways must develop; and, in degradable systems, the polymer must undergo progressive chain cleavage and structural relaxation. Because each of these steps can be tuned, the release curve can often be moderated relative to the sharp profile of an immediate-release product. Even when a microsphere system shows some initial burst, the majority of the dose can still be distributed across a much longer time window. This is especially valuable when a formulation goal is not simply delayed release, but a more sustained and usable release profile with less abrupt exposure.
Why Microspheres Are Especially Valuable for Injectable Sustained Delivery?
Among long-acting injectable concepts, microspheres are especially valuable because they combine a particulate format with well-established biodegradable polymer options such as Poly(lactic-co-glycolic acid) (PLGA), Polylactic Acid (PLA), and Polycaprolactone (PCL). A microsphere suspension can often be administered with simpler handling than a preformed implant, while still delivering prolonged release from the deposited particles. The particle format also allows the same general platform concept to be adapted across different APIs, concentrations, and release targets. For this reason, microspheres are often evaluated early when a team wants sustained injectable release but has not yet committed to an implant, hydrogel, or in situ depot architecture.
How Microspheres Achieve Long-Acting Release?
Release from polymer microspheres is rarely governed by a single mechanism. Instead, it usually reflects an evolving balance among matrix diffusion, water uptake, polymer relaxation, pore development, and erosion. A useful microsphere formulation therefore depends on understanding not only what the polymer is, but also how its structure changes during exposure to the release medium. For some systems diffusion dominates early and degradation later. For others, the internal microenvironment becomes the key driver of release acceleration. Appreciating these layered mechanisms helps prevent oversimplified design decisions and leads to better control over burst release and long-term release duration.
Diffusion-Controlled Release from Polymer Matrices
In diffusion-controlled release, dissolved drug molecules move through the hydrated polymer matrix and any water-filled pores that form within it. This mechanism is particularly important in the early phase of release, especially for drugs located near the particle surface or within more permeable regions of the microsphere. The effective diffusion rate depends on matrix tortuosity, polymer chain mobility, hydration level, and the drug's own solubility and molecular size. A dense, hydrophobic matrix generally slows diffusion, whereas higher porosity or greater water uptake increases the likelihood of faster drug escape. In practice, formulators often seek a controlled early diffusion phase that avoids an excessive initial burst while still allowing the release profile to begin in a predictable and reproducible way.
Polymer Degradation and Erosion-Controlled Release
In biodegradable microspheres, polymer degradation becomes increasingly important as release progresses. Hydrolytic cleavage of ester bonds reduces molecular weight, weakens matrix integrity, and eventually creates new aqueous pathways through which the drug can leave the particle. Polymers such as PLGA often exhibit progressive bulk degradation, meaning water can penetrate the particle before significant mass loss becomes visible. As the chains become shorter and more mobile, the matrix can become more permeable, and release may accelerate. In slower-degrading systems such as PCL-rich microspheres, this process is delayed, which can stretch the release window but may also slow full payload liberation. Release control therefore depends not only on whether a polymer is degradable, but on how its degradation kinetics interact with the drug's mobility inside the particle.
Water Ingress, Pore Formation, and Burst Release Behavior
Initial burst release is one of the defining challenges in microsphere development. It often arises from drug located at or near the particle surface, but that is not the only cause. Water ingress into the microsphere can rapidly hydrate superficial regions, dissolve exposed payload, and generate pore networks that accelerate outward transport. If the internal drug distribution is nonuniform, if solvent removal created channels, or if the polymer-drug compatibility is poor, the burst can become more pronounced. Burst release is not always inherently negative; in some product concepts a modest early phase is acceptable or even desirable. The real challenge is making it intentional and controlled rather than accidental. That requires coordinated attention to particle structure, polymer choice, loading strategy, and processing conditions.
How Microsphere Size and Internal Structure Affect Release Duration?
Particle size has a direct effect on diffusion length and surface-area-to-volume ratio. Smaller microspheres generally release faster because they offer shorter travel distances for dissolved drug and proportionally more surface area. Larger particles often prolong release, but they may also create steeper internal gradients in water penetration and degradation. Internal structure matters just as much. A hollow or highly porous particle behaves differently from a dense, homogeneous one. Internal phase separation, residual voids, and surface roughness can all alter how quickly water enters and how rapidly the drug redistributes inside the matrix. Therefore, release duration cannot be predicted from polymer identity alone. The final microsphere architecture is an equally important determinant.
Polymer Materials Commonly Used for Long-Acting Microspheres
Choosing the polymer for a microsphere system is not a box-checking exercise. The material defines degradation rate, mechanical integrity, hydrophobicity, internal microenvironment, solvent compatibility, and process behavior. Even when two materials are both categorized as biodegradable polyesters, their release behavior can differ substantially because of differences in crystallinity, glass transition temperature, end groups, or monomer ratio. For long-acting microspheres, the key is not simply whether a polymer is biodegradable, but whether its full property set matches the release target, payload sensitivity, and manufacturing route.
PLGA as the Benchmark Polymer for Sustained-Release Microspheres
PLGA is widely regarded as the benchmark polymer for sustained-release microspheres because it offers a useful balance of biodegradability, formulation history, and tunable release behavior. By adjusting the lactide:glycolide ratio, formulators can influence hydrophobicity, water uptake, and degradation pace. A higher glycolide fraction usually promotes faster water access and degradation, while higher lactide content often slows these processes and can extend release duration. PLGA also provides broad compatibility with classical solvent evaporation routes, making it a practical starting point for many injectable microsphere projects. However, PLGA is not universally optimal. Acidic degradation products, internal pH shifts, and protein instability risks must be considered carefully, especially for sensitive payloads.
PLA and PCL for Slower Degradation and Longer Release Windows
PLA and PCL are often selected when slower degradation is desired. PLA generally degrades more slowly than many PLGA grades because of its greater hydrophobicity and higher crystallinity tendency, though exact behavior depends on stereochemistry and molecular characteristics. PCL is even slower in many cases, which can be useful when a longer release window is required. The trade-off is that very slow matrix evolution may reduce release completeness for some APIs if the drug remains too strongly retained in the polymer. Therefore, slower degradation is not automatically better. It must be aligned with the desired release window, the drug's diffusivity, and the acceptable duration of residual polymer presence.
Natural, Hybrid, and Functional Polymer Systems
Although polyester systems dominate classical long-acting microspheres, natural polymers and hybrid polymer systems can be useful in specific design contexts. Polysaccharide-derived materials, protein-based matrices, or hybrid networks that combine biodegradable polyesters with hydrophilic components may be considered when bioadhesion, swelling behavior, or payload compatibility requires more than a simple hydrophobic matrix. Functional polymers can also be introduced to modify charge, hydration, or interfacial properties. The caution is that each added functionality affects not only release but also manufacturability, solvent selection, particle integrity, and storage behavior. As a result, hybrid systems can broaden formulation options, but they require tighter control of both material chemistry and process design.
How Molecular Weight, Monomer Ratio, and End Groups Shape Performance?
Molecular weight affects chain entanglement, matrix density, viscosity during processing, and the rate at which degradation alters the structure. Higher molecular weight polymers often yield denser matrices and slower initial diffusion, but they can also complicate processing because of higher solution viscosity. Monomer ratio influences hydrophobicity and degradation kinetics, especially in a copolymer system such as PLGA. End groups are also important. Acid-terminated polymers may hydrate and degrade differently than ester-capped materials, and these differences can influence not only release duration but also the internal microenvironment around the payload. In practice, seemingly small changes in molecular specification can produce substantial changes in release behavior, which is why microsphere development often depends on iterative material screening rather than nominal polymer class alone.
Need a Custom Microsphere Formulation Strategy?
From polymer screening and drug encapsulation to release-profile optimization and particle engineering, our team supports long-acting microsphere development for diverse payload classes.
Drug Loading Strategy and API Compatibility in Microsphere Systems
A microsphere formulation succeeds only when the drug can be loaded in a way that is both efficient and stable. This is where many early concepts fail: the polymer may look promising on paper, but the payload partitions poorly, degrades during processing, migrates toward the surface, or becomes trapped in a nonreleasing state. The right loading strategy depends on API polarity, solubility, molecular size, structural fragility, and compatibility with both the polymer and the manufacturing solvent system. Therefore, payload type is not a secondary consideration; it is one of the main drivers of microsphere design.
Small-Molecule Loading in Polymer Microspheres
Small molecules are often the most straightforward class to load, but success still depends heavily on polymer affinity and process route. Hydrophobic small molecules may partition effectively into hydrophobic polymer phases, which can improve encapsulation and support sustained release. However, if the drug crystallizes during solvent removal or migrates toward the particle surface, burst release can become severe. More hydrophilic small molecules may be harder to retain and may require modified processes or internal aqueous domains, which can increase the risk of porosity and leakage. Therefore, even for small molecules, the central question is not only how much drug can be incorporated, but where in the particle it finally resides after processing.
Peptide and Protein Encapsulation Challenges
Peptides and proteins introduce a different set of constraints. They may be sensitive to solvent exposure, shear, interfaces, dehydration stress, and acidic degradation products generated by the polymer matrix. In classical double-emulsion systems, proteins can denature at water-oil interfaces or redistribute unevenly within the particle. Later in release, the internal microenvironment of degrading polyesters may further destabilize the payload. Because of these risks, protein-loaded microspheres often require careful excipient support, process protection, and deeper attention to the polymer's degradation profile. The challenge is not simply to entrap the macromolecule, but to preserve structural integrity and achieve a controlled release profile without inducing aggregation or chemical instability.
Hydrophilic versus Hydrophobic Payload Compatibility
Hydrophilic and hydrophobic payloads interact with microsphere matrices in fundamentally different ways. Hydrophobic molecules often show stronger affinity for hydrophobic polyester matrices, which can improve retention but sometimes hinder release completeness if the interactions are too strong. Hydrophilic molecules may be more prone to loss during processing or early release because they partition into aqueous pathways more readily. This difference affects not only loading efficiency but also pore formation, internal drug distribution, and the need for formulation aids. As a result, the same polymer grade can perform very differently depending on whether the payload favors the organic phase, the aqueous phase, or neither strongly enough to stabilize the desired microstructure.
Drug–Polymer Interactions That Affect Loading and Release
Drug–polymer compatibility influences almost every important performance outcome in a microsphere system. Strong hydrogen bonding, hydrophobic association, ionic effects, or steric mismatch can alter encapsulation efficiency, internal phase behavior, and the final physical state of the drug. If compatibility is poor, the payload may phase separate, enrich near the surface, or crystallize into domains that release too quickly or too slowly. If compatibility is too strong, the drug may remain overly retained in the matrix. Understanding these interactions early can help avoid unnecessary reformulation cycles. In many cases, microsphere performance is better predicted by the quality of drug–polymer matching than by generic assumptions about the polymer alone.
Microsphere Preparation Methods and Process Trade-Offs
Preparation method is not just a manufacturing detail; it directly shapes particle structure, drug distribution, solvent residuals, and ultimately release performance. Two microsphere batches made from the same polymer and drug can behave very differently if they were produced by different solvent removal rates, emulsification intensities, or phase architectures. Method selection should therefore be treated as a core design decision rather than a downstream scale-up issue. The best process is the one that produces the target microstructure consistently while preserving drug stability and supporting practical manufacturing.
Single Emulsion, Double Emulsion, and Solvent Evaporation Methods
Solvent evaporation-based methods remain among the most widely used routes for microsphere preparation. In a single-emulsion approach, a drug-polymer organic phase is emulsified in an external phase and then hardened by solvent removal, making it especially useful for hydrophobic drugs. Double-emulsion approaches are often used for more hydrophilic drugs, peptides, and proteins because they allow an internal aqueous phase to carry the payload before secondary emulsification. While versatile, these methods introduce multiple stress points, including shear, interfacial exposure, solvent exchange, and the possibility of uneven internal void formation. Process parameters such as emulsifier concentration, stirring intensity, solvent volatility, and polymer concentration strongly affect final particle attributes.
Spray Drying and Other Rapid Particle-Forming Approaches
Spray drying can provide a comparatively rapid route to particle formation and is often attractive for its process efficiency and potential scalability. It may be useful when developers want tighter control over drying kinetics or prefer a route that avoids multi-step emulsion hardening. However, spray drying can introduce thermal and dehydration stress, which may be problematic for sensitive payloads. The method also influences particle density, surface topology, and internal distribution differently from classical emulsion routes. Therefore, it is not simply a faster version of solvent evaporation; it is a structurally distinct manufacturing pathway that must be judged on how it shapes the resulting release behavior.
Microfluidic and Advanced Routes for Size Uniformity
Microfluidic methods and related advanced particle-generation strategies are gaining attention because they can improve uniformity in droplet formation and therefore narrow the final size distribution. Greater uniformity can simplify downstream interpretation of release profiles because particle-to-particle variability is reduced. These methods are particularly attractive when a program values tighter control of size, surface characteristics, and reproducibility. The trade-off is that process throughput, equipment complexity, and formulation window may differ from more traditional batch approaches. For some programs this is worth the added complexity; for others, conventional methods remain more practical.
Residual Solvent, Scale-Up, and Batch-Consistency Concerns
Long-acting microsphere development does not end once a promising lab-scale release curve is observed. Residual solvent control, batch-to-batch reproducibility, and scale-dependent changes in emulsification and drying all become critical as the process matures. Solvent removal kinetics often change with scale, which can alter porosity, surface drug enrichment, and particle density. A formulation that works well in a small batch may therefore behave differently when mixing, heat transfer, or solvent stripping conditions are modified. For this reason, process scalability must be evaluated alongside particle performance from an early stage. Robust microsphere development requires attention to manufacturability as well as release design.
| Preparation Method | Suitable Payload Type | Primary Advantage | Main Risk Point | Scale-Up Note |
|---|
| Single Emulsion | Mainly hydrophobic small molecules | Simple route for matrix-type microspheres | Surface enrichment and burst release if phase distribution is poor | Mixing intensity strongly influences size and morphology |
| Double Emulsion | Hydrophilic molecules, peptides, proteins | Supports loading of water-favoring payloads | Interfacial stress, leakage, and internal void formation | More sensitive to process variability across scales |
| Spray Drying | Broad use depending on payload stability | Rapid particle formation and practical process efficiency | Drying stress and altered particle density | May support scale-up well, but particle structure must be revalidated |
| Microfluidic Routes | Programs prioritizing size uniformity | Narrow size distribution and improved reproducibility | Equipment and throughput limitations | Requires early evaluation of production feasibility |
How to Evaluate Microsphere Quality and Release Performance?
Because microspheres are structure-sensitive systems, their evaluation must go beyond a simple assay and particle size measurement. A useful assessment framework links particle attributes to release outcomes and identifies the failure modes most likely to compromise long-acting behavior. That means developers need to examine not only what the particles contain, but how the drug is distributed, what physical state it occupies, how the matrix changes during release, and how storage conditions may alter reconstitution or release reproducibility. Strong characterization helps transform a microsphere project from empirical trial-and-error into rational formulation development.
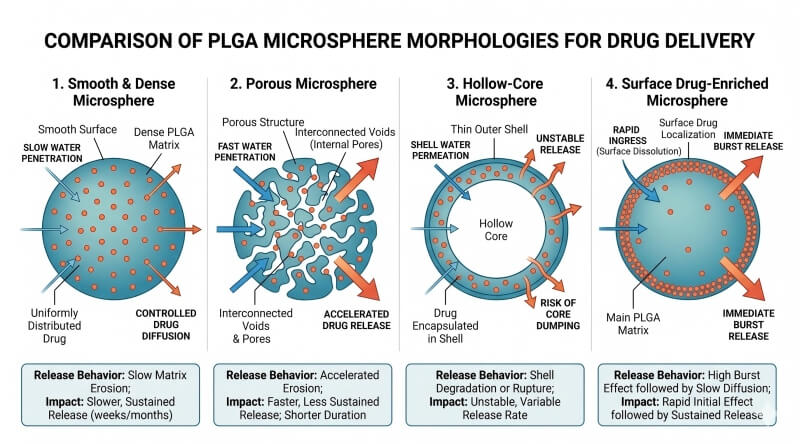 Fig. 2. Structural features that shape PLGA microsphere release performance (BOC Sciences Authorized).
Fig. 2. Structural features that shape PLGA microsphere release performance (BOC Sciences Authorized).
Particle Size Distribution, Morphology, and Surface Structure
Particle size distribution is one of the most informative quality attributes in a microsphere system because it directly influences diffusion distance, injectability, suspension behavior, and release uniformity. Morphology adds another layer of meaning. Smooth, dense particles often behave differently from rough or porous ones, even if their average size is similar. Surface cracks, internal cavities, and irregular geometry can all indicate process-related issues that later appear as burst release or unstable release profiles. Therefore, microscopy and size analysis should be interpreted together, not in isolation.
Drug Loading, Encapsulation Efficiency, and Solid-State Properties
Drug loading and encapsulation efficiency determine how much therapeutic can realistically be delivered by the system, but they do not tell the full story. The payload's solid-state properties inside the microsphere also matter. A drug may be molecularly dispersed, amorphous, phase-separated, or partially crystalline, and each state affects release differently. Likewise, a high encapsulation number can still hide a poor internal distribution if the drug is concentrated near the surface. A meaningful evaluation therefore integrates quantitative loading data with information about physical state and intraparticle distribution.
In Vitro Release Profile Interpretation and Burst-Release Analysis
In vitro release testing is central to microsphere development, but the resulting curve must be interpreted mechanistically rather than descriptively. A rapid initial phase may indicate surface-associated drug, excessive porosity, or poor drug–polymer compatibility. A long lag phase may suggest overly dense matrices or insufficient water access. A late acceleration phase often reflects degradation-driven pathway formation. The best release profile is not necessarily the slowest one; it is the one that aligns with the intended duration and remains reproducible across batches. Burst analysis is especially important because excessive early release can undermine the value of the entire long-acting design.
Stability, Storage Risk, and Reconstitution Considerations
Microspheres must maintain their intended structure before use, not just after administration. Storage can change residual moisture, promote drug migration, affect polymer aging, or alter suspension behavior. If the system is supplied for reconstitution, redispersibility becomes a critical attribute because poor reconstitution can change the effective particle population administered. Sensitive payloads may also degrade during storage if the internal environment is unstable. Therefore, stability assessment should include not only chemical assay, but also physical integrity, particle behavior after storage, and release performance after the product has been reconstituted or redispersed.
When Microspheres Are the Right Choice in Long-Acting Delivery?
Microspheres are not the answer to every sustained-release problem, but they are often one of the most flexible first platforms to evaluate. They are especially attractive when a project needs a particulate long-acting system with adjustable release duration, biodegradable polymer options, and a clear path to tuning particle properties through formulation and process parameters. Their strengths become most evident when the product concept requires sustained injectable release without moving immediately into a larger implantable device format. At the same time, their limitations must be acknowledged early so that development effort is directed toward the right platform.
- Best-Fit Scenarios for Polymer Microsphere Platforms: Microspheres are well suited when a program needs a long-acting particulate system that can be customized through polymer selection and particle engineering. They can be particularly attractive for biodegradable injectable products, local depot-like release from dispersed particles, and formulations where the release window must be tuned through composition and structure rather than through a preformed device geometry. They are also useful when development teams want flexibility in screening multiple polymer grades and loading levels before committing to a more complex application form such as a structured implant.
- Common Limitations and How to Mitigate Them: The major limitations of microspheres include burst release, incomplete release, payload instability during fabrication or storage, scale-up variability, and the difficulty of achieving consistent internal morphology. These issues can often be mitigated, but rarely by a single intervention. Burst control may require improved drug distribution, denser matrices, or altered solvent removal. Payload stability may depend on excipient protection, gentler processes, or a different polymer grade. Poor reproducibility may require tighter control over emulsification, drying, or particle-size uniformity. The key point is that microsphere problems are usually multivariable and should be approached through integrated material-process optimization rather than isolated adjustments.
- When to Switch to Depot, Hydrogel, or Implant Systems: There are situations in which microspheres are not the best long-acting platform. If a project needs a more coherent bulk-forming matrix after administration, a polymer hydrogel or in situ depot may be more suitable. If the target profile requires highly defined geometry, very prolonged release, or a true reservoir architecture, an implant may offer better control. If the objective is deeper systemic circulation control or intracellular delivery, polymer nanoparticles may be more appropriate. Choosing microspheres therefore makes the most sense when their particulate, biodegradable, and tunable nature aligns directly with the intended product profile.
Services
Advanced Polymer Synthesis and Microsphere Formulation Services
At BOC Sciences, developing effective polymer microspheres requires more than access to raw materials. It depends on coordinated control over polymer architecture, encapsulation strategy, particle formation, and analytical characterization. We support microsphere projects through custom polymer and copolymer synthesis, material modification, formulation screening, and release-behavior evaluation. Whether a project requires biodegradable polyester selection, functional polymer design, or process optimization for particle uniformity and reproducibility, BOC Sciences provides integrated technical support to connect polymer chemistry with practical sustained-release formulation outcomes.

Custom Polymer & Copolymer Synthesis
- Selection and synthesis of biodegradable and functional polymers for microsphere applications.
- Control over molecular weight, monomer ratio, and end-group chemistry.
- Support for polyester, polyamine, and amphiphilic polymer platforms.
- Optimization of structure to match sustained-release targets.

Encapsulation & Formulation Strategy
- Drug–polymer compatibility screening for small molecules and biomacromolecules.
- Process selection across emulsion, solvent evaporation, spray drying, and advanced particle routes.
- Burst-release mitigation and release-window tuning support.
- Guidance on payload distribution and matrix-structure optimization.

Microsphere Characterization & Performance Evaluation
- Particle size, morphology, and surface-structure assessment.
- Drug loading, encapsulation efficiency, and solid-state analysis.
- In vitro release testing and burst-phase interpretation.
- Stability and reconstitution risk evaluation for long-acting formulations.

Scale-Up and Process Optimization
- Assessment of solvent handling, drying behavior, and batch consistency.
- Process transfer support from exploratory screening to more robust production workflows.
- Analytical control of key polymer and particle attributes.
- Development support for reproducible long-acting microsphere manufacturing.
Discuss Your Microsphere Project with Us
Talk with our team about polymer selection, encapsulation strategy, particle design, and release-profile optimization for sustained and long-acting microsphere systems.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
Why do polymer microspheres often show burst release?
Burst release usually results from drug enrichment near the particle surface, rapid hydration of superficial regions, or pore structures formed during processing. It often indicates that drug distribution, matrix density, or solvent-removal conditions need further optimization.
-
How does the PLGA lactide:glycolide ratio affect release duration?
The ratio influences hydrophobicity, water uptake, and degradation pace. In general, compositions that hydrate and degrade more quickly tend to release faster, while more hydrophobic compositions can support slower matrix evolution and longer release windows.
-
Are polymer microspheres suitable for peptides and proteins?
They can be, but the formulation must account for interfacial stress, solvent exposure, internal acidification, and structural fragility. Payload protection and careful process selection are usually essential.
-
What particle size range is practical for sustained-release microspheres?
There is no universal size target because the right range depends on injectability, desired diffusion distance, release duration, and suspension behavior. In practice, size is chosen as part of the overall release design rather than as a standalone specification.
-
When are microspheres better than in situ depots?
Microspheres are often preferable when preformed particle control, defined size distribution, and particulate suspension behavior are more valuable than bulk phase-transition behavior after administration. Depots may be better when a cohesive formed matrix is required at the site of administration.

 Fig. 1. Diffusion, degradation, and burst release behavior in polymer microspheres (BOC Sciences Authorized).
Fig. 1. Diffusion, degradation, and burst release behavior in polymer microspheres (BOC Sciences Authorized). Fig. 2. Structural features that shape PLGA microsphere release performance (BOC Sciences Authorized).
Fig. 2. Structural features that shape PLGA microsphere release performance (BOC Sciences Authorized).