Polymeric Inserts for Long-Acting Drug Delivery: Material Selection and Site-Specific Design
Polymer-based inserts are a distinct long-acting drug delivery platform because they are designed to remain at a defined local site long enough to improve residence, regulate the local microenvironment, and extend drug release beyond what conventional liquids, semisolids, or rapidly cleared local formulations can usually achieve. Unlike systems that depend mainly on immediate spreading or passive diffusion, polymeric inserts use matrix architecture, hydration, mucoadhesion, swelling, erosion, or structural persistence to hold the dosage form in place and modulate how drug leaves the system over time. This makes them especially valuable when the local site is affected by rapid clearance, fluid turnover, leakage, motion, or short contact time. In practical formulation work, the performance of an insert depends less on the label "insert" itself than on how well the material, geometry, and release logic match the site of administration and the intended long-acting delivery goal.
Resources
Polymeric Inserts as a Retention-Based Drug Delivery Platform
Polymeric inserts are best understood as retention-oriented dosage systems rather than simply shaped local formulations. Their value comes from the ability to remain where they are placed and create a controlled interface between the payload and the local tissue for longer than a conventional dose would normally persist. In long-acting delivery, that retention function is often the prerequisite for meaningful release control. If a dosage form cannot remain in place, even a carefully selected release polymer may not provide a useful sustained profile. For that reason, insert design starts with residence and only then moves into release kinetics, material choice, and payload compatibility.
What Counts as a Polymeric Insert?
A polymeric insert is a local drug delivery system with a defined form that is intentionally placed at a specific site and designed to remain there for a controlled period while releasing drug through matrix-based or structure-based mechanisms. Depending on the application, the insert may be thin and flexible, compact and swellable, partially erodible, or more structurally persistent. What unifies these systems is not one route of administration, but the use of polymer design to shape residence, hydration, handling, and release. In that sense, inserts are not limited to one anatomical site and should not be treated as a narrow ophthalmic-only concept.
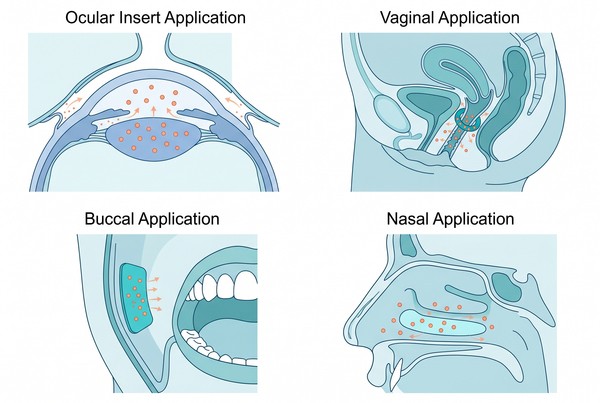 Fig. 1. Main application forms of polymeric inserts for long-acting drug delivery (BOC Sciences Authorized).
Fig. 1. Main application forms of polymeric inserts for long-acting drug delivery (BOC Sciences Authorized).
How Inserts Differ from Patches, Gels, Films, and Implants?
Inserts differ from patches because they are usually intended to sit within or against a local cavity or confined mucosal surface rather than remain primarily on external skin with adhesive backing. They differ from gels because they begin as a more defined dosage form rather than a spreadable mass, even when they later swell or soften. They differ from films because their performance is usually built around local residence and three-dimensional matrix behavior rather than only thin-layer dissolution. They differ from implants because they are generally less invasive, more site-dependent, and more closely tied to local retention than to deep tissue placement or device-like structural persistence. These boundaries matter because each platform solves a different formulation problem.
Why Retention Is the Core Value of Insert Systems?
For inserts, retention is the primary functional advantage because it allows the dosage form to stay in contact with the local site long enough for controlled release to matter. Without adequate residence, the drug may be lost rapidly through tear turnover, saliva, fluid exchange, leakage, or other clearance forces before the matrix has time to deliver the intended profile. The most useful insert systems therefore do more than hold drug. They hold position, maintain contact, and control how the surrounding environment interacts with the formulation.
Why Long-Acting Inserts Are Driven by Site-Specific Challenges?
Long-acting insert systems are shaped less by route names and more by the physical realities of the local environment. Fluid turnover, mucus renewal, motion, local space constraints, and user comfort all influence whether a polymeric insert can remain in place and function as intended. That is why insert development is inherently site-specific. The same material or geometry that works well in one setting may fail in another because the dominant clearance forces are different. Understanding those local constraints is essential before selecting the polymer or the release mechanism.
Rapid Clearance in Ocular and Nasal Environments
Ocular and nasal sites are challenging because the applied formulation can be removed quickly by tears, blinking, mucus flow, or drainage. In these environments, a long-acting insert must resist rapid clearance without creating excessive bulk or discomfort. This often makes swelling behavior, local fit, and surface interaction more important than simple drug loading. A useful insert in these settings needs to remain localized while controlling hydration and release within a short but meaningful residence window.
Leakage and Residence Limits in Vaginal and Intrauterine Systems
Vaginal and intrauterine systems face a different set of constraints, including fluid exchange, leakage, limited usable volume, and the need for stable residence in a changing local environment. Inserts in these contexts often rely on a balance of structural integrity and adaptation to the surrounding tissue rather than only dissolution. Excessive swelling may reduce comfort or alter placement stability, while insufficient residence may shorten the release window. As a result, polymer selection must account for both local retention and the mechanical behavior of the hydrated system.
Saliva, Motion, and Surface Renewal in Buccal Inserts
Buccal inserts must function in an environment defined by saliva, tissue movement, speaking, swallowing, and frequent surface renewal. These factors can shorten residence and disrupt uniform release if the system does not adhere well or if hydration progresses too quickly. Mucoadhesion, flexibility, and dimensional stability therefore become critical. A buccal insert that releases drug well in a static test may perform poorly if it detaches too early or softens excessively during actual use.
Why Local Environment Shapes Insert Design?
The local environment shapes insert design because it determines which failure mode appears first. In some sites, the main risk is early washout. In others, it is leakage, discomfort, deformation, or variable hydration. These site-specific pressures explain why insert development should not begin with a generic dosage-form template. It should begin with a realistic understanding of what the surrounding environment will do to the system over time.
| Insert Type | Typical Site of Use | Main Retention Mechanism | Main Release Mechanism | Key Polymer Requirement | Main Formulation Challenge |
|---|
| Ocular Insert | Ocular surface | Local fit and surface interaction | Diffusion, swelling, or erosion | Hydration control with acceptable comfort | Rapid clearance and limited tolerated bulk |
| Vaginal / Intrauterine Insert | Mucosal or cavity site | Residence within a fluid-exposed environment | Matrix release, swelling, or gradual erosion | Stable hydrated structure and local compatibility | Leakage, position stability, and variable environment |
| Buccal Insert | Oral mucosa | Mucoadhesion and flexible contact | Diffusion and hydration-mediated release | Adhesion with manageable softening | Saliva, motion, and early detachment |
| Nasal / Localized Insert | Nasal cavity or confined local site | Surface contact and form retention | Hydration-controlled matrix release | Rapid hydration without premature loss | Mucus turnover and clearance variability |
Where Polymeric Inserts Are Used in Long-Acting Drug Delivery?
Polymeric inserts appear across multiple local delivery settings, but the reason they are used is rarely identical from one site to another. In some cases, the main goal is to extend residence at a rapidly cleared surface. In others, it is to create a matrix that fits a confined local space and releases drug over time. This variety is exactly why the topic should be framed as polymeric inserts in long-acting delivery rather than as a single-site dosage form. The platform concept is broad, but the design logic is always local.
Ocular Inserts for Sustained Local Delivery
Ocular inserts are a clear example of how polymeric inserts can improve long-acting local delivery by addressing short surface residence and rapid drainage. In this setting, the insert serves as a controlled-release interface that may dissolve, swell, or remain intact long enough to improve local exposure compared with rapidly cleared liquid formulations. Comfort, hydration rate, flexibility, and limited tolerated size all shape ocular insert design. Because the ocular surface is sensitive to both bulk and residence behavior, material selection must be especially careful.
Vaginal and Intrauterine Inserts for Extended Retention
Vaginal and intrauterine insert systems can use polymer structure to improve local residence and support more sustained release within fluid-exposed environments. Here, the dosage form may benefit from a combination of shape stability, controlled swelling, and surface interaction. Long-acting behavior depends not only on drug diffusion through the matrix, but also on whether the insert can maintain its position and function under variable local conditions. A polymer that hydrates too quickly or loses mechanical coherence too early may shorten the effective release period.
Buccal Inserts for Mucoadhesive Drug Delivery
Buccal inserts often rely heavily on mucoadhesion because the oral environment exposes the system to movement, saliva dilution, and repeated mechanical stress. The polymer matrix must therefore achieve enough adhesion and flexibility to remain in place while still allowing predictable hydration and release. This makes buccal systems especially useful for demonstrating how retention and release are intertwined in insert design. Good drug release alone is not enough if the dosage form detaches prematurely.
Nasal and Other Localized Insert Systems
Nasal and other localized insert systems show the same basic pattern: the dosage form is selected because the local site benefits from a form-retaining system rather than a rapidly cleared liquid or an adhesive external patch. Depending on the target site, the insert may need fast hydration, moderate swelling, or more stable structure. These platforms are best viewed as local engineering solutions built around the needs of a specific environment rather than as one standardized dosage family.
What Polymers Actually Do in Insert Formulations?
The most useful way to think about polymers in insert systems is not as a list of materials, but as a set of functional tools. A polymer can anchor the insert at the site, regulate water uptake, determine whether the matrix keeps its shape, and decide how quickly the drug can leave the system. This is why insert formulation is fundamentally a material-function problem. The polymer is not a passive carrier. It is the main factor that turns a local dosage form into a retention-controlled and release-controlled platform.
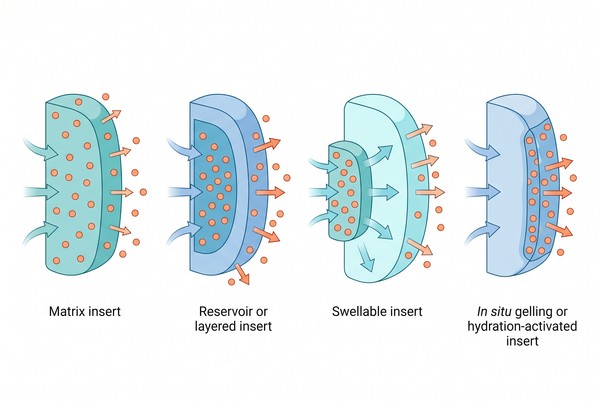 Fig. 2. How polymer matrices control retention and sustained release in inserts (BOC Sciences Authorized).
Fig. 2. How polymer matrices control retention and sustained release in inserts (BOC Sciences Authorized).
Mucoadhesion and Surface Anchoring
One of the most important roles of a polymer in insert systems is to support surface interaction strong enough to resist early displacement. Mucoadhesive behavior helps the dosage form remain at the target site long enough for controlled release to occur. Materials from natural polymers or bioinspired derivatives are often considered when surface interaction is a priority, especially when the local environment is wet or dynamic. This anchoring function is frequently more important than simple drug entrapment because it directly affects whether the intended release profile can be realized in practice.
Swelling, Hydration, and Microenvironment Control
Swelling and hydration behavior determine how fast the matrix absorbs fluid, how its dimensions change, and how the drug begins to move through the system. Hydrophilic materials such as Polyethylene glycol or local-matrix components based on Alginate can strongly affect how the insert interacts with moisture. Controlled hydration may help generate a release channel, improve local conformity, or maintain a favorable microenvironment around the payload. Uncontrolled hydration, by contrast, can accelerate deformation, leakage, or overly rapid release.
Matrix Formation, Mechanical Integrity, and Handling
Polymers also determine whether the insert can be manufactured, handled, inserted, and retained without breaking or softening too quickly. Materials associated with films or matrix-forming systems, including those related to Cellulose, help illustrate the importance of balancing formability and mechanical behavior. The matrix must be strong enough to keep the intended geometry, yet responsive enough to hydrate or erode in a controlled way. This is especially important when local comfort and dimensional stability need to be balanced in the same design.
Erosion, Degradation, and Release Modulation
When long-acting performance depends on gradual structural change, the polymer controls not only residence but also how the dosage form loses mass or becomes more permeable over time. Biodegradable polymers such as Poly(lactic-co-glycolic acid) (PLGA), Polylactic Acid (PLA), or Polycaprolactone (PCL) can be relevant when structural persistence and gradual release need to be coordinated. The development question is not simply whether the insert erodes, but whether the pace of erosion matches the intended release duration and the local site's tolerance for the changing matrix.
Need Support for Long-Acting Insert Design?
From polymer selection and mucoadhesive matrix design to retention control and release optimization, we support long-acting insert development across localized delivery platforms.
Insert Architecture and Long-Acting Release Logic
Insert performance is shaped as much by architecture as by material identity. Two systems made from similar polymers can behave very differently if one is a simple matrix, another contains a layered reservoir, and another swells into a gel-like structure after placement. For long-acting delivery, architecture determines how far drug must travel, how the environment penetrates the insert, and how the structure evolves during residence. This is why insert design should always connect geometry and internal organization to release logic.
Matrix Inserts for Sustained Drug Release
Matrix inserts distribute the drug throughout the polymer body and release it as water enters, the matrix hydrates, and the payload diffuses outward. This format is often attractive because it is structurally straightforward and compatible with a wide range of material strategies. However, matrix systems require close control of polymer-drug compatibility, thickness, and hydration rate. If the drug is enriched near the surface or if water ingress is too rapid, the release may become more front-loaded than intended.
Reservoir and Layered Inserts for Controlled Release
Reservoir and layered designs create more explicit separation between the drug-rich region and the surrounding release barrier. This can improve control when a more defined release pattern is needed or when the dosage form must combine different functional layers, such as a retentive surface and a slower-permeating core. These systems can be especially useful when sustained release must be balanced with a specific orientation or directional transport requirement. Their added design value, however, comes with greater manufacturing and consistency demands.
Swellable and In Situ Gelling Inserts for Prolonged Residence
Some insert systems depend on swelling or local gel formation to increase contact area, improve fit, or transform into a more persistent hydrated structure after placement. These strategies can be particularly relevant when prolonged local residence is more important than rigid structural persistence. Systems related in logic to polymer hydrogels illustrate how hydration can become a performance tool rather than only a side effect. The key challenge is to ensure that swelling supports retention and release without producing deformation severe enough to impair comfort or consistency.
Drug Distribution and Geometry Effects on Release Duration
Release duration is strongly affected by where the drug is located within the insert and by how thick, wide, or layered the system is. A thin insert with strong surface loading may release very differently from a thicker insert with deeper internal distribution. Geometry also changes the balance between exposed surface area and internal diffusion path length. As a result, long-acting behavior is rarely a simple function of polymer identity alone. It emerges from how drug placement and insert structure interact.
How to Match Polymer Choice to Payload and Residence Goal?
Insert development becomes much more efficient when polymer selection starts from the residence goal and payload profile rather than from a favored material family. Some payloads are best suited to fast-hydrating matrices that create a short but meaningful sustained window. Others require longer residence, stronger shape retention, or slower structural change. The right insert therefore comes from matching polymer function to the combination of local environment, drug properties, and intended duration. This matching step is often where technically promising ideas either become practical or begin to fail.
Fast-Dissolving vs Prolonged-Retention Inserts
Fast-dissolving inserts are useful when the formulation needs to release promptly after placement while still benefiting from better local residence than a liquid system. Prolonged-retention inserts are more appropriate when the dosage form must remain structurally or functionally present for a longer period to support continuous release. The decision between these two directions affects material hydrophilicity, shape stability, and the amount of drug that can be delivered effectively before the system loses performance.
Hydrophilic vs Poorly Soluble Payloads
Hydrophilic payloads may benefit from matrices that manage water uptake without allowing immediate washout, while poorly soluble payloads may require internal distribution strategies that prevent phase separation and uneven release. The insert must therefore support both payload stability and a workable diffusion pathway. A material that retains one type of payload well may release another too slowly or too quickly, which is why polymer choice has to be connected directly to payload behavior rather than treated as a generic platform decision.
Sensitive Molecules and Moisture-Dependent Stability
Sensitive molecules bring extra constraints because the insert often operates in a hydration-dependent environment. A matrix that performs well mechanically may still expose the payload to excessive moisture, interfacial stress, or local pH shifts during storage or use. In these cases, the formulation strategy must consider not just residence and release, but also what the polymer environment does to molecular integrity over time. This is especially important when the insert is expected to remain in place long enough for gradual release to matter.
Balancing Residence Time, Comfort, and Release Window
A practical insert must balance residence and release against site comfort and ease of use. A system that stays in place very well but feels too bulky, swells excessively, or becomes uncomfortable may not be a viable choice. Likewise, a very comfortable insert that releases too quickly may fail the long-acting objective. The best formulations are usually those that match the minimum necessary residence with the most efficient release design, rather than maximizing every parameter at once.
Development, Testing, and Failure Modes of Polymeric Inserts
The most informative way to evaluate polymeric inserts is to examine not only how well they release drug, but how they fail. Insert systems often lose value through early detachment, swelling-induced deformation, packaging instability, or inconsistent residence rather than through a purely pharmacotechnical release problem. Strong development work therefore combines structure, handling, hydration, and release testing. This approach reveals whether the platform truly behaves as a long-acting local system or only performs well under simplified conditions.
- Mucoadhesion, Residence, and Detachment Risk: Residence testing should ask not only whether the insert can adhere or remain in place initially, but whether that position is maintained long enough to support the planned release window. A system may exhibit strong early adhesion yet still detach when hydration changes the matrix or when the local environment exerts repeated mechanical stress. Detachment risk is therefore one of the most important development filters for this platform class.
- Swelling, Deformation, and Mechanical Failure: Swelling is often beneficial, but excessive swelling can change fit, thickness, surface pressure, and release behavior in ways that undermine the platform. Likewise, a matrix that is initially flexible may become too weak after hydration, while a strong matrix may crack or lose uniformity during drying and storage. Mechanical failure in insert systems is often dynamic rather than static, which means testing has to reflect the transition from dry handling to hydrated use.
- Release Testing and Duration Assessment: In vitro release testing should be interpreted alongside residence behavior, because a long release curve is not meaningful if the insert cannot remain in place under realistic conditions. Duration assessment should therefore combine matrix release logic with likely local exposure time. This is especially important when comparing matrix, reservoir, and swellable systems, since each may show a different relationship between laboratory release time and practical functional residence.
- Moisture, Packaging, and Reproducibility Challenges: Many insert systems are highly sensitive to ambient moisture because hydration directly affects flexibility, swelling onset, adhesion, and payload stability. Packaging is therefore part of formulation design, not an afterthought. Reproducibility also depends on controlling thickness, drug distribution, residual moisture, and polymer aging. If these factors drift, the same nominal insert can behave differently in residence and release even before the user applies it.
When Polymeric Inserts Are the Right Platform — and When They Are Not?
Polymeric inserts are at their best when the delivery problem is fundamentally about local residence and controlled release in a site that does not favor rapid loss of structure. They are less useful when the environment does not support practical retention, when a simpler dosage form can already deliver the needed performance, or when the required duration and geometry point more clearly toward another platform. Choosing inserts well therefore depends on understanding both their functional strengths and their natural limits.
- Best-Fit Scenarios for Insert-Based Long-Acting Delivery: Polymeric inserts are especially suitable when a program needs a local system that can remain at the site, maintain a defined contact area, and provide sustained release through matrix hydration, swelling, erosion, or barrier-controlled transport. They are often a strong fit when retention is the main challenge and when a form-retaining structure offers advantages over liquids, semisolids, or thin films.
- When Films, Hydrogels, or Patches Are Simpler: Some delivery goals do not require the structural presence of an insert. Films may be simpler when a thin format is sufficient. Gel-based systems may be more practical when spreading and conformal coverage are more important than defined shape. External patches may be preferable when the delivery site is the skin surface and adhesion, not local cavity residence, is the main requirement.
- When Implants or Depots Offer Better Duration Control: If the target product profile requires much longer duration, larger payload capacity, or highly defined geometry-driven control, an implant or depot may outperform an insert. In these cases, the insert format may not provide enough structural persistence or release precision to justify the added site-specific formulation complexity.
Services
Advanced Polymer Synthesis and Insert Formulation Services
At BOC Sciences, we approach polymeric inserts as material-driven delivery platforms rather than generic local dosage forms. Our development perspective connects retention behavior, matrix architecture, local-site compatibility, and release control into one formulation strategy, helping teams move from broad insert concepts to more practical polymer choices and testable product designs. Through polymer synthesis, material screening, structure-oriented formulation support, and performance evaluation, BOC Sciences provides technical support for insert systems that need to balance local residence, handling properties, and long-acting release behavior in a realistic development workflow.

Custom Polymer & Insert Material Design
- Material screening for mucoadhesive, swellable, and biodegradable insert systems.
- Selection of polymers based on retention target, hydration behavior, and release logic.
- Support for matrix-forming and structure-supporting polymer combinations.
- Design guidance for local-site-compatible polymer platforms.

Insert Formulation and Structure Development
- Development support for matrix, layered, and swellable insert architectures.
- Drug loading strategy optimization for different payload classes.
- Geometry and dimensional design aligned with residence requirements.
- Assistance in balancing comfort, retention, and release duration.

Release and Residence Evaluation
- Assessment of swelling, hydration, and matrix transformation behavior.
- In vitro release testing linked to residence and structural change.
- Drug–polymer compatibility review for long-acting insert systems.
- Support for identifying release-limiting and residence-limiting factors.

Stability, Packaging, and Development Support
- Evaluation of moisture sensitivity, packaging needs, and storage-related risks.
- Support for reproducibility across material batches and insert dimensions.
- Assessment of handling properties before and after hydration.
- Technical guidance for moving from concept screening to robust insert design.
Need a Consultation?
BOC Sciences supports polymeric insert development with material selection, retention design, matrix engineering, and long-acting release strategy guidance.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What are polymeric inserts in drug delivery?
Polymeric inserts are shaped local dosage systems that rely on polymer matrices to remain at a target site and release drug through controlled hydration, diffusion, swelling, erosion, or related mechanisms.
-
How do polymeric inserts provide long-acting drug release?
They provide long-acting performance by combining local residence with material-controlled release. The insert stays in place long enough for the polymer matrix to regulate how fast the payload leaves the system.
-
Which polymers are commonly used in insert systems?
Common choices include hydrophilic, mucoadhesive, swellable, and biodegradable polymers. The right material depends on the local environment, the required residence time, and the desired release mechanism.
-
How do polymeric inserts differ from hydrogels or implants?
Inserts are usually local, form-retaining dosage systems designed around residence at a defined site. Hydrogels may emphasize hydrated matrix behavior more broadly, while implants are usually more device-like and structurally persistent.
-
When are polymeric inserts a better choice than patches?
They are often better when the delivery site is a local cavity or mucosal surface where a retained insert can provide better contact and controlled release than an external adhesive patch.

 Fig. 1. Main application forms of polymeric inserts for long-acting drug delivery (BOC Sciences Authorized).
Fig. 1. Main application forms of polymeric inserts for long-acting drug delivery (BOC Sciences Authorized). Fig. 2. How polymer matrices control retention and sustained release in inserts (BOC Sciences Authorized).
Fig. 2. How polymer matrices control retention and sustained release in inserts (BOC Sciences Authorized).