Polymeric Implants for Long-Acting Drug Delivery: Material Selection and Formulation Design
Polymeric implants are an important platform for long-acting drug delivery because they combine material structure, drug–polymer compatibility, and controlled mass transport in a single dosage form. Unlike short-duration carriers that rely mainly on rapid dissolution or dispersion, implantable polymer systems are designed to maintain structural integrity and regulate drug release over extended periods through diffusion, polymer degradation, swelling, pore evolution, or a combination of these mechanisms. By adjusting polymer composition, molecular weight, crystallinity, geometry, wall thickness, and fabrication route, formulators can build implant systems that better match the stability, solubility, and release requirements of challenging active pharmaceutical ingredients (APIs). For research teams evaluating long-acting delivery strategies, polymeric implants offer a practical framework for balancing release duration, formulation robustness, and manufacturability.
Resources
Polymeric Implants for Long-Acting Drug Delivery
Polymeric implants are solid or in situ formed depot systems engineered to release an active compound gradually after placement in a target environment. In long-acting formulation design, they are especially valuable when the goal is to reduce dosing frequency, protect the API inside a structured matrix, and create a more predictable release pathway than can often be achieved with fast-dissolving dosage forms. Compared with dispersed carriers such as polymer nanoparticles or hydrated networks such as injectable polymer hydrogels, implants provide a more geometry-defined and often more durable release architecture. Their performance depends on how the polymer phase interacts with water, how the drug is distributed within the device, and how the internal microstructure changes over time.
What Are Polymeric Implants in Drug Delivery?
A polymeric implant is an implantable dosage system in which a drug is incorporated into, surrounded by, or separated by a polymer phase that controls the rate of release. The polymer may act as a diffusion barrier, a degrading matrix, a structural shell, or a phase-transforming medium. In practical formulation terms, implants are selected when the API requires prolonged release, localized retention, or a more stable solid-state environment than is available in solution or suspension systems. They are often designed as rods, cylinders, membranes, pellets, extrudates, monoliths, or injectable in situ forming depots that solidify after administration. The central formulation question is not simply whether a polymer can carry the drug, but whether that polymer can regulate release in a stable and reproducible manner throughout the intended duration.
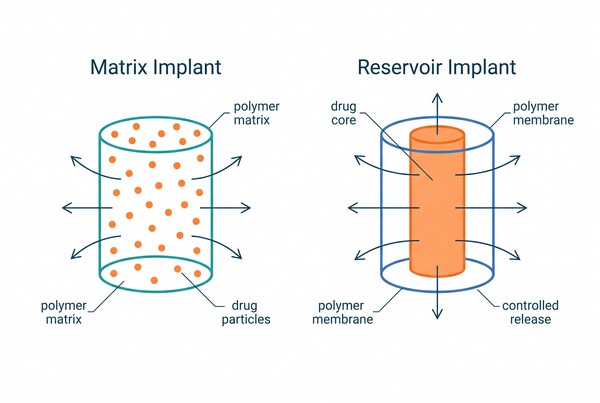 Fig. 1. Schematic of matrix and reservoir implant release pathways (BOC Sciences Authorized).
Fig. 1. Schematic of matrix and reservoir implant release pathways (BOC Sciences Authorized).
How Polymeric Implants Differ from Other Long-Acting Systems?
Polymeric implants differ from microspheres, hydrogels, and non-implant depots mainly in how geometry and structure determine mass transfer. Polymer microspheres provide high surface area and can release rapidly unless the polymer and particle morphology are tightly controlled. Hydrogels are useful when water-rich environments and soft mechanics are desired, but their mesh-based release behavior is distinct from the dense or semi-dense matrices seen in many implant systems. Implant formats offer a more controlled external geometry, allowing formulators to manipulate surface area, thickness, diffusion path length, and matrix density more directly. This is why implants are often attractive when long-duration release, structural persistence, or a defined device architecture is required.
Why Polymeric Implants Are Useful for Long-Acting Release?
The value of polymeric implants lies in their ability to decouple immediate drug availability from the total loaded dose. Instead of exposing the full API payload at once, the implant can meter release through a barrier mechanism, a degrading matrix, or a progressive pore-forming network. This is especially important for APIs that are poorly soluble, chemically sensitive, or difficult to keep in a stable dispersed state over time. The implant approach also gives formulators multiple control points, including polymer composition, device dimensions, drug loading pattern, and process conditions. Those variables make polymeric implants highly tunable, but they also mean formulation success depends on understanding the full structure–property–release relationship rather than choosing a polymer by name alone.
Types of Polymeric Implants and Their Structural Logic
Not all polymeric implants work in the same way. The internal arrangement of the drug and polymer often determines whether release is dominated by diffusion, membrane transport, swelling, or degradation. For that reason, implant classification is not just a descriptive exercise. It directly affects how formulators estimate burst release risk, how they scale the device, and which analytical methods are most informative during development. The most useful practical distinction is between matrix systems, reservoir systems, and in situ forming systems, followed by whether the carrier is biodegradable or non-biodegradable.
Matrix Implant Systems
In matrix implants, the drug is distributed throughout the polymer body. Release occurs as dissolved drug migrates through the matrix, diffuses through water-filled pores, or becomes available after polymer erosion and microstructural change. Matrix systems are often simpler to process than reservoir systems because they do not require separate core-shell assembly. They also support broad formulation flexibility, especially with poly(D,L-lactide-co-glycolide), polylactic acid, and poly(caprolactone) type materials. However, because the drug is present near the surface as well as deeper in the matrix, matrix implants can be more sensitive to initial burst release, internal pore evolution, and residual unreleased drug if the device does not become sufficiently porous during degradation.
Reservoir Implant Systems
Reservoir implants contain a drug-rich core surrounded by a rate-controlling polymer membrane. Their advantage is conceptual simplicity at the release interface: once the core environment is established, release may be tuned by membrane permeability, implant surface area, and wall thickness. In such systems, polymer selection is driven not only by biocompatible degradation behavior but also by barrier function, mechanical integrity, and stability under fabrication conditions. Reservoir designs are useful when a more linear or membrane-regulated release profile is desired, but they place greater emphasis on shell uniformity, sealing quality, and the consistency of the internal core formulation.
Biodegradable and Non-Biodegradable Implant Systems
Biodegradable implants rely on polymer cleavage, erosion, or matrix opening to support eventual clearance of the device. Materials such as lactide- and glycolide-based polyesters are widely used because their ester bonds undergo hydrolysis and their degradation behavior can be adjusted through monomer ratio and molecular weight. Non-biodegradable implants instead preserve their structure over much longer periods and depend mainly on diffusion through a stable membrane or matrix. The practical formulation difference is that biodegradable systems must be optimized for changing microstructure over time, while non-biodegradable systems require closer control of steady barrier performance and long-term structural persistence.
Preformed and In Situ Forming Implants
Preformed implants are fabricated before use as solid devices such as rods, films, cylinders, or extruded segments. This allows tighter geometric control and clearer device characterization before release testing. In situ forming implants begin as polymer solutions, suspensions, or precursor mixtures and then solidify or precipitate after placement because of solvent exchange, temperature change, or another trigger. Their appeal lies in simplified placement and adaptable shape formation, but they are more sensitive to solvent effects, phase inversion kinetics, and early burst release linked to rapid water ingress and pore generation. The choice between preformed and in situ forming systems therefore depends on how much control is needed over device geometry versus placement flexibility.
Which Polymer Materials Are Most Suitable for Long-Acting Implants?
Choosing the right polymer for a long-acting implant is not simply a matter of selecting a biodegradable or non-biodegradable material. The polymer must match the intended release duration, implant architecture, API properties, fabrication route, and the balance between early release control and long-term structural behavior. In practice, the most suitable implant polymers are those that provide a predictable combination of matrix integrity, water uptake behavior, degradation or permeation control, and manufacturability. Some materials are especially useful for degradable matrix implants, while others perform better as reservoir membranes, structural coatings, or hydrophilic modifiers. For this reason, polymer selection should be based on release mechanism and formulation purpose rather than on material popularity alone.
PLGA: Tunable Degradation with Broad Formulation Flexibility
PLGA remains one of the most useful polymer families for long-acting implants because it offers a practical balance between degradability, processability, and release tunability. By adjusting the lactide-to-glycolide ratio, formulators can influence hydrophobicity, water penetration, chain mobility, and the overall pace of hydrolytic breakdown. This makes PLGA highly adaptable for matrix implants, monolithic rods, and in situ forming depots where the release profile depends on both diffusion and progressive matrix opening. PLGA is especially valuable when the formulation goal is to design a biodegradable implant with a defined but adjustable release window. At the same time, its performance must be interpreted carefully, because internal acid generation, pore evolution, and drug distribution heterogeneity can all change the release shape in ways that are not obvious from composition alone.
PCL: Slower Erosion and Better Long-Term Structural Persistence
Polycaprolactone (PCL) is often selected when longer structural persistence and slower matrix change are more important than faster biodegradation. Its semicrystalline nature and lower hydrolysis rate make it useful in implants that need to maintain shape and barrier function over extended durations. PCL can work well in both matrix and membrane-based designs, especially when the intent is to reduce rapid matrix collapse or to build a reservoir system with a more stable rate-controlling layer. Because PCL erodes slowly, it is particularly attractive for formulations that need a prolonged release tail without rapid loss of device integrity. The tradeoff is that slow erosion can also prolong incomplete-release risk if the internal transport pathways remain too restricted, so PCL-based implants often require more deliberate control of geometry, loading state, and drug solubility.
PLA and Related Polyesters: Where Higher Hydrophobicity Helps
PLA and related polyester materials are useful when a more hydrophobic matrix is needed to slow water ingress and moderate early drug escape. Compared with more rapidly hydrating systems, these materials may offer better control over initial diffusion and improved structural retention in solid implants. They are especially helpful when the API is sensitive to rapid aqueous exposure or when the formulation objective is to delay matrix opening relative to more hydrophilic alternatives. However, greater hydrophobicity does not automatically produce a better implant. If the polymer becomes too resistant to hydration or too slow to form connected transport channels, the formulation may show extended tail release or leave part of the payload trapped in the matrix. For this reason, PLA-type systems are most effective when hydrophobicity is used as a tuning tool rather than treated as an end in itself.
EVA, Silicone, and Other Non-Degradable Polymers for Reservoir Designs
Non-degradable polymers such as EVA, silicone, and selected polyurethanes are particularly useful in reservoir implant designs because they act as long-term diffusion barriers rather than degrading matrices. In these systems, the polymer does not need to erode in order to release the drug. Instead, release is governed mainly by membrane permeability, wall thickness, implant surface area, and the physical state of the internal drug core. This makes non-degradable materials attractive when a stable, membrane-controlled release mechanism is preferred over a dynamically changing biodegradable matrix. Their main formulation strength is structural consistency across time, but that strength also means the designer must place more emphasis on membrane uniformity, sealing integrity, and long-term barrier reproducibility. They are best suited to applications where controlled diffusion through a persistent polymer shell is more valuable than matrix breakdown.
How Molecular Weight, Monomer Ratio, and Crystallinity Change Implant Behavior?
Even within the same polymer family, implant behavior can change substantially depending on molecular weight, monomer composition, and crystallinity. Higher molecular weight typically increases chain entanglement and slows relaxation, which can reduce early release but may also make processing more difficult. Monomer ratio, especially in copolyesters, changes hydrophobicity, degradation pace, glass transition behavior, and the way water penetrates the matrix. Crystallinity further affects permeability by creating domains that are less accessible to water and dissolved drug, often improving structural persistence while potentially prolonging late-stage release. These variables are not secondary material descriptors; they are central release-control parameters. In implant design, they should be screened with the same importance as drug loading and geometry because they determine whether the matrix behaves as a fast-opening depot, a slow-eroding monolith, or a stable long-term barrier.
| Polymer Family | Primary Role in Long-Acting Implants | Key Strength | Main Limitation | Typical Formulation Use |
|---|
| PGA | Fast-degrading biodegradable polyester for matrix design and release acceleration | Promotes relatively rapid matrix hydration and degradation-driven release | May degrade too quickly for implants requiring extended structural persistence | Fast-opening matrix implants, blend component in copolyester systems, and release-rate adjustment studies |
| PLA | Hydrophobic biodegradable polyester for sustained matrix implants | Slower water ingress and better structural retention than faster-eroding aliphatic polyesters | Can show prolonged tail release if matrix opening is too limited | Monolithic implant rods, sustained-release matrices, and hydrophobic polyester blends |
| PLGA | Biodegradable copolyester for tunable long-acting implant matrices | Broad control over degradation rate and release duration through composition adjustment | Can show burst release, acidic microenvironment effects, and incomplete release if not optimized | Biodegradable matrix implants, extruded rods, and in situ forming depot systems |
| Polyethylene Glycol (PEG) | Hydrophilic modifier, blend component, and porosity regulator | Improves water uptake and helps tune internal transport pathways | Excess incorporation may accelerate initial release and weaken long-duration control | Polymer blends, pore-forming modulation, hydrophilic channel tuning, and release-profile adjustment |
| Ethylene-Vinyl Acetate | Non-degradable membrane or reservoir implant material | Stable diffusion-barrier performance for prolonged release systems | Does not provide degradable matrix clearance | Reservoir implants, membrane-controlled rods, and long-term diffusion barrier layers |
| Polyvinyl Alcohol | Hydrophilic film-forming polymer and release-modifying component | Useful for coatings, blends, and controlled hydration adjustment | Water sensitivity may increase early drug escape if poorly balanced | Coatings, matrix blends, hydration regulators, and pore-structure modification systems |
| Polyurethanes | Mechanically durable membrane, shell, or structural implant polymer | Good mechanical stability with tunable permeability for reservoir designs | Performance depends strongly on segment chemistry and transport balance | Reservoir shells, rate-controlling membranes, flexible implant housings, and diffusion-controlled devices |
| Chitosan | Natural cationic polymer for hybrid, hydrated, or functional implant systems | Provides bioadhesive and ionic interaction capability in structured release platforms | Usually requires blending or structural reinforcement for durable long-acting implants | Hybrid matrices, functional coatings, polyelectrolyte-modified implants, and combination depot systems |
| Hyaluronic Acid (HA) | Hydrophilic natural polymer for hydrated matrices or combination implant systems | Supports water-rich environments and functional modification in soft depot designs | Alone it is typically too water-responsive for long-term solid implant persistence | Hydrated hybrid implants, soft depot combinations, surface-functional layers, and matrix modification platforms |
Need Polymer Screening for Implant Formulation Development?
Whether you are comparing PLGA-type matrices, slow-eroding PCL barriers, or functional polymer blends for implantable depots, our team supports material selection, release-oriented design, and structure–property evaluation for long-acting delivery projects.
Drug Release Mechanisms in Polymeric Implants
Drug release from polymeric implants is rarely controlled by a single mechanism from start to finish. The dominant pathway may change as the device hydrates, the drug dissolves, the polymer swells or degrades, and the internal porosity evolves. A robust implant formulation therefore requires more than measuring cumulative release. It requires interpreting why release takes a certain shape and which structural events control each phase. In practical terms, most systems display a sequence involving initial wetting, early surface-associated release, matrix or membrane transport, and later-stage structural transformation. Understanding those transitions is essential when trying to reduce burst, extend duration, or avoid incomplete release.
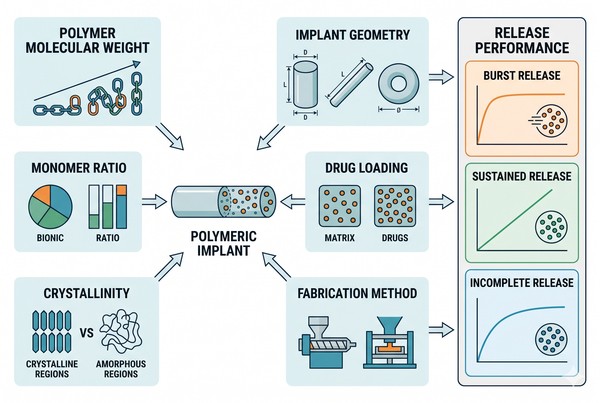 Fig. 2. Key formulation variables shaping implant release performance (BOC Sciences Authorized).
Fig. 2. Key formulation variables shaping implant release performance (BOC Sciences Authorized).
Diffusion-Controlled Release
Diffusion-controlled release dominates when the drug dissolves into the aqueous phase within or adjacent to the polymer and then migrates through a polymer membrane, a dense matrix, or a network of water-filled channels. In reservoir systems, this may be approximated as membrane transport controlled by wall thickness and permeability. In matrix systems, diffusion depends on how fast water enters, whether the drug is molecularly dispersed or crystalline, and whether the polymer already contains pathways that permit solute transport. A dense and hydrophobic matrix slows diffusion, while porosity, hydrophilic additives, or phase separation can accelerate it.
Degradation- and Erosion-Controlled Release
Degradation-controlled release becomes important when the implant matrix initially restricts mobility and the drug cannot leave efficiently until the polymer undergoes hydrolysis, chain scission, softening, or pore formation. This is common in biodegradable polyester implants, especially when the drug has low diffusivity in the intact matrix. As the polymer breaks down, the matrix may become more permeable, pores may interconnect, and trapped drug may finally access the external medium. In some devices, degradation is the main driver of the later release phase, even if the early phase is diffusion-dominated. The transition point depends on polymer chemistry, device dimensions, and water penetration depth.
Swelling, Water Uptake, and Pore Formation
Water uptake is often the hidden control variable behind implant performance. Once water enters the polymer body, it can dissolve surface-near drug, plasticize the matrix, alter chain mobility, and initiate internal pore formation. In hydrophilic or partially hydrophilic systems, swelling may open transport pathways earlier than expected. In more hydrophobic matrices, water ingress may be delayed and then accelerate once microcracks or localized pores appear. Formulators should not treat water uptake as a secondary effect. It governs whether the implant behaves as a stable barrier, a slowly opening depot, or a rapidly transitioning porous matrix.
Why Burst Release Happens
Burst release is usually caused by a combination of surface-enriched drug, near-surface pores, rapid solvent exchange, or weak drug–polymer compatibility that drives phase segregation during processing. In matrix implants, drug located at or near the external surface can dissolve and escape quickly upon wetting. In in situ forming systems, fast phase inversion can create large channels that accelerate the initial escape of dissolved drug. High loading, poor solid-state dispersion, and excessive porosity all increase this risk. Reducing burst release therefore requires attention to particle distribution, device surface condition, solvent removal kinetics, and the miscibility of the API with the polymer phase.
Why Incomplete Release and Long Tail Release Occur
Incomplete release occurs when a fraction of the API remains trapped in a matrix that never develops sufficiently connected pathways for transport, or when the drug becomes immobilized through recrystallization, poor solubilization, or strong local interactions. Long tail release often appears when the device architecture is too resistant to water penetration, when the remaining drug resides in poorly connected domains, or when the external barrier remains intact long after the therapeutically useful release phase. These issues are common in dense matrices and slow-eroding devices and should be assessed early during formulation screening rather than after scale-up.
Formulation Design: Matching API Properties with Implant Architecture
A polymeric implant succeeds only when the polymer, the API, and the fabrication route are compatible with one another. Even a well-known implant polymer will perform poorly if the drug crystallizes during processing, partitions to the surface, or destabilizes the matrix microstructure. That is why implant development must start with the physicochemical profile of the API, not with a list of popular polymers. Solubility, pKa, hygroscopicity, dose requirement, melting behavior, thermal sensitivity, and target release duration all determine which implant architecture is realistic. Formulators should then use those API constraints to choose between matrix, reservoir, or in situ forming systems and to define the most informative screening variables.
| Formulation Variable | What It Changes | Risk If Too Low | Risk If Too High | Optimization Focus |
|---|
| Polymer molecular weight | Chain mobility, erosion rate, matrix integrity | Premature softening or faster release | Difficult processing and delayed release | Balance manufacturability and release duration |
| Drug loading | Concentration gradient and internal domain structure | Insufficient dose density | Burst release and phase separation | Maintain uniform distribution without surface enrichment |
| Crystallinity | Water ingress and diffusional resistance | Weak barrier control | Long tail release or incomplete release | Use crystallinity as a transport tuning lever |
| Surface area | Drug flux and hydration rate | Undershooting target release | Excessively rapid release | Match geometry to desired daily output |
| Wall thickness | Diffusion path length in reservoir devices | Fast membrane transport | Overly restricted release | Control rate without causing dose lag |
| Porosity / pore formers | Water access and channel connectivity | Poor total release | High burst release | Create controlled, not uncontrolled, pathways |
| Fabrication route | Drug state, morphology, residual solvent, geometry | Poor structural consistency | Thermal or processing stress on API | Select a route aligned with API stability |
Matching Drug Solubility with Polymer Polarity
Drug solubility relative to the polymer and the release medium strongly shapes implant behavior. A highly hydrophobic drug in a hydrophobic matrix may show low burst and slow release, but it may also become too strongly retained if water access is limited. A more polar drug may partition toward water-rich regions quickly, especially if the matrix contains hydrophilic domains or solvent-induced pores. The goal is not simply to maximize affinity between drug and polymer. It is to create enough compatibility for stable loading while still allowing predictable release once the implant hydrates.
Drug State inside the Implant
The API can exist as a molecular dispersion, amorphous domains, fine crystals, or large segregated particles. Each state gives a different release signature. Molecularly dispersed drug may show smooth early release if the matrix remains intact, but it may also recrystallize over storage if the system is unstable. Crystalline drug can act as a long-term reservoir, yet oversized crystals or uneven particle placement can cause local channels and heterogeneous depletion. During process development, the internal physical state of the drug should therefore be treated as a critical design attribute, not an incidental result of fabrication.
Geometry as a Release Control Tool
Implant geometry affects release more directly than many formulators expect. Diameter, length, membrane area, thickness, and aspect ratio all influence flux, hydration rate, and the path length available for drug escape. A smaller device may release faster simply because its surface-area-to-volume ratio is higher. A thicker membrane can slow transport in a reservoir system even when the internal core composition is unchanged. Geometry should be screened as early as polymer composition because it can often deliver a cleaner release adjustment than drastic changes in chemistry.
Choosing the Right Fabrication Route
Common fabrication routes for polymeric implants include hot-melt extrusion, compression molding, solvent casting, phase inversion, and in situ forming precipitation systems. Each route changes morphology in a different way. Hot-melt processing may improve uniformity but requires thermal compatibility with the API. Solvent-based methods may protect heat-sensitive compounds but can introduce pore formation, residual solvent concerns, and rapid initial release if solvent removal is not well controlled. In situ forming routes are attractive for minimally invasive placement, yet they demand careful control of solvent exchange rate and polymer precipitation behavior. The right process is the one that yields the target internal structure reproducibly, not merely the one that is easiest at bench scale.
How to Reduce Burst Release without Blocking Total Release
Burst control should not be pursued by making the matrix uniformly impermeable. That can solve the first-day profile while creating poor total release later. A better strategy is to reduce surface-near enrichment, improve drug dispersion, optimize polymer molecular weight, use geometry to control flux, and moderate porosity rather than eliminating it. In some systems, blending polymers with different transport characteristics can smooth the early release phase while preserving longer-term matrix opening. The correct balance depends on whether the implant is intended to behave as a barrier device, a degrading monolith, or an in situ forming porous depot.
Evaluation Strategy and Common Development Pitfalls
Reliable implant development depends on evaluating not just how much drug is released, but why the release profile takes its observed form. Many implant programs fail not because the polymer was unsuitable in principle, but because the wrong development signals were prioritized. A release curve alone cannot distinguish membrane control from pore-assisted diffusion or tell whether a late-stage slowdown comes from depleted drug or a transport bottleneck. The evaluation strategy must therefore combine release testing with structural, thermal, and chemical characterization so that formulation adjustments are guided by mechanism rather than guesswork.
Critical Quality Attributes for Polymeric Implants
The most useful critical quality attributes include drug loading uniformity, implant geometry, surface condition, residual solvent level where relevant, polymer molecular weight distribution, thermal behavior, internal morphology, and release reproducibility. For reservoir systems, membrane thickness consistency and seal integrity are especially important. For degrading matrix implants, porosity development, polymer mass loss, and drug state evolution deserve close attention. These attributes connect directly to performance and provide more meaningful guidance than generic pass/fail specifications.
Why In Vitro Release Testing Must Be Interpreted Carefully
In vitro release testing is indispensable, but it should not be treated as a perfect surrogate for every use environment. Medium composition, sink conditions, agitation, sampling interval, and device placement all affect the observed profile. A release method that is too aggressive may exaggerate burst and underrepresent barrier effects, while a method with poor sink conditions may artificially flatten later release. The purpose of in vitro testing during development is to reveal mechanism, rank formulations, and identify instability or variability. It works best when paired with parallel imaging and polymer characterization rather than used in isolation.
Mechanical Integrity, Stability, and Process Reproducibility
Long-acting implants must maintain sufficient structural integrity throughout fabrication, handling, storage, and the intended release window. A brittle device may crack and create uncontrolled surface area. A softening device may collapse or alter diffusion pathways unexpectedly. Storage stability matters as much as release duration because drug crystallization, moisture uptake, or polymer relaxation before use can shift the profile substantially. Process reproducibility is equally critical: small changes in solvent evaporation, extrusion temperature, or particle dispersion can translate into large changes in porosity and burst release.
Common Pitfalls in Implant Development
Frequent pitfalls include choosing polymers by reputation rather than API compatibility, treating drug loading as a simple scale-up variable, overlooking surface enrichment after processing, and trying to eliminate burst release by making the device too impermeable. Another common mistake is assuming that one successful implant geometry will translate across different APIs. In reality, drug solubility, solid-state behavior, and partitioning can completely change the governing mechanism. The best development programs therefore build a small but mechanism-focused screening matrix rather than testing many conditions without a clear structure–property hypothesis.
A Practical Selection Framework
A useful decision path begins with the API: determine dose density, thermal sensitivity, solid-state risk, and desired release window. Then decide whether the system needs a degrading matrix, a persistent membrane barrier, or an in situ forming depot. Next, choose a polymer family whose transport and processing behavior can support that architecture. Finally, screen the minimum set of variables that most strongly affect mechanism: molecular weight, composition, loading, geometry, and fabrication route. This approach reduces unnecessary experimentation and aligns material selection with release logic from the start.
Services
Advanced Polymer Synthesis and Formulation Services
At BOC Sciences, we support long-acting implant development from the perspective of polymer design, release control, and formulation problem solving. Our team works across biodegradable polyesters, functional polymer modification, release-oriented matrix design, and analytical characterization to help researchers match polymer properties with implant architecture and API requirements. Whether the project involves a PLGA-type degrading matrix, a slow-permeation barrier built around poly-ε-caprolactone, or a customized material screening workflow based on polymer materials, we provide practical support focused on drug–polymer compatibility, process feasibility, and release performance.

Custom Polymer & Copolymer Synthesis
- Tailored synthesis of biodegradable polyester and functional copolymer candidates.
- Control over molecular weight, composition, and architecture for release-oriented screening.
- Support for matrix-forming, membrane-forming, and specialty implant polymers.
- Adaptation of polymer structures to API compatibility and target duration needs.

Polymer Functionalization & Material Optimization
- End-group and side-chain modification to tune hydrophobicity and interaction strength.
- Polymer blending strategies for burst control and matrix opening balance.
- Material screening support for barrier layers, degradable matrices, and release modifiers.
- Development of customized polymer candidates for implant-specific formulation logic.

Implant Formulation Development
- Screening of drug loading, geometry, and polymer ratios for long-acting release.
- Evaluation of matrix, reservoir, and in situ forming implant design routes.
- Optimization of process variables affecting morphology, porosity, and surface enrichment.
- Support for release testing strategy and structure–property interpretation.

Analytical Characterization & Scale-Up Support
- Characterization of molecular weight, thermal behavior, and drug–polymer state.
- Morphology and consistency analysis for reproducible implant fabrication.
- Support for process transfer from exploratory screening to scalable routes.
- Quality-focused evaluation of residual solvent, batch variation, and release reproducibility.
Do You Need A Consultation?
BOC Sciences helps researchers evaluate polymer materials, implant architectures, and release-control strategies for long-acting drug delivery projects with a formulation-first technical approach.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What is a polymeric implant in drug delivery?
A polymeric implant is a dosage system in which a polymer matrix or membrane controls drug release over an extended period. Depending on the design, release may be governed by diffusion, polymer degradation, swelling, or membrane permeability, making these systems useful for long-acting and structurally controlled drug delivery.
-
What is the difference between matrix and reservoir implants?
In a matrix implant, the drug is distributed throughout the polymer body, so release depends on diffusion and matrix change over time. In a reservoir implant, the drug core is surrounded by a polymer membrane, and release is controlled mainly by membrane permeability, thickness, and implant surface area.
-
Why are PLGA-type polymers widely used in long-acting implants?
PLGA-type polymers are widely used because they combine biodegradability with adjustable release behavior. By changing monomer ratio, molecular weight, and device geometry, formulators can tune water uptake, degradation rate, and matrix opening. This flexibility makes PLGA suitable for many long-acting implant and depot formulation strategies.
-
When is PCL a better choice than faster-degrading polyesters?
PCL is often preferred when a formulation needs slower erosion and longer structural persistence. Its semicrystalline character can help maintain implant integrity and extend release duration. This makes it useful for matrix or membrane designs where slower polymer change is more desirable than the faster breakdown seen in some polyesters.
-
What usually causes burst release in polymeric implants?
Burst release is commonly caused by drug located near the implant surface, rapid water penetration, uncontrolled pore formation, or poor drug–polymer compatibility. Process-related factors such as solvent exchange rate, uneven drug distribution, and excessive porosity can also accelerate early drug escape and distort the intended release profile.
-
How can formulators reduce burst release?
Formulators usually reduce burst release by improving drug dispersion, limiting surface enrichment, optimizing polymer molecular weight, and controlling porosity without blocking total release. Geometry can also be adjusted to reduce early flux. The goal is to moderate initial escape while preserving enough matrix opening for continued long-term release.
-
What factors most strongly affect implant release kinetics?
Implant release kinetics are mainly affected by polymer composition, molecular weight, crystallinity, drug loading state, device geometry, membrane thickness, and fabrication route. These variables influence water ingress, diffusion path length, degradation rate, and internal pore formation, all of which determine whether release is smooth, delayed, burst-prone, or incomplete.
-
What tests are most informative during implant development?
The most informative tests usually include in vitro release studies, morphology analysis, thermal characterization, drug-state evaluation, polymer molecular weight tracking, and storage stability assessment. Together, these methods help explain not only how much drug is released, but also how implant structure and polymer change drive release behavior.

 Fig. 1. Schematic of matrix and reservoir implant release pathways (BOC Sciences Authorized).
Fig. 1. Schematic of matrix and reservoir implant release pathways (BOC Sciences Authorized). Fig. 2. Key formulation variables shaping implant release performance (BOC Sciences Authorized).
Fig. 2. Key formulation variables shaping implant release performance (BOC Sciences Authorized).