In Situ Forming Polymeric Depots for Long-Acting Drug Delivery: Formulation Design and Release Control
Polymeric in situ forming depots are long-acting drug delivery systems designed to be administered as low-viscosity liquids, solutions, or suspensions that transform into a gelled or solidified reservoir after administration. This combination of injectability before use and depot persistence after administration makes the platform especially attractive when formulators need extended release without relying on preformed implants or large particulate suspensions. By selecting suitable polymer materials, trigger mechanisms, and formulation variables, these systems can be engineered to control depot formation, internal structure, drug distribution, and long-term release behavior. Their practical value lies in the ability to combine relatively simple administration with programmable release, adaptable depot architecture, and compatibility with both localized and systemic long-acting delivery goals.
Resources
Polymeric In Situ Forming Depots for Long-Acting Drug Delivery
In situ forming depots occupy a distinctive position in long-acting formulation science because they separate administration from final depot structure. Instead of manufacturing a fully solid implant or a preformed matrix before administration, the formulator delivers a precursor system that converts into a depot in the target environment. This apparently simple distinction has major formulation consequences. It changes injectability requirements, introduces strong coupling between solvent exchange and matrix formation, and makes early-stage release behavior highly sensitive to formulation composition and application conditions. As a result, successful in situ depot development depends on understanding not only materials, but also the kinetics of transformation from a fluid precursor into a functional long-acting matrix.
What Defines a Polymeric In Situ Forming Depot?
A polymeric in situ forming depot is a drug delivery system in which a polymer-containing formulation is administered in a flowable state and then forms a semi-solid or solid drug reservoir after placement. The precursor may be a true solution, a polymer suspension, or a multi-component formulation that responds to environmental triggers such as water influx, temperature, pH, ions, or chemical reaction. Once the depot forms, drug release proceeds through diffusion, matrix relaxation, swelling, degradation, erosion, or combinations of these mechanisms depending on polymer type and internal structure. The defining feature is therefore not merely prolonged release, but post-administration depot formation. This matters because the earliest stage of performance often determines the later release profile. If solvent leaves the system too quickly, if the polymer precipitates unevenly, or if the drug partitions into the surrounding medium before the matrix stabilizes, the final depot may show burst release, porosity-related variability, or inconsistent long-term behavior even when the bulk composition appears rational.
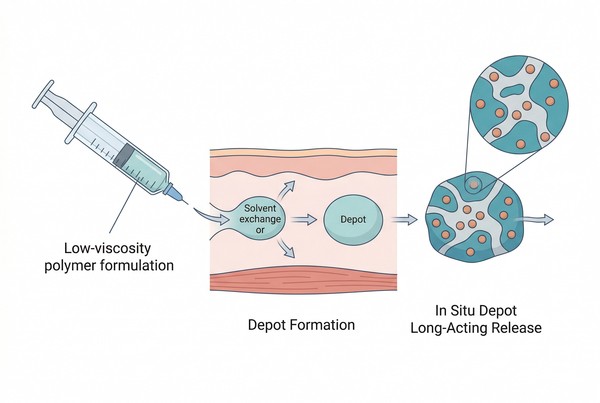 Fig. 1. In situ depots form after administration through triggered matrix solidification or gelation (BOC Sciences Authorized).
Fig. 1. In situ depots form after administration through triggered matrix solidification or gelation (BOC Sciences Authorized).
Why In Situ Depots Are Attractive for Long-Acting Delivery?
In situ depots are attractive because they combine long-acting release potential with practical administration advantages. A precursor formulation with relatively low viscosity can often be handled, filled, and delivered more easily than a preformed implant or dense particulate suspension. This reduces the need for invasive placement while preserving the possibility of forming a structurally persistent depot after administration. For many projects, that balance between administration simplicity and depot-based release is the core reason the platform is considered. Their flexibility also makes them useful complements to existing long-acting systems. They can sometimes bridge the gap between large implants and dispersed particles by providing a localized matrix without requiring the manufacturing of a rigid device. In other cases, they are chosen because the release profile can be tuned by polymer selection, trigger mechanism, solvent system, or matrix architecture. This ability to predefine depot behavior through formulation design is one of the platform's main strengths.
How In Situ Depots Differ from Implants, Microspheres, and Hydrogels?
In situ depots differ from preformed implants because the final structure is created after administration rather than before it. They differ from microspheres because the system is typically delivered as a continuous precursor formulation rather than as a suspension of already hardened particles. They also differ from polymer hydrogels in an important way: although some in situ depots ultimately form gel-like structures, many rely on precipitation, phase inversion, or matrix solidification rather than classic swelling-network gelation as the primary depot-forming mechanism. These differences are not just academic. They determine how the formulation is manufactured, how it behaves during administration, and how early release begins. A preformed implant may offer more stable geometry from the outset, while an in situ depot may offer easier injection. A hydrogel may provide a milder water-rich environment, while a phase-inverted depot may provide stronger hydrophobic retention. Platform comparison should therefore begin with structure formation logic rather than with broad labels alone.
What Makes Low-Viscosity Depot Precursors Useful in Practice?
Low-viscosity precursor systems are useful because they simplify several practical aspects of formulation and delivery. They are generally easier to process, can often be filled more reproducibly, and can be administered through conventional needles without the force required for dense implants or highly structured gels. This improves manufacturing flexibility and often makes the platform more attractive when the product concept depends on injection efficiency rather than surgical placement. However, low viscosity is only helpful if it is paired with controlled depot formation after administration. A precursor that is easy to inject but slow to solidify may lose drug before the depot stabilizes, while one that begins to precipitate too early may become difficult to administer. Good in situ depot design therefore treats low-viscosity handling and post-injection structural locking as a coupled formulation problem.
How Polymeric In Situ Forming Depots Are Fabricated?
Fabrication strategy is one of the most important design choices in this platform because it determines how the precursor becomes a depot and what internal structure is created during that transformation. The same polymer can produce very different release behavior depending on whether the depot forms by solvent exchange, thermal transition, ionic response, chemical crosslinking, or a mixed pathway. Formulators therefore need to evaluate fabrication route as a structural design variable rather than a procedural detail.
Solvent Exchange and Phase Inversion Systems
Solvent exchange and phase inversion are among the most recognized in situ depot formation routes. In these systems, a polymer is dissolved in a water-miscible or partially miscible solvent and injected into an aqueous environment. As solvent diffuses outward and water diffuses inward, polymer solubility decreases and the matrix precipitates to form a depot. The speed and uniformity of that exchange largely determine the eventual porosity, drug distribution, and early burst release of the system. This route is attractive because it can create solid or semi-solid depots from relatively simple precursor formulations. At the same time, it is one of the most sensitive approaches from a release-control perspective. Fast solvent exchange may lead to a porous outer shell, rapid drug loss, or heterogeneous depot formation, while slower exchange may improve densification but change injectability and solidification kinetics. In practice, solvent-driven systems require especially careful matching of polymer, solvent, and payload behavior.
Thermally Triggered In Situ Gelling Systems
Thermally triggered systems remain fluid or less structured before administration and become more ordered as temperature changes after placement. These systems are attractive when the formulation goal is to combine easy injection with gel or semi-solid formation without relying heavily on solvent loss. In many cases, the transformation is driven by changes in polymer–water interactions, hydrophobic association, or chain organization that become stronger at the target temperature. Their main advantage is that they can reduce dependence on large solvent fluxes during depot formation, which may be beneficial for sensitive payloads. Their limitation is that gelation behavior can be highly sensitive to concentration, composition, and thermal history. If the transition is too weak, the depot may not persist; if it is too aggressive, the precursor may become difficult to handle or inject. For this reason, thermally triggered systems often demand narrow control over formulation variables.
pH-, Ion-, and Stimulus-Responsive Depot Formation
Stimulus-responsive in situ systems use changes in pH, ionic environment, or other triggers to initiate matrix formation. These approaches expand depot design beyond simple precipitation or heating by allowing polymers to respond to more specific environmental signals. In ion-responsive systems, multivalent ions can induce gelation or association. In pH-responsive systems, ionizable groups alter polymer solubility or network formation behavior. Other designs may incorporate chemical or external triggers to activate crosslinking or structural transition. The appeal of these systems lies in versatility. They can be tailored to the desired administration route, matrix strength, or release window more flexibly than some single-mechanism formulations. Their main challenge is reproducibility under realistic use conditions, because the trigger intensity can change with local environment, tissue conditions, and formulation aging. This makes trigger robustness a central part of development rather than a secondary confirmation experiment.
Suspension-Based and Solution-Based Depot Formulations
In situ depot precursors can be formulated as polymer solutions, drug suspensions, polymer suspensions, or combined systems in which multiple phases contribute to final structure formation. Solution-based systems often provide more uniform initial composition, while suspension-based systems may enable higher loading or altered release pathways when solid drug or polymer domains are already present before administration. The choice between them depends on solubility, payload stability, viscosity, and the mechanism used to form the depot. This distinction matters because suspended material can affect solvent transport, nucleation of solidification, and final pore structure. A solution precursor may form a smoother and more homogeneous matrix, while a suspension may create more complex internal domains that influence release and degradation. Good design therefore requires thinking about formulation state before injection as a determinant of final depot morphology.
How Fabrication Route Affects Final Depot Structure?
Fabrication route determines whether the final depot becomes dense or porous, uniform or phase-separated, rigid or gel-like. These structural outcomes directly influence how water enters the system, how fast the drug diffuses, and how degradation or erosion later contributes to release. Two formulations with the same polymer and same payload can therefore behave very differently if their depot formation pathways differ. This is why fabrication route should be selected with the intended release profile in mind. If the target is a denser matrix with delayed opening, the chosen mechanism should support controlled solidification. If a softer depot or more hydrated structure is preferred, another formation pathway may be more suitable. In situ depots are fundamentally structure-evolving systems, so fabrication and performance cannot be separated.
| Depot Formation Approach | Trigger Type | Typical Precursor State | Key Advantage | Main Limitation | Best-Fit Use |
|---|
| Solvent exchange / phase inversion | Water ingress and solvent loss | Polymer solution or suspension | Simple route to solidifying depots | High burst and morphology sensitivity | Dense or semi-solid long-acting depots |
| Thermally triggered gelation | Temperature change | Flowable polymer solution | Less dependence on solvent escape | Narrow gelation window | Injectable gel-like depots |
| Ion- or pH-responsive formation | Local chemical environment | Solution or mixed precursor | Versatile depot design | Trigger robustness can vary | Stimulus-responsive local systems |
| Chemically activated depot formation | Reactive crosslinking or bonding | Multi-component precursor | Potentially strong matrix integrity | Higher formulation complexity | Longer-lasting structured depots |
Need a Custom In Situ Depot Formulation Strategy?
From polymer screening and trigger selection to depot fabrication and release tuning, we support the design of in situ forming systems for long-acting drug delivery.
What Materials Are Used to Build In Situ Forming Depots?
Material selection in this platform is more than a matter of choosing a polymer that degrades slowly or gels efficiently. The polymer must be compatible with the precursor state, must respond appropriately to the intended formation trigger, and must produce a depot with the right mechanical persistence and release behavior after administration. For this reason, polymer selection should be evaluated alongside solvent compatibility, matrix-forming ability, and payload tolerance.
Biodegradable Polymers for Depot Formation
Biodegradable polymers are central to many in situ depot designs because they allow the matrix to persist long enough for long-acting release and then gradually break down over time. Polyesters such as PLGA, PLA, and PCL are especially important because their hydrophobicity, degradation rate, and processability can be tuned through composition and molecular characteristics. In solvent-triggered depot systems, these materials frequently serve as the structural backbone of the formed matrix. Their usefulness comes from the ability to support a relatively dense depot and to contribute to long-term release through degradation-assisted structural evolution. Their limitation is that they often introduce sensitivity to solvent selection, internal acidification, and morphology variability during precipitation. In practice, their performance depends as much on matrix formation history as on polymer identity itself.
Functional Polymers for Tunable Release Behavior
Functional polymers are valuable when the formulation needs more than passive matrix formation. By incorporating ionizable groups, associative segments, reactive sites, or specific interaction motifs, these polymers can influence release behavior more deliberately. Some materials are selected because they alter how quickly the precursor solidifies. Others are chosen because they improve payload retention, change hydration behavior, or support more responsive release under defined conditions. This is one of the reasons in situ depot systems are attractive: drug release can be shaped in advance through polymer function rather than only through bulk geometry. However, more functionality also means more coupled variables. A polymer that improves retention may reduce injectability, while a polymer that improves solidification may destabilize a sensitive payload. Functional design should therefore remain focused on the dominant release problem rather than adding complexity without clear benefit.
Stimuli-Responsive Polymers in In Situ Systems
Stimuli-responsive polymers are used when depot formation or later release should depend on temperature, pH, ions, or other environmental changes. These materials expand the design space because they allow the precursor to remain easier to handle before use and transform more selectively after administration. In some systems the stimulus primarily controls gelation or solidification; in others it also changes swelling, porosity, or the onset of drug release. Their usefulness is greatest when the trigger is reliable and when the response improves either administration or release control in a meaningful way. If the response is weak, inconsistent, or too sensitive to minor environmental variation, the resulting depot may become less predictable rather than more advanced. Robustness is therefore a critical part of materials assessment for this class.
Polymer Blends and Multi-Component Depot Matrices
Polymer blends are often used when one material alone cannot provide the desired balance of injectability, solidification behavior, depot persistence, and release control. A second polymer may be introduced to adjust viscosity, change precipitation rate, improve matrix hydration, strengthen the formed depot, or alter drug–matrix interaction. Multi-component systems can also help resolve the common trade-off between easy injection and stable post-injection structure. The challenge with blends is that they introduce additional compatibility questions. Polymer–polymer interaction, phase behavior during depot formation, and selective partitioning of the drug into one domain can all change performance. Blends are therefore most useful when they solve a clearly identified structural or release problem rather than being added only to broaden formulation options.
How Drug Release Happens in In Situ Forming Depot Systems?
Drug release from in situ depots is unusual because the earliest release behavior begins before the matrix is fully established. The precursor transforms while the payload is already present, which means release can be shaped by solvent exchange, precipitation, pore formation, and matrix densification almost immediately after administration. As a result, long-term release behavior is inseparable from depot formation history. A formulation that looks identical after full solidification may still release differently if it reached that structure through a different transformation path.
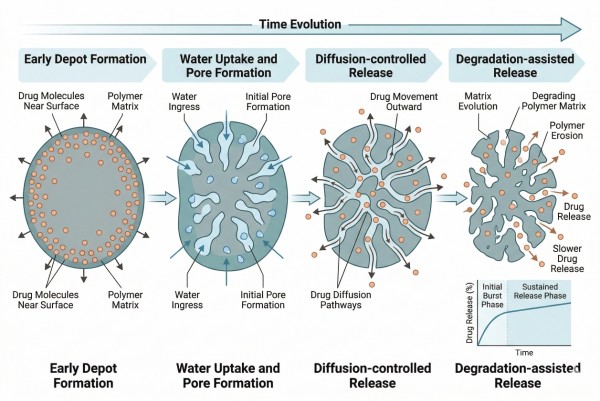 Fig. 2. Early formation, pore development, and matrix evolution together shape in situ depot release profiles (BOC Sciences Authorized).
Fig. 2. Early formation, pore development, and matrix evolution together shape in situ depot release profiles (BOC Sciences Authorized).
Initial Release During Depot Formation
Initial release in in situ depots often occurs during the same period in which the depot is forming. In solvent-based systems, outward solvent flux and inward water penetration can carry dissolved or weakly retained payload toward the external environment before the matrix fully solidifies. In gel-forming systems, slow network development can allow the drug to escape while the gel is still structurally immature. This is one of the main reasons burst release is a recurring challenge in in situ formulations. Reducing early release requires more than slowing diffusion after the depot has formed. It requires understanding what happens during formation itself. Polymer concentration, trigger rate, API partitioning, and local microstructure all affect how much drug is lost in this initial stage. Long-acting design is therefore strongest when early release is treated as a formation-stage phenomenon, not just as a later matrix property.
Diffusion Through the Newly Formed Matrix
Once a depot has formed sufficiently, diffusion becomes one of the main release pathways. The effective rate depends on matrix density, water content, pore connectivity, and the extent to which the payload is dissolved, dispersed, or associated with the polymer phase. In denser depots, diffusion may be significantly restricted; in more hydrated or porous depots, the payload may continue to move out relatively quickly even though the system appears macroscopically solid. For long-acting performance, diffusion should ideally be slowed by matrix structure rather than by accidental formulation artifacts. A depot that diffuses slowly only because it is inconsistently formed may not be reproducible batch to batch. A depot that diffuses slowly because its internal structure is intentionally designed is much more likely to be a useful formulation platform.
Swelling, Pore Formation, and Matrix Relaxation
Even in systems that form relatively dense depots, the matrix often changes over time. Water uptake can enlarge transport pathways, precipitated regions can reorganize, and internal pores or channels can emerge as solvent leaves and the depot equilibrates. These structural changes alter the effective diffusion environment and can shift the release profile from a more restricted early stage to a more permeable later stage. Matrix relaxation is especially important in systems that do not remain rigid after formation. A depot that initially seems suitable for long-acting release may become significantly more permissive as it swells or partially reorganizes. This is why swelling and structural evolution should be evaluated together with release data, not treated as secondary material observations.
Polymer Degradation and Long-Term Release Control
In biodegradable in situ depots, long-term release is often influenced by polymer degradation in addition to diffusion. As ester bonds cleave or the matrix loses integrity, the depot becomes more permeable and may release remaining payload through a combination of erosion, fragmentation, and accelerated internal transport. This can be useful for extending release beyond the timescale of simple diffusion alone, especially in polyester-based systems. At the same time, degradation introduces additional complexity because its rate depends on polymer composition, depot geometry, internal water distribution, and local accumulation of degradation products. For long-acting design, degradation should not simply be viewed as a desirable endpoint. It is a dynamic variable that must be matched to the intended release window and payload stability requirements.
How Functional Polymers Predetermine Release Behavior?
One of the strengths of in situ depots is that release behavior can be influenced in advance through functional polymer selection. Hydrophilic segments can change water ingress, charged groups can alter payload retention, and responsive chains can shift the timing of matrix transition or relaxation. This makes it possible to design systems in which release is not only a passive consequence of matrix formation, but an actively engineered property of the polymer network. The most effective release design uses these functions to shape the dominant mechanism rather than adding them for complexity alone. A functional polymer is useful when it solves a specific problem such as burst release, weak retention, or unstable matrix evolution. It is less useful when it introduces additional formulation variables without improving the actual long-acting release logic.
What Parameters Matter Most When Evaluating In Situ Forming Depots?
Evaluation is a central part of in situ depot development because these systems cannot be judged by release profile alone. A depot may release slowly but still fail if it cannot be injected reproducibly, if its matrix is mechanically weak, or if its transformation behavior changes across storage or manufacturing conditions. The most informative evaluation strategy therefore combines administration performance, matrix quality, payload release, and formulation readiness into a single development framework.
- Biocompatibility and biodegradation behavior: The polymer, trigger mechanism, and resulting degradation products should be compatible with the intended long-acting use. This includes evaluating whether the matrix persists appropriately and whether its structural breakdown remains consistent with the formulation goal.
- Syringeability and injection performance: The precursor must pass through a practical delivery device without excessive force, clogging, or premature solidification. This parameter is essential because in situ depots only work when the transformation happens at the right time and place.
- Gel strength and depot integrity: Once formed, the depot must maintain enough structural cohesion to remain localized and retain the payload. Weak gel or matrix strength often leads to poor long-acting performance even if transformation appears successful.
- Texture and structural consistency: Texture reflects how the depot forms and whether its internal and bulk properties are reproducible. A formulation that forms structurally inconsistent depots is unlikely to provide predictable release across batches or conditions.
- Release profile and long-acting performance: Release testing should capture both early formation-stage behavior and later sustained release. This is especially important in in situ systems because burst release and later matrix-controlled release may arise from different physical causes.
- Sterility and formulation readiness: Practical long-acting depot systems must be compatible with appropriate sterile handling, filling, and storage logic. Sterility is not just a manufacturing consideration; it is part of whether the formulation is realistically deployable.
How to Design In Situ Forming Depots for Different Pharmaceutical Needs?
In situ depots are not a one-format solution. Their usefulness depends on the payload class, the intended delivery objective, and the administration route. A system designed for a hydrophobic small molecule may be built around solvent-driven dense matrix formation, while a system for a sensitive biomacromolecule may need gentler triggering and a more hydrated or protective environment. The most effective design strategy is therefore to begin with pharmaceutical need rather than with a preferred depot chemistry.
Small Molecules in Long-Acting Depot Systems
Small molecules are often well suited to in situ depots when they can be retained within a forming polymer matrix and released gradually through diffusion and degradation. Hydrophobic small molecules are especially compatible with polyester-based systems because they can partition into the forming depot and remain associated with the matrix during the early release period. In these cases, solvent exchange and matrix density become the main controls on long-acting behavior. The main design challenge for small molecules is avoiding rapid loss during the formation stage. If the drug partitions too strongly into the surrounding fluid during phase inversion, burst release can dominate the profile. This is why small-molecule depot design must consider drug solubility, polymer affinity, solvent composition, and depot hardening rate together rather than treating the payload as a passive passenger in the matrix.
Peptides and Proteins in In Situ Formulations
Peptides and proteins present a different challenge because the depot must support not only sustained release, but also payload stability throughout fabrication, storage, and matrix formation. Solvent-rich systems can be difficult for sensitive biomolecules if the precursor environment or phase inversion process introduces interfacial or chemical stress. For this reason, in situ designs for biomacromolecules often require more cautious selection of solvent, polymer, and trigger mechanism than small-molecule systems do. A useful depot for peptides or proteins should form under conditions that preserve structure while still creating enough matrix integrity for sustained release. This may favor gentler gelling systems, buffered environments, or multi-component formulations that separate depot formation from direct payload exposure to harsh conditions. As a result, biomacromolecular in situ depot design often overlaps with broader strategies used in polymer carriers for protein and peptide drug delivery.
Multi-Drug and Combination Depot Designs
One advantage of in situ depots is that they can support multiple drug moieties in a single system when the formulation is designed carefully. This makes them attractive for long-acting regimens where synchronized or staged release of more than one component may be useful. However, combination loading introduces additional complexity because each drug may partition differently during depot formation and may influence the local microstructure in its own way. For these systems, the key design question is whether the depot can maintain adequate structural control while accommodating more than one release logic. A matrix that works well for one compound may destabilize another or separate them unevenly during phase inversion. Multi-drug depots are therefore best approached as a structure-distribution problem as much as a loading problem.
Local Versus Systemic Delivery Goals
In situ depots can be designed for either localized or systemic long-acting delivery, but these goals create different priorities. Localized systems may emphasize depot integrity, controlled spatial retention, and slower local diffusion. Systemic systems may place more emphasis on total release window, overall payload availability, and reproducibility of depot transformation. The same polymer system may not serve both objectives equally well. This means delivery goal should be defined early in development rather than inferred from release data later. A depot meant to remain localized can tolerate different structural properties than one meant to support prolonged systemic exposure. Matching matrix behavior to delivery objective is therefore one of the most important early design decisions.
What Are the Main Development Challenges in In Situ Forming Depots?
The main development risks in polymeric in situ forming depots do not usually come from one isolated variable. They arise because injection, depot formation, internal structure development, and early drug release all occur in a tightly coupled sequence. A formulation may appear acceptable in terms of polymer choice or nominal composition, yet still fail because the depot forms too quickly, too slowly, or too heterogeneously under practical administration conditions. For this reason, troubleshooting in situ depots requires understanding how precursor properties, phase transition behavior, payload partitioning, and matrix evolution interact during the first hours after injection.
- Burst release caused by rapid phase inversion: Burst release is one of the most characteristic failure modes in solvent-triggered in situ depots because phase inversion can begin before the payload is securely retained inside the forming matrix. When solvent leaves the formulation too rapidly and water enters too quickly, the polymer may precipitate into a porous, channel-rich structure that allows dissolved or weakly associated drug to escape at an early stage. In many cases, the problem is not simply that release is fast. The problem is that the depot forms in a way that exposes the payload before a stable diffusion barrier has been established.
- Uncontrolled depot morphology after injection: Depot morphology is often far less predictable in practice than it appears during conceptual formulation design. After injection, the precursor can spread, collapse, precipitate asymmetrically, or form nonuniform internal domains depending on solvent exchange rate, injection site conditions, and the balance between polymer mobility and local fluid penetration. As a result, the depot may not become the dense, coherent reservoir expected from the nominal composition, and release behavior may shift substantially from batch to batch.
- Payload instability in solvent-rich environments: In situ depot systems are often developed around solvents or precursor environments that are acceptable for polymer handling but not automatically favorable for all payloads. This becomes a major issue for peptides, proteins, and other sensitive molecules that may be exposed to interfacial stress, local concentration changes, altered polarity, or prolonged contact with reactive or dehydrating conditions during depot formation. Even when the final depot offers controlled release, the payload may already have lost part of its structural integrity during the formation process.
- Inconsistent injectability and syringe performance: A formulation cannot function as a useful in situ depot if it cannot be administered reproducibly. Inconsistent injectability usually arises when precursor viscosity, solvent composition, dwell time in the syringe, or early structuring behavior make flow sensitive to small operational changes. A system may pass a simple syringeability check under one condition and still fail during real handling because the precursor thickens over time, responds to temperature changes, or begins partial phase transition before complete administration.
- Batch variability and process sensitivity: In situ depots are often more process-sensitive than they appear because the final product is defined partly by what happens after injection, not only by what is filled into the container. Small changes in polymer molecular weight, solvent composition, precursor water content, mixing history, residual particulates, or filling conditions can alter phase inversion behavior and internal matrix formation. These shifts may not be obvious in routine appearance testing, yet they can produce significant differences in burst release, depot shape, or long-term release kinetics.
When In Situ Forming Depots Are the Right Long-Acting Platform?
The most important question at the end of in situ depot development is not whether the platform is scientifically interesting. It is whether the platform is the right match for the formulation objective. In situ depots offer clear advantages in certain situations, especially when an injectable precursor that transforms into a localized reservoir is more practical than administering a preformed implant or a discrete particulate system. At the same time, they are not automatically the best route for every long-acting project, especially when burst release, solvent stress, or post-injection variability become dominant risks. Good platform choice depends on aligning the depot's transformation logic with the real problem that needs to be solved. If the project needs easy administration, flexible depot geometry, and tunable post-injection structure, an in situ system may be highly attractive. If it needs maximum structural uniformity before administration or a release mechanism less sensitive to local formation conditions, another platform may be stronger. The decision should therefore be made by comparing platform behavior against product goals rather than by assuming that every injectable depot concept belongs in the same category.
When In Situ Depots Outperform Preformed Implants?
In situ depots usually outperform preformed implants when administration flexibility and minimally invasive delivery are major priorities. A flowable precursor can often be delivered more easily than a rigid or semi-rigid implant, especially when the desired depot does not need a fixed prefabricated geometry before injection. This can be particularly useful when the formulation must adapt to local site conditions or when a lower-viscosity precursor simplifies filling, handling, and injection relative to a solid implant format. They may also offer an advantage when the project benefits from a depot that forms directly in place rather than being inserted as a finished object. In those cases, the ability to create the matrix after administration can be a true functional benefit rather than just a formulation novelty. However, this advantage only holds when depot formation remains predictable. If the final structure is too variable or too sensitive to local conditions, the apparent convenience of injection can be outweighed by poor reproducibility in the actual long-acting profile.
When Microspheres or Hydrogels May Be Better Options?
Microspheres may be better when the project requires a more fully defined particulate system before administration, especially if release control depends on established particle engineering rather than post-injection phase transition. They can also be advantageous when the main challenge is to control matrix degradation in a known particulate architecture instead of managing solvent exchange and depot formation in vivo. If early burst from in situ formation remains difficult to suppress, a microsphere route may offer a more stable path to reproducible long-acting release. Hydrogels may be better when the payload needs a more water-rich, milder environment or when the desired long-acting behavior depends more on swelling, mesh-size control, and soft matrix residence than on dense polymer precipitation. This is especially relevant for certain biomacromolecules and sensitive payloads that do not tolerate solvent-rich precursor conditions well. In short, in situ depots are not the "next step up" from every other platform. They are best viewed as one route among several, with clear strengths but also distinct boundaries.
How to Judge Whether an In Situ Depot Is Truly Long-Acting?
An in situ depot is truly long-acting only when its post-injection matrix meaningfully controls payload release over the intended timeframe. A precursor that forms a visible depot is not automatically successful. A system that reduces dosing convenience concerns but still loses a large fraction of drug during the first phase inversion window is not genuinely long-acting in formulation terms. The real test is whether the depot can retain the payload after formation and continue releasing it in a controlled way instead of simply delaying immediate escape by a short interval. This judgment should be based on the full release logic of the system: early burst, matrix development, medium-term retention, long-term structural evolution, and the relationship between depot persistence and payload stability. If most of the formulation performance depends on the first few hours and very little on the later depot structure, then the system may function as an injectable precipitation event rather than a meaningful long-acting platform. A true in situ long-acting depot should show that the matrix formed after injection remains pharmacotechnically relevant across the full intended release window.
A Practical Framework for Platform Selection
A practical platform decision can be built around four questions. First, does the project truly benefit from a precursor that forms a depot after injection, or would a preformed system be more reproducible? Second, can the payload tolerate the precursor environment and the transformation pathway required to create the depot? Third, is burst release controllable enough that the formed matrix still governs a meaningful fraction of the release profile? Fourth, does the expected depot morphology remain sufficiently consistent under realistic administration conditions? If the answer to these questions is largely yes, then an in situ depot is likely worth pursuing because its administration flexibility and tunable formation behavior may provide real long-acting value. If the answer is largely no, then the route may still be scientifically interesting but not developmentally efficient. In that case, the right decision is often to move earlier toward microspheres, hydrogels, implants, or another platform instead of continuing to optimize a system whose core limitations are already visible. That is the real purpose of platform selection: not to defend a concept, but to choose the route most likely to produce a robust long-acting formulation.
Services
Advanced In Situ Depot Design and Formulation Services
BOC Sciences provides integrated support for polymeric in situ forming depot development, from polymer and solvent selection to depot fabrication design, release evaluation, and formulation optimization. Our technical focus is on the coupled variables that determine whether an in situ precursor becomes a useful long-acting product, including injectability, transformation kinetics, depot integrity, internal morphology, payload compatibility, and release reproducibility. By connecting polymer chemistry with formation-stage behavior, we help teams design depot systems that are better aligned with both scientific and development goals.

Polymer and Solvent Selection
- Screening of biodegradable and functional polymers for in situ depot formation.
- Selection of solvent systems and trigger mechanisms for controlled transformation.
- Matching precursor composition to injectability and depot persistence goals.
- Support for long-acting depot materials across multiple routes.

Depot Fabrication Strategy Design
- Optimization of phase inversion, gelling, and stimulus-responsive formation routes.
- Development of solution- and suspension-based depot formulations.
- Control of depot formation rate and internal matrix structure.
- Design of systems for prolonged and adaptable release behavior.

Release and Performance Evaluation
- Assessment of syringeability, gel strength, texture, and depot consistency.
- Evaluation of burst release, matrix evolution, and long-term release behavior.
- Correlation of material properties with transformation-stage performance.
- Support for rational troubleshooting of in situ depot systems.

Formulation Optimization Support
- Reduction of burst release and morphology-related variability.
- Improvement of precursor stability and depot reproducibility.
- Adjustment of matrix composition for different payload classes.
- Integrated support from early screening to long-acting depot design.
Need Support with Polymeric In Situ Depot Development?
Discuss polymer selection, trigger strategy, depot formation, and long-acting release design with a team focused on advanced polymer-based drug delivery systems.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What is a polymeric in situ forming depot?
A polymeric in situ forming depot is a formulation injected as a solution or suspension that forms a gel or solid depot after administration. The formed depot then releases the payload over an extended period through diffusion, matrix evolution, degradation, or a combination of these processes.
-
Why are in situ forming depots useful for long-acting drug delivery?
These systems are useful because they combine minimally invasive administration with depot-based long-acting release. Their precursor formulations are often easier to inject than preformed implants, while the resulting depot can provide prolonged retention and controllable release depending on polymer type, trigger mechanism, and internal structure.
-
What controls drug release in polymeric in situ depots?
Drug release is controlled by several coupled factors, including depot formation rate, early solvent exchange, internal porosity, matrix diffusion, swelling, and polymer degradation. Functional polymers can also be selected to influence how quickly the depot forms and how the release profile evolves over time.
-
What are the most important evaluation parameters for in situ depot systems?
Key parameters include biocompatibility, biodegradation, syringeability, gel strength, texture, release profile, and sterility. These factors help determine whether the formulation can be administered practically, form a stable depot after injection, and support a consistent long-acting release profile.
-
When are in situ forming depots better than preformed implants or microspheres?
In situ depots may be better when easy injection, simpler administration, or adaptable depot formation is more important than using a preformed solid dosage form. However, they are not always superior. If burst release, solvent sensitivity, or morphology variability cannot be controlled, other long-acting platforms may be more suitable.

 Fig. 1. In situ depots form after administration through triggered matrix solidification or gelation (BOC Sciences Authorized).
Fig. 1. In situ depots form after administration through triggered matrix solidification or gelation (BOC Sciences Authorized). Fig. 2. Early formation, pore development, and matrix evolution together shape in situ depot release profiles (BOC Sciences Authorized).
Fig. 2. Early formation, pore development, and matrix evolution together shape in situ depot release profiles (BOC Sciences Authorized).