PLGA for Long-Acting Drug Delivery: Selection, Release Mechanisms, and Formulation Design
Poly(lactic-co-glycolic acid) (PLGA) is one of the most practical and extensively used biodegradable polymers for long-acting drug delivery because it gives formulators a rare combination of tunable degradation, broad processing flexibility, and compatibility with multiple sustained-release platforms. By adjusting copolymer ratio, molecular weight, end-group chemistry, and matrix structure, PLGA systems can be designed to regulate water uptake, erosion, pore formation, and payload diffusion across weeks or months. This makes PLGA a central material choice for depot formulations, microspheres, nanoparticles, implants, and in situ forming systems intended to improve exposure control, reduce dosing frequency, and support more stable formulation performance for small molecules, peptides, proteins, and selected macromolecular payloads.
Resources
PLGA for Long-Acting Drug Delivery
In long-acting formulation development, PLGA is often the material that turns a conceptual sustained-release strategy into an experimentally workable dosage form. Unlike inert matrix excipients that mainly provide bulk or viscosity, PLGA functions as an active structural framework whose composition, chain length, end-group type, and morphology directly shape drug retention, release rate, degradation pattern, and processing window. Because PLGA can be fabricated into microspheres, implants, films, depots, and polymer nanoparticles, it serves as both a foundational polymer and a practical bridge between material screening and delivery-platform selection.
What PLGA Is and Why It Matters?
PLGA is a synthetic aliphatic copolyester prepared from lactic acid- and glycolic acid-derived monomer units. Its value in long-acting delivery comes from hydrolytically cleavable ester bonds distributed throughout the backbone. As water enters the polymer matrix, these bonds gradually break, reducing chain length, increasing matrix porosity, and eventually converting the solid structure into smaller oligomers and monomers. Because the lactic acid component is more hydrophobic and sterically hindered than the glycolic acid component, the relative ratio of the two units strongly influences hydration, chain mobility, and degradation speed. This compositional tunability is a major reason PLGA remains a preferred material for formulation scientists who need predictable ways to modulate release behavior without completely changing platform type.
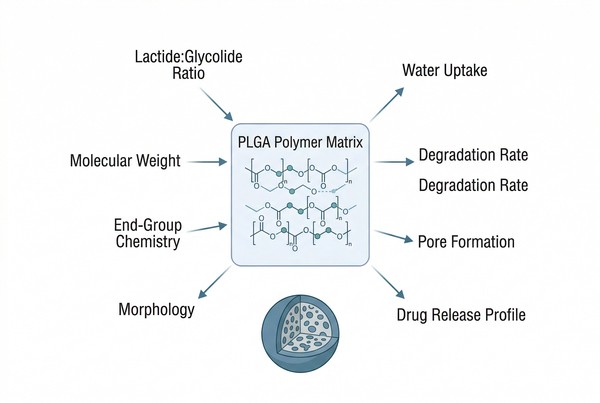 Fig. 1. PLGA composition, molecular weight, and end groups jointly influence degradation and release behavior (BOC Sciences Authorized).
Fig. 1. PLGA composition, molecular weight, and end groups jointly influence degradation and release behavior (BOC Sciences Authorized).
Why PLGA Is Widely Used in Sustained Release Formulations?
PLGA is widely used because it combines several traits that are difficult to obtain simultaneously in one polymer family. First, it offers adjustable degradation kinetics through lactide:glycolide ratio, molecular weight, and end-group selection. Second, it can be processed through common formulation routes such as solvent evaporation, phase separation, spray drying, and in situ solvent exchange. Third, it is versatile enough to accommodate both diffusion-dominant and degradation-dominant release profiles depending on matrix design. Fourth, PLGA can be paired with related materials such as polylactic acid (PLA), poly(caprolactone) (PCL), PEG derivatives, or surface modifiers when a pure PLGA system does not fully meet formulation needs. These features make it useful not only as a polymer choice, but also as a formulation-design platform.
How PLGA Differs from Other Biodegradable Polymers?
Compared with slower-degrading hydrophobic polyesters such as PCL, PLGA generally provides faster water penetration and a more practical route to intermediate release windows. Compared with highly crystalline homopolymers, PLGA often offers lower crystallinity and greater compositional flexibility, which supports finer control of release behavior. Compared with highly hydrophilic polymers or hydrogel-forming matrices, PLGA usually provides stronger barrier properties for water-soluble payloads and a more compact depot structure. It also differs from injectable polymer hydrogels in that drug release is more often linked to matrix degradation, pore evolution, and hydrophobic partitioning rather than solely to swelling and mesh-size control.
Why PLGA Is Often the Starting Material?
PLGA is frequently the starting point for long-acting development because it helps teams answer several early formulation questions before committing to a more specialized polymer strategy:
- Can the payload be retained inside a degrading polyester matrix?
- Is the target release window closer to diffusion-controlled release or erosion-assisted release?
- Does the drug tolerate solvent-based processing?
- Is a particulate depot more suitable than a hydrogel, a polymer-based gene delivery platform, or a polymer–drug conjugate design?
Even when PLGA is not the final material, screening with PLGA can reveal whether the project is fundamentally limited by polymer hydrophobicity, acidic degradation products, burst release, or drug–matrix incompatibility. In that sense, PLGA functions as both a candidate material and a diagnostic material.
Key PLGA Properties That Affect Formulation Performance
PLGA does not behave like a single fixed material. Formulation performance changes substantially when the copolymer ratio, molecular weight, end-group chemistry, and solid-state morphology change. Those variables alter the rate and pattern of water ingress, internal acid accumulation, chain mobility, mechanical integrity, and drug diffusion pathways. For long-acting systems, the practical challenge is not only choosing PLGA, but selecting the specific PLGA grade whose material profile matches the intended payload, process, and release window.
- Lactide:glycolide ratio: The monomer ratio strongly affects hydrophobicity, hydration behavior, and degradation pace. Increasing lactide content often slows water uptake and can lengthen matrix persistence, while increasing glycolide content may speed hydration and promote earlier matrix opening. This ratio is therefore a primary lever for shifting the balance between initial diffusion and later erosion-assisted release.
- Molecular weight: Molecular weight influences chain entanglement, viscosity, and the time required for hydrolysis to reduce the matrix into more mobile fragments. Higher-molecular-weight PLGA usually supports longer structural integrity and can extend release duration, whereas lower-molecular-weight grades often hydrate and degrade more quickly. It also affects processing by changing solution rheology, droplet formation, and particle hardening behavior.
- End-group chemistry: Acid-terminated and ester-capped PLGA grades do not hydrate or degrade in the same way. Acid end groups generally increase hydrophilicity and can accelerate water penetration and autocatalytic degradation, while end-capped grades often slow early hydration. This difference becomes important when trying to reduce burst release, control lag time, or manage interactions with ionizable drugs.
- Glass transition and morphology: The glass transition temperature and matrix morphology determine how rigid or mobile the depot remains during storage and release. Dense, glassy structures generally slow diffusion more effectively, while porous or partially relaxed matrices can accelerate water ingress and pore connectivity. Even with the same PLGA chemistry, morphology can create large differences in release behavior and batch consistency.
| PLGA Attribute | What It Changes | Typical Release Impact | Key Development Risk |
|---|
| Lactide:glycolide ratio | Hydrophobicity, hydration, degradation pace | Shifts release from slower matrix opening to faster erosion-assisted release | Choosing a ratio that accelerates water uptake too early |
| Molecular weight | Chain entanglement, viscosity, matrix integrity | Higher molecular weight usually supports longer release duration | Excessive viscosity and difficult particle formation |
| End-group chemistry | Surface polarity, water uptake, autocatalysis | Acid end groups can speed hydration and degradation | Greater burst release or faster internal acid buildup |
| Morphology and porosity | Diffusion path length, pore connectivity | More porous matrices often release faster at early time points | Uncontrolled morphology after solvent removal |
Main PLGA Delivery Platforms for Long-Acting Formulations
PLGA is not limited to one dosage-form concept. The same polymer family can support multiple long-acting platforms, each with a different dominant release mechanism, processing route, and optimization logic. Because platform architecture determines whether release is controlled mainly by diffusion, matrix degradation, interfacial area, or solvent exchange, the correct development question is not only "Which PLGA grade should I use?" but also "Which PLGA-based platform best fits the payload and the intended release profile?"
PLGA Microspheres for Sustained Drug Release
PLGA microspheres are among the most established depot architectures for prolonged release because they offer a large design space for particle size, polymer grade, internal porosity, and drug distribution. Drug can be dissolved, dispersed, or emulsified into the polymer phase before hardening. Release from microspheres often reflects an interplay of surface-associated diffusion, pore formation, polymer hydration, and bulk degradation. They are especially useful when the goal is to create a free-flowing particulate depot rather than a preformed implant. Microspheres are also a frequent entry point for projects seeking to reduce dosing frequency without building a device-like dosage form.
PLGA Nanoparticles for Controlled Delivery
PLGA nanoparticles provide a smaller interfacial structure and are typically used when colloidal dispersion, local transport, or intracellular delivery considerations matter. Their high surface area can improve interaction with biological environments, but it also increases the chance of faster release if the payload partitions readily into aqueous media. Compared with larger depots, nanoparticles usually require stricter control over surface charge, stabilizer choice, and storage stability. For users comparing platform options, it is often helpful to review polymer nanoparticles for drug delivery and polymer nanoparticles for gene delivery as complementary platform strategies rather than assuming all PLGA systems behave like microspheres.
PLGA Implants for Long-Duration Release
PLGA implants and compact solid matrices provide a lower-surface-area depot than particulate systems and can support prolonged release when matrix integrity is maintained for extended periods. Because implant geometry, thickness, and drug distribution can be tightly controlled, implants are useful when more gradual erosion or lower burst release is desired. Their design logic often centers on diffusion path length, matrix density, and internal acid accumulation rather than simply on particle size. PLGA films, rods, and compressed matrices fall into this broader category when the formulation goal is a structurally persistent depot.
In Situ Forming PLGA Depot Systems
In situ forming PLGA depots are generated after administration through solvent exchange or phase inversion. The liquid formulation becomes a semi-solid or solid depot as solvent leaves and aqueous fluid enters. This approach can simplify administration compared with preformed implants, but it introduces its own complexity: solvent choice, phase separation rate, drug partitioning, and depot densification all strongly affect burst release and subsequent matrix behavior. In these systems, the polymer grade is only one part of the problem; solvent kinetics and depot microstructure are equally important.
Need a PLGA Material Strategy for Long-Acting Formulation Development?
From copolymer ratio and molecular weight selection to microsphere, implant, and depot design, we support PLGA screening, synthesis, and formulation optimization for sustained-release projects.
How Drug Release Happens in PLGA-Based Formulations?
Drug release from PLGA systems is rarely governed by a single event. Instead, it usually emerges from a sequence of overlapping processes: initial desorption or diffusion from near-surface regions, water penetration into the matrix, pore generation, chain cleavage, internal acid accumulation, and eventual erosion-assisted drug escape. Understanding this sequence is essential because two formulations can contain the same PLGA grade and the same drug, yet show very different release profiles if their internal structure, loading state, or processing history differ.
Initial Drug Diffusion from PLGA Matrices
Early release is commonly influenced by drug located close to the particle or depot surface, or by drug dissolved in water-accessible regions that provide short diffusion paths. This is the origin of the familiar "initial burst" seen in many PLGA systems. Burst release becomes more pronounced when the drug is poorly retained in the polymer phase, when particles are highly porous, or when the manufacturing route leaves payload enriched near the exterior. For highly water-soluble compounds, even a relatively dense PLGA matrix may not fully suppress early escape if the drug partitions quickly into the external medium.
Water Penetration and Pore Formation in PLGA
Once exposed to aqueous media, PLGA begins to absorb water. Water uptake does not only trigger hydrolysis; it also changes the internal transport environment. Microvoids can form, existing pores can connect, and previously isolated drug domains may become exposed to liquid channels. This matrix opening phase is critical because it often determines when the system transitions from a burst-dominated regime to a slower period of more distributed release. The pace of pore generation depends on polymer composition, particle density, process-induced morphology, and the presence of soluble additives or dispersed drug domains that leave behind channels as they dissolve.
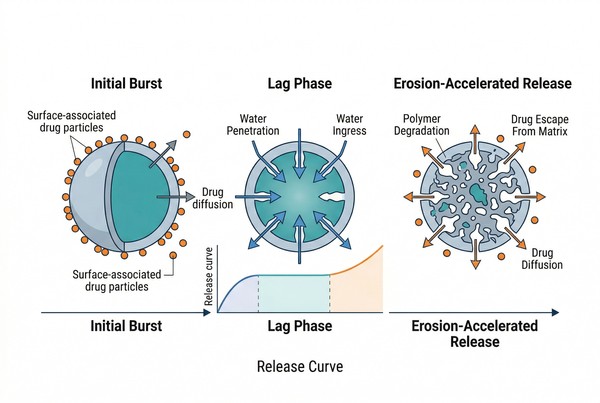 Fig. 2. A typical PLGA release profile can include burst, lag, and erosion-accelerated release phases (BOC Sciences Authorized).
Fig. 2. A typical PLGA release profile can include burst, lag, and erosion-accelerated release phases (BOC Sciences Authorized).
Polymer Degradation and Bulk Erosion Behavior
PLGA usually undergoes bulk erosion rather than pure surface erosion. Water penetrates the matrix faster than the polymer mass is lost from the exterior, so hydrolysis occurs throughout the bulk. As the molecular weight drops, the matrix loses strength and becomes more permeable. In confined structures, acidic oligomers may accumulate internally and accelerate further degradation through autocatalysis. This is a defining feature of many PLGA depots: degradation can become spatially heterogeneous, causing one region of the matrix to collapse or open faster than another. Release therefore reflects not only average degradation rate, but also how degradation products redistribute inside the depot.
Why PLGA Often Shows Multi-Phase Release?
Many PLGA formulations show a multi-phase profile with an early burst, a slower intermediate region or lag phase, and a later increase as erosion becomes significant. The exact shape depends on how much payload is surface accessible, how quickly the matrix hydrates, and how long it takes for internal connectivity to develop. A long lag phase may indicate a dense matrix with slow pore interconnection, while a rapid second phase may reflect aggressive internal degradation or high water uptake. For development purposes, recognizing which phase is problematic is more useful than asking whether release is simply "fast" or "slow."
How Drug Properties Influence PLGA Release Profiles?
Drug release in PLGA is always co-determined by the payload. Water solubility, crystal state, ionization, molecular size, and affinity for the polymer phase all matter. A hydrophobic small molecule with good matrix compatibility may remain dispersed in PLGA and release mainly as polymer mobility increases or erosion progresses. A hydrophilic peptide may localize in aqueous microdomains, creating faster escape pathways and greater sensitivity to pore formation. Charged or unstable payloads can be affected by internal acid generation, moisture exposure, or interface stress created during emulsification. That is why release control in PLGA is fundamentally a drug–polymer design problem, not a polymer-only problem.
Applications of PLGA in Long-Acting Drug Delivery
PLGA is used across several payload classes because its matrix can be tuned to provide both structural retention and time-dependent opening. Yet the reasons for selecting PLGA differ by application. For small molecules, PLGA often solves duration and solubility-distribution problems. For peptides and proteins, it is frequently used to build a protective depot but requires extra attention to internal microenvironment and processing stress. For macromolecular or charged payloads, PLGA may act as a structural carrier, but often needs formulation modification or combination design to achieve acceptable loading and release control.
PLGA for Small Molecule Sustained Release
Small molecules are often the most direct fit for PLGA, especially when the project requires prolonged exposure from a depot platform. Poorly water-soluble compounds may be retained effectively inside the hydrophobic matrix, while moderately soluble compounds may require tighter control over particle morphology and surface enrichment. Small-molecule projects often use PLGA to shift from rapid systemic exposure to controlled matrix-mediated release. In these cases, the central questions are drug–polymer compatibility, crystalline versus amorphous dispersion within the matrix, and whether the release window should depend more on diffusion or degradation. Users working with hydrophobic payloads may also benefit from comparing PLGA depots with broader strategies for polymers for small molecule drug delivery and polymeric micelles.
PLGA for Peptide and Protein Delivery
PLGA is attractive for peptide and protein depots because it can create sustained-release reservoirs for molecules that would otherwise clear rapidly. However, this application is technically more demanding. Proteins and peptides may face interfacial stress during emulsion formation, moisture-induced instability during storage, and chemical stress from the evolving acidic microenvironment generated by PLGA degradation. Release can also become heterogeneous if the biomolecule partitions into water-rich internal pockets. For these payloads, formulation success often depends on stabilizers, solid-state control, buffer engineering, and careful assessment of how processing affects bioactive structure. The broader design logic overlaps with challenges discussed in polymer carriers for protein and peptide drug delivery.
PLGA for Nucleic Acid Carrier Design
PLGA can contribute to nucleic acid delivery, but usually not as a simple standalone matrix in the same way it is used for hydrophobic small molecules. Nucleic acids are hydrophilic, charged, and structurally sensitive, which makes direct entrapment into a hydrophobic polyester more complex. In practice, PLGA may be used as a structural shell, a composite carrier component, or part of a hybrid particulate architecture where surface modification, complexation layers, or auxiliary cationic materials provide the primary nucleic acid association. For teams considering whether PLGA is suitable for a charged biomacromolecule project, it is often helpful to compare polyester-based systems against the broader framework of polymers for nucleic acid delivery, polymer systems for mRNA delivery, and polymer-based gene delivery platforms.
PLGA for Injectable Depot Formulations
One of the most valuable application spaces for PLGA is the injectable depot. PLGA enables depot structures that can be formed from discrete microspheres, dense implants, or in situ precipitating solutions. The material is especially useful when a project needs a compact biodegradable reservoir rather than a highly swollen hydrogel. Injectable depots built from PLGA can be tuned through particle size, density, polymer grade, and solvent system to emphasize early suppression of burst, delayed opening, or progressive erosion. Selection within this category depends on whether the desired profile calls for a free-flowing suspension, a compact depot, or a less invasive phase-transitioning system.
How Application Needs Change PLGA Design Choices?
The same PLGA grade is rarely ideal for every application class. Small molecules may benefit from stronger hydrophobic retention, peptides may need gentler processing and better microenvironment control, and hybrid macromolecular carriers may require functional layers beyond the PLGA matrix itself. Application context changes which variables matter most: in one project molecular weight may dominate, while in another end-group chemistry or solvent selection becomes the deciding factor. That is why application-driven PLGA design should begin with payload behavior and release objective before polymer grade is finalized.
| Application Type | Why PLGA Is Considered | Main Formulation Focus | Common Limitation |
|---|
| Small molecules | Good depot compatibility and release tunability | Drug–polymer affinity, morphology, release window | Burst release from surface-enriched drug |
| Peptides and proteins | Supports prolonged release from biodegradable matrices | Stability, internal pH control, water-domain management | Payload degradation during processing or storage |
| Nucleic acids or sensitive macromolecules | Useful in hybrid or protected carrier designs | Association strategy, surface modification, composite structure | Low direct loading into hydrophobic PLGA matrices |
| Injectable depots | Flexible platform options from particles to in situ systems | Depot formation, injectability, long-term release control | Process-dependent variability and solvent effects |
Common PLGA Formulation Challenges and How to Evaluate Them
PLGA is versatile, but it also introduces recurring development problems that can undermine release predictability if they are not diagnosed correctly. Most failures trace back to a small number of material-process interactions: excessive surface drug, rapid hydration, unstable payload state, poor polymer compatibility, or morphology drift during fabrication. The goal is not only to observe the release profile, but to understand why the profile looks the way it does. Mechanistic evaluation prevents the common mistake of changing multiple variables at once without knowing which one actually controls the problem.
Why Burst Release Is Common in PLGA Formulations?
Burst release is common when a substantial fraction of the drug sits near the surface, is weakly retained in the matrix, or becomes exposed through rapid solvent loss and pore formation. High surface area, inadequate polymer density, and water-soluble payload domains can all accelerate early release. In microspheres, burst can also arise from process conditions that leave drug at the oil-water interface during particle hardening. The right response depends on the root cause: a polymer-grade change may help if hydration is too fast, but if the drug is simply segregating to the surface, process redesign is often more effective than switching to another PLGA ratio.
How Acidic Degradation Affects Payload Stability?
As PLGA hydrolyzes, acidic oligomers and monomers accumulate inside the matrix. In dense or poorly diffusing systems, this can lower the local microenvironment pH well before the external medium reflects the change. Such acidification can accelerate degradation of acid-sensitive payloads, alter solubility state, or change ionic interactions inside the depot. The issue is especially important for peptides and proteins, but it can also affect small molecules that undergo hydrolysis, rearrangement, or phase changes under acidic conditions. Evaluating this problem requires more than cumulative release data; it also requires chemical stability measurements and, where relevant, assessment of degradation-related matrix changes.
Why Some Drugs Show Low Loading in PLGA?
Low loading often reflects poor thermodynamic compatibility between the payload and the polymer phase. A hydrophilic drug may prefer the external aqueous phase during emulsification, while a crystalline hydrophobic drug may phase separate rather than remain evenly dispersed. Low loading can also be caused by high diffusion loss during solvent removal, inadequate viscosity in the polymer phase, or an unfavorable drug physical state before encapsulation. Solving low loading therefore requires checking not only formulation ratios, but also drug solubility in each process phase, polymer-phase affinity, and the timing of solidification.
How Processing Conditions Affect Particle Morphology?
Processing determines whether a PLGA system becomes dense, porous, collapsed, hollow, smooth, rough, or phase-separated. Emulsification intensity, solvent volatility, polymer concentration, external phase composition, drying rate, and hardening kinetics all influence morphology. Because morphology directly controls diffusion path length and pore connectivity, it is often the hidden variable behind unexplained release changes between nominally similar batches. Morphology should therefore be treated as a core formulation attribute rather than as a secondary cosmetic feature.
Why Batch Variability Occurs in PLGA Systems?
Batch variability usually arises when critical material and process attributes are not tightly connected to release outcomes. Small shifts in polymer molecular weight distribution, residual solvent, emulsification shear, external phase osmolarity, or drying history can produce measurable differences in particle size, porosity, and drug distribution. That is why early PLGA development benefits from a structured evaluation plan that links polymer properties, processing conditions, morphology, and release data instead of treating them as separate workstreams. When teams need broader platform comparison, choosing the right drug delivery system can also clarify whether a polymeric depot remains the best fit.
How to Select and Optimize PLGA for a Specific Release Goal?
Effective PLGA selection is a staged decision process rather than a one-time grade choice. The best formulation programs first define the target release window, dosage-form logic, and payload risks, then choose a short list of polymer grades that bracket the likely design space. Optimization proceeds by linking each material attribute to a measurable outcome: burst release, lag time, cumulative release, payload stability, morphology, and process reproducibility. This approach is more efficient than broad trial-and-error because it turns PLGA development into a controlled structure–property–performance exercise.
How to Match PLGA Grade to Release Duration?
Start with the intended duration and the dominant release mechanism you want to emphasize. If the target profile requires longer matrix integrity, a higher-molecular-weight or more hydrophobic grade may be a logical starting point. If earlier matrix opening is acceptable or desired, a faster-hydrating composition may be better. End-group choice should then be used to fine-tune hydration and degradation pace rather than as an afterthought. Selection is strongest when those variables are screened with release data, morphology data, and degradation data together.
How to Choose Between Microspheres and Depots?
Platform choice should reflect how the depot will be made, delivered, and controlled. Microspheres are useful when a suspension-based particulate depot is desirable and particle engineering is central to optimization. Dense implants or compact depots make sense when a lower-surface-area structure is beneficial. In situ systems may be attractive when a preformed implant is impractical, but they require closer attention to solvent exchange and depot formation kinetics. Choosing among these options early can prevent wasted effort screening PLGA grades for a platform that is fundamentally misaligned with the release goal.
How to Adjust Particle Size and Porosity?
Particle size and porosity are two of the most direct release-control levers in PLGA particulate systems. Smaller particles increase surface area and often accelerate early release. More porous structures shorten effective diffusion paths and allow water to penetrate more rapidly. Denser particles can reduce burst but may create longer lag phases or increase sensitivity to internal acid accumulation. Optimization therefore involves finding a morphology that balances early retention, medium-term release continuity, and acceptable manufacturability rather than maximizing any single attribute in isolation.
How to Evaluate Release and Formulation Stability?
Meaningful evaluation goes beyond a single cumulative release curve. A useful PLGA assessment package includes release sampling over appropriate early and late time points, particle size or depot morphology, residual solvent where relevant, polymer molecular weight change during aging or release, and payload stability inside the matrix. For biomacromolecules, structural integrity and aggregation should be checked alongside release. For small molecules, crystalline state and chemical stability often matter just as much as total loading. A formulation is only truly optimized when release behavior and payload quality remain aligned throughout the intended use period.
When PLGA Is Not the Best Polymer Choice?
PLGA is not always the right answer. If the payload is highly acid-sensitive, poorly retained in hydrophobic matrices, or better controlled by swelling rather than degradation, another material family may be more suitable. Water-rich matrices may outperform PLGA for some proteins, and functional cationic polymers may be better for nucleic acid complexation. In other projects, hybrid systems that combine PLGA with buffering, surface modification, or secondary polymers may solve the problem more effectively than a pure PLGA matrix. Good material strategy recognizes when PLGA offers true control and when it creates avoidable complexity.
Services
PLGA Material and Formulation Support Services
BOC Sciences provides integrated support for PLGA material selection, formulation design, and analytical evaluation so development decisions are made from a connected structure–property–performance perspective. Our service approach focuses on the practical variables that determine whether a PLGA system becomes a reliable long-acting formulation, including polymer grade choice, payload compatibility, process optimization, release control, reproducible material characterization, and scale-up planning for advanced biodegradable polymers.

Custom PLGA & Copolymer Design
- Selection and customization of lactide:glycolide ratios.
- Control of molecular weight and end-group chemistry.
- Development of PLGA-related polyester and copolymer variants.
- Material matching for depot, implant, and particle systems.

PLGA Formulation Development
- Microsphere, nanoparticle, and in situ depot formulation support.
- Optimization of drug loading, morphology, and burst control.
- Screening of solvent systems, stabilizers, and process variables.
- Design of formulations for small molecules and biomacromolecules.

Analytical Characterization for PLGA Systems
- Molecular weight, composition, and end-group assessment.
- Particle size, morphology, and porosity characterization.
- Drug loading, encapsulation efficiency, and residual solvent testing.
- Release-profile and degradation-behavior evaluation.

Scale-Up and Supply Support
- Process transfer from lab-scale screening to larger batches.
- Improvement of batch-to-batch reproducibility in PLGA systems.
- Supply of biodegradable polymers and related materials.
- Integrated support for material, process, and formulation continuity.
Ready to Evaluate PLGA for Your Long-Acting Formulation?
Discuss polymer selection, depot design, release optimization, and formulation troubleshooting with a team focused on biodegradable polymer systems for drug delivery.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What makes PLGA suitable for long-acting drug delivery?
PLGA is suitable because its degradation rate, matrix density, and platform format can all be tuned through copolymer ratio, molecular weight, and end-group chemistry. This allows formulators to build microspheres, implants, depots, or nanoparticles with more controlled release behavior than a fixed excipient matrix usually provides.
-
Does a higher PLGA molecular weight always mean slower release?
No. Higher molecular weight often helps maintain matrix integrity longer, but release still depends on porosity, particle size, drug distribution, end-group type, and payload compatibility. A highly porous or surface-enriched system can still release quickly even when a relatively high-molecular-weight PLGA grade is used.
-
Why do PLGA formulations often show burst release?
Burst release usually appears when drug is concentrated near the surface, retained weakly in the matrix, or exposed by rapid water ingress and pore formation. It is therefore often a combined material-and-process issue rather than a simple polymer problem, especially in microspheres and in situ depot systems.
-
Is PLGA equally suitable for small molecules and proteins?
Not equally. PLGA is often more straightforward for small molecules because matrix retention is easier to control. Proteins and peptides can also be formulated with PLGA, but they typically require tighter control over interface stress, moisture exposure, internal acidification, and stabilization within the degrading polyester environment.
-
How should PLGA grades be screened in early development?
Early screening usually works best when a focused set of PLGA grades is chosen to bracket copolymer ratio, molecular weight, and end-group chemistry. Each candidate should then be compared using loading, morphology, release profile, degradation behavior, and payload stability, rather than by cumulative release alone.

 Fig. 1. PLGA composition, molecular weight, and end groups jointly influence degradation and release behavior (BOC Sciences Authorized).
Fig. 1. PLGA composition, molecular weight, and end groups jointly influence degradation and release behavior (BOC Sciences Authorized). Fig. 2. A typical PLGA release profile can include burst, lag, and erosion-accelerated release phases (BOC Sciences Authorized).
Fig. 2. A typical PLGA release profile can include burst, lag, and erosion-accelerated release phases (BOC Sciences Authorized).