Implantable Polymeric Wafers for Long-Acting Drug Delivery: Structural Design and Release Strategy
Polymer-based wafers represent a distinctive class of implantable drug delivery systems because their performance is strongly defined by geometry, local placement, and matrix-controlled release. Unlike particulate systems that disperse after administration or larger device-like implants that rely on bulk structure, polymeric wafers use thin, planar, or layered architectures to provide localized coverage and sustained drug exposure from a defined surface. This makes them especially relevant when the development goal is long-acting local delivery supported by controllable diffusion distance, manageable structural persistence, and tunable release behavior. In practical formulation work, the value of a wafer lies not only in what polymer it contains, but in how that polymer behaves inside a thin implantable structure over time.
Resources
What Are Implantable Polymeric Wafers?
Implantable polymeric wafers are thin or moderately thick solid delivery systems designed for localized placement and sustained drug release from a defined geometry. Their most important feature is not simply that they are implantable, but that they operate as structure-defined dosage forms in which surface area, thickness, internal organization, and matrix evolution directly influence performance. This makes wafers fundamentally different from general implant bodies, even when both rely on similar material classes.
Core Definition of an Implantable Polymeric Wafer
A polymeric wafer is typically a planar or near-planar implantable system in which the drug is incorporated into, layered within, or associated with a polymer matrix that remains localized after placement. The wafer format is especially useful when a project needs a defined contact area and a predictable release pathway from a relatively thin solid structure. In contrast to more volumetric implants, the wafer is often chosen because its geometry can be tuned as part of the release strategy rather than treated as a secondary physical detail.
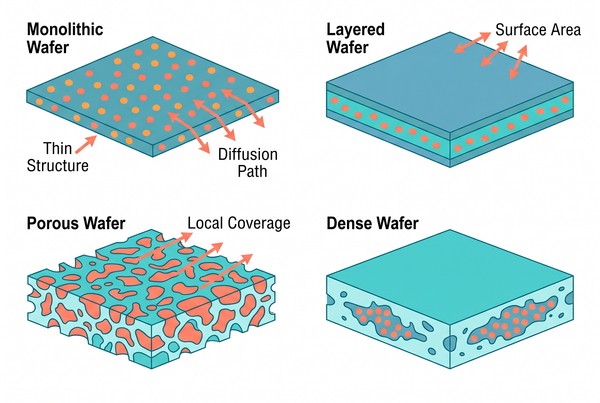 Fig. 1. Structural formats and geometry logic of implantable polymeric wafers (BOC Sciences Authorized).
Fig. 1. Structural formats and geometry logic of implantable polymeric wafers (BOC Sciences Authorized).
Why Wafers Are Considered Geometry-Defined Delivery Systems?
Wafers are geometry-defined because their release behavior is strongly affected by thickness, exposed area, layer sequence, and internal density. In many systems, a small change in thickness has a meaningful effect on diffusion distance, hydration pattern, and burst behavior. That relationship between geometry and performance is stronger than in many bulk implants, which is why wafer development usually requires close coordination between materials selection and structural design.
How Wafers Differ from Conventional Implant Bodies?
Conventional implant bodies are often optimized for bulk structural persistence, device-like dimensions, or reservoir-based control within a larger form factor. By comparison, wafers usually emphasize local coverage, shorter diffusion paths, simpler placement geometry, and a stronger dependence on matrix behavior across a thin structure. They also differ from inserts or films because they are intended as implantable systems rather than primarily surface-retained or easily removable dosage forms.
What Makes Wafer Systems Relevant to Localized Long-Acting Delivery?
Wafers are relevant to localized long-acting delivery because they can create prolonged exposure at a specific site without requiring a large bulk implant. Their thin profile may help improve placement flexibility while still allowing the formulator to design sustained release through matrix diffusion, degradation, or layer-specific transport. When localized control matters more than broad distribution, wafer systems often provide a useful balance between structural precision and formulation adaptability.
Why Consider a Polymeric Wafer Instead of Another Long-Acting Platform?
The decision to develop a wafer usually comes from a platform-selection question rather than from a purely material question. Developers do not choose wafers simply because they are implantable; they choose them when a localized, geometry-defined solid system offers advantages that are not as easy to achieve with dispersed particles, bulk implants, or hydration-driven matrices. In that sense, a wafer is often best viewed as a strategic format choice rather than a default long-acting solution.
- What Makes a Wafer Different from Bulk Implants and Depots: Compared with bulk implants, wafers typically offer thinner structures, larger relative surface area, and more direct control over local contact patterns. Compared with depots, they are preformed solid systems rather than materials that form after administration. This can make wafer performance easier to interpret structurally, since geometry and internal matrix design are established before placement rather than emerging from an in situ transformation process.
- Where Wafers Offer Better Localized Control: Wafers can be especially attractive when the delivery goal depends on local coverage, controlled directional release, or maintaining a defined solid form at the site of placement. Because the dosage form can be predesigned to match a local environment, wafers often provide more explicit structural control than dispersed particles and more localized simplicity than larger implant bodies. This makes them particularly useful in projects where the release area itself is part of the design logic.
- When a Wafer Is Too Simple — or Too Limiting: A wafer can also be too limiting when the project needs very large payload capacity, highly complex release barriers, or very long structural persistence beyond what a thin solid matrix can conveniently support. If the system requires a much larger volume, a bulk implant may be more practical. If localized solid geometry is not important, a depot or hydrogel-based alternative may provide a more flexible route. Wafer development is most successful when the format itself solves a real design problem rather than serving as a thinner version of another platform.
How Geometry Makes Wafers Unique in Long-Acting Drug Delivery?
Geometry is not just a packaging detail in wafer systems; it is one of the core determinants of release behavior. Because wafers are thin and planar relative to many implantable systems, their performance is highly sensitive to diffusion length, exposed surface area, and local fit. This means geometry can be used intentionally as a release-design tool, but it can also become a source of inconsistency if it is not tightly controlled.
- Thin, planar structures change diffusion paths: In thin planar systems, the path a dissolved molecule must travel to leave the matrix may be relatively short compared with that in bulk implants. This can accelerate early release unless the matrix is dense enough or the drug is distributed in a way that restrains transport. At the same time, thin structures can make release more predictable when geometry is consistent and the matrix is well designed.
- Surface area-to-thickness ratio acts as a release design tool: The surface area-to-thickness ratio can be used deliberately to tune how quickly a wafer hydrates and how strongly early release contributes to the total profile. A higher ratio often favors faster interaction with the surrounding medium, while thicker structures can extend diffusion paths and reduce the relative dominance of the initial phase. This makes geometric proportion a key design variable in wafer development.
- Shape affects conformability, placement, and local coverage: Shape affects not only release but also how the wafer sits at the target location. A geometry that provides better local conformity may improve contact and stabilize release, while a poorly matched shape can reduce effective coverage or alter the surrounding environment in ways that affect transport. For this reason, geometry in wafer systems should be treated as both a placement variable and a performance variable.
- Geometry can improve or reduce release consistency: The same sensitivity that makes geometry useful can also create risk. Small changes in thickness, edge profile, porosity distribution, or layer uniformity may cause noticeable variation in release. If the drug distribution is uneven across a thin wafer, the geometry can amplify rather than smooth that inconsistency. Therefore, geometry control must be approached as part of quality design, not only as a fabrication outcome.
How Polymeric Wafers Achieve Controlled Release?
Controlled release in implantable wafers usually emerges from a combination of matrix diffusion, surface-controlled depletion, and structural change over time. Unlike some larger implant systems where a distinct reservoir may dominate performance, wafer systems often depend heavily on how the thin matrix handles water entry, drug redistribution, and gradual evolution of the polymer structure. This is why release in wafers is best understood as a geometry-coupled materials process.
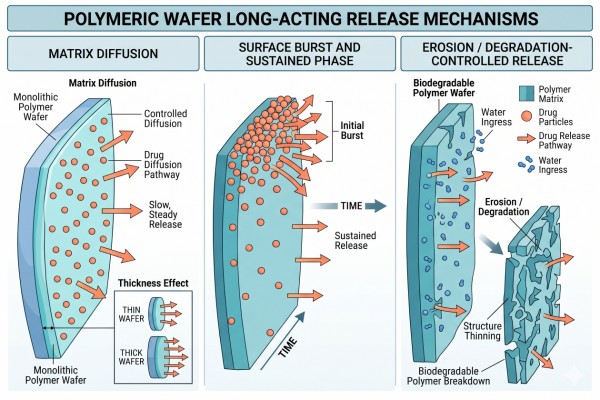 Fig. 2. How polymer matrices and wafer geometry control long-acting release (BOC Sciences Authorized).
Fig. 2. How polymer matrices and wafer geometry control long-acting release (BOC Sciences Authorized).
Matrix Diffusion Through Thin Polymer Structures
In many wafer systems, the dominant release pathway is diffusion through a relatively thin polymer matrix. As water enters the structure, drug molecules become mobile and migrate toward the exposed surface. The rate of transport depends on matrix density, polymer-drug compatibility, hydration behavior, and the available diffusion distance. Thin structures can support sustained release, but only when diffusion is sufficiently moderated by matrix design. In practice, this means the wafer must be engineered so that the drug is not simply released because the structure is thin, but because the matrix provides a controlled resistance to transport over the intended time window. Even modest changes in internal density or drug dispersion can shift the balance between an acceptable early phase and an overly rapid loss of payload.
Surface-Area Effects on Burst and Sustained Phases
Because wafers often expose a relatively large area compared with their thickness, surface-associated drug can contribute strongly to an initial burst phase. This makes surface enrichment and edge effects especially important. A well-designed wafer balances the early phase with the longer diffusion tail so that the system does not lose too much payload before the intended sustained-release period begins. The challenge is that surface area in wafers is not only a dimensional feature but also a release amplifier: if the drug is distributed unevenly near the surface, the large exposed area can magnify burst release far more than in bulkier implants. For that reason, formulation strategy must control both geometry and drug localization if a stable sustained phase is expected to follow the initial release period.
Erosion and Degradation in Biodegradable Wafer Systems
In systems built from biodegradable polymers, erosion or degradation can gradually open new transport pathways and shift the dominant release mechanism over time. The key development challenge is matching the rate of structural change to the desired duration. If degradation is too fast, release may become front-loaded; if too slow, release completion may be incomplete or overly delayed for the project goal. In wafer systems, this balance is especially important because the structure is thin enough that partial loss of integrity can quickly change the available diffusion path and effective release area. As a result, degradation is not just a material property but a structural event that reshapes the release profile as the wafer evolves during implantation.
Polymer Materials Used in Implantable Wafers
Material selection in implantable wafers is not simply about choosing a polymer that can form a thin sheet. The material must also support the intended balance among mechanical integrity, hydration response, release duration, and manufacturability. Some polymers are chosen because they maintain structure well, others because they soften, swell, or degrade in useful ways, and others because they can be combined into composite systems that offer more precise local control than a single matrix alone.
Biodegradable Polyesters and Related Systems
Polyesters and related degradable systems are often relevant in wafer design because they offer a practical route to gradual structural evolution and extended release. Materials such as Polylactic Acid (PLA), Polycaprolactone (PCL), and related copolymeric systems can help control how the wafer retains its structure and how quickly it opens diffusion pathways. Their usefulness depends on how the degradation pace aligns with the intended local release window. Beyond simple degradability, these materials are valued because they often provide a workable balance between processability and mechanical persistence in thin structures. In wafer systems, that balance matters more than it does in many thicker implants, since even a moderate change in matrix integrity can alter release timing, edge stability, and local handling behavior.
Hydrophilic and Swelling Polymer Components
Hydrophilic materials may be introduced when controlled hydration, softening, or internal water-channel formation is needed. Components such as Polyethylene glycol, Gelatin, or Dextran-related systems can influence internal mobility and matrix hydration in ways that reshape release behavior. Their use in wafers requires careful balance, since excessive water affinity can weaken structural integrity or shorten the sustained phase. In practical formulation terms, these materials are most useful when the development goal is not simply to make the wafer more hydrophilic, but to create a predictable hydration response that supports the desired transport profile. If swelling becomes too strong or too uneven, the same components that help release can also generate deformation, premature softening, or unstable release behavior.
Composite Wafer Structures for Tuned Local Release
Composite wafer structures are useful when a single polymer cannot provide the required combination of strength, hydration control, and release modulation. A composite design may combine a more persistent structural phase with a more hydrophilic or more permeable component to tune local transport. This is especially attractive when the project requires directional release, layered functionality, or better burst control than a uniform matrix can provide. Composite systems also expand the design space for localized delivery because they allow one part of the wafer to govern shape retention while another part shapes water entry, drug mobility, or interface behavior at the implantation site. The value of this approach lies in decoupling functions that would otherwise compete inside one polymer, making the wafer easier to optimize for both structural performance and long-acting release.
Need Support for Implantable Wafer Development?
From polymer selection and wafer architecture to localized release control, we support implantable wafer development through material screening, structure design, and formulation strategy.
What Structural Wafer Designs Are Actually Used?
Wafer systems can be built in several distinct structural forms, and each form changes how the drug is retained, how the environment enters the matrix, and how sustained release evolves over time. For this reason, architecture selection is often just as important as polymer selection. The most successful designs are those in which structure and material are chosen together rather than sequentially.
| Wafer Design Type | Structural Feature | Main Release Mechanism | Key Strength | Main Limitation | Best-Fit Development Goal |
|---|
| Monolithic Wafer | Drug distributed through one continuous matrix | Matrix diffusion with possible degradation contribution | Simple design and scalable structure | Burst risk if distribution is surface-biased | Straightforward sustained local release |
| Layered Wafer | Distinct functional layers | Directional or sequential release | Better control over transport pathway | Higher manufacturing complexity | Multiphase or spatially controlled delivery |
| Porous Wafer | Internal void structure with higher fluid access | Accelerated hydration and diffusion | Potentially faster activation and tunable transport | More sensitive to burst and structural instability | Systems needing faster matrix wetting |
| Dense Wafer | Low-porosity compact matrix | Restricted diffusion and slower hydration | Stronger release restraint | May delay useful release too strongly | Longer, more controlled release tails |
| Flexible Wafer | More compliant structure | Depends on matrix composition and thickness | Improved local conformity in some placements | Potential softening or deformation risk | Sites requiring adaptable contact |
| Rigid Wafer | Higher dimensional stability | Often diffusion-dominant with structural persistence | Strong handling and shape retention | May be less adaptable to local fit | Defined placement and stable geometry |
Monolithic Wafers for Simple Matrix Release
Monolithic wafers place the drug throughout one continuous polymer structure, making them one of the most direct formats for studying matrix-controlled release. This design is often attractive because fabrication can be comparatively straightforward and the relationship between polymer choice, drug dispersion, and release behavior is easier to interpret than in more complex architectures. At the same time, monolithic systems require especially good control over internal uniformity. If the drug is distributed unevenly, if solvent removal creates concentration gradients, or if the surface becomes enriched during processing, the apparent simplicity of the system can quickly translate into unstable release behavior. In practice, monolithic wafers are most useful when the goal is a well-defined sustained-release profile that does not require strong directional control or multiple functional layers.
Layered Wafers for Sequential or Directional Delivery
Layered wafers are valuable when one part of the structure needs to perform a different function from another, such as supporting mechanical integrity, restricting diffusion, or staging the order of release. This architecture can be used to direct release preferentially toward one exposed face, to delay the exposure of part of the payload, or to separate incompatible components within the same wafer body. Because each layer may have a different composition, porosity, or thickness, the system offers much finer control than a uniform matrix, but it also becomes more sensitive to fabrication consistency and interfacial defects. Small problems at the boundary between layers can change water ingress, delamination risk, or the timing of release transitions. As a result, layered wafers are especially attractive for projects that need more precise release programming, but they demand a higher level of structural discipline during design and processing.
Porous vs Dense Wafers for Different Transport Profiles
Porous and dense wafers represent two ends of a transport-control spectrum, and the choice between them has major consequences for how the system behaves after implantation. Porous wafers allow fluid to enter more rapidly, which can speed up matrix wetting, mobilize drug sooner, and shorten the lag before measurable release begins. This can be advantageous when the formulation requires faster activation, but it also increases the likelihood of burst release and can weaken structural stability if the pore network becomes too open. Dense wafers, by contrast, slow water penetration and restrict drug movement more strongly, often supporting a longer and more restrained release phase. However, if the matrix is too dense, the system may become overly diffusion-limited and fail to deliver the payload within the desired timeframe. In real development work, many successful wafers use an intermediate structure that balances fluid access with sufficient resistance to avoid uncontrolled early loss.
Flexible vs Rigid Wafers for Site-Specific Placement
Flexible and rigid wafers are not simply different in feel; they reflect different assumptions about how the dosage form is expected to behave at the site of placement. A more flexible wafer may adapt better to irregular local surfaces, maintain broader contact, and reduce mechanical mismatch in environments where conformity matters. This can improve positional stability and help preserve a consistent release interface. Rigid wafers, on the other hand, often offer better handling stability and shape retention during placement, which may be important when geometry must remain highly defined. The trade-off is that a rigid system can be less forgiving in confined or uneven local environments and may not maintain the same degree of surface adaptation. Choosing between flexibility and rigidity is therefore not a secondary materials question; it is part of deciding how the wafer should interact mechanically with its intended implantation site over the full release period.
How to Match a Wafer Design to Molecule Type and Delivery Goal?
A wafer system is only useful when the molecule and delivery goal actually fit the constraints of a thin implantable matrix. This is why formulation strategy should begin with the payload and exposure objective rather than with a preferred structure. Drug loading, distribution, stability, and the intended local release pattern all influence whether a wafer is practical or whether another long-acting platform is a better choice.
When Hydrophobic Molecules Fit Wafer Systems Well
Hydrophobic molecules often fit wafer systems well because they can usually be retained more effectively in hydrophobic or moderately permeable polymer matrices, which helps prevent excessively rapid loss after implantation. This makes wafer systems attractive when the goal is to achieve prolonged local exposure from a relatively thin solid structure. At the same time, strong affinity between the molecule and the matrix is not automatically beneficial. If the interaction is too strong, diffusion may become too slow, and a large fraction of the payload may remain trapped in the wafer longer than intended. The real formulation challenge is therefore not simply to embed the molecule in the wafer, but to create a matrix in which retention and mobility are balanced well enough to support a meaningful sustained-release profile across the target duration.
What Changes When the Payload Is Hydrophilic or Unstable
Hydrophilic or unstable payloads usually narrow the formulation window because they are more sensitive to water entry, interfacial stress, migration during processing, and uneven distribution within thin structures. In wafer systems, these issues are amplified by the limited thickness of the matrix, since drug localization near the surface can quickly translate into burst release or chemical instability. Hydrophilic molecules may require tighter control of hydration pathways, more protective microenvironments, or layered architectures that isolate the payload from the most stressful parts of the structure. Unstable molecules raise additional concerns related to solvent exposure, residual moisture, and polymer–payload compatibility over time. For these reasons, designing wafers for hydrophilic or labile payloads usually involves more than choosing a more hydrophilic polymer; it requires a broader strategy for controlling both distribution and local matrix conditions after implantation.
Why High Loading Is Not Always an Advantage in Thin Wafers
High drug loading may seem desirable because it increases the amount of active material delivered per unit size, but in thin wafer systems it often introduces structural and release-related penalties. As loading rises, the risk of drug aggregation, crystallization, nonuniform dispersion, and surface enrichment also increases. In a geometry-defined matrix, those changes can quickly translate into brittleness, curling, weak edge integrity, and a more pronounced initial release phase. A highly loaded wafer may therefore appear efficient on paper while performing poorly in practice because the structure no longer supports controlled release. In many cases, a lower but more uniform loading level provides better long-acting performance, especially when the development goal is not maximum payload density but a reproducible balance among structural stability, manageable burst, and a sustained release tail.
How Localized Exposure Goals Change Formulation Priorities
When the primary objective is localized exposure rather than broad systemic distribution, formulation priorities change in important ways. The wafer no longer needs to maximize total drug flux as aggressively as a systemic platform might; instead, it must provide the right amount of exposure at the right local site for the right duration. This often makes placement behavior, local contact area, directional release, and structural persistence more important than absolute loading efficiency alone. A formulation designed for localized goals may benefit from denser matrices, more deliberate geometry, or slower transport if those features improve positional control and reduce unnecessary early loss. In other words, the "best" wafer for localized delivery is not simply the most drug-rich or the fastest-releasing design, but the one whose material and structure are aligned most closely with the intended local exposure pattern.
What Research Applications Can Polymeric Wafers Support?
Polymeric wafers support a broad range of research activities because they provide a controllable model for localized sustained release in a geometry-defined implantable format. They are useful not only as candidate delivery systems, but also as research tools for examining how structure, polymer properties, and drug positioning affect long-term release behavior. This makes them relevant to both formulation development and broader delivery-platform research.
Localized Small-Molecule Delivery Research
Wafers are well suited for localized delivery research involving small molecules when a project needs sustained exposure near a defined site rather than broad systemic distribution. Their structured geometry makes it easier to study how thickness, surface area, and matrix composition influence drug movement compared with more dispersed delivery formats. This is especially useful in early formulation screening, where researchers want to understand whether a molecule can be retained locally and released in a controlled manner without relying on a large implant body or repeated administration. Because the wafer is a relatively simple yet highly tunable structure, it can serve as both a prototype dosage form and an experimental platform for comparing how local release changes with material and design adjustments.
Sustained Delivery Studies for Potent or Site-Limited Molecules
Potent molecules and site-limited delivery concepts are particularly relevant to wafer-based research because the dosage form can concentrate a relatively small drug load into a localized structure while still supporting sustained release. This makes wafers attractive when the scientific question is less about delivering a large total dose and more about maintaining useful exposure over time in a confined region. In these studies, researchers can explore how matrix density, degradability, and geometry influence the duration of action without having to transition immediately to more complex implant formats. Wafers are therefore valuable in projects where local control, dose efficiency, and prolonged exposure need to be studied together in a compact implantable system.
Polymeric Wafer Platforms for Combination and Multi-Layer Release Research
Layered and composite wafers are especially useful in combination-release research because they allow different drugs, barriers, or functional layers to be organized within one defined implantable structure. This makes it possible to study sequential release, directional transport, or multi-phase delivery without requiring an entirely separate device architecture for each variable. In research settings, these systems can reveal how one layer modifies hydration, how another affects diffusion resistance, and how the combined structure shapes overall release timing. For teams exploring dual-payload systems or staged local delivery concepts, wafers provide a practical platform for examining whether multiple release functions can be integrated into one thin geometry while preserving structural integrity and reproducibility.
Material–Structure–Release Relationship Studies in Implantable Systems
Polymeric wafers are also highly valuable as research models for studying the relationship between polymer properties, internal structure, and long-acting release behavior in implantable systems. Because the geometry is relatively simple but still highly sensitive to thickness, porosity, layering, and drug distribution, wafers make it easier to isolate how one variable changes performance. Researchers can use them to compare degradable and nondegradable matrices, evaluate how swelling alters transport, or examine how structural defects influence burst release and long-term behavior. In this sense, wafers are not only candidate products but also experimental tools that help clarify the design rules governing localized implantable drug delivery.
Services
Advanced Polymer Synthesis and Wafer Formulation Services
At BOC Sciences, implantable polymeric wafers are approached as structure-defined delivery platforms in which geometry, material selection, processability, and localized release behavior must be developed together rather than separately. Our support is designed to help teams move from broad wafer concepts to more practical material choices and testable architectures through polymer synthesis, matrix screening, structural design support, and performance-oriented formulation evaluation. By connecting polymer function with thin-structure mechanics, release strategy, and localized delivery goals, BOC Sciences helps developers build wafer systems that are not only scientifically interesting, but also technically actionable.

Custom Polymer & Wafer Material Design
- Selection of biodegradable, hydrophilic, and composite-forming polymers for wafer systems.
- Material matching based on structural integrity, hydration behavior, and release duration.
- Support for tuning polymer roles beyond drug loading alone.
- Technical guidance for localized long-acting wafer concepts.

Wafer Architecture and Formulation Development
- Support for monolithic, layered, porous, dense, flexible, and rigid wafer designs.
- Optimization of geometry, thickness, and drug distribution strategy.
- Guidance on matching architecture to release goals and payload type.
- Formulation workflows aligned with structure-defined delivery logic.

Release and Performance Evaluation
- Assessment of diffusion, degradation, and burst-release behavior in thin matrices.
- Analysis of structure–release relationships in wafer systems.
- Support for localized delivery evaluation and release-duration optimization.
- Interpretation of how material and geometry interact during sustained release.

Processing, Stability, and Handling Support
- Evaluation of cracking, curling, softness, and handling-related risks.
- Assessment of moisture sensitivity, storage behavior, and structural reproducibility.
- Support for process development in thin geometry systems.
- Technical guidance from concept screening to robust wafer design.
Need a Consultation?
Discuss your implantable wafer project with BOC Sciences to evaluate material selection, structural design, and localized sustained-release strategy.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What is an implantable polymeric wafer in drug delivery?
An implantable polymeric wafer is a thin or planar solid dosage system placed at a localized site to provide sustained release from a geometry-defined matrix. Its performance depends not only on the polymer itself, but also on thickness, surface area, internal structure, and how the wafer evolves after implantation.
-
How do polymeric wafers provide long-acting release?
Polymeric wafers usually provide long-acting release through a combination of matrix diffusion, surface-controlled depletion, and gradual structural change such as erosion or degradation. Because the dosage form is thin, release is strongly influenced by geometry, drug distribution, hydration rate, and how the polymer matrix changes over time.
-
What polymers are commonly used in implantable wafer systems?
Common materials include biodegradable polyesters, hydrophilic matrix modifiers, and composite-forming polymers selected for structural integrity, hydration control, and release modulation. The most suitable polymer depends on the intended duration, local placement conditions, payload compatibility, and whether the wafer needs to remain dense, swell, or gradually degrade.
-
How do wafers differ from films, inserts, or bulk implants?
Wafers differ from films and inserts because they are implantable, geometry-defined systems intended for localized sustained release after placement. They differ from bulk implants because their behavior is more strongly shaped by thin-planar structure, surface-area effects, and local coverage, rather than by large-volume device architecture and reservoir size.
-
What research applications can polymeric wafers support?
Polymeric wafers can support localized small-molecule delivery research, sustained-release studies for potent molecules, combination-release investigations, and material–structure–release correlation work. They are useful both as candidate long-acting systems and as experimental platforms for studying how implantable matrix geometry and polymer design influence localized delivery behavior.

 Fig. 1. Structural formats and geometry logic of implantable polymeric wafers (BOC Sciences Authorized).
Fig. 1. Structural formats and geometry logic of implantable polymeric wafers (BOC Sciences Authorized). Fig. 2. How polymer matrices and wafer geometry control long-acting release (BOC Sciences Authorized).
Fig. 2. How polymer matrices and wafer geometry control long-acting release (BOC Sciences Authorized).