Stimuli-Responsive Polymer Micelles: Design Strategies, Trigger Mechanisms, and Controlled Release Applications
Stimuli-responsive polymer micelles occupy a distinctive position in advanced polymer delivery because they combine the nanoscale advantages of self-assembled core-shell carriers with a second layer of control built into the polymer structure itself. Their defining feature is not simply that they can carry a payload, but that they can alter core integrity, corona hydration, interfacial behavior, or linker stability when exposed to a relevant trigger. In practical formulation terms, this means the same carrier can be designed to remain relatively stable during preparation and transport, yet become more permeable, destabilized, or disassembled when local conditions shift in a predictable way. The value of this design logic depends on whether the trigger is translated into a useful structural response rather than into uncontrolled instability. A responsive micelle must therefore be judged by more than the presence of a pH-sensitive group or a reducible linkage. The real question is whether the trigger changes the micelle at the right rate, in the right environment, and with enough structural contrast to improve release behavior without sacrificing formulation reproducibility. That requirement makes stimuli-responsive micelles a materials design problem, a colloid problem, and a process problem at the same time.
Resources
What Are Stimuli-Responsive Polymer Micelles?
Stimuli-responsive polymer micelles are self-assembled nanostructures built from amphiphilic copolymer systems or related macromolecular architectures that contain a triggerable structural element in the core, shell, or connecting domain. Under a defined stimulus, such as pH variation, reductive environment, enzymatic cleavage, temperature shift, or externally applied energy, the balance of forces that maintains the micelle can be altered. That change may accelerate payload diffusion, disrupt the core, detach a shielding shell, or induce complete structural rearrangement. Responsive behavior is therefore meaningful only when it is tied to a mechanistically interpretable change in assembly state.
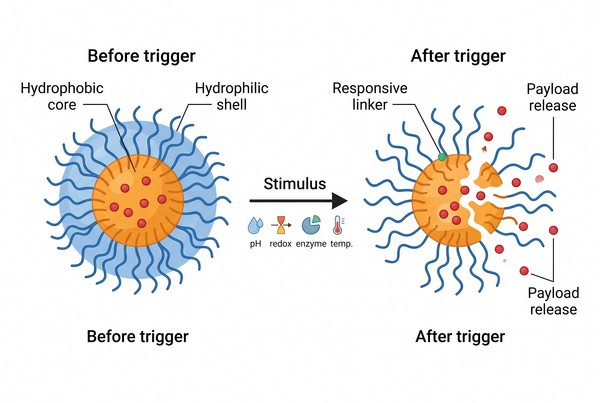 Fig. 1. Stimuli-responsive polymer micelle before and after triggering (BOC Sciences Authorized).
Fig. 1. Stimuli-responsive polymer micelle before and after triggering (BOC Sciences Authorized).
A stimuli-responsive polymer micelle is a self-assembled carrier whose colloidal structure or release behavior changes in response to a defined environmental cue. In most systems, the micelle is formed from amphiphilic chains that first organize into a hydrophobic core and hydrophilic shell, then incorporate a triggerable motif such as an ionizable segment, a cleavable linker, or a conformationally responsive block. The trigger does not merely decorate the carrier; it modifies the free-energy balance that stabilizes the assembly. The resulting response can involve shell collapse, core swelling, bond cleavage, or altered permeability, depending on where the responsive element is placed.
How They Differ from Conventional Polymeric Micelles?
Conventional polymer micelles are already dynamic because self-assembled structures are sensitive to solvent composition, concentration, and interchain interactions. Responsive micelles go further by building a programmed structural switch into the carrier. Instead of relying only on passive core partitioning and spontaneous leakage, they are designed so that a specific stimulus alters chain ionization, cleaves a junction, changes hydrophilic-hydrophobic balance, or weakens core packing. This gives the formulator an extra level of control, but also introduces additional complexity in synthesis, preparation, and stability testing because the triggerable feature must remain useful without making the system unstable before activation.
Why Responsiveness Is Introduced into Micellar Systems?
Responsiveness is introduced when passive micellar behavior is not sufficient to balance transport stability and desired release. A standard micelle may improve dispersion of a hydrophobic compound, yet still release too early, too slowly, or without enough selectivity once conditions change. By contrast, a responsive micelle can be designed so that exposure to a local cue increases permeability, reduces core cohesion, or causes structural breakup only after the carrier reaches the intended microenvironment. The practical objective is not maximal sensitivity, but controlled asymmetry: relatively stable behavior before triggering and a predictable structural shift after triggering.
Why Responsive Design Matters in Polymer Micelle Development?
The need for responsive design becomes clearer when passive micellar delivery is examined critically. Ordinary micelles can solubilize hydrophobic molecules, reduce apparent aggregation, and create small colloidal carriers, but they do not automatically solve the problem of when and where release should occur. In many formulations, the main limitation is not loading itself but the mismatch between transport stability and release timing. Responsive design addresses this mismatch by introducing a stimulus-dependent mechanism that translates a local environmental change into altered micelle behavior rather than relying solely on passive diffusion and dilution-dependent dynamics.
Limits of Passive Micellar Delivery
Passive micelles can be highly useful, especially for poorly soluble compounds, but their release behavior is often governed by a combination of diffusion, repartitioning, and gradual structural relaxation. That can be adequate for some formulations, yet it becomes limiting when the payload must remain associated during one stage and release more efficiently during another. A passive micelle may appear colloidally stable while still leaking a weakly compatible solute, or it may retain the payload so strongly that release remains incomplete. Responsive design is intended to reduce that uncertainty by making release depend less on uncontrolled background kinetics and more on a deliberately engineered transition.
Spatiotemporal Control of Payload Release
Spatiotemporal control means that the release event is linked to a relevant condition rather than to time alone. In responsive micelles, this may involve protonation at lower pH, cleavage of disulfide or diselenide bonds in a reductive environment, enzymatic degradation of a polymer segment, or temperature-driven dehydration of a shell-forming block. The aim is not only to speed release, but to confine the release mechanism to the environment where it is most useful. In formulation design, that distinction is important because a carrier that releases everywhere is not truly responsive, even if its chemistry contains a triggerable group.
Balancing Stability Before Trigger and Release After Trigger
The central design problem in responsive micelles is balance. If the triggerable feature is too weak, the micelle behaves almost like a passive carrier. If it is too strong or too exposed, premature destabilization can occur during storage, dilution, or processing. A useful responsive system therefore needs two compatible states: a pre-trigger state with sufficient core cohesion and shell stabilization, and a post-trigger state with a clear route to enhanced release or structural rearrangement. This dual-state requirement is one reason responsive micelles are more challenging to optimize than ordinary self-assembled carriers, even when the underlying polymer chemistry seems straightforward.
Endogenous Triggers: pH-, Redox-, and Enzyme-Responsive Polymer Micelles
Endogenous triggers are among the most widely studied because they rely on chemical or biochemical differences already present in local environments rather than on externally applied equipment. In responsive polymer micelles, endogenous triggering often works by changing ionization state, cleaving labile bonds, or altering polymer-solvent interactions under selective conditions. pH-responsive and redox-responsive systems remain especially common, while enzyme-responsive systems add another layer of specificity through substrate recognition. More recent discussions also include ROS- and hypoxia-associated motifs as extensions of the same logic.
pH-Responsive Polymer Micelles
pH-responsive polymer micelles are usually designed around ionizable polymer segments or acid-labile linkers that change the assembly state when proton concentration shifts. The key design variable is not simply whether a group can be protonated, but whether that protonation is strong enough to alter hydrophilic-hydrophobic balance, interchain repulsion, or core cohesion at the desired pH window. In practice, pH-sensitive motifs may be placed in the core, shell, or interfacial junction, and each location produces a different structural response. Core-localized ionizable segments tend to increase water penetration and weaken hydrophobic packing, while shell-localized responsive groups can alter colloidal stabilization or expose previously shielded domains. Acid-cleavable linkers, by contrast, convert a pH signal into bond breakage and more discrete structural disassembly. For this reason, pH-responsive micelles are most effective when the target pH range, trigger location, and intended mode of structural change are aligned, rather than when pH sensitivity is introduced in a generic way.
Redox-Responsive Polymer Micelles
Redox-responsive polymer micelles typically rely on chemically labile bonds such as disulfides, diselenides, thioethers, or related motifs that can be cleaved or transformed under reductive or oxidative conditions. Their technical advantage lies in the fact that a single bond can serve as both a stabilizing structural element before triggering and a programmed failure point after triggering. When placed between hydrophilic and hydrophobic blocks, redox-cleavage can shift the amphiphilic balance of the entire chain and drive micelle disassembly. When used as a core crosslink or internal connector, the same chemistry can preserve micelle integrity during transport and then accelerate internal loosening or fragmentation upon activation. The main design challenge is matching cleavage kinetics to the intended formulation window. If the bond is too labile, structural weakening may occur prematurely during preparation or storage; if it is too inert, the micelle may show little functional contrast after exposure to the target reducing or oxidizing environment.
Enzyme-Responsive Polymer Micelles
Enzyme-responsive polymer micelles are designed so that a specific enzymatic transformation changes micelle stability, shell behavior, or payload accessibility. This is commonly achieved through peptide linkers, ester-containing segments, or other enzyme-labile motifs that are selectively cleaved in the presence of a target enzyme. Compared with broader triggers such as pH or temperature, enzyme responsiveness offers a more chemically specific route to structural activation, but that specificity also creates additional constraints. The chosen substrate must be accessible to the enzyme within the assembled micelle, the cleavage rate must be fast enough to influence release on a useful timescale, and the resulting structural change must be large enough to alter transport or retention behavior. If the substrate is buried too deeply or if cleavage only removes a small peripheral fragment, the micelle may remain functionally unchanged despite successful chemistry. Enzyme-responsive systems therefore require closer coupling between substrate placement, micelle architecture, and post-cleavage structural consequences than many other trigger types.
ROS- and Hypoxia-Related Extensions
ROS- and hypoxia-related responsive micelles extend endogenous trigger design beyond conventional pH and redox systems by exploiting oxidative stress or low-oxygen-associated chemical transformations. ROS-responsive systems often use sulfur-, selenium-, boronic ester-, or thioketal-containing motifs that become more hydrophilic, cleave, or otherwise destabilize the assembly after oxidation. Hypoxia-related systems are more indirect in many cases, often relying on reducible groups or chemistry that becomes activated under oxygen-poor conditions rather than by oxygen depletion alone. These approaches are attractive because they aim to connect polymer micelle behavior to more selective biochemical conditions, but they also increase the burden of validation. It is not enough to demonstrate that a motif is ROS-sensitive or hypoxia-related in isolation; the formulator must show that the specific trigger level is sufficient to change micelle structure, permeability, or release under experimentally relevant conditions. As a result, these advanced endogenous designs are most convincing when the responsive chemistry is integrated into a clearly interpretable structural transition rather than treated as an abstract microenvironment marker.
Exogenous Triggers: Temperature-, Light-, and Ultrasound-Responsive Micelles
Exogenous triggers differ from endogenous triggers because the release event is initiated or amplified by an externally applied condition rather than by the local medium alone. This can offer improved timing control, but it also demands a more rigorous connection between stimulus exposure and micelle response. For responsive micelles, temperature, light, and ultrasound are among the most discussed external triggers. Each can alter micelle behavior through a different physical route, and each requires additional consideration of exposure uniformity, process compatibility, and the structural reversibility of the response.
Temperature-Responsive Polymer Micelles
Temperature-responsive polymer micelles are typically based on blocks whose solvation changes sharply near a defined transition temperature, such as lower critical solution temperature or upper critical solution temperature behavior. In these systems, a small thermal shift can change chain hydration, segmental mobility, or corona conformation, which in turn alters micelle size, interfacial properties, or even assembly state. A responsive temperature transition can be placed in the shell-forming block to regulate hydration and steric stabilization, or in the core-associated segment to modify internal packing and payload mobility. The success of the design depends on whether the transition occurs in a narrow and application-relevant range. If the transition is too broad, the system behaves more like a gradually drifting colloid than a true triggerable carrier. If the transition temperature is poorly positioned relative to processing or storage conditions, the micelle may undergo unwanted pre-trigger restructuring. Temperature-responsive systems are therefore most useful when the thermal transition is both compositionally tunable and tightly connected to a meaningful change in colloidal or release behavior.
Light-Responsive Polymer Micelles
Light-responsive polymer micelles use photo-cleavable, photo-isomerizable, photo-crosslinkable, or photo-oxidizable groups to convert irradiation into a structural change in the carrier. The main technical attraction of this strategy is temporal precision, because the trigger can be applied at a chosen time rather than being passively dictated by the environment. However, the responsive chemistry must be placed where photoreaction alters micelle structure in a measurable way. A photo-labile linker can induce shell shedding or block separation, an isomerizable chromophore can alter amphiphilicity or packing geometry, and a light-sensitive crosslink can shift core integrity after irradiation. The central challenge is that photochemical activation must compete with optical limitations such as penetration depth, local energy distribution, and possible nonuniform exposure across the dispersion. For this reason, light-responsive micelles are strongest when the structural effect of photoreaction is large, the responsive moiety is strategically positioned, and the irradiation conditions are carefully matched to the optical properties of the system.
Ultrasound-Responsive Polymer Micelles
Ultrasound-responsive polymer micelles generally rely on mechanical, cavitational, or localized thermal effects induced by acoustic energy rather than on direct covalent trigger chemistry alone. In some designs, ultrasound destabilizes the micelle by increasing local shear or permeability, while in others it accelerates release by perturbing a structurally primed core or shell. This makes ultrasound less of a purely chemical trigger and more of a process-coupled activation method. Its effectiveness therefore depends on both carrier design and exposure conditions, including frequency, intensity, duration, pulse pattern, and the surrounding medium. A major technical issue is distinguishing genuine ultrasound-triggered structural activation from nonspecific release acceleration caused by heating or agitation. For that reason, convincing ultrasound-responsive micelle systems require careful control experiments showing that acoustic exposure changes the micelle in a defined, reproducible way rather than merely disrupting a marginally stable colloid.
Why External Triggers Need Extra Process Control?
External triggers offer stronger temporal control than endogenous signals, but they also introduce a second layer of process engineering that can become a major source of variability. A responsive micelle exposed to nonuniform heating, partial light penetration, or inconsistent acoustic energy may not undergo a single coherent transition; instead, it may form mixed populations of intact, partially destabilized, and fully triggered assemblies. This heterogeneity can complicate both structural interpretation and release analysis. In addition, external triggering must be compatible with the formulation's preparation history, concentration range, and surrounding medium, because the same stimulus can produce different outcomes depending on shell hydration, particle density, or payload distribution at the time of exposure. External-trigger systems are therefore not defined only by responsive chemistry, but by the reproducibility of stimulus application itself. Without controlled exposure geometry and well-defined activation conditions, even an elegant responsive design may fail to deliver a technically meaningful or scalable response.
Need custom polymers for responsive micelle design?
BOC Sciences provides tailored copolymer synthesis, trigger-responsive polymer design, and formulation support for pH-, redox-, and multi-responsive micellar systems.
How Responsive Polymer Micelles Are Designed?
Responsive micelle design is most effective when the trigger is treated as a structural variable rather than as an isolated functional group. The same responsive chemistry can behave very differently depending on whether it is placed in the core, shell, or linker region of the assembly. A useful design must therefore answer two connected questions: what event should the stimulus induce, and which part of the micelle must change to produce that event? Triggerable design becomes meaningful only when bond cleavage, ionization, hydration change, or shell detachment is directly linked to an interpretable change in carrier structure.
Stimuli-Cleavable Linkers and Triggerable Bonds
Cleavable linkers such as disulfides, diselenides, ketals, hydrazones, and other labile junctions are widely used because they convert a trigger into a discrete structural event. Their effect depends strongly on placement. A cleavable bond inserted between hydrophilic and hydrophobic blocks can change overall amphiphilicity after cleavage, whereas a similar bond used as a core crosslink can hold the micelle together before trigger and accelerate breakdown after trigger. This makes linker design more than a synthetic choice. It is a way to decide whether the stimulus should cause shell shedding, core decompaction, crosslink loss, or full micelle destabilization.
Responsive Core Design
Responsive core design focuses on altering the internal microenvironment that controls loading and retention. If the trigger changes core hydrophobicity, crosslink density, or internal packing, the payload may be released because the core becomes less compatible or more permeable. This strategy is particularly useful for poorly soluble compounds loaded through partitioning into the micelle interior. The core can be made pH-sensitive through ionizable hydrophobic segments, redox-sensitive through cleavable crosslinks, or oxidation-sensitive through sulfur- or selenium-containing motifs. The key design question is how much core transformation is needed to alter release without causing premature instability during preparation or storage.
Responsive Shell Design
Shell design becomes important when the desired response involves altered hydration, colloidal stability, surface accessibility, or shield removal rather than immediate core breakdown. A responsive corona may contract, dehydrate, lose charge, detach, or expose a previously hidden domain after stimulation. Such changes can influence particle-particle interaction, permeability to surrounding medium, and how rapidly the payload escapes from the core. Shell-responsive designs are especially useful when the pre-trigger state must remain well dispersed and sterically stabilized, but the post-trigger state should become more interactive or less shielded. This allows the response to regulate both transport behavior and release kinetics simultaneously.
Mixed and Multi-Block Architectures
Mixed and multi-block architectures expand design freedom by allowing different chain segments to perform different functions. One block may drive self-assembly, another may provide hydration, and a third may host the responsive motif or a detachable segment. These architectures are powerful because they let the designer separate stability from responsiveness to a certain extent, but they also increase synthetic and compositional complexity. The more blocks or mixed components included, the more carefully the assembly pathway must be controlled. In responsive micelles, architectural sophistication is valuable only if it creates clearer trigger translation rather than merely adding features that are difficult to characterize or reproduce.
Single-, Dual-, and Multi-Responsive Micelles
Responsive micelles are often described by the number of triggers they are designed to recognize, but the real distinction is functional rather than numerical. A single-responsive system links one environmental cue to one dominant structural transition. A dual-responsive system is intended to sharpen control by requiring two chemical or physical conditions, or by allowing one trigger to complement the limitations of another. Multi-responsive systems extend this logic further, but increasing trigger count does not automatically improve formulation quality. The value of additional responsiveness depends on whether it improves selectivity or simply multiplies design burden.
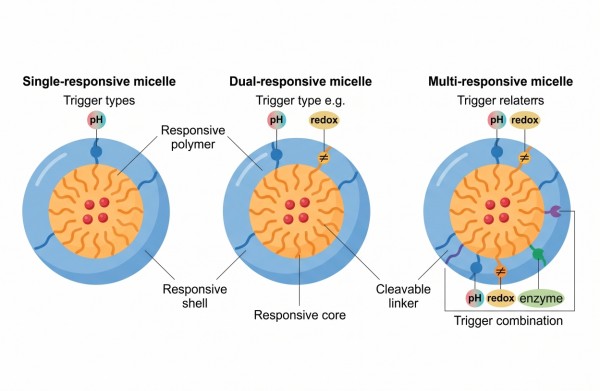 Fig. 2. Design logic of single- and multi-responsive micelles (BOC Sciences Authorized).
Fig. 2. Design logic of single- and multi-responsive micelles (BOC Sciences Authorized).
Why Single-Trigger Systems May Be Insufficient?
Single-trigger systems are attractive because they are conceptually clean and often easier to synthesize and characterize. However, one trigger alone may not always provide enough contrast between the transport state and the release state. A pH-responsive micelle, for example, may respond across a broad pH range rather than only where the designer wants the transition to occur. In such cases, a single trigger may improve responsiveness without providing enough selectivity. This does not make the design wrong; it simply means that one stimulus may be too general to deliver the degree of structural discrimination needed for the intended application.
Dual pH/Redox-Responsive Micelles
Dual pH/redox-responsive systems are among the most common because they combine an environmental shift in ionization with a cleavage event at a reducible linkage. This pairing can be effective when pH alone does not destabilize the micelle sufficiently or when redox cleavage alone is too slow to produce a useful release contrast. By assigning one trigger to shell or interfacial change and another to core or linker cleavage, the system can produce a more structured transition between stable transport and triggered release. The design challenge is coordination: the two triggers must work together rather than generate partially conflicting structural responses.
Multi-Responsive Design and Its Trade-Offs
Multi-responsive systems are often appealing because they suggest high selectivity and fine control, yet they also create a combinatorial increase in design variables. Each additional trigger adds not only another chemical function but another possible failure mode, another characterization requirement, and another reproducibility challenge. If one stimulus alters the micelle in a way that changes sensitivity to the next, the overall response may become harder to interpret. Multi-responsive design is most useful when different triggers clearly govern different structural roles, such as one controlling shell exposure and another controlling core disassembly. Without that separation of function, extra complexity may add little real value.
When Added Complexity Is Justified?
Additional responsive complexity is justified when one trigger cannot reasonably satisfy the full formulation objective. This may occur when the carrier must remain highly stable during preparation and dispersion but then undergo a sharper transition after exposure to a more selective environment. It may also be useful when the release event depends on both a chemical and a physical change. By contrast, if a single-responsive design already produces adequate structural contrast and reproducible performance, adding more triggers can be counterproductive. In responsive micelle development, complexity should be added only when it creates a measurable formulation advantage, not simply because the chemistry is possible.
Preparation and Characterization of Responsive Micelles
Responsive micelles must be prepared in a way that preserves both self-assembly and triggerability. A system can fail not only because the chemistry is poorly chosen, but because the preparation route suppresses the intended structure or the characterization package is too weak to show whether triggering truly changes the micelle. This is especially important for responsive carriers because an apparent release effect may come from poor baseline stability rather than from a stimulus-dependent mechanism. Reliable evaluation therefore requires both route-sensitive preparation and characterization methods capable of distinguishing pre-trigger and post-trigger states.
Preparation Routes for Responsive Polymer Micelles
Responsive polymer micelles must be prepared under conditions that preserve both self-assembly integrity and the chemical functionality of the triggerable motif. This requirement makes route selection more restrictive than for passive micelles, because solvent choice, pH history, temperature exposure, concentration gradient, and purification conditions can all alter whether the responsive segment remains latent or is partially activated before use. Direct dissolution may be suitable when the polymer is already compatible with the target aqueous medium and the responsive group is stable throughout hydration. Dialysis or controlled solvent exchange is often preferred when gradual micellization is needed to avoid kinetic trapping or premature aggregation. Thin-film hydration can be useful for hydrophobic payload incorporation, but it also risks exposing labile linkers or ionizable segments to concentrated local environments during film formation and redispersion. For responsive systems, the preparation method should therefore be chosen not only by polymer solubility, but by whether it preserves trigger threshold, reproducibility, and the intended relationship between pre-trigger stability and post-trigger structural change.
How to Verify Trigger Sensitivity?
Trigger sensitivity should be verified through a comparative framework rather than by observing release in a single stimulated sample. A meaningful evaluation begins with establishing a stable baseline under non-trigger conditions, then exposing the same system to a well-defined trigger intensity, duration, and medium composition. The goal is to show that the stimulus causes a measurable change beyond normal drift, dilution effects, or time-dependent leakage. Depending on the design, that change may involve bond cleavage, altered shell hydration, particle swelling, reduced colloidal stability, faster release, or full micelle disassembly. Just as important, trigger sensitivity must be assessed against matched controls, because many responsive formulations appear "active" simply because they were already unstable. A convincing responsive system therefore demonstrates not only post-trigger change, but also a clear contrast between the pre-trigger state and the activated state under experimentally controlled conditions.
Size, Morphology, and Structural Transition Analysis
Structural characterization of responsive micelles should be designed to capture transition, not just static particle properties. Dynamic light scattering is useful for detecting hydrodynamic size shifts, distribution broadening, or aggregation after trigger exposure, but it cannot by itself show whether the response involves shell contraction, core loosening, or fragmentation into smaller structures. Electron microscopy, cryogenic microscopy, or related imaging tools help reveal whether the spherical morphology is preserved, distorted, fused, or lost after stimulation. Scattering-based methods add another layer by probing internal organization, core-shell dimensions, or changes in interfacial density that may not be obvious from average particle size alone. For responsive systems, the strongest evidence comes from combining these methods so that the trigger-induced transition can be interpreted mechanistically. In other words, structural analysis should answer not only "did the size change," but "how did the micelle reorganize and which part of the structure was most affected."
Drug Loading, Retention, and Triggered Release Testing
Loading and release evaluation in responsive micelles should be treated as a coupled problem rather than as two separate datasets. A formulation that loads well but leaks extensively before triggering provides weak evidence for true responsiveness, while a system that remains stable but shows minimal post-trigger release may be over-engineered without functional advantage. Proper assessment therefore requires at least three comparisons: retained loading before trigger exposure, passive leakage under matched non-trigger conditions, and release acceleration or structural loss after trigger application. The release medium itself must also be chosen carefully, because apparent trigger sensitivity can be exaggerated or obscured by sink conditions, protein-free media, or overly simplified buffer systems. For robust interpretation, triggered release data should be supported by structural evidence that the micelle actually changed in the expected way. Only then can faster release be attributed to responsive design rather than to poor baseline stability, formulation drift, or analytical artifact.
Stability Challenges and Design Trade-Offs
The main challenge in responsive micelles is not adding a triggerable group but managing the trade-offs that group creates. Responsiveness often weakens at least one part of the system before activation, whether through lower core cohesion, less stable shell shielding, or greater sensitivity to environmental variation. In other words, a triggerable design introduces a controlled vulnerability. The formulator must then decide how much instability can be tolerated before triggering and how much structural contrast is required after triggering to justify the design. This balance is one of the defining technical problems of responsive micelle development.
Premature Triggering and Off-Target Release
Premature triggering occurs when the responsive motif is activated, at least partially, under conditions encountered before the intended release stage. This can result from pH drift during processing, reductive exposure during storage, oxidative background conditions, or nonselective environmental fluctuations that alter shell hydration or bond stability. The practical consequence is loss of payload before the carrier reaches the relevant environment. Preventing premature response therefore requires both chemical selectivity and process discipline. A micelle can only be considered meaningfully responsive if the triggerable feature remains mostly silent under handling conditions and becomes active only when the required stimulus is clearly present.
Trigger Sensitivity vs Storage Stability
A highly sensitive responsive group may produce a dramatic release effect, but the same sensitivity can shorten shelf stability or increase batch variability. Conversely, a more stable group may survive processing well but respond too slowly or too weakly when activated. This is not a flaw in one particular chemistry; it is a general trade-off in responsive systems. Designers therefore need to select sensitivity windows rather than maximize sensitivity blindly. The right design is the one that preserves acceptable stability under expected storage and handling conditions while still creating a measurable, formulation-relevant transition once the target trigger is introduced.
Core Integrity vs Fast Disassembly
Fast disassembly can increase release contrast, but if the core becomes too weak even before triggering, the micelle may show poor retention and unconvincing baseline performance. On the other hand, an overly cohesive core may protect the payload well yet resist the trigger so strongly that little structural change occurs. This means core design must balance pre-trigger retention against post-trigger transformability. Crosslinking, mixed-core strategies, or block design may help manage that balance, but each solution introduces its own process and characterization burden. The best responsive core is not the weakest or the strongest, but the one whose cohesion changes in a controlled and measurable way.
Manufacturing and Reproducibility Constraints
Responsive micelles often face reproducibility challenges because their behavior depends not only on composition but also on subtle preparation details such as solvent history, pH adjustment sequence, mixing profile, and purification conditions. When multiple responsive features are present, this sensitivity can intensify. A formulation that behaves convincingly in one preparation run may shift in size, trigger threshold, or release profile when small process differences occur. For this reason, manufacturing feasibility should be considered early. A responsive system that cannot be prepared reproducibly may be academically interesting but still unsuitable for robust formulation development.
Controlled Release Applications of Stimuli-Responsive Polymer Micelles
Application value should be judged by whether responsiveness solves a real formulation problem rather than by the novelty of the chemistry alone. In practice, responsive micelles are most useful when passive self-assembly provides a good starting carrier but additional control is needed over when, where, or how fast release occurs. This is why they appear most often in systems involving poorly soluble small molecules, nucleic-acid-associated designs, and multifunctional carriers where one structural event must be coordinated with another. Responsive micelles are strongest when the trigger changes performance in a way the nonresponsive system cannot easily match.
Poorly Soluble Small Molecules
Hydrophobic small molecules remain one of the most natural application classes for responsive micelles because these compounds already benefit from hydrophobic core loading in ordinary micellar systems. Responsive design adds a second control layer by making that core less stable, more permeable, or more weakly associated only under the selected trigger. This can be useful when passive micelles provide good loading but insufficient contrast between the transport state and the release state. In such systems, the responsive element is most effective when it changes the compatibility between the loaded compound and the core environment rather than merely destabilizing the particle in a nonspecific way.
Gene and Nucleic Acid Delivery
Responsive micellar strategies are also valuable in nucleic-acid-associated systems, especially when the carrier must protect the cargo during one stage and enable a more rapid or more selective release during another. In these systems, responsiveness may be introduced through ionizable segments, reducible motifs, detachable shells, or mixed architectures that balance shielding with later release. The challenge is that nucleic-acid systems often require multiple functions at once: condensation, stabilization, surface compatibility, and controlled decomplexation. Responsive design can help coordinate these steps, but only if the trigger changes association strength and structural accessibility in a predictable and experimentally verifiable manner.
Combination Delivery and Theranostic Systems
Combination and theranostic designs often require one carrier to perform more than one task, such as protecting a payload, co-loading another functional component, or changing behavior under a secondary signal. Responsive micelles are attractive here because structural switching can be coordinated with release, signal exposure, or shell transformation. For example, a core-crosslinked micelle may remain compact during handling and then disassemble under a trigger that also changes the accessibility of a second functional domain. These systems can be powerful, but they also concentrate complexity. Their usefulness depends on whether the responsive event truly coordinates multiple functions rather than simply producing a more elaborate but not more effective carrier.
When Responsive Micelles Are the Right Choice?
Responsive micelles are the right choice when a passive micelle already makes sense for the payload class, but the project still requires better separation between pre-release stability and activated release. They are less compelling when passive systems already provide adequate performance or when a matrix-based particle would solve the problem more directly. In selection terms, responsive micelles are best viewed as precision upgrades to self-assembled carriers rather than as universal replacements for all polymer delivery systems. Their value lies in turning a relevant environmental difference into a controlled structural response, not in making the formulation concept more complicated than necessary.
Services
Advanced Polymer Micelle Synthesis and Formulation Services
BOC Sciences supports responsive polymer micelle development from trigger-responsive polymer design to route-specific formulation optimization. Our capabilities include custom amphiphilic polymer synthesis, architecture control, responsive linker incorporation, end-group modification, and process matching for micellar systems prepared through direct dissolution, solvent exchange, thin-film hydration, or other controlled assembly workflows. We help align polymer composition, trigger mechanism, and self-assembly pathway so that the resulting micelles combine meaningful responsiveness with reproducible preparation, structural consistency, and practical formulation performance.

Custom Responsive Polymer Design

Triggerable Functionalization & Process Matching
- Incorporation of cleavable linkers, ionizable motifs, and responsive interfacial segments.
- Process matching for direct dissolution, dialysis, solvent exchange, and film-based preparation routes.
- Design of Polyethylene glycol-containing systems for corona engineering and shell control.
- Method-specific refinement to improve trigger sensitivity without sacrificing colloidal stability.

Responsive Micelle Preparation & Formulation Development
- Selection of preparation routes for Polymer micelles based on responsive chemistry and solubility profile.
- Optimization of solvent composition, hydration sequence, and processing conditions for reproducible self-assembly.
- Evaluation of route-dependent particle size, dispersity, and trigger-sensitive behavior.
- Integration of polymer design with controlled release strategy and practical formulation workflow.

Analytical Characterization Support
- Characterization of route-sensitive self-assembled micellar systems before and after triggering.
- Comparative assessment of size, structural transition, and release behavior under defined stimulus conditions.
- Evaluation of formulation consistency, baseline stability, and trigger-driven response contrast.
- Documentation support to improve reproducibility and process-transfer readiness.
Do You Need A Consultation?
Discuss your responsive polymer micelle project with BOC Sciences to identify the right balance among trigger mechanism, polymer design, self-assembly behavior, and controlled release performance.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What are stimuli-responsive polymer micelles?
Stimuli-responsive polymer micelles are self-assembled polymer carriers designed to change structure or release behavior when exposed to a specific trigger such as pH, redox conditions, enzymes, or temperature.
-
What is the difference between pH-responsive and redox-responsive micelles?
pH-responsive micelles rely on ionization or acid-labile groups, while redox-responsive micelles usually depend on reducible linkages such as disulfides. They respond to different environmental cues and therefore require different design logic.
-
Why are dual-responsive polymer micelles used?
Dual-responsive micelles are used when one trigger alone does not provide enough selectivity or control. Combining triggers can improve release precision, but it also increases structural complexity and development burden.
-
How do you evaluate triggered release in polymer micelles?
Triggered release is evaluated by combining structural characterization with release testing before and after stimulation. Useful methods often include DLS, microscopy, and quantitative release analysis under defined trigger conditions.
-
When are responsive micelles better than conventional micelles?
Responsive micelles are more suitable when the formulation needs better separation between transport stability and release activation. If passive micelles already provide sufficient loading and release behavior, a responsive design may not be necessary.

 Fig. 1. Stimuli-responsive polymer micelle before and after triggering (BOC Sciences Authorized).
Fig. 1. Stimuli-responsive polymer micelle before and after triggering (BOC Sciences Authorized). Fig. 2. Design logic of single- and multi-responsive micelles (BOC Sciences Authorized).
Fig. 2. Design logic of single- and multi-responsive micelles (BOC Sciences Authorized).