How Polymer Micelles Form and How to Prepare Them: Mechanisms, Methods, and Formulation Strategy
Polymer micelles are self-assembled colloidal systems formed by amphiphilic macromolecules under selective solvent conditions. Their practical value lies in the fact that molecular design, solvent environment, and process history jointly determine whether the final dispersion becomes a useful nanoscale carrier or a poorly controlled aggregate population. For formulation work, it is therefore not enough to ask whether a polymer can form micelles in principle. What matters is how micellization proceeds, which route controls the transition from dissolved chains to core-shell assemblies, and how that route affects size, dispersity, loading behavior, and colloidal robustness in downstream use.
Resources
What Are Polymer Micelles?
In formulation science, polymer micelles occupy an important position because they translate polymer architecture directly into nanoscale function. A useful micelle system usually begins with an amphiphilic copolymer or other tailored polymer whose solvophobic segments can associate into a confined core while solvophilic chains stabilize the surrounding interface. Yet the presence of amphiphilicity alone does not guarantee a robust formulation. The final assembly depends on how the polymer encounters the solvent, how quickly the environment changes, and whether the system reaches an equilibrated or kinetically trapped state during preparation and post-processing.
Definition of Polymer Micelles
Polymer micelles are nanoscale core-shell assemblies generated when amphiphilic macromolecules organize in a selective medium. In water, the hydrophobic segments cluster to minimize unfavorable solvent contact, while the hydrophilic segments remain exposed to create a stabilized corona. Compared with conventional surfactant micelles, polymeric systems usually exhibit lower CMC values, slower chain exchange, and broader opportunities for structural tuning through block length, composition, and architecture. Those features make polymer micelles particularly relevant when a formulation requires improved colloidal persistence, controllable internal microenvironment, and closer integration between material design and processing strategy.
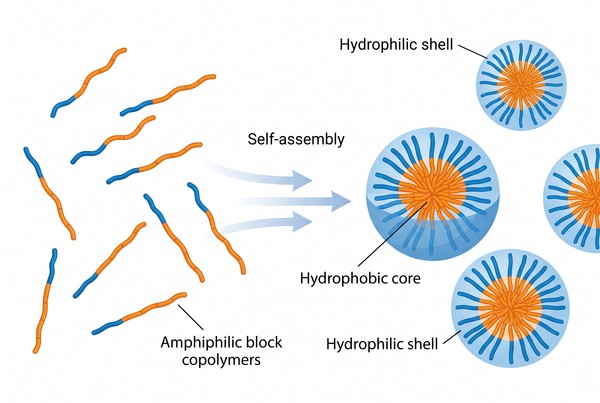 Fig. 1. Self-assembly of amphiphilic polymers into core-shell micelles (BOC Sciences Authorized).
Fig. 1. Self-assembly of amphiphilic polymers into core-shell micelles (BOC Sciences Authorized).
Core-Shell Architecture and Molecular Basis of Assembly
The usefulness of polymer micelles is rooted in their compartmentalized architecture. The core functions as a solvophobic domain whose polarity, packing density, and mobility are dictated by hydrophobic block chemistry, while the corona controls hydration, steric stabilization, and interaction with the external medium. This division of roles means that a micelle is not just a particle but a dynamic, chemically differentiated nanostructure. Because core and shell emerge from the same chain under different solvation conditions, subtle changes in block ratio, dispersity, or end-group chemistry can shift micelle size, morphology, and preparation sensitivity in significant ways.
Why Formation and Preparation Must Be Discussed Together?
In micelle development, formation is not just a conceptual event that happens once the polymer touches water. It governs nearly every practical outcome that follows. If the polymer chains assemble too quickly, the system may become kinetically trapped before reaching a uniform state. If the solvent quality changes too gradually, the system may allow structural rearrangement but at the cost of low throughput. If the polymer is only marginally amphiphilic, the resulting particles may be broad in size or unstable upon dilution. In other words, preparation is the way formulation scientists intentionally steer the route from dissolved polymer to functional micelle population.
How Polymer Micelles Form in Selective Solvent Environments?
Polymer micelles form when amphiphilic chains are exposed to a solvent environment that favors one segment of the chain while penalizing another. In aqueous media, hydrophobic domains lose favorable contact with the solvent and begin to associate, whereas hydrophilic segments remain solvated and occupy the particle interface. That transition reduces the overall free-energy penalty of the dispersed state. However, micellization does not proceed identically for every system. The rate of solvent change, the initial chain conformation, and the opportunity for molecular rearrangement all determine whether the resulting assemblies are compact, polydisperse, metastable, or poorly formed from the outset.
Amphiphilicity as the Primary Driver of Micellization
Amphiphilicity is the fundamental requirement for polymer micelle formation because it creates opposing solvation preferences within the same chain. If the solvophobic block is too weak, the polymer remains molecularly dispersed or forms loose aggregates with poor cohesion. If it is too dominant, the system may collapse into larger aggregates or precipitated domains before stable micelles can develop. A useful micelle-forming polymer therefore needs an appropriate balance between attractive association in the core-forming segment and sufficient solvated volume in the shell-forming segment. That balance governs not only whether micelles form, but also how sensitive they will be to the subsequent preparation route.
Interfacial Energy Reduction and Core Formation
Core formation is usually interpreted as a response to unfavorable solvent exposure of the hydrophobic block, but the process is more nuanced than simple collapse. The system lowers interfacial free energy by clustering solvophobic segments into a shared domain while extending solvophilic segments outward to stabilize the interface. At the same time, local packing interactions such as van der Waals association, hydrogen bonding, or aromatic stacking may strengthen the core and alter chain mobility. These contributions are important because they help explain why polymers with similar hydrophilic-hydrophobic ratios can still produce markedly different micelle compactness, loading behavior, and resistance to structural rearrangement.
Critical Micelle Concentration and the Onset of Assembly
The critical micelle concentration, or CMC, is often used to describe the concentration range where self-assembly becomes thermodynamically favored. In polymeric systems, CMC values are usually much lower than in conventional surfactant micelles, which is one reason these carriers are valued for dispersion-based formulation. Still, the practical meaning of CMC should not be overstated. A low CMC suggests improved resistance to dilution, but it does not alone guarantee tight cargo retention, low exchange kinetics, or stability in complex media. Those properties also depend on core cohesion, corona structure, and how the micelles were created during the preparation process.
What Determines Micelle Structure During Formation?
Once micellization begins, several material and environmental variables act together to define the final particle population. Some govern the thermodynamic preference for spherical micelles, while others influence how quickly the system can reorganize before it becomes kinetically trapped. Because polymer micelles are pathway-sensitive assemblies, the same nominal polymer can yield different size distributions or morphologies under different solvent histories. Understanding those determinants is essential before choosing a preparation method, because method selection should follow the assembly behavior of the system rather than rely on procedural convenience or general popularity of a given laboratory technique.
Polymer Composition, Molecular Weight, and Block Ratio
Polymer composition strongly influences both the driving force and geometric outcome of self-assembly. A longer hydrophobic block generally strengthens core formation and can reduce CMC, but it may also slow rearrangement and promote larger aggregates if the chain collapses too quickly. A longer hydrophilic block typically enhances steric stabilization and corona hydration, yet excessive shell dominance can weaken association and broaden assembly behavior. Molecular weight and block ratio therefore affect more than size alone; they shape preferred curvature, chain packing, interfacial coverage, and the degree to which a system can tolerate different preparation histories without losing colloidal definition.
Solvent Quality, pH, Ionic Strength, and Temperature
The medium surrounding the polymer determines how strongly each segment is solvated and how sharply the system crosses into the micellization regime. Solvent quality dictates whether the hydrophobic block remains dissolved long enough for controlled assembly or collapses too abruptly into broad aggregates. pH can alter ionization state, shell hydration, and electrostatic repulsion, while ionic strength may screen charged segments and reduce corona expansion. Temperature affects chain mobility and solvation and can even induce assembly transitions in responsive systems. For this reason, the solvent environment should be treated as an active design parameter, not just a background condition.
Pathway Dependence and Kinetic Trapping Effects
A critical feature of polymer micelles is that the route to the final solvent composition often matters as much as the final composition itself. Rapid exposure to water can generate small, kinetically trapped assemblies, whereas gradual solvent exchange may allow chain rearrangement toward a more equilibrated state. Dry-film rehydration introduces locally concentrated polymer domains that must redisperse before uniform micelles can emerge. Because of this pathway dependence, two processes using the same polymer and nominal concentration can still produce different particle populations. Method development must therefore consider whether the goal is equilibrium-driven assembly, useful metastability, or a controlled compromise between both.
Need the right polymer and process route for micelle development?
BOC Sciences provides polymer synthesis, block copolymer design, and formulation support to help match self-assembly behavior with the most suitable preparation strategy for your target micelle system.
Why Self-Assembly Mechanism Determines Method Selection?
Preparation methods do not merely package a pre-existing micelle concept into a laboratory workflow. They control the way solvent quality changes, how quickly local supersaturation develops, how much time polymer chains have to reorganize, and whether co-loaded species are incorporated before or after core formation. That is why method selection should start with a mechanistic question: how does this polymer prefer to assemble under realistic solvent conditions? Only after that is understood can a route be chosen rationally to improve reproducibility, loading opportunity, and practical scalability without forcing the system into an inherently unstable or poorly defined state.
Thermodynamic Assembly Versus Kinetically Frozen Structures
Some preparation routes allow the polymer system to approach a more relaxed, lower-energy state, whereas others freeze the structure rapidly into a metastable but still useful micelle population. Neither outcome is automatically superior. Thermodynamically favored assemblies may support better long-term consistency if equilibration is achievable, but kinetically trapped micelles can also be valuable when they preserve a desirable size or encapsulation state that would otherwise reorganize. The important point is to recognize which regime a method promotes. Without that understanding, researchers may misinterpret apparent success in one process as a general property of the polymer itself.
Solubility Mapping as the First Process Decision
Before selecting any preparation method, it is essential to map polymer solubility across relevant aqueous, organic, and mixed-solvent environments. If the polymer disperses and assembles cleanly in the final aqueous medium, direct dissolution may be appropriate. If both blocks require a common organic solvent before controlled transfer into water, dialysis or another solvent exchange route becomes more logical. If a hydrophobic additive must first associate with the polymer in a shared organic phase, thin-film hydration or evaporation-based strategies may provide better control. Solubility behavior therefore acts as the first technical filter for method selection.
Cargo Compatibility and Process Sensitivity
A preparation method that works well for empty micelles may fail once a cargo is introduced, because the additive changes solvent preference, core packing, interfacial tension, or chain mobility. Hydrophobic compounds often benefit from co-dissolution with the polymer prior to aqueous assembly, while more labile species may require short processing times and mild solvent exposure. In some cases, the cargo itself alters the preferred assembly pathway and shifts the resulting particle size or morphology. For robust development, polymer choice, loading logic, and preparation route should be evaluated as a combined system rather than in isolated sequential steps.
Practical Route Design for Polymer Micelle Preparation
In polymer micelle development, the practical route used to generate self-assembly is often as important as the polymer structure itself. Preparation methods differ in how rapidly solvent quality changes, how uniformly polymer chains are redistributed, and whether the system has time to reorganize before becoming kinetically trapped. As a result, method selection should be based on polymer solubility, cargo compatibility, and the level of control required over size, dispersity, loading, and post-preparation stability, rather than on laboratory convenience alone.
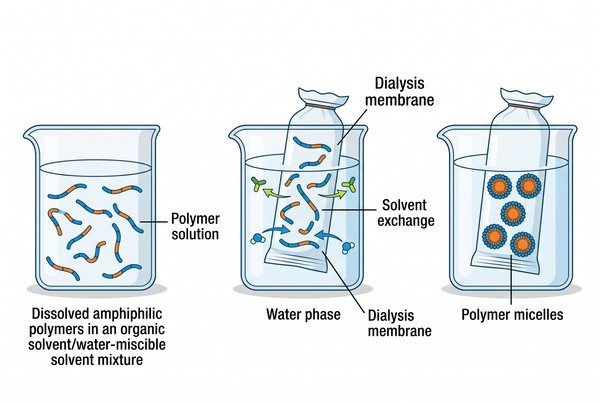 Fig. 2. Dialysis-driven solvent exchange for polymer micelle preparation (BOC Sciences Authorized).
Fig. 2. Dialysis-driven solvent exchange for polymer micelle preparation (BOC Sciences Authorized).
Direct Dissolution
Direct dissolution is most suitable when the amphiphilic polymer can disperse and reorganize directly in the final aqueous medium without first requiring a common organic solvent. Its technical advantage is process simplicity, which makes it useful for early screening and fast composition comparison. However, this route is highly sensitive to incomplete wetting, local overconcentration, and overly rapid hydrophobic collapse, especially for polymers with strong core-forming blocks. In practice, stirring intensity, hydration temperature, addition sequence, and polymer concentration determine whether the system forms narrow micelles or broad aggregates with hidden batch-to-batch variability.
Dialysis-Mediated Solvent Exchange
Dialysis is a controlled solvent-transition method in which the polymer is first dissolved in a water-miscible organic solvent and then gradually exposed to water through membrane-mediated exchange. Its main strength is that the changing solvent environment gives the polymer chains time to reorganize during assembly, which often improves size uniformity and reduces uncontrolled precipitation relative to abrupt aqueous transfer. At the same time, dialysis is not inherently robust unless membrane cutoff, exchange rate, solvent choice, polymer concentration, and external volume ratio are well controlled. The process is therefore technically powerful but operationally slower and more parameter-sensitive than direct dissolution.
Emulsification and Solvent Evaporation
Emulsification and solvent evaporation become relevant when the polymer or hydrophobic payload cannot be handled effectively by direct aqueous assembly. In this route, polymer dissolved in an organic phase is emulsified into an external medium, and subsequent solvent removal drives formation of colloidal structures. For micelle-oriented systems, the key technical issue is that droplet-scale mass transfer can dominate the final outcome: droplet size distribution, solvent diffusion rate, interfacial stabilization, and evaporation profile all affect whether the system yields discrete micelles, loose aggregates, or more compact nanoparticulate structures. This route is therefore useful, but only when emulsion behavior is treated as a critical quality variable rather than a background step.
Thin-Film Hydration
Thin-film hydration is particularly valuable when the polymer and a poorly soluble hydrophobic compound must first be co-dissolved in a shared volatile solvent before assembly in water. After solvent removal, the resulting film is hydrated and reorganized into micellar structures. The technical challenge is that film morphology strongly affects the hydration pathway: thick or uneven films may redisperse slowly and leave polymer-rich domains that generate broad particle populations. Hydration temperature, aqueous phase volume, agitation energy, and residual solvent content all influence whether the polymer reaches a true micellar state or remains partly trapped in incompletely redispersed aggregates.
Freeze-Drying and Reconstitution Control
Freeze-drying should not be viewed only as a storage step, because for many polymer micelle systems the real technical question is whether the original colloidal structure can survive freezing, sublimation, and rehydration without irreversible reorganization. Cryoconcentration, ice crystal formation, and interparticle compression may disrupt the corona or induce fusion-like interactions during dehydration. Reconstitution behavior then becomes a decisive indicator of process success. Studies on polymeric micelles show that lyophilization outcomes depend strongly on cryoprotectant identity, excipient ratio, and redispersion conditions, and some formulations can recover particle size well while others show persistent size growth or loading redistribution after drying.
Comparative Evaluation of Established Preparation Routes
Comparative evaluation of these routes should focus on pathway control rather than simply operational ease. Direct dissolution is efficient but least forgiving when polymer hydration is incomplete. Dialysis offers finer solvent-transition control but imposes time and throughput penalties. Emulsification and solvent evaporation are useful for difficult hydrophobic systems, yet their outcomes are strongly coupled to emulsion quality. Thin-film hydration supports early polymer-cargo association but is vulnerable to redispersion heterogeneity. Freeze-drying adds storage flexibility but introduces a second structural stress event during reconstitution. A rigorous method comparison should therefore include DLS size distribution, redispersion behavior, dilution stability, and loaded-state integrity rather than relying only on initial particle appearance.
| Method | Typical Use Case | Core Strength | Main Limitation | Most Important Variable |
|---|
| Direct Dissolution | Polymers that assemble directly in aqueous medium | Simple workflow and fast screening | Can broaden quickly with poor dispersibility | Polymer concentration and hydration conditions |
| Dialysis | Organic-solution to water transfer systems | Controlled solvent transition and improved uniformity | Slow throughput and parameter sensitivity | Exchange rate and initial solvent choice |
| Emulsification / Solvent Evaporation | Hydrophobic co-loading and solvent-dependent polymers | Useful for difficult loading scenarios | Droplet and evaporation variability | Emulsion stability and solvent removal profile |
| Thin-Film Hydration | Polymer-cargo co-dissolution before hydration | Supports pre-association of hydrophobic components | Film and redispersion heterogeneity | Film quality and hydration intensity |
| Freeze-Drying | Dry intermediate and reconstitution workflows | Storage and transport flexibility | Structural stress during dehydration | Reconstitution quality after lyophilization |
Advanced Process Engineering Approaches for Polymer Micelle Assembly
Advanced preparation strategies are valuable when conventional methods no longer provide sufficient control over mixing, solvent transition, structural reproducibility, or trigger-dependent assembly behavior. These approaches do not replace classical routes in every case, but they extend the design space by allowing tighter control over mass transfer, phase behavior, and assembly timing. For technically demanding systems, they can reduce operator dependence, improve scale translation logic, or create micellization pathways that are difficult to access through standard batch processing alone.
Microfluidic-Assisted Fabrication
Microfluidic-assisted fabrication improves polymer micelle preparation by controlling the contact between polymer solution and antisolvent at micrometer scale, which narrows the mixing window and reduces uncontrolled local supersaturation. This is especially useful for pathway-sensitive systems where manual addition or bulk stirring creates high run-to-run variability. Published studies show that flow-rate ratio, total flow rate, channel geometry, organic solvent type, and drug concentration can all affect final micelle size and uniformity, which makes microfluidics a true process-engineering tool rather than merely a miniaturized mixer. Its main advantage is reproducibility, but it requires systematic parameter mapping before meaningful scale translation is possible.
Supercritical Fluid Processing
Supercritical fluid processing is attractive because it can modify solvent removal and phase-transition behavior without relying on conventional high-boiling organic solvent workflows. In polymer micelle systems, the technique has been explored as a way to prepare assemblies with improved handling of thermally sensitive and poorly soluble compounds. One reported comparison found that self-assembled micelles prepared through supercritical-fluid technology showed better performance than a thin-film hydration route in an oral delivery setting, illustrating that the method can influence not only particle formation but also downstream functional behavior. Its limitation is that pressure-sensitive operating windows and polymer compatibility must be understood in detail before the route becomes practically transferable. :contentReference[oaicite:10]{index=10}
Stimuli-Triggered Micelle Formation
Stimuli-triggered assembly uses changes in pH, temperature, ionic strength, redox environment, or other external inputs to induce micellization only after the polymer reaches a defined condition set. This is technically useful when formulation handling requires the polymer to remain processable in one state and assemble only during a later trigger step. From a process perspective, the challenge is not merely achieving trigger response but controlling how sharply and uniformly the transition occurs across the full dispersion volume. If trigger application is spatially or temporally uneven, the result can be broad particle populations or mixed morphologies. This route is most successful when responsive polymer design and process timing are optimized together.
PEG-Assisted Method
PEG-assisted preparation strategies use PEG-containing segments or PEG-mediated interfacial effects to moderate aggregation during assembly and strengthen corona formation in the final micelle. Technically, PEG is useful because it can improve hydration, reduce interparticle adhesion during solvent transition, and promote more stable shell development, especially in systems that otherwise aggregate during rapid aqueous exposure. At the same time, PEG-assisted design is not universally beneficial: if the hydrophilic fraction becomes too dominant, core association may weaken and the micelles can become overly dynamic. The method is therefore best viewed as an interfacial control strategy that links formulation processability with final colloidal stabilization, not simply as a generic additive trick.
How to Select the Right Preparation Strategy?
Method selection becomes rational only when polymer behavior, cargo properties, and development goals are evaluated together. The most appropriate route is not necessarily the simplest or the most advanced; it is the one that produces reproducible, well-defined micelles under conditions compatible with both the material and the intended application. A useful decision framework starts with solubility mapping, then considers whether the system benefits from gradual solvent transition, co-dissolution of a hydrophobic additive, dry-state handling, or highly controlled mixing. From there, method choice can be narrowed by comparing reproducibility, throughput, storage needs, and downstream formulation requirements.
Choose According to Polymer and Solvent Compatibility
The first screening question is whether the polymer can assemble directly in the target medium or requires a common solvent before transfer. Polymers that disperse and organize cleanly in water are natural candidates for direct dissolution, whereas solvent-dependent systems often perform better in dialysis or other exchange-based routes. If the polymer shows strong pathway sensitivity, microfluidic or staged solvent control may be necessary. This compatibility analysis is especially important for materials related to biodegradable polymers, polyesters, or more specialized architectures whose assembly window is narrower than simple aqueous dispersibility tests might suggest.
Choose According to Cargo and Functional Objective
Empty micelles for self-assembly studies and loaded micelles for formulation development do not always require the same route. Hydrophobic additives often benefit from co-dissolution with the polymer, which favors film-hydration or solvent-evaporation strategies. Systems that emphasize sustained release may need tighter core organization and careful comparison with polymer micelle platforms for sustained and targeted drug delivery. Formulations oriented toward poorly soluble compounds should also be considered alongside polymeric micelles for poorly soluble drugs, because loading strategy and preparation method are tightly connected.
Choose According to Reproducibility and Platform Context
Preparation routes should be compared not only by initial particle size but also by repeatability, storage behavior, and how well the route fits the broader delivery platform. A method that performs acceptably at very small scale may create hidden redevelopment costs later if process transfer becomes necessary. In some cases, it is also useful to compare micelle preparation with adjacent carrier platforms such as polymer nanoparticles or to review broader carrier choices through choosing the right drug delivery system: polymer vs lipid vs inorganic carriers. That wider context helps determine whether micelles are truly the right vehicle.
Services
Advanced Polymer Micelle Synthesis and Formulation Services
BOC Sciences supports polymer micelle development from molecular design to process-oriented formulation optimization. Our capabilities cover custom amphiphilic polymer and copolymer synthesis, block architecture control, end-group modification, and route-specific preparation strategy design for self-assembled micellar systems. We help clients align polymer composition, solubility profile, and assembly pathway with practical fabrication routes such as direct dissolution, solvent exchange, thin-film hydration, and advanced controlled assembly workflows. In addition, BOC Sciences provides integrated support in material screening, process refinement, and analytical evaluation to improve reproducibility, structural consistency, and formulation readiness for polymer micelle research and development.

Custom Amphiphilic Polymer Design

Polymer Functionalization & Process Matching
- End-group and side-chain modification to improve interfacial stabilization and shell hydration.
- Process matching for direct dissolution, dialysis, solvent exchange, and film-based preparation routes.
- Polyethylene glycol-containing polymer design to support corona engineering and controlled assembly.
- Method-specific refinement to improve micelle uniformity, redispersion behavior, and structural robustness.

Micelle Preparation & Formulation Development
- Selection of suitable fabrication routes for polymer micelles based on polymer solubility and assembly behavior.
- Optimization of solvent composition, hydration conditions, exchange sequence, and processing parameters.
- Evaluation of route-dependent particle size, dispersity, and reconstitution performance.
- Integration of polymer design with practical preparation workflows for reproducible micelle fabrication.

Analytical Characterization Support
- Characterization of polymers, copolymers, and route-sensitive self-assembled systems.
- Comparative analysis of preparation methods from a structure-property-process perspective.
- Assessment of formulation consistency, redispersion quality, and colloidal stability.
- Documentation support to improve reproducibility and process-transfer readiness.
Do You Need A Consultation?
Discuss your polymer micelle project with BOC Sciences to identify the right combination of polymer architecture, self-assembly pathway, and preparation strategy for your formulation objectives.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
How do polymer micelles form?
Polymer micelles form when amphiphilic polymers self-assemble in a selective solvent. The hydrophobic segments associate to reduce unfavorable solvent contact, while the hydrophilic segments remain solvated and stabilize the assembly.
-
What is the most common method for preparing polymer micelles?
Common methods include direct dissolution, dialysis, and thin-film hydration. The best choice depends on polymer solubility, cargo properties, desired particle uniformity, and whether process reproducibility or scale-up is important.
-
Why does the preparation method affect micelle properties?
Preparation controls the self-assembly pathway, solvent environment, and mixing history. These factors influence particle size, dispersity, loading behavior, structural uniformity, and the balance between thermodynamic assembly and kinetic trapping.
-
Is dialysis always better than direct dissolution?
No. Dialysis often improves control over solvent exchange and can help produce more uniform micelles, but it is slower and less efficient for throughput. Direct dissolution is simpler when the polymer and cargo can assemble well in aqueous conditions.
-
When should microfluidics be considered for polymer micelles?
Microfluidics is useful when tight control over mixing, reproducibility, and process consistency is needed. It is especially relevant for systems that are sensitive to assembly conditions or may later require better scale translation.

 Fig. 1. Self-assembly of amphiphilic polymers into core-shell micelles (BOC Sciences Authorized).
Fig. 1. Self-assembly of amphiphilic polymers into core-shell micelles (BOC Sciences Authorized). Fig. 2. Dialysis-driven solvent exchange for polymer micelle preparation (BOC Sciences Authorized).
Fig. 2. Dialysis-driven solvent exchange for polymer micelle preparation (BOC Sciences Authorized).