A Practical Guide to Polymer Micelles for Nucleic Acid Delivery
Polymer micelles provide a versatile materials framework for nucleic acid delivery when the design goal extends beyond simple nanoparticle formation to controlled condensation, shielding, intracellular transport, and timely release of DNA, RNA, or oligonucleotide cargoes. Unlike conventional micellar systems developed for hydrophobic small molecules, nucleic acid-loaded polymer micelles are often built around electrostatic self-assembly between charged polymer segments and negatively charged nucleic acids, forming polyion complex micelles or micelleplexes with tunable core-shell organization. By controlling block composition, charge density, hydrophilic corona structure, degradability, and assembly conditions, these systems can be tailored to improve cargo protection, circulation stability, uptake behavior, endosomal escape, and intracellular decomplexation. As a result, polymer micelles have become an important design option for researchers developing non-viral nucleic acid delivery platforms with more adaptable structure-property relationships than many one-component polyplex systems.
Resources
How Polymer Micelles Support Nucleic Acid Delivery?
Polymer micelles used for nucleic acid delivery should not be understood as a simple extension of hydrophobic drug-loaded micelles. In most nucleic acid systems, the critical design problem is not how to solubilize a poorly water-soluble payload inside a hydrophobic core, but how to condense, protect, transport, and release a highly charged macromolecular cargo with minimal structural loss. This difference changes the entire materials logic of the platform. Instead of relying only on amphiphilic partitioning, many relevant systems are based on electrostatic complexation between nucleic acids and charged blocks within polymeric micelles, often producing polyion complex micelles or micelleplexes with a protective hydrophilic corona and a functionalized condensed core.
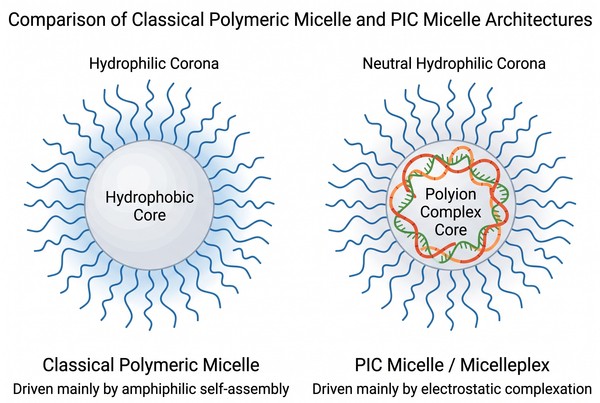 Fig. 1. Comparison of classical and PIC micelle architectures (BOC Sciences Authorized).
Fig. 1. Comparison of classical and PIC micelle architectures (BOC Sciences Authorized).
In the context of nucleic acid delivery, polymer micelles are nanoscale assemblies formed from carefully designed block or graft polymers that organize into core-shell structures capable of associating with plasmid DNA, siRNA, mRNA, antisense oligonucleotides, or related polyanionic biomacromolecules. The loaded structure may arise through amphiphilic self-assembly, electrostatic condensation, or a hybrid of both. What matters functionally is that the polymer framework creates a colloidally stable particle able to maintain a compact size, preserve nucleic acid integrity, and support transport across extracellular and intracellular barriers. This makes the term "polymer micelle" broader in nucleic acid delivery than in classical solubilization chemistry.
How PIC Micelles and Micelleplexes Differ from Classical Micelles?
Classical micelles are usually driven by hydrophobic association of amphiphilic blocks above a critical micelle concentration, producing a hydrophobic interior and a hydrophilic exterior. By contrast, many nucleic acid micellar systems contain a core formed partly or primarily by electrostatic interaction between a cationic or ionizable polymer segment and the negatively charged phosphate backbone of the cargo. Such assemblies are often described as polyion complex (PIC) micelles or micelleplexes. This distinction matters because particle stability, assembly reversibility, salt sensitivity, and release behavior are governed not only by hydrophobic effects but also by charge neutralization, ionic screening, and the kinetics of decomplexation.
Why Nucleic Acid Delivery Requires a Different Micelle Design Logic?
A hydrophobic drug micelle can often succeed by improving apparent solubility and slowing premature leakage. A nucleic acid micelle must do much more. It must condense a large and highly hydrated cargo, shield surface charge sufficiently to reduce unfavorable interactions in biological fluids, remain intact under dilution, avoid irreversible over-binding, and then release the nucleic acid in the correct intracellular compartment. For this reason, polymer selection, molecular weight, block architecture, charge density, and the balance between stability and disassembly all become central design variables. Researchers therefore evaluate polymer micelles for nucleic acids less as passive carriers and more as responsive self-assembled delivery systems.
Why Nucleic Acids Are Difficult to Deliver with Polymer Micelles?
The value of polymer micelles in nucleic acid delivery becomes clear only after understanding the physicochemical barriers associated with DNA and RNA cargoes. These materials do not diffuse across lipid membranes like many small hydrophobic compounds, and they do not remain stable in complex biological environments without protection. As a result, micelle design has to solve several linked problems at once: condensation, shielding, persistence in relevant media, cellular entry, and productive intracellular release. A polymer micelle that performs well in one stage but fails at another is rarely useful as a platform.
High Negative Charge, Large Size, and Limited Membrane Transport
Nucleic acids possess a densely charged phosphate backbone that creates strong hydration and strong electrostatic repulsion from negatively charged cell membranes. Their size also increases transport difficulty. Plasmid DNA is particularly demanding because of its large contour length and structural rigidity relative to smaller oligonucleotides, while mRNA and long RNA cargoes combine high molecular weight with conformational flexibility and sensitivity to shear and enzymatic degradation. Polymer micelles are therefore expected to mediate condensation into smaller particles, reduce net surface charge exposure, and produce a structure more compatible with endocytic uptake.
Nuclease Degradation and Poor Stability in Physiological Fluids
Naked RNA is rapidly degraded in many environments, and unprotected DNA or oligonucleotides may also suffer structural damage or loss of function. A useful polymer micelle must reduce direct exposure of the cargo to enzymes, salts, and competing biomacromolecules. This requirement is not met simply by generating a particle of the correct size. The assembly must present sufficient shielding through a neutral or weakly interactive corona, while the inner complex should maintain enough binding strength to preserve cargo association during handling, storage, and biological contact. At the same time, the protective effect cannot be so strong that it prevents later release.
Rapid Clearance, Opsonization, and Short Circulation Time
Once introduced into biological media, nanoparticles experience adsorption, ionic screening, and interactions with proteins and cells that can destabilize the assembly or alter its fate. Strongly exposed cationic surfaces are often associated with rapid nonspecific interactions, aggregation risk, and unfavorable biodistribution. For this reason, polymer micelles used for nucleic acid delivery commonly rely on hydrophilic corona-forming segments such as poly(ethylene glycol) analogs or other neutral chains that reduce direct interfacial contact. The corona is not merely decorative. It is a major structural component controlling colloidal stability, stealth behavior, and the extent to which the micelle retains its intended architecture after dilution.
Intracellular Barriers and Endosomal Entrapment
Even after uptake, the platform still faces substantial intracellular barriers. Many polymer micelles enter cells by endocytic pathways, which means the cargo may remain trapped inside endosomes or progress to lysosomal compartments where degradation or functional loss becomes more likely. Efficient design therefore requires a workable strategy for endosomal escape or membrane destabilization at the right stage. The micelle must also support the correct final delivery endpoint. Some cargoes require cytosolic availability, while plasmid DNA additionally faces the challenge of reaching the nucleus for expression-related applications. These constraints explain why effective micelle design focuses on the whole transport sequence, not just particle formation.
Which Types of Nucleic Acids Can Be Delivered by Polymer Micelles?
Not all nucleic acids impose the same design requirements on polymer micelles. Differences in size, flexibility, charge density, intracellular site of action, and sensitivity to degradation strongly influence what type of polymer architecture is appropriate. A practical resource page should therefore distinguish among major cargo classes rather than treating all DNA and RNA as interchangeable payloads. This is also where platform selection becomes more realistic, because the best-performing micellar design for one nucleic acid format may be suboptimal for another.
Plasmid DNA and the Need for Condensation plus Intracellular Access
Plasmid DNA is one of the most demanding nucleic acid cargos for polymer micelles because it combines large hydrodynamic size, high linear charge density, and substantial conformational sensitivity during complexation. Efficient delivery usually begins with electrostatic condensation, where cationic or ionizable polymer segments reduce DNA coil dimensions and form compact nanostructures that are small enough for stable dispersion and cellular uptake. However, the design challenge is not simply to maximize binding strength. Overly strong polymer–DNA association can generate kinetically trapped complexes that resist intracellular decondensation, thereby limiting transcriptional accessibility after internalization. For this reason, plasmid-oriented micelles often rely on a balance between condensation capacity and reversibility, achieved by tuning amine density, block length, hydrophilic shielding, and the presence of degradable or environment-responsive linkages.
Another key distinction is that plasmid DNA generally requires not only protection during extracellular transport but also access to the appropriate intracellular compartment after uptake. That means micelle design must consider multiple sequential barriers: colloidal stability in buffered or protein-containing environments, resistance to premature dissociation, efficient endocytic uptake, endosomal destabilization, and subsequent decomplexation under intracellular conditions. In practice, hydrophobic core-forming segments can improve particle integrity, while detachable cationic domains, acid-labile junctions, or redox-cleavable motifs can help reduce binding strength after entry into the cell. Thus, plasmid DNA delivery by polymer micelles is best understood as a coupled problem of macromolecular condensation, transport stability, and timely intracellular unpacking rather than as a simple encapsulation task.
siRNA and Other Short Interfering RNA Cargoes
siRNA is substantially smaller than plasmid DNA, but its reduced size does not make delivery straightforward. Short duplex RNA still presents a highly anionic surface, is vulnerable to nuclease attack, and requires preservation of its structural integrity during assembly, storage, and cellular uptake. Because siRNA acts in the cytosol rather than the nucleus, the central design requirement for polymer micelles is usually to create a carrier that is sufficiently stable to protect the duplex during extracellular transit, yet sufficiently labile to release it after endosomal escape. This shifts formulation emphasis toward compact particle formation, low-polydispersity assembly, steric shielding, and efficient post-endocytic disassembly.
In many siRNA systems, the corona layer plays a particularly important role because it influences serum interaction, colloidal longevity, and nonspecific adsorption. Meanwhile, the cationic or ionizable block must be strong enough to maintain complex integrity but not so persistent that it suppresses functional release in the cytosol. Buffering capacity, membrane-disruptive segments, and pH-responsive charge transitions are often evaluated in this context because endosomal escape remains a major rate-limiting step. As a result, siRNA is often used as a model cargo for studying how polymer micelle composition affects the relationship among protection, uptake, escape, and release. A high-performing siRNA micelle is therefore not defined only by loading efficiency, but by its ability to coordinate these transport events without generating excessive aggregation or irreversible binding.
mRNA and the Need for Strong Protection with Timely Cytosolic Release
mRNA introduces an additional level of formulation complexity because it is long, structurally labile, and highly sensitive to both enzymatic degradation and processing-induced damage. Compared with shorter oligonucleotides, mRNA can be more vulnerable to shear, interfacial stress, pH extremes, and prolonged exposure to strongly cationic environments during particle formation. Polymer micelles intended for mRNA-associated delivery systems therefore need to support mild assembly conditions, minimize structural perturbation during loading, and maintain sufficient extracellular protection against hydrolysis and nuclease exposure. This often places stricter demands on solvent selection, mixing conditions, ionic strength control, and the reversibility of polymer–cargo interactions.
At the same time, strong protection alone is not enough. Since mRNA must become accessible in the cytosol, the micelle must also permit efficient release after uptake. This requirement creates a classic stability–release tradeoff: a formulation that is too loose may leak or degrade prematurely, whereas a formulation that is too stable may suppress translation-relevant availability after internalization. For that reason, mRNA-compatible micelles frequently incorporate intracellularly activated release mechanisms such as protonation-driven weakening of interactions, hydrolysable or redox-cleavable bonds, or ionizable blocks whose affinity changes across biological compartments. In practical terms, mRNA delivery pushes polymer micelle design toward architectures that combine gentle complexation, robust colloidal protection, and precisely staged disassembly behavior rather than maximum condensation strength alone.
ASOs and Other Oligonucleotide Therapeutics
Smaller oligonucleotide therapeutics, including antisense oligonucleotides (ASOs) and related short single-stranded formats, often require a different polymer micelle design logic from that used for plasmid DNA or mRNA. Because these cargos are shorter and generally impose lower condensation demands, maximizing electrostatic compaction is not always the primary objective. Instead, delivery performance may depend more strongly on controlling nonspecific interactions, maintaining colloidal stability in physiologically relevant media, and enabling productive intracellular exposure without excessive retention within overly stable complexes. In such cases, the shell chemistry, hydration behavior, and association reversibility of the micelle can become more important than absolute loading density.
Another important consideration is that smaller oligonucleotides may tolerate or even benefit from carrier systems that emphasize surface presentation, adaptive association, or transient complexation rather than deep, highly compacted sequestration. Polymer micelles can be useful here because their interfacial properties are tunable: corona composition can reduce aggregation and unwanted protein binding, while ionizable segments can modulate affinity as the particle moves across extracellular and intracellular environments. For ASOs and similar cargos, the key question is often not whether the polymer can condense the nucleic acid as tightly as possible, but whether it can provide enough protection and uptake support while still allowing the oligonucleotide to disengage in a functionally relevant time window. This makes reversibility, colloidal behavior, and microenvironment-responsive association especially important design parameters.
Why Different Nucleic Acids Need Different Micelle Designs?
The central design lesson is that polymer micelles should be matched to nucleic acid class rather than chosen as a one-size-fits-all carrier. Payload size affects condensation behavior; charge density affects complex strength; structural fragility affects loading method selection; and the intracellular site of action determines what kind of release profile is most desirable. A well-designed resource page therefore guides users toward material-property matching instead of simply listing possible cargoes.
| Nucleic Acid Type | Main Delivery Barrier | Preferred Micelle Feature | Main Release Requirement | Key Evaluation Readout |
|---|
| Plasmid DNA | Large size and difficult condensation | Strong but reversible electrostatic complexation | Release after uptake with support for downstream intracellular transport | Condensation quality, particle stability, functional release |
| siRNA | Rapid degradation and poor membrane transport | Compact PIC micelles with efficient shielding | Cytosolic release after endosomal escape | Serum stability, uptake, endosomal escape, release efficiency |
| mRNA | High instability and large hydrated structure | Protective corona with low-damage loading conditions | Fast enough cytosolic decomplexation for translation access | Integrity retention, protection, intracellular availability |
| ASO/Oligonucleotides | Modest size but limited productive uptake | Controlled association and surface shielding | Accessible intracellular release without irreversible binding | Binding balance, cellular uptake, release profile |
Looking for Custom Polymer Micelle Design for Nucleic Acid Delivery?
From cationic and ionizable block copolymers to shielding corona engineering and micelle characterization, our team supports custom polymer design and formulation development for nucleic acid delivery research.
What Micelle Structures Are Used for Nucleic Acid Delivery?
The term "polymer micelle" covers several structurally distinct assemblies in nucleic acid delivery. Clarifying these forms is essential because performance differences often originate from how the core is organized, how the shell is built, and what interactions dominate assembly. In practice, the most informative distinction is between conventional amphiphilic micelles used mainly for hydrophobic partitioning and electrostatically assembled micellar systems built around nucleic acid condensation.
Conventional Amphiphilic Polymeric Micelles
Conventional polymeric micelles are typically formed by amphiphilic block copolymers with a hydrophobic block that creates an internal core and a hydrophilic block that forms an external corona. These systems are most familiar in hydrophobic drug delivery, but they can still play a role in nucleic acid-related design when hydrophobic association stabilizes an otherwise electrostatic complex or when the micelle carries auxiliary functional components. Their main value in nucleic acid delivery is often supportive rather than primary, unless the nucleic acid is associated indirectly through conjugation or through a hybrid assembly mechanism.
Polyion Complex Micelles with Charged Core Formation
PIC micelles are especially relevant because their core forms through electrostatic association between oppositely charged species, typically a cationic polymer block and a nucleic acid cargo. A neutral hydrophilic block extends into the surrounding medium and acts as a stabilizing corona. This structure can provide a better balance between condensation and colloidal shielding than some simpler polyplex formats. Because the core is charge-derived, the assembly is sensitive to block ratio, ionic strength, pH, sequence length, and mixing order. These variables strongly influence whether the resulting micelle is compact, stable, and releasable.
Micelleplexes and Other Core-Shell Electrostatic Assemblies
The term micelleplex is commonly used for assemblies that exhibit both micellar and polyplex-like features. In these systems, electrostatic nucleic acid binding may coexist with amphiphilic stabilization, hydrophobic association, or additional crosslinking strategies. Such hybrid organization can be useful when electrostatic complexation alone does not provide adequate stability, especially under dilution or in protein-rich environments. Micelleplexes are therefore valuable when researchers need a more structurally reinforced system without abandoning the fundamental logic of charged cargo condensation.
Hybrid and Mixed Micelle Systems
Mixed micelle approaches combine two or more polymers to tune performance variables that are difficult to optimize within a single macromolecule. One component may contribute stronger nucleic acid binding, another may improve corona shielding, and a third may enhance membrane interaction or assembly robustness. This modular strategy is useful when the ideal balance between stability and release cannot be obtained from one polymer composition alone. However, mixed systems also introduce new complexity because component ratio, co-assembly efficiency, and batch reproducibility must be controlled carefully.
How Nucleic Acids Are Incorporated into Polymer Micelles?
Incorporation of nucleic acids into polymer micelles is fundamentally different from loading a hydrophobic molecule into a preformed core. The process is governed by electrostatic condensation, block organization, competing interactions with solvent and salts, and the dynamic balance between stable complex formation and later release. Understanding these loading mechanisms is essential for selecting the right polymer composition and for interpreting characterization data correctly.
Electrostatic Complexation and N/P Ratio Control
In many systems, the starting point is electrostatic complexation between the negatively charged nucleic acid and a polymer segment bearing protonatable, cationic, or permanently charged groups. The ratio between polymer amines and nucleic acid phosphates, often discussed in terms of N/P ratio, strongly affects condensation, particle size, charge exposure, and colloidal stability. A low ratio may lead to incomplete complexation and poor protection, while an excessively high ratio may increase nonspecific interactions or cause overly strong binding that slows release. Rational loading therefore depends on identifying a practical operating window rather than maximizing charge excess.
Core-Shell Organization in PIC Micelles
Once electrostatic association occurs, the polymer may reorganize into a micellar structure in which the condensed nucleic acid and charged segments occupy the core region while a neutral block forms the shell. This core-shell arrangement helps reduce direct exposure of the charged complex to the surrounding medium and can limit aggregation. The quality of this organization depends on polymer architecture, chain mobility, molecular weight distribution, and solution conditions during assembly. A well-formed shell improves colloidal persistence, while a poorly developed shell can leave the complex vulnerable to destabilization.
Role of Hydrophobic Segments in Micelle Stabilization
Hydrophobic blocks may serve several functions even when the main loading mechanism is electrostatic. They can reinforce structural cohesion, reduce exchange dynamics, and help maintain integrity under dilution. In some designs, hydrophobic association creates an additional stabilizing domain that complements the polyion complex core. The challenge is to use hydrophobicity strategically. Too little may give insufficient stabilization, while too much may increase kinetic trapping and hinder intracellular unpacking. Productive design therefore treats hydrophobic content as a structural tuning parameter, not as the primary loading principle.
Controlled Decomplexation and Intracellular Release
A polymer micelle is not successful merely because it binds nucleic acids tightly. Successful delivery also requires controlled decomplexation at the correct stage. This may be achieved through environmental pH response, ionizable groups, bond cleavage, competitive intracellular interactions, or reduction in complex stability after endosomal escape. The desired outcome is a system that remains intact during extracellular transport but becomes permissive to release once the cargo reaches a more favorable intracellular environment.
How Polymer Micelles Are Designed for Effective Nucleic Acid Delivery?
Polymer micelle design for nucleic acid delivery is a problem in balancing multiple coupled variables rather than optimizing a single parameter. The carrier must condense the cargo, maintain a stable colloidal interface, respond appropriately to biological conditions, and avoid becoming so inert that release never occurs. This makes block copolymer design, architecture control, and functional segment selection central to platform performance. A strong materials strategy considers the full transport pathway from initial assembly through intracellular release.
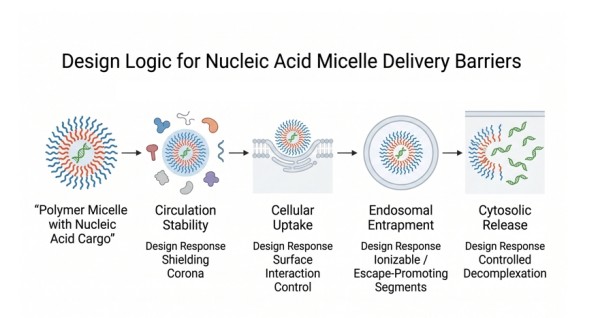 Fig. 2. Design logic for nucleic acid micelle delivery barriers (BOC Sciences Authorized).
Fig. 2. Design logic for nucleic acid micelle delivery barriers (BOC Sciences Authorized).
Cationic versus Ionizable Segments
Cationic polymer segments can provide strong nucleic acid condensation and facilitate assembly, but permanently high charge density may also increase undesirable interactions in complex media. Ionizable segments offer an alternative strategy by remaining less interactive under some conditions and becoming more protonated in acidic environments. This can improve the balance between extracellular compatibility and intracellular activation. From a materials standpoint, the choice between cationic and ionizable blocks should be based on the intended loading environment, desired stability profile, and release trigger strategy rather than on charge strength alone.
Hydrophilic Corona Design for Shielding and Circulation
The hydrophilic corona is a major determinant of micelle behavior in aqueous media. It limits interparticle association, reduces direct interaction between the condensed core and surrounding proteins or salts, and helps preserve a more uniform colloidal state. Corona design involves more than selecting a hydrophilic polymer. Chain length, density, architecture, and compatibility with the core region all affect whether the shell provides effective shielding without impeding required cellular interaction. In practice, the corona should be optimized for protection and persistence while still allowing productive uptake when the system reaches its target environment.
Hydrophobic Blocks for Structural Stability
Incorporating hydrophobic segments into a nucleic acid micelle can stabilize the assembly, slow disintegration under dilution, and help define the morphology of the nanoparticle. However, hydrophobic stabilization must be adjusted carefully. Excessive hydrophobic content may lead to particles that are too rigid or too slow to release their cargo. Moderate hydrophobic reinforcement is often more useful than dominant hydrophobic driving force, especially for systems intended to decomplex in response to intracellular conditions.
Stimuli-Responsive Elements for Triggered Release
Responsive chemistry can be introduced to support release at a more favorable stage of the delivery sequence. Acid-labile, redox-sensitive, hydrolytically cleavable, or ionization-dependent motifs may be incorporated into the polymer backbone, side chains, or linkers. The purpose is not simply to make the carrier "smart," but to decouple extracellular stability from intracellular release. Properly chosen stimuli-responsive features allow the micelle to remain robust during handling and transport while becoming more permissive to nucleic acid release after environmental change.
Design Strategies for Endosomal Escape
Endosomal escape remains a major performance bottleneck. Polymer chemists often address this through buffering groups, membrane-active segments, ionizable domains, or architectures that alter interfacial behavior under acidic conditions. The most effective strategy depends on the nucleic acid class and the rest of the micelle design. In some systems, escape is promoted by protonation-driven changes in osmotic or membrane interaction behavior; in others, a hydrophobic or conformational transition helps destabilize the endosomal membrane. What matters is that the design strategy aligns with the assembly's overall stability and release profile.
How Nucleic Acid-Loaded Polymer Micelles Are Prepared?
Preparation methods strongly influence the final architecture of nucleic acid-loaded polymer micelles. Because these systems depend on delicate balances among electrostatic attraction, chain mobility, ionic strength, and hydrophobic association, processing history can alter particle size, dispersity, shell development, and binding strength even when the same polymer and cargo are used. For this reason, preparation is not a secondary procedural detail but a fundamental part of platform design.
Direct Complexation in Aqueous Media
One of the most common routes is direct mixing of polymer and nucleic acid in aqueous solution. This approach can be simple and gentle, particularly for sensitive RNA cargoes, but it is highly sensitive to concentration, mixing rate, ionic background, and order of addition. Rapid local charge neutralization may create heterogeneous structures if mixing is not controlled. When optimized, however, direct complexation can generate reproducible PIC micelles with minimal solvent burden and a relatively straightforward workflow.
Dialysis and Solvent Exchange Methods
Dialysis and solvent exchange are useful when the polymer contains both charged and hydrophobic segments that require a more gradual transition into the final assembly environment. These methods can promote more ordered self-assembly and may help produce narrower particle distributions. Their main trade-off is that process duration and solvent history must be managed carefully, especially for fragile nucleic acids. The advantage lies in greater control over how the polymer reorganizes as solvent quality changes.
Thin-Film or Post-Assembly Loading Routes
In some cases, polymers are pre-organized or partially assembled before nucleic acid association is introduced. Post-assembly strategies can be useful when one wants to combine hydrophobic stabilization with subsequent electrostatic loading, but they are not universally appropriate. The feasibility depends on whether the preformed structure remains accessible to the nucleic acid and whether loading occurs without destabilizing the micelle. These approaches are most valuable when the final platform is intentionally hybrid in nature.
Mixed Micelle and Microfluidic Assembly Approaches
Mixed polymer systems may be assembled by co-dissolution, staged mixing, or controlled co-injection. Microfluidic approaches are particularly attractive when precise control over local mixing and reproducibility is required. By narrowing the distribution of assembly conditions, microfluidic preparation can reduce batch variability and provide a more reliable basis for comparing formulations. This is especially important for nucleic acid systems, where subtle differences in mixing history can lead to major differences in performance.
How to Characterize Nucleic Acid-Loaded Polymer Micelles?
Characterization of polymer micelles for nucleic acid delivery must go beyond confirming nanoscale size. Because the platform succeeds only if it protects cargo, persists under relevant conditions, enters cells, and eventually releases the nucleic acid, evaluation should connect particle properties to functional delivery behavior. A well-designed characterization workflow therefore combines colloidal measurements, binding analysis, stability testing, and biologically relevant readouts.
Particle Size, PDI, and Surface Charge
Dynamic light scattering and related methods remain useful because they reveal whether the micelles fall within a relevant size range and whether the population is reasonably uniform. Surface charge also provides clues about the degree of shielding and the likely interfacial behavior of the particles. However, these values must be interpreted with caution. A particle can appear well sized in simple buffer yet perform poorly if the charge is overly exposed or if the assembly reorganizes in salt-containing or protein-rich media.
Nucleic Acid Binding, Condensation, and Protection
A more discriminating analysis examines whether the nucleic acid is effectively condensed, retained, and protected. Binding studies, gel-based assays, and nuclease exposure tests help determine whether the micelle provides meaningful cargo protection without creating irreversible sequestration. These measurements are especially important because they capture a feature that cannot be inferred reliably from size alone: the quality of the polymer-cargo interaction.
Serum Stability, Dilution Stability, and Structural Robustness
Micelles intended for realistic use conditions should be examined under dilution and in media containing salts or biomolecular components. Such testing helps reveal whether the corona remains effective, whether aggregates form, and whether the nucleic acid stays associated. Dilution stability is especially important for micellar systems because self-assembled structures can reorganize when concentration changes. In nucleic acid micelles, this issue is compounded by charge screening and competitive interactions.
Cellular Uptake, Endosomal Escape, and Release Behavior
Ultimately, characterization should include functional evaluation related to uptake and release. A platform that protects nucleic acid perfectly but fails to release it productively is not well optimized. Fluorescence-based trafficking studies, endosomal localization analysis, and release-sensitive assays help determine whether the micelle supports the intended intracellular sequence. These studies are often where differences among apparently similar formulations become most visible.
When Polymer Micelles Are the Right Choice for Nucleic Acid Delivery?
Polymer micelles are one option within a broader non-viral delivery toolbox, and they are most valuable when their structural advantages match the needs of the nucleic acid cargo. A useful conclusion for this page is therefore not that polymer micelles are always superior, but that they become highly relevant when a project requires a tunable balance among condensation, shielding, structural stability, and controlled release. Platform selection should follow the properties of the payload and the barriers it must overcome.
When Micelles Offer Clear Advantages over Simpler Polyplexes
Polymer micelles are especially attractive when direct polyplexes do not provide enough colloidal refinement or surface shielding. The addition of a stabilizing corona can reduce aggregation tendency, improve particle persistence, and create a more tunable interface. Micellar design is also useful when the platform needs multiple coordinated functions, such as nucleic acid condensation, responsive release, and shell-mediated stability in the same structure.
When Lipid Systems or Other Carriers May Be More Suitable
In some cases, lipid systems, simpler polyplexes, or other nanocarriers may be more straightforward choices. If a project does not benefit from micellar corona design or if the selected nucleic acid and route of use are better matched to another carrier logic, forcing a micelle format may add unnecessary complexity. Good platform selection therefore depends on matching assembly mechanism to delivery need rather than on default preference for one carrier family.
Payload-Barrier-Platform Matching Principles
The most reliable way to decide whether polymer micelles are appropriate is to analyze the payload and the main delivery bottlenecks together. Large or fragile cargoes may benefit from gentle loading and strong protection. Highly dilution-sensitive systems may need hydrophobic reinforcement or improved corona design. Cargoes requiring rapid cytosolic release may benefit from ionizable or stimuli-responsive segments. In every case, the question is whether the micellar structure solves a real barrier more effectively than a simpler alternative.
What to Check Before Choosing a Micellar Delivery Platform
Before committing to a polymer micelle strategy, researchers should evaluate whether the polymer can provide the right balance of binding and release, whether the intended preparation route is compatible with the nucleic acid, whether the corona is adequate for the relevant environment, and whether the proposed endosomal escape logic is realistic. These checks help prevent a common mistake in formulation development: selecting a visually appealing nanostructure without confirming that its transport logic matches the cargo's actual needs.
Services
Advanced Polymer Micellar Synthesis and Formulation Services
At BOC Sciences, we support nucleic acid delivery research by combining polymer synthesis, block copolymer design, functional modification, and formulation-focused materials characterization. For polymer micelle projects, our team can help tailor charged, ionizable, hydrophilic, hydrophobic, biodegradable, or stimuli-responsive polymer segments to align with specific condensation, shielding, and release requirements. We also support the development of copolymers, functional polymers, and custom self-assembling systems prepared through controlled polymerization and post-polymer modification workflows, helping researchers refine both material structure and delivery performance.

Custom Polymer & Copolymer Synthesis
- Design of cationic, ionizable, amphiphilic, and biodegradable polymer backbones.
- Control over molecular weight, dispersity, and block composition for self-assembly behavior.
- Support for linear, block, graft, and multi-segment polymer architectures.
- Development of materials suitable for PIC micelles and micelleplex systems.

Polymer Functionalization & Bioconjugation
- Introduction of charged, ionizable, hydrophilic, or responsive side groups.
- End-group modification for ligand attachment or interface tuning.
- Linker design for controlled destabilization or triggered release behavior.
- Optimization of corona-forming segments for shielding and colloidal stability.

Nanocarrier Formulation Development
- Optimization of polymeric micelles, PIC micelles, and mixed micelle systems.
- Adjustment of assembly parameters including ratio, solvent history, and mixing conditions.
- Support for particle size, PDI, zeta potential, and stability optimization.
- Evaluation of formulation robustness and release-oriented design logic.

Polymer Characterization & Supply Support
- Material analysis through structure and molecular property assessment.
- Support for custom polymer supply from exploratory to larger research quantities.
- Assistance with reproducible batch design for formulation studies.
- Integration of polymer development with downstream delivery platform evaluation.
Do You Need A Consultation?
BOC Sciences supports polymer micelle design for nucleic acid delivery with tailored materials, formulation insight, and structure-focused development strategies.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What is the difference between a polymer micelle and a PIC micelle for nucleic acid delivery?
A classical polymer micelle is mainly organized by amphiphilic self-assembly around a hydrophobic domain, while a PIC micelle forms a core through electrostatic association between a charged polymer segment and a nucleic acid. For nucleic acid delivery, PIC micelles are often more relevant because condensation and shielding are central design requirements.
-
Why are polymer micelles used for siRNA or mRNA delivery?
They can help condense and protect fragile nucleic acids, reduce direct charge exposure, and provide a tunable shell that improves colloidal behavior. Properly designed micelles may also support endosomal escape and controlled intracellular release, which are both critical for productive RNA delivery.
-
Are stronger cationic polymers always better for nucleic acid micelles?
No. Stronger binding may improve condensation and protection, but it can also make intracellular release more difficult and increase undesirable interactions. The better design is usually one that balances stability with timely decomplexation rather than maximizing charge interaction alone.
-
What should be measured when evaluating nucleic acid-loaded micelles?
Useful evaluation goes beyond size and zeta potential. Researchers should also assess nucleic acid binding, condensation quality, protection against degradation, stability under dilution and relevant media conditions, uptake behavior, endosomal escape, and the ability of the cargo to become accessible after internalization.
-
When are polymer micelles preferred over simpler polyplexes?
They are often preferred when a project needs stronger colloidal control, a protective hydrophilic corona, or a more tunable combination of stability and release. If a simple polyplex already provides sufficient performance, a micellar system may not always be necessary.
-
Can polymer micelles be designed for both nucleic acid loading and triggered release?
Yes. This is commonly approached by combining charged or ionizable binding segments with degradable, pH-responsive, redox-responsive, or other stimuli-sensitive features. The goal is to maintain cargo association during transport while making release more favorable under intracellular conditions.

 Fig. 1. Comparison of classical and PIC micelle architectures (BOC Sciences Authorized).
Fig. 1. Comparison of classical and PIC micelle architectures (BOC Sciences Authorized). Fig. 2. Design logic for nucleic acid micelle delivery barriers (BOC Sciences Authorized).
Fig. 2. Design logic for nucleic acid micelle delivery barriers (BOC Sciences Authorized).