Polymer Micelles vs Polymer Microspheres vs Polymer Nanoparticles: How to Choose the Right Drug Delivery Platform
Selecting the right polymer-based delivery platform is rarely a matter of choosing the most advanced material or the smallest particle size. In practice, the decision depends on how the carrier is built, how the payload is incorporated, how release is expected to occur, and how the formulation will be prepared, stored, and reproduced. Polymer micelles, polymer microspheres, and polymeric nanoparticles are often discussed together because all three can improve payload handling and controlled delivery, yet they operate through fundamentally different structural and process logics.
Resources
How to Choose the Right Polymer-based Delivery Platform?
Although polymer micelles, polymer microspheres, and polymeric nanoparticles all belong to the broader family of polymer-based delivery systems, they should not be treated as interchangeable. Polymer micelles are typically formed through the self-assembly of amphiphilic chains into soft nanoscale core-shell colloids. Polymer microsphere systems are usually matrix-based or reservoir-like particles in the micrometer range and are often selected when sustained retention or prolonged release is required. Polymer nanoparticles occupy a broader category that includes nanoscale solid or semisolid carriers engineered through precipitation, emulsification, or related processes.
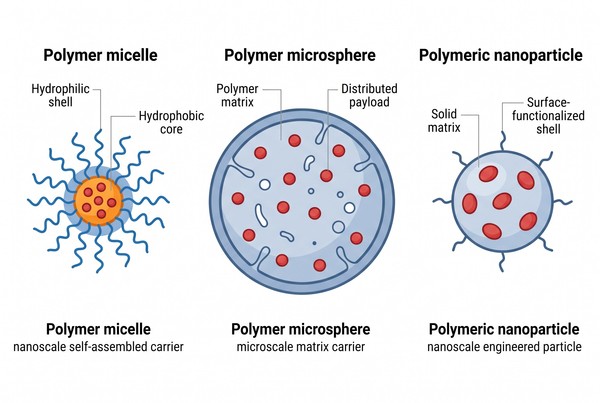 Fig. 1. Structural comparison of micelles, microspheres, and nanoparticles (BOC Sciences Authorized).
Fig. 1. Structural comparison of micelles, microspheres, and nanoparticles (BOC Sciences Authorized).
What Defines a Polymer Micelle?
A polymer micelle is a self-assembled colloid that forms when amphiphilic macromolecules are placed in a selective solvent and spontaneously segregate into a solvophobic core and a solvated shell. This architecture is especially useful when a hydrophobic internal domain is needed to host poorly water-soluble compounds while the shell maintains aqueous dispersibility and steric stabilization. Because micelles are dynamic assemblies rather than rigid particles, their behavior is closely tied to polymer composition, critical micelle concentration, chain exchange kinetics, and the preparation pathway used during formation. From an application standpoint, polymer micelles are particularly well suited to nanoscale solubilization tasks, especially for hydrophobic small molecules that require improved apparent aqueous compatibility without being locked into a dense solid matrix. They are also attractive when relatively small particle size, soft colloidal behavior, and tunable surface hydration are important, such as in systems that emphasize dispersion stability, interfacial stealth properties, or flexible incorporation of functional surface groups.
What Defines a Polymer Microsphere?
Polymer microspheres are generally micrometer-scale particles in which the payload is embedded within a polymer matrix or distributed in a reservoir-like internal structure. They are not defined by self-assembly in the same sense as polymer micelles. Instead, they are more commonly produced through emulsification, solvent evaporation, spray drying, or related solidification approaches. Their larger size and matrix-based organization make them especially relevant when a formulation must slow release over a longer time window, remain localized, or function as a durable particulate depot rather than a highly dynamic nanoscale colloid. In practical application, polymer microspheres are often favored when long-acting release, site retention, or matrix-governed payload diffusion is more important than achieving the smallest possible particle size. They are therefore especially useful in systems where prolonged release kinetics, reduced dosing frequency, local delivery persistence, or sustained matrix erosion behavior are desired, and where a robust particulate depot offers more benefit than rapid nanoscale redistribution.
What Defines a Polymeric Nanoparticle?
Polymeric nanoparticles form a broader category than polymer micelles because they can include nanospheres, nanocapsules, and other solid or semisolid nanoscale carriers with diverse internal structures. Some are matrix-like, some contain internal cavities, and some are surface-modified to tune interfacial behavior. Their advantage lies in engineering flexibility: size, surface charge, porosity, degradation profile, and loading mode can often be tuned more independently than in simple micellar systems. This makes polymeric nanoparticles particularly valuable when the payload, release profile, or particle surface requirements extend beyond what soft self-assembled micelles can reliably provide. In application terms, they are often selected when the development goal requires stronger structural definition at the nanoscale, broader compatibility with different classes of payloads, or more extensive control over particle surface properties and release behavior. They are especially useful when nanoscale delivery must be combined with matrix protection, controlled degradation, surface functionalization, or more versatile loading options than hydrophobic core partitioning alone can offer.
Why These Platforms Are Structurally Different Rather Than Simply Different in Size?
It is tempting to compare these carriers only by particle diameter, but size alone does not capture their most important distinctions. The defining difference is structural state. Polymer micelles are soft self-assembled colloids whose internal organization depends on amphiphilic segregation in solution. Polymeric nanoparticles are usually more solid or semisolid nanoscale constructs with stronger particle identity. Microspheres are larger matrix-based structures designed to hold and release payloads through diffusion, polymer relaxation, and degradation over longer timescales. Once this distinction is clear, differences in loading, release, and process behavior become easier to understand.
Self-Assembled Soft Carriers vs Matrix-Based Particles
Polymer micelles derive their structure from reversible self-assembly in solution, which means their colloidal identity is thermodynamically and kinetically linked to solvent conditions and polymer architecture. By contrast, polymer microspheres and many polymeric nanoparticles are more strongly defined by the fabrication route and the resulting solidified particle structure. This matters because self-assembled micelles can reorganize, exchange polymer chains, or release weakly associated cargo more readily than matrix-based particles. In other words, the softness and reversibility of the micelle are not minor details; they are central to how the system behaves under dilution, storage, and biological exposure.
Nanometer-Scale Systems vs Micrometer-Scale Systems
Particle size affects more than transport distance or visual classification. Nanoscale carriers such as polymer micelles and polymeric nanoparticles present a high interfacial area and can be engineered for rapid dispersion, colloidal suspension, and broad surface accessibility. Micrometer-scale carriers such as microspheres create a very different mass-transport regime: diffusion distances are longer, matrix erosion becomes more relevant, and local retention can be more pronounced. As a result, microspheres are often a better fit when the goal is prolonged, matrix-controlled release, whereas nanoscale systems are more often selected when dispersibility, small size, or interfacial engineering are primary design priorities.
Why Structural State Changes Downstream Formulation Logic?
Once the carrier structure is fixed, the rest of the formulation logic follows. A self-assembled micelle requires attention to amphiphilic balance, dilution stability, core compatibility, and chain dynamics. A nanoparticle requires attention to particle formation, surface engineering, and structural integrity. A microsphere requires control of matrix architecture, residual solvent, particle size distribution, and release heterogeneity across larger particles. These are not merely different laboratory workflows; they are different development philosophies. Choosing the wrong platform at the outset can force the project into unnecessary complexity or lock the formulation into a release mechanism that is poorly matched to the payload.
How Payload Loading Works in Each Platform?
Payload incorporation is one of the clearest ways to distinguish these systems. Each platform creates a different internal environment and therefore supports a different loading logic. Polymer micelles usually rely on partitioning into a hydrophobic core, polymeric nanoparticles allow broader matrix and surface loading options, and microspheres typically depend on entrapment within a larger polymer matrix. These differences strongly affect loading efficiency, release profile, structural stability, and the kinds of compounds that can be handled without major reformulation.
Core Partitioning in Polymer Micelles
In polymer micelles, payload loading usually occurs through partitioning of a poorly water-soluble molecule into the hydrophobic core during or after self-assembly. This mode is particularly useful for compounds that benefit from a nanoscale hydrophobic microenvironment but do not require a rigid particle matrix for protection. The quality of loading depends on drug-polymer compatibility, core mobility, and how the micelle was prepared. If compatibility is weak, the system may still appear colloidally stable while showing low retained loading or rapid leakage. For this reason, loading in micelles should be understood as a thermodynamic and kinetic problem, not merely as a solubilization event.
Matrix and Surface Loading in Polymeric Nanoparticles
Polymeric nanoparticles offer broader loading pathways. Depending on particle design, the payload may be molecularly dispersed in a solid matrix, localized in a nanocapsule-like compartment, adsorbed at the surface, or chemically associated with the particle-forming polymer. This flexibility is one reason nanoparticles are often selected when the project requires more than simple hydrophobic core loading. However, that broader loading freedom comes with a greater burden of process optimization. Nanoprecipitation conditions, solvent removal, polymer concentration, and surface chemistry can all alter how much payload is incorporated and whether the resulting structure is robust enough for downstream use.
Matrix Entrapment in Polymer Microspheres
Polymer microspheres usually load payloads by entrapment within a larger polymer matrix during emulsification, phase separation, spray drying, or related particle-forming processes. The matrix can be designed to slow release over a relatively long time by increasing diffusion distance and controlling polymer degradation or erosion. This is a very different loading logic from micelles: instead of creating a nanoscale internal core for partitioning, microspheres create a bulk polymer environment that physically retains the compound within a larger structure. That can be highly effective for sustained release, but it also makes microsphere formation more sensitive to solidification conditions, particle porosity, and internal payload distribution.
| Platform | Structural State | Typical Size Range | Main Loading Logic | Main Release Logic | Best-Fit Use Case | Main Limitation |
|---|
| Polymer Micelles | Soft self-assembled core-shell colloids | Nanoscale | Hydrophobic core partitioning | Micelle dissociation, diffusion, repartitioning | Poorly soluble small molecules and soft nanodispersion | Dilution sensitivity and cargo leakage risk |
| Polymer Microspheres | Micrometer-scale matrix or reservoir particles | Microscale | Matrix entrapment | Diffusion, erosion, degradation, depot-like release | Longer-duration release and local retention | Broader particle size and slower process development |
| Polymeric Nanoparticles | Solid or semisolid nanoscale particles | Nanoscale | Matrix, cavity, or surface-associated loading | Diffusion, swelling, degradation, surface-controlled effects | Broad payload fit and surface-engineered nanocarriers | Higher structural and process complexity |
How Release Behavior and Stability Differ Across the Three Systems?
Release performance is rarely determined by one variable alone. It emerges from the coupling of carrier structure, payload location, and the environment surrounding the particle. Polymer micelles, microspheres, and polymeric nanoparticles all control release differently because their internal organizations are different. Understanding those release mechanisms is essential before choosing a carrier platform, especially when the project requires either immediate colloidal performance, intermediate structural protection, or extended matrix-mediated release.
Release from Polymer Micelles
Micellar release is usually influenced by three related processes: diffusion of the payload out of the core, repartitioning between the core and surrounding medium, and changes in micelle integrity under dilution or environmental stress. This means polymer micelles are especially effective when the payload benefits from core solubilization and moderate nanodispersion control, but they can become less reliable when very prolonged retention is required without additional stabilization. Core chemistry, block ratio, and preparation method all matter because they govern how tightly the cargo is held within the hydrophobic domain and how quickly the assembly can reorganize.
Release from Polymeric Nanoparticles
In polymeric nanoparticles, release is often more closely tied to particle matrix properties, polymer degradation, swelling behavior, porosity, and interfacial transport. Because the particle identity is usually stronger than in micelles, the system can provide more structural protection for the payload and more opportunities to modulate release using polymer composition or surface design. At the same time, the release profile can become more complex because multiple transport mechanisms may operate simultaneously. This makes polymeric nanoparticles attractive when the formulation requires more than rapid nanodispersion, but it also increases the analytical burden during optimization.
Release from Polymer Microspheres
Microspheres are often selected specifically because they can create longer diffusion pathways and stronger matrix-based retention than nanoscale systems. Release commonly reflects a combination of initial diffusion, matrix relaxation, pore formation, and polymer degradation or erosion, depending on the material and fabrication method. This makes microspheres especially valuable when the formulation goal emphasizes sustained release rather than nanoscale solubilization. The tradeoff is that release heterogeneity can become significant if particle size distribution, internal porosity, or payload distribution vary across the microsphere population, which means process control is central to microsphere performance.
Need help selecting the right polymer delivery platform?
BOC Sciences provides material screening, custom polymer design, and formulation support for polymer micelles, polymer microspheres, and polymeric nanoparticles based on payload type, release profile, and process requirements.
How Preparation and Manufacturing Logic Compare?
Preparation route is often the hidden factor that determines whether a platform choice remains practical. The same payload may appear compatible with more than one delivery system on paper, yet process requirements can quickly narrow the realistic options. Polymer micelles are often favored when self-assembly can be achieved through relatively mild solvent transition routes. Polymeric nanoparticles demand stronger control over precipitation or particle formation. Microspheres require solidification or matrix-forming methods that are suitable for larger particles and longer release objectives. Comparing manufacturing logic early helps prevent unnecessary redevelopment later.
Typical Preparation of Polymer Micelles
Polymer micelles are often prepared by direct dissolution, dialysis, solvent exchange, thin-film hydration, or related routes that guide amphiphilic polymers into self-assembled core-shell structures. The advantage of these methods is that they can be operationally mild and particularly suitable for hydrophobic-core loading. However, preparation route strongly affects size distribution, loading retention, and kinetic stability. Because micelles remain soft self-assembled colloids rather than rigid particles, their final properties are tightly linked to how solvent quality changes during formation. Process selection is therefore inseparable from material selection when micelles are under consideration. From an application perspective, these routes are especially valuable when the formulation goal is to improve the apparent solubility of poorly water-soluble small molecules, maintain nanoscale dispersion, or create a soft carrier with tunable surface hydration. In such cases, the chosen preparation pathway can directly influence whether the final micellar system remains suitable for colloidal delivery, functional surface modification, or controlled incorporation of hydrophobic actives.
Typical Preparation of Polymeric Nanoparticles
Polymeric nanoparticles are often prepared by nanoprecipitation, emulsification-solvent evaporation, ionic or polymeric complexation, and other particle-forming routes that create a stronger nanoscale entity than a simple self-assembled micelle. This broader process toolbox allows more control over particle structure and surface properties but also creates more process variables. Solvent choice, mixing intensity, stabilizer content, polymer concentration, and purification strategy can all alter the final particle identity. As a result, nanoparticles may provide broader engineering flexibility than micelles, but they often demand more extensive process refinement before the system becomes reproducible. In application terms, this added process complexity is often justified when the payload requires stronger nanoscale protection, more versatile loading modes, or more deliberate surface engineering than micelles can usually offer. Polymeric nanoparticles are therefore especially useful in projects where delivery performance depends on matrix integrity, controlled degradation, interfacial functionality, or broader compatibility with structurally diverse payloads rather than on hydrophobic core solubilization alone.
Typical Preparation of Polymer Microspheres
Polymer microspheres are usually produced through matrix-forming or solidification approaches such as emulsification, solvent removal, phase separation, or spray-drying routes. Because the goal is to create micrometer-scale particulate reservoirs rather than soft nanoscale assemblies, process variables such as droplet size control, solidification rate, matrix porosity, and residual solvent become especially important. Microsphere preparation can be more demanding when narrow particle distributions or uniform internal loading are required, but the resulting platform can be far more suitable when prolonged release is a primary objective. This is why microspheres often remain competitive despite their larger size and more involved fabrication. From an application standpoint, these manufacturing routes are most valuable when the delivery system must support extended release, local retention, or depot-like behavior over a longer timescale. In such settings, the matrix architecture created during microsphere fabrication directly determines diffusion distance, erosion behavior, and release uniformity, making process control essential for applications where sustained performance matters more than nanoscale dispersion.
Which Payloads and Development Goals Fit Best?
Platform choice becomes much clearer when the decision starts with the payload and the intended release goal instead of with the carrier name. Some systems primarily need better dispersion of a poorly soluble small molecule. Others require stronger structural protection, more surface engineering, or a longer release window. Still others must remain compatible with a very specific manufacturing or storage strategy. These differences mean the same platform should not be recommended for every payload merely because it performs well in one category.
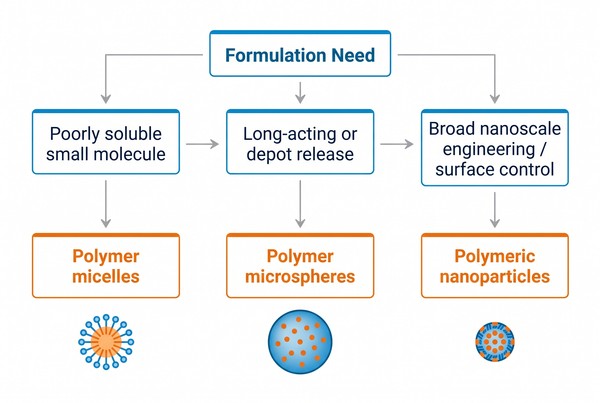 Fig. 2. Selection framework for polymer drug delivery platforms (BOC Sciences Authorized).
Fig. 2. Selection framework for polymer drug delivery platforms (BOC Sciences Authorized).
Poorly Water-Soluble Small Molecules
Polymer micelles are often the most intuitive starting point for poorly water-soluble small molecules because the hydrophobic core provides a direct route to apparent solubility enhancement and nanoscale dispersion. When the project is centered on hydrophobic loading and relatively soft colloidal delivery, micelles can offer a more elegant solution than larger matrix particles. That said, if the compound requires stronger structural retention, more aggressive surface engineering, or less dynamic release behavior, polymeric nanoparticles may become the more robust option. The best choice depends on whether the challenge is mainly solubilization or broader structural control. In application terms, polymeric micelles for poorly soluble drugs are especially attractive when rapid formulation screening, improved aqueous handling, and nanoscale delivery of lipophilic actives are the primary objectives. Polymeric nanoparticles become more relevant when the same class of compounds also requires stronger particle integrity, more tunable release, or more deliberate surface functionality, while polymer microspheres for long-acting drug delivery are generally considered only when the small molecule must be delivered over a prolonged period through matrix-controlled retention rather than through nanoscale colloidal dispersion.
Proteins, Peptides, and More Sensitive Payloads
More sensitive payloads such as proteins and peptides often impose stricter requirements on preparation conditions, interfacial exposure, and structural protection. In such cases, polymeric nanoparticles or specially engineered matrix systems may offer better control than simple hydrophobic-core micelles, particularly when the formulation must avoid destabilizing interfaces or weakly retained loading states. Microspheres can also be valuable when a longer local release window is needed, provided the fabrication route does not compromise payload integrity. This means the platform decision should weigh not only release duration but also how the payload experiences the preparation pathway itself. From an application perspective, this category is less about maximizing loading through a hydrophobic domain and more about preserving structural integrity during fabrication, storage, and release. Polymeric nanoparticles are often favored when a sensitive payload needs nanoscale protection and surface-tunable delivery behavior, whereas microspheres may be selected when sustained local exposure is more important than nanoscale circulation. Polymer micelles can still be relevant, but usually only when the system is specifically designed to avoid destabilizing interfaces and to support gentle association or encapsulation conditions, as seen in polymer carriers for protein and peptide drug delivery.
Long-Acting or Depot-Oriented Objectives
When the development target is sustained release over an extended period, matrix-forming systems usually become more attractive than dynamic self-assembled micelles. Microspheres are often strong candidates here because their larger size and polymer matrix can create slower transport pathways and longer retention. Polymeric nanoparticles for long-acting drug delivery can also support sustained release in some designs, especially when matrix degradation and particle architecture are engineered carefully. Micelles, by contrast, usually require additional stabilization or specialized design to reach similar persistence. For this reason, the intended release window is one of the most decisive questions in choosing among these platforms. In practical application, polymer microspheres are often the most natural fit when the goal is depot-like behavior, prolonged matrix-mediated diffusion, or reduced dosing frequency over a relatively long period. Polymeric nanoparticles may be chosen instead when sustained release is still needed but must be combined with nanoscale particle behavior or more advanced interfacial engineering. Polymer micelles are generally less suitable as the default option in this category unless the project specifically benefits from a soft nanocarrier and can tolerate the additional design work needed to improve long-term retention.
A Practical Selection Framework for Early Development
Early platform selection is most effective when it follows a structured logic instead of broad preference. A useful framework begins with the payload, then moves to the required release behavior, and finally considers process complexity and reproducibility. This order matters because many formulation difficulties arise from trying to force a payload into a structurally unsuitable carrier. The goal is not to prove one platform superior to the others, but to reduce mismatch between the carrier's intrinsic behavior and the project's actual development target.
Start with the Payload
The first question should always be what the payload most urgently needs from the carrier. If the primary problem is poor aqueous solubility for a hydrophobic small molecule, a micellar core may be sufficient and highly efficient. If the payload requires stronger protection, more elaborate surface control, or a more matrix-like environment, polymeric nanoparticles or microspheres may be more appropriate. Starting with the payload keeps the project anchored to the real formulation bottleneck instead of to the appeal of a particular platform name. In application terms, this means formulation selection should begin with whether the cargo needs solubilization, structural shielding, prolonged retention, or controlled exposure. A small hydrophobic compound with low apparent solubility may justify starting from polymer micelles, while a more sensitive or structurally complex payload may immediately shift the platform toward nanoparticles or microspheres because the application demand is driven less by dispersion and more by preservation, localization, or sustained release.
Match the Needed Release Window
The second question is how long the formulation must retain and release the payload. If the goal is soft nanoscale solubilization and moderate release control, polymer micelles may fit naturally. If the goal is stronger nanoscale retention or engineered particle-mediated release, polymeric nanoparticles are often preferable. If the project requires extended or depot-like release, microspheres generally deserve priority consideration. This step is crucial because release mechanism is one of the hardest variables to retrofit after the platform has already been chosen. From an application perspective, release window often determines whether the system should behave as a transient nanocarrier, a more persistent engineered particle, or a long-acting matrix reservoir. Choosing the wrong release architecture early can lead to repeated reformulation later, especially when the intended use case depends on prolonged exposure, localized retention, or reduced dosing frequency rather than on rapid nanoscale delivery alone.
Check Process Burden and Platform Transferability
The third question concerns process reality. A platform that performs well only under a narrow, operator-sensitive preparation window may not be the most strategic choice, even if early laboratory data look promising. Micelles can be elegant but may be sensitive to dilution and loading leakage. Nanoparticles can be highly tunable but process-intensive. Microspheres can support long release but require more attention to particle-size distribution and matrix quality. Comparing process burden early helps determine whether the selected system is likely to remain workable beyond initial proof-of-concept experiments. In practical application, this consideration becomes especially important when the project is expected to move from exploratory formulation toward reproducible development. A platform that looks ideal for a small feasibility study may become less attractive if it is difficult to reproduce, purify, dry, redisperse, or scale in a consistent way for the intended delivery setting.
Know When to Switch Platforms
One of the most useful development skills is recognizing when a carrier should no longer be optimized and should instead be replaced. If a micellar system consistently shows insufficient retention, switching to nanoparticles may be more productive than endless micelle reformulation. If a nanoparticle system cannot produce the required release duration, a microsphere route may be the more rational move. Platform switching is not a failure; it is a design decision that acknowledges structural mismatch between the carrier and the actual formulation need. In application terms, this step prevents a project from being trapped in the wrong delivery logic simply because the first platform appeared promising at an early stage. A rational switch can save time and improve technical fit when the application ultimately requires stronger matrix control, more persistent release, more reliable nanoscale protection, or a different balance between dispersibility and retention.
Services
Advanced Polymer Synthesis and Formulation Services
BOC Sciences supports platform selection and formulation development across polymer micelles, polymer microspheres, and polymeric nanoparticles from a material-design perspective. Our capabilities cover custom polymer and copolymer synthesis, architecture optimization, end-group functionalization, route-specific formulation planning, and process-oriented material screening. Whether the goal is amphiphilic self-assembly, matrix-based sustained release, or nanoscale particle engineering, we help align polymer composition, fabrication logic, and formulation targets so that material choice remains consistent with real development requirements rather than with generic platform assumptions.

Custom Polymer & Copolymer Design
- Design of amphiphilic polymers for micelle-forming systems and controlled nanoscale assembly.
- Selection of matrix-forming materials for microsphere and nanoparticle fabrication.
- Support for architecture control, molecular weight adjustment, and functional group placement.
- Optimization of ATRP and RAFT polymerization routes when controlled polymer structures are required.

Material Screening & Platform Matching
- Screening of polymers for self-assembly, particle formation, and matrix-retention behavior.
- Matching of polymer type with micelle, microsphere, or nanoparticle fabrication logic.
- Selection support for biodegradable polymers used in sustained-release and particle-based systems.
- Evaluation of solubility, process compatibility, and carrier fit before formulation refinement.

Formulation Development Support
- Optimization of self-assembly routes for polymer micelles.
- Process planning for polymer microsphere systems intended for prolonged release.
- Particle-engineering support for polymer nanoparticles with tailored size and interfacial behavior.
- Integration of material selection, loading logic, and fabrication route into a reproducible formulation strategy.

Analytical Characterization Support
- Comparative analysis of polymers, micelles, microspheres, and nanoparticle systems from a structure-property perspective.
- Assessment of process-dependent particle size, matrix quality, and colloidal behavior.
- Support for route comparison, formulation consistency, and redispersion or release-related evaluation.
- Documentation assistance to improve reproducibility and platform-transfer readiness.
Do You Need A Consultation?
Discuss your delivery platform strategy with BOC Sciences to identify the right balance among polymer micelles, polymer microspheres, and polymeric nanoparticles for your formulation and process goals.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
What is the main difference between polymer micelles and polymeric nanoparticles?
Polymer micelles are self-assembled soft colloids formed by amphiphilic polymers, while polymeric nanoparticles are broader particle systems with more matrix-like or engineered nanostructures. Their loading and release behavior are therefore not the same.
-
When are polymer microspheres a better choice than polymer micelles?
Polymer microspheres are often more suitable when long-duration release, depot behavior, or local retention is required. They are generally more aligned with matrix-controlled sustained delivery than dynamic micellar systems.
-
Which platform is best for poorly water-soluble small molecules?
Polymeric micelles are often the first platform considered for poorly soluble small molecules because their hydrophobic core can improve apparent solubility and nanoscale dispersion.
-
Are polymeric nanoparticles more versatile than polymer micelles?
In many cases, yes. Polymeric nanoparticles can offer broader control over particle structure, surface engineering, and payload handling, although that added flexibility may come with higher formulation complexity.
-
Can these three platforms be used for the same drug?
Yes, but they are usually not equally suitable. The best choice depends on payload properties, desired release profile, particle size needs, and manufacturing constraints.

 Fig. 1. Structural comparison of micelles, microspheres, and nanoparticles (BOC Sciences Authorized).
Fig. 1. Structural comparison of micelles, microspheres, and nanoparticles (BOC Sciences Authorized). Fig. 2. Selection framework for polymer drug delivery platforms (BOC Sciences Authorized).
Fig. 2. Selection framework for polymer drug delivery platforms (BOC Sciences Authorized).