How to Choose the Right Polymer for Long-Acting Drug Delivery Formulations?
Choosing the right polymer for long-acting drug delivery formulations requires more than selecting a familiar excipient or a commonly cited biodegradable material. In practice, polymer choice controls how the formulation absorbs water, how the drug is distributed and stabilized, how the matrix changes with time, and how reliably release can be tuned across a defined duration. The same active pharmaceutical ingredient (API) may behave very differently in a degrading polyester matrix, a hydrophilic hydrogel network, a membrane-controlled reservoir system, or an in situ forming depot. For this reason, effective polymer selection must connect API properties, delivery platform, polymer chemistry, processing route, and release objectives into one coherent design framework. This resource outlines that framework and shows how to evaluate polymer candidates for long-acting systems with a material-science-based approach.
Resources
What is Long-Acting Drug Delivery Formulations?
Long-acting drug delivery systems depend on polymer materials that can do more than simply hold an API in place. A suitable polymer must support the intended release mechanism, tolerate the chosen fabrication route, maintain compatibility with the drug, and preserve enough structural or diffusional control to produce a useful release profile over time. In some formulations, the polymer primarily acts as a degrading matrix. In others, it behaves as a barrier membrane, a hydrated network, a self-assembled carrier, or a phase-transforming depot precursor. The challenge is that no single polymer family works best for every API or every delivery platform. Instead, the right choice emerges from understanding which material properties matter most for the target formulation and which trade-offs are acceptable.
Polymer Selection in Long-Acting Drug Delivery
Polymer selection in long-acting formulation development refers to the systematic process of matching a polymer or polymer combination to the intended API, dosage architecture, release window, and manufacturing pathway. This decision includes more than choosing between biodegradable and non-biodegradable materials. It also requires attention to molecular weight, monomer composition, hydrophobicity, crystallinity, thermal behavior, swelling tendency, drug–polymer interactions, and the way processing changes internal morphology. A polymer may appear attractive in isolation yet fail during formulation because the API partitions to the surface, crystallizes during storage, or becomes trapped in a matrix that does not open sufficiently over time.
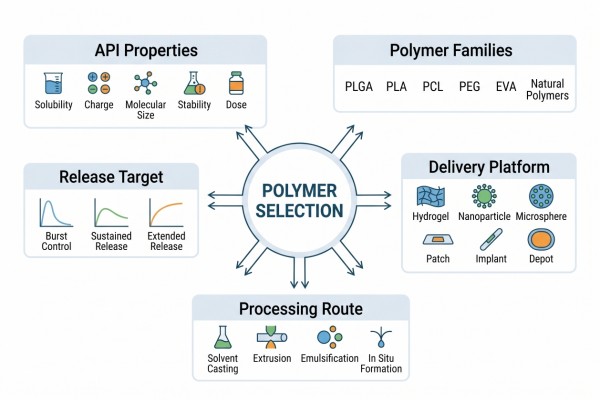 Fig. 1. Framework linking API properties, polymers, and release targets (BOC Sciences Authorized).
Fig. 1. Framework linking API properties, polymers, and release targets (BOC Sciences Authorized).
How Polymer Choice Shapes Release and Stability?
Polymer choice determines release control because the polymer defines the internal transport environment experienced by the drug. Dense hydrophobic matrices slow water ingress and diffusion, while hydrophilic regions accelerate wetting and pore formation. Degradable polyesters add another level of complexity because chain scission changes porosity and local microenvironment during release. Stability is also affected because the polymer can either protect the API from moisture, oxygen, and interfacial stress or amplify those same risks. Formulation feasibility depends on whether the polymer tolerates extrusion, solvent casting, emulsification, coating, or in situ phase inversion without destabilizing the payload or generating uncontrollable morphology.
What Determines Polymer Selection in Long-Acting Drug Delivery?
There is no universally best polymer for long-acting drug delivery because different systems rely on different release logics. A polymer suited to a biodegradable implant may not be ideal for a transdermal patch or a micellar carrier for poorly soluble compounds. A matrix designed for a hydrophobic small molecule may fail for a peptide because the peptide responds differently to water, pH shifts, and process stress. The most reliable selection strategy therefore starts with the therapeutic payload and desired performance, then narrows materials according to platform-specific constraints rather than brand familiarity or literature frequency alone.
Key Criteria for Choosing the Polymer for Long-Acting Drug Delivery Formulations
Before comparing specific polymer families, it is important to define the core decision criteria that make one material more suitable than another. In long-acting systems, the goal is not merely to prolong release. The polymer must also provide consistent device or carrier performance, acceptable manufacturability, and a release pattern that can be tuned without introducing unnecessary instability. A useful selection process therefore evaluates polymer candidates through a series of practical questions: How does the polymer interact with the API? How does it handle water? Does it erode, swell, or remain largely intact? Can it be processed into the intended platform? And does it support the desired balance between early release control and longer-term availability?
Release Mechanism Fit
The first question is whether the polymer supports the intended release mechanism. Some materials are best for diffusion-controlled systems, where the polymer acts as a stable matrix or membrane. Others are chosen because they degrade gradually and open transport pathways over time. Hydrogels and hydrophilic modifiers may depend on swelling and mesh size, while micelles rely on self-assembly stability and payload retention. Selecting a polymer without identifying the dominant release mechanism can lead to profiles that are burst-prone, overly delayed, or incomplete.
Drug–Polymer Compatibility
Compatibility between the API and the polymer affects loading, internal distribution, physical stability, and release reproducibility. Strong but balanced interactions can help maintain molecular dispersion or fine particle distribution. Poor compatibility may cause phase separation, drug migration to the surface, or crystallization during processing and storage. Compatibility also influences whether the polymer environment protects the API or exposes it to unfavorable microconditions. In long-acting systems, these effects often become more visible with time, making early compatibility screening essential.
Water Uptake, Barrier Function, and Structural Persistence
Water ingress is one of the most important yet often underestimated formulation variables. It determines whether the system behaves as a stable barrier, a gradually opening matrix, or a rapidly hydrated depot. Polymers with strong barrier function may reduce initial burst but risk prolonged tail release if the device remains too impermeable. Highly hydrophilic systems can facilitate transport yet compromise long-duration control. The right balance depends on whether the formulation requires persistent shape retention, controlled swelling, or programmed degradation.
Manufacturing and Processing Compatibility
A polymer should never be chosen without considering the fabrication route. Some materials perform well in solvent-based processing but are unsuitable for thermal methods. Others tolerate extrusion or compression but not emulsification or self-assembly conditions. Process compatibility also determines residual solvent risk, internal morphology, and drug-state evolution. In long-acting formulation development, the question is not just whether the polymer can form the target system, but whether it can do so reproducibly without undermining API stability or release control.
Tunability across the Screening Space
The best polymer candidates are not only functional, but tunable. Materials that allow systematic adjustment through molecular weight, composition, blend ratio, crosslinking density, or membrane thickness are especially valuable because they support rational optimization rather than trial-and-error reformulation. Tunability matters because early screening rarely identifies the final composition immediately. A polymer family with useful internal design space is often more valuable than one with attractive baseline behavior but limited room for targeted refinement.
Which API Properties Should Guide Polymer Selection?
Polymer selection should begin with the API because release performance is ultimately governed by how the drug behaves inside the formulation, not simply by how the polymer behaves on its own. Drug solubility, ionization, molecular size, dose requirement, thermal tolerance, and sensitivity to moisture or interfaces all influence whether a polymer is likely to support or undermine the intended release profile. Two polymers with similar degradation behavior may produce very different results if the API partitions differently, dissolves at different rates, or becomes unstable under one processing route but not another.
Solubility, Partitioning, and Polymer Affinity
Solubility determines whether the drug prefers the polymer phase, the aqueous phase, or an interfacial environment between them. A hydrophobic drug may be retained effectively in a polymer matrix but released too slowly if water penetration remains limited. A more hydrophilic drug may partition rapidly into incoming water and escape before a sustained profile is established. Affinity between the drug and polymer influences whether the API stays uniformly distributed or migrates during solvent removal, thermal processing, or storage. These relationships must be evaluated early because they strongly affect both burst release and total release.
Molecular Size, Charge, and Diffusion Constraints
Molecular size and charge shape transport behavior. Small neutral molecules may diffuse quickly if the polymer network hydrates or becomes porous. Larger peptides, proteins, and nucleic-acid-related payloads often face stronger diffusional limitations and may require more hydrated or interactive polymer environments. Charged payloads can also bind to oppositely charged polymers, altering both loading stability and release. These interactions can be useful when controlled carefully, but they may also trap the API too strongly or destabilize carrier formation if the charge balance is poorly designed.
Chemical Stability, Moisture Sensitivity, and Processing Tolerance
Some APIs tolerate organic solvents and moderate thermal stress well, while others degrade rapidly under those same conditions. Moisture-sensitive compounds may require hydrophobic matrices or low-water processing environments. Proteins and peptides are particularly vulnerable to unfolding, aggregation, or interfacial stress, while certain small molecules are more sensitive to oxidation or solid-state transitions. A polymer that seems attractive from a release perspective may still be a poor choice if it forces the drug through damaging process conditions or exposes it to an unfavorable internal microenvironment during storage and release.
Dose Requirement and Drug Loading Limits
Long-acting delivery often requires compact systems with meaningful dose density. This makes drug loading a central selection criterion. A polymer that supports excellent release control at low loading may become unstable at higher loadings because the API crystallizes, segregates, or creates uncontrolled pore formation. The required total dose also influences which platform is realistic. High-dose systems may favor implants, depots, or patches with large loading capacity, while low-dose potent compounds may be compatible with micelles, nanoparticles, or thin inserts. Polymer choice should therefore be informed by how much drug must be carried and how uniformly that dose can be maintained.
Why API Properties Must Be Screened before Polymer Families
Screening API properties first prevents formulation teams from narrowing polymer candidates too early based on generic expectations. It keeps the selection process anchored to the real constraints of the molecule rather than to a preferred material family. This approach also improves efficiency: once the API behavior is understood, it becomes easier to eliminate polymer classes that are unlikely to provide sufficient compatibility, durability, or process fit. Starting with the drug makes downstream polymer selection more rational and reduces the risk of spending time optimizing fundamentally mismatched material routes.
How Polymer Choice Changes for Different Drug Types?
Once core API attributes are defined, the next step is to recognize that different classes of therapeutics demand different selection logic. Even when the target is the same release duration, polymer requirements change with molecular size, solubility, charge, and structural fragility. This is why a polymer that performs well for a hydrophobic small molecule cannot automatically be assumed suitable for peptides, proteins, or charged biomolecular payloads. Drug-type-specific selection allows formulators to move beyond broad polymer categories and focus on the carrier environments most likely to preserve activity while supporting controllable long-acting release.
Polymer Selection for Small Molecule Drugs
Small molecule drugs often provide the broadest material options, but that does not make selection simple. Hydrophobic small molecules may benefit from polyester matrices, membrane-controlled systems, or polymeric micelles when solubility enhancement is needed. More water-soluble compounds may require stronger barrier control or reduced hydrophilic content to avoid rapid loss. For small molecules, the key questions are whether the polymer can prevent surface enrichment, whether the drug remains physically stable inside the matrix, and whether the release mechanism supports complete delivery rather than only good initial control.
Polymer Selection for Peptides and Proteins
Peptides and proteins require gentler formulation environments because they are sensitive to moisture, interfaces, pH shifts, and thermal stress. Hydrated carriers such as injectable polymer hydrogels may be useful when the design goal is to protect higher-order structure in a water-rich environment. Biodegradable matrices and microspheres can also be considered, but the internal microenvironment must be controlled carefully to avoid destabilization. For these payloads, polymer selection should prioritize structural protection, compatibility with mild processing, and the ability to support diffusion or matrix opening without causing aggregation or loss of activity.
Polymer Selection for Nucleic Acid and Charged Payloads
Nucleic acid and other charged payloads introduce a different selection logic because electrostatic interactions often become central to loading and release. Functional polymers such as polyethyleneimine, linear, cationic derivatives, or charge-bearing hybrid systems can improve payload association, but excessive binding may hinder release or compromise stability. In these cases, the polymer must do more than slow transport. It must also maintain charge balance, structural integrity, and activity preservation. Long-acting designs for charged payloads often benefit from hybrid approaches that combine structural polymers with functional or ionic modifiers rather than relying on one material alone.
Polymer Selection for Poorly Soluble Compounds
Poorly soluble compounds require polymers that can either maintain them in a dispersed state or create transport environments that avoid rapid precipitation and incomplete release. Amphiphilic carriers, hydrophobic matrices, and controlled-porosity systems all become relevant depending on platform. The goal is not only to encapsulate the drug, but to preserve release-driving conditions throughout the dosing window. A highly hydrophobic polymer may initially stabilize loading well but still fail if the compound remains locked in the matrix. Material selection for poorly soluble drugs therefore requires an explicit balance between retention, hydration control, and eventual transport out of the dosage system.
Which Polymer Families Are Most Useful for Long-Acting Drug Delivery?
After defining API constraints, formulation teams can compare polymer families more productively. The best materials for long-acting drug delivery are not those with the strongest reputation, but those whose transport behavior, degradation profile, processing characteristics, and compatibility space match the intended platform. In practice, polyester systems remain central because of their tunable degradation and solid-device versatility, while hydrophilic modifiers, non-biodegradable barrier materials, and functional natural polymers play important roles in specialized or hybrid designs. Comparing these families side by side helps identify where each one contributes most effectively.
| Polymer Family | Primary Role in Long-Acting Formulations | Key Strength | Main Limitation | Typical Formulation Use |
|---|
| Poly(D,L-lactide-co-glycolide) and related PLGA systems | Biodegradable matrix-forming copolyesters | Broad tunability of degradation and release behavior | May show burst release, acidic microenvironment effects, or incomplete release if poorly optimized | Long-acting implants, microspheres, in situ depots, wafers, and matrix-based sustained-release systems |
| Polylactic acid and related polyesters | Hydrophobic biodegradable structural matrices | Slower water ingress and better structural retention | Can prolong terminal release if matrix opening is too limited | Monolithic devices, sustained matrices, films, and slower-opening implant platforms |
| Polycaprolactone and slow-eroding polyesters | Longer-lasting structural and membrane-forming materials | Strong persistence in extended-release architectures | Very slow erosion may reduce total release efficiency | Implants, inserts, transdermal systems, and long-duration barrier layers |
| Hydrophilic modifiers including polyethylene glycol and polyvinyl alcohol | Water-uptake regulators and transport modifiers | Useful for tuning hydration, porosity, and release pathways | Excess use may accelerate early release or weaken barrier control | Hydrogels, matrix blends, coatings, and release-adjusted hybrid formulations |
| Ethylene-vinyl acetate, polyurethanes, and other non-biodegradable polymers | Membrane and reservoir control materials | Stable diffusion-barrier performance | Do not rely on degradable matrix clearance | Reservoir devices, patches, inserts, and persistent membrane-controlled systems |
| Functional natural polymers such as chitosan and hyaluronic acid | Interactive or hydrated components in specialized systems | Biofunctional and adaptable in hybrid carrier environments | Often need combination with stronger structural polymers for long-duration use | Hybrid depots, microneedles, inserts, hydrogels, and site-specific long-acting systems |
Poly(lactic-co-glycolic acid) (PLGA) and related PLGA-type materials are widely used because they offer a practical combination of degradability, structural versatility, and formulation tunability. By changing lactide-to-glycolide ratio, molecular weight, and end-group characteristics, formulators can shift water uptake, matrix opening, and release duration across a broad range. PLGA is especially useful in implants, microspheres, depots, and wafers where degradation contributes directly to sustained delivery. Its main advantage is flexibility. Its main challenge is that release profiles are highly sensitive to microstructure, local acid generation, and the internal distribution of the API.
Polylactic acid (PLA) and related hydrophobic polyesters are often selected when slower hydration and stronger structural retention are needed. These materials can reduce very rapid early transport and help maintain device integrity in solid implants or slower-opening matrices. They are especially valuable when a more persistent polymer phase is needed than fast-degrading systems can provide. The limitation is that excessive hydrophobicity or crystallinity can slow matrix opening too much, leaving the formulation with extended tail release or incomplete drug recovery unless geometry and loading are tuned carefully.
PCL for Long-Acting Formulations
Poly-ε-caprolactone (PCL) is useful when very slow structural change and long-term barrier persistence are desired. As a semicrystalline polyester, it often provides stronger long-duration device integrity than more rapidly degrading alternatives. PCL is well suited to inserts, implants, transdermal architectures, and certain membrane-based designs. Because it erodes slowly, it can support extended release but may also create a prolonged late phase if the system does not develop sufficient transport pathways. For that reason, PCL often works best when geometry, thickness, and drug state are controlled deliberately.
PEG and Hydrophilic Polymer Modifiers
Hydrophilic modifiers such as polyethylene glycol (PEG) and polyvinyl alcohol are not always the main structural materials in long-acting systems, but they are often critical to tuning water access, mesh properties, pore generation, and release smoothness. In hydrogels, depots, and hybrid matrices, these polymers can improve hydration and modify internal transport paths. However, they must be used carefully. Too much hydrophilic content may accelerate ingress and burst release, while too little may leave the matrix too restrictive for useful total release.
EVA and Other Non-Biodegradable Polymers
Ethylene-vinyl acetate (EVA), polyurethanes, and other non-biodegradable polymers are valuable when a stable membrane or reservoir barrier is more important than matrix erosion. These materials are particularly useful in patches, inserts, and diffusion-controlled devices where release is regulated by permeability, wall thickness, and surface area. Their key strength is barrier consistency over time. Their limitation is that they do not provide degradation-driven clearance or matrix opening, so long-term performance depends heavily on device engineering and membrane uniformity.
Chitosan, Hyaluronic Acid, and Functional Natural Polymers
Functional natural polymers such as chitosan and hyaluronic acid are especially relevant in hydrated or interactive long-acting systems. Their roles often include mucoadhesion, charge-based interaction, localized retention, or soft-matrix formation rather than acting as the sole structural backbone of a long-duration solid implant. These materials are valuable in microneedles, inserts, hydrogels, and hybrid depot systems where responsiveness or local interface behavior matters. In many cases, they work best when combined with stronger structural polymers that provide the long-term mechanical or diffusional control needed for sustained release.
Need Help Matching Polymer Properties with Your Release Target?
BOC Sciences supports polymer screening, release-oriented formulation design, and material optimization for long-acting delivery systems across small molecules, peptides, proteins, and functional payload platforms.
Which Polymer Properties Actually Determine Long-Acting Release Behavior?
Material names alone do not explain release performance. Within the same polymer family, small changes in composition or physical structure can substantially alter hydration, diffusion, barrier strength, degradation rate, and total release efficiency. This is why polymer selection should always move beyond family-level labels toward property-level evaluation. Molecular weight, monomer ratio, crystallinity, thermal transitions, hydrophobicity, swelling tendency, and drug–polymer compatibility all determine how the matrix or carrier behaves after formulation and during release. A strong long-acting design depends on understanding these variables as active release controls rather than passive material descriptors.
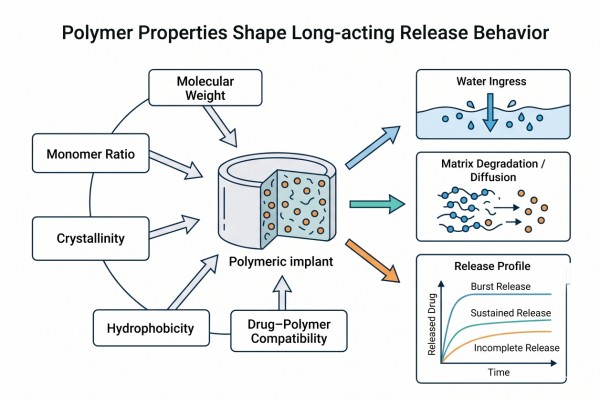 Fig. 2. How polymer properties shape long-acting release behavior (BOC Sciences Authorized).
Fig. 2. How polymer properties shape long-acting release behavior (BOC Sciences Authorized).
Molecular Weight and Chain Mobility
Molecular weight affects chain entanglement, matrix density, mechanical persistence, and the pace of structural relaxation. Higher molecular weight usually slows polymer mobility and may reduce early release, but it can also complicate processing or delay matrix opening too much. Lower molecular weight materials may hydrate and erode faster, which sometimes helps total release but may increase burst or compromise structural control. Molecular weight is therefore best treated as a primary tuning lever for both manufacturability and release kinetics.
Monomer Ratio and Degradation Speed
In copolymer systems, monomer ratio directly changes hydrophobicity, hydrolysis behavior, and internal transport development. For lactide/glycolide materials, composition affects how quickly water penetrates, how rapidly the matrix softens or degrades, and how the local environment evolves during release. A suitable monomer ratio should match the intended duration rather than simply maximize or minimize degradation. The right balance is the one that supports the target release window without destabilizing the API or causing uncontrolled late-stage release.
Crystallinity, Glass Transition, and Water Ingress
Crystallinity restricts transport in ordered regions and usually enhances structural persistence, but it can also delay matrix opening or extend terminal release if overemphasized. Glass transition is important because it affects chain mobility at formulation and storage conditions. Materials near or below their glass transition often provide stronger barrier properties, while more mobile matrices may absorb water and reorganize more rapidly. Together, crystallinity and thermal behavior influence how the dosage system responds to hydration and how stable the internal drug distribution remains over time.
Hydrophobicity, Swelling, and Pore Formation
Hydrophobicity determines how quickly water enters the matrix and whether the system behaves as a resistant barrier or a rapidly opening transport network. Swelling can be highly useful in hydrogels and depot systems where controlled expansion helps diffusion, but uncontrolled swelling may destabilize release. Pore formation plays a similar dual role: it can enable total release from otherwise dense matrices, yet excessive or poorly timed porosity often drives burst release. Selecting the right polymer therefore means selecting the right hydration pathway, not simply choosing the most hydrophobic or the most water-compatible option.
Drug–Polymer Compatibility and Internal Drug State
A polymer may look promising from a degradation perspective and still fail because the API does not remain in a useful internal state. The drug may be molecularly dispersed, amorphous, finely crystalline, or phase-separated in large domains, and each state produces a different release pattern. Compatibility influences whether the API stays distributed during processing, whether it migrates during storage, and whether the matrix supports complete release later. Careful characterization of the internal drug state is essential when comparing candidate polymers, especially for high-loading or poorly soluble systems.
Matching Polymer with Delivery Platform and Formulation Type
Polymer choice should always be evaluated in the context of the delivery platform because the same material can behave very differently across hydrogels, microspheres, patches, wafers, depots, and implant systems. Platform architecture determines surface area, diffusion path length, hydration mode, fabrication stress, and the way the polymer contributes to release. A material that performs well in one geometry may fail in another because the dominant transport mechanism changes. For this reason, platform-specific selection is one of the most important steps in long-acting formulation design.
Injectable hydrogels rely on water-rich polymer networks that entrap the API and regulate release through mesh size, swelling behavior, and crosslink density. They are especially useful when mild handling conditions and localized retention are priorities. The polymer selection focus here is on gelation mechanism, hydration control, and whether the network can support sustained transport without rapid dissolution or collapse.
In nanoparticle systems, the polymer must support both carrier stability and release control at small size scales. Surface chemistry, matrix density, and payload retention become especially important. Materials that work in implants or depots may not translate directly because nanoparticle performance depends more strongly on colloidal stability, particle morphology, and the balance between retention and dispersibility.
Polymeric micelles are useful for poorly soluble compounds because amphiphilic polymers create core-shell carriers that improve apparent solubility and protect the drug from rapid precipitation. Selection here focuses on self-assembly stability, critical micelle behavior, core compatibility, and whether the polymer architecture can sustain drug retention long enough to achieve a meaningful extended-release effect.
In situ depots begin as polymer-containing liquids or semisolids and convert into depots after placement. The selection challenge is not only matrix composition but also phase transformation behavior. Polymer candidates must be screened for solvent compatibility, precipitation kinetics, early pore formation, and burst-release risk because the structure is generated during administration rather than fully preformed.
Microspheres demand polymer choices that balance encapsulation efficiency, particle morphology, internal porosity, and degradation behavior. High surface area can make burst release more difficult to control, so polymer properties that stabilize the internal drug distribution become especially important. Materials with tunable degradation and strong process compatibility are often favored in this platform.
Microneedle systems require polymers with precise mechanical behavior, controlled dissolution or persistence, and compatibility with microfabrication. Depending on the design, the polymer may need to dissolve in a defined time window, remain intact as a depot-forming element, or work as part of a hybrid transdermal system. Mechanical integrity and reproducible molding performance are therefore central selection criteria.
Inserts are site-specific solid or semi-solid systems where local environment, fluid exposure, and residence time influence polymer choice. The selected material must balance shape retention, local compatibility, and sustained release without excessive swelling or premature fragmentation. Surface properties and hydration behavior are particularly important because the insert operates in a confined local environment.
Patch systems depend on adhesive performance, matrix diffusion, skin contact stability, and sometimes reservoir membrane control. Polymer choice therefore includes not only release behavior but also film formation, flexibility, and compatibility with skin-facing design. Materials that function well in implants may be poor choices here if they do not provide appropriate film mechanics or controlled transdermal transport.
Implantable wafers require polymers that can form stable thin geometries while still supporting controlled erosion or diffusion. Wafer design places strong emphasis on thickness uniformity, surface area, local matrix behavior, and API distribution across a relatively flat structure. Suitable polymers are those that maintain reliable release without cracking, dose dumping, or highly uneven matrix opening.
Implants are among the most structurally defined long-acting systems, and polymer selection must support device geometry, sustained release, and long-term microstructural evolution. Depending on the architecture, the polymer may act as a degrading matrix, a persistent shell, or both. Suitable materials are those that maintain shape long enough to control release yet evolve in a way that does not trap the API or cause uncontrolled burst behavior.
A Practical Decision Framework for Choosing the Right Polymer
A useful polymer selection strategy should end in a practical screening pathway, not only in a theoretical comparison. The most effective framework is one that narrows material options according to mechanism, platform, and API constraints while minimizing unnecessary screening work. Instead of testing large numbers of polymers without structure, formulation teams can define a smaller decision tree based on release objective, compatibility risk, and platform fit. This helps identify promising candidates earlier and makes later optimization more efficient.
- Step 1: Define the Release Window and Formulation Objective: Start by identifying the intended duration, acceptable early release level, and the overall release profile needed from the system. This determines whether the design should emphasize membrane control, degradation, swelling, or a combination of mechanisms. Polymer selection becomes much more consistent once the release target is defined in mechanistic rather than only descriptive terms.
- Step 2: Screen API Constraints before Screening Polymers: Use the API to eliminate clearly unsuitable material routes early. Compounds that are heat-sensitive, highly water-soluble, highly hydrophobic, strongly charged, or high-dose each require different material environments. Screening these constraints first makes polymer selection more purposeful and prevents unnecessary optimization around materials that are fundamentally mismatched.
- Step 3: Narrow the Polymer Family by Mechanism, Not by Trend: Select polymer families according to the release mechanism and platform logic they support. For example, matrix-forming degradable polyesters are useful for many implants, microspheres, and wafers, while barrier-oriented materials may be stronger choices for reservoir patches or inserts. This step reduces the tendency to default to well-known polymers even when the formulation mechanism points elsewhere.
- Step 4: Evaluate the Minimum Critical Material Variables First: Early screening should focus on the variables most likely to change release behavior: molecular weight, polymer composition, hydrophilic content, and geometry-related factors. Screening too many secondary variables too early often obscures the main structure–property relationship. A smaller mechanistic screening set usually delivers clearer decisions than broad but shallow experimentation.
- Step 5: Match Polymer Choice with the Intended Delivery Platform: Reconfirm that the material still makes sense after platform selection. A polymer that is attractive in theory may become impractical once fabrication method, particle size, membrane thickness, gelation route, or film mechanics are considered. This step keeps material decisions integrated with formulation architecture instead of treating polymer identity as an isolated variable.
- Step 6: Identify Failure Signals Early in Formulation Screening: Common early warning signs include strong burst release, visible phase separation, poor drug distribution, unstable self-assembly, incomplete release, and morphology drift after processing or storage. These signals usually indicate that polymer choice, loading state, or platform architecture needs to be reconsidered. Recognizing them early saves time and leads to more informative reformulation.
A polymer is a strong candidate when it aligns with the required release mechanism, supports the intended delivery platform, remains compatible with the API throughout processing and storage, and offers enough tunability to optimize without changing the whole formulation concept. The best long-acting systems are built on polymers that are not only functional, but predictable and adjustable within a clearly defined design space.
Services
Advanced Polymer Synthesis and Formulation Services
At BOC Sciences, we support long-acting drug delivery development by helping researchers connect polymer structure with formulation performance. Our capabilities span polymer selection, custom synthesis, functional modification, release-oriented material screening, and analytical characterization for sustained and controlled delivery platforms. Whether a project requires a degradable polyester matrix, a membrane-forming polymer for a device-based system, or a hybrid material strategy that balances stability with tunable transport, BOC Sciences provides practical support grounded in polymer chemistry and formulation logic. We work with researchers to clarify structure–property relationships, refine material choice, and improve consistency across early formulation development.

Custom Polymer & Copolymer Synthesis
- Tailored synthesis of biodegradable and functional polymer candidates for long-acting delivery.
- Control over molecular weight, composition, and architecture for release-oriented screening.
- Support for matrix-forming, membrane-forming, and specialty formulation polymers.
- Design of polymer materials aligned with API compatibility and target duration.

Polymer Functionalization & Material Optimization
- End-group and side-chain modification for tuning hydrophilicity, charge, and interaction strength.
- Blend and modifier strategies for burst control, porosity management, and structural balance.
- Support for hybrid material design across implants, depots, hydrogels, and transdermal systems.
- Optimization of polymer candidates for improved formulation feasibility.

Long-Acting Formulation Development Support
- Material screening for platform-specific long-acting delivery systems.
- Evaluation of matrix, membrane, depot, and self-assembled carrier strategies.
- Assessment of drug loading, geometry, and formulation-variable effects on release behavior.
- Structure–property interpretation to support efficient formulation decision making.

Analytical Characterization & Technical Evaluation
- Characterization of molecular weight, thermal behavior, crystallinity, and drug state.
- Analysis of internal morphology and release-related microstructural change.
- Evaluation of batch consistency, process effects, and formulation stability risks.
- Support for building a stronger experimental basis for polymer selection.
Do You Need A Consultation?
BOC Sciences helps researchers evaluate polymer families, platform fit, and release-control strategies for long-acting drug delivery formulations through practical material and formulation support.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
How do I choose the right polymer for a long-acting drug formulation?
The right polymer is chosen by matching API properties, release target, delivery platform, and processing route. A useful selection workflow starts with drug solubility, stability, charge, and dose, then narrows polymer families according to release mechanism, compatibility, and platform-specific formulation requirements.
-
Is PLGA always the best polymer for sustained release?
No. PLGA is versatile and widely used, but it is not automatically the best choice for every API or platform. Other materials may be more suitable when slower structural persistence, stronger membrane control, softer hydrated environments, or different processing conditions are required for stable long-acting release.
-
What polymer properties matter most in long-acting release systems?
The most important properties usually include molecular weight, monomer composition, hydrophobicity, crystallinity, swelling tendency, and drug–polymer compatibility. These variables determine water ingress, diffusion path development, degradation behavior, and whether the internal drug state supports smooth sustained release or causes burst and incomplete delivery.
-
How does drug solubility affect polymer selection?
Drug solubility affects how the API partitions between the polymer and incoming aqueous phase, which directly shapes burst risk, loading stability, and total release. Highly hydrophobic drugs may need amphiphilic or release-opening strategies, while more soluble compounds often require stronger barrier control to prevent rapid initial escape.
-
When should non-biodegradable polymers be preferred?
Non-biodegradable polymers are often preferred when the formulation depends on stable membrane or reservoir-controlled diffusion rather than gradual matrix erosion. They are particularly useful in patches, inserts, and other device-based systems where barrier consistency, film integrity, and sustained permeability are more important than degradable clearance.
-
Why can the same polymer behave differently in different delivery platforms?
The same polymer can perform differently because each platform changes geometry, surface area, hydration mode, fabrication stress, and dominant transport mechanism. A polymer used in a microsphere, implant, hydrogel, or patch experiences different structural demands, so release behavior must always be evaluated in platform context.
-
Which polymers are more suitable for peptides and proteins?
Peptides and proteins often need polymers that support mild processing, reduced interfacial stress, and controlled internal environments. Hydrogels, hybrid systems, and carefully designed biodegradable matrices can be useful, but the final choice depends on how well the polymer preserves structural integrity while still enabling sustained release.
-
How can formulators reduce burst release during polymer screening?
Burst release can often be reduced by improving drug dispersion, limiting surface enrichment, controlling porosity, optimizing hydrophilic content, and adjusting molecular weight or geometry. The goal is not to block release completely, but to moderate early transport while preserving enough matrix or carrier evolution for continued delivery.

 Fig. 1. Framework linking API properties, polymers, and release targets (BOC Sciences Authorized).
Fig. 1. Framework linking API properties, polymers, and release targets (BOC Sciences Authorized). Fig. 2. How polymer properties shape long-acting release behavior (BOC Sciences Authorized).
Fig. 2. How polymer properties shape long-acting release behavior (BOC Sciences Authorized).