How Polymer Micelles Support Protein and Peptide Delivery?
Protein and peptide delivery places unusual demands on polymer micelles because these cargos are far more structurally sensitive than many hydrophobic small molecules and often respond strongly to solvent exposure, interfacial stress, ionic conditions, and dehydration history. A useful micellar system must therefore do more than create a nanoscale carrier: it must preserve conformational integrity where possible, reduce premature leakage, maintain colloidal stability during processing, and support a release pathway that matches the physicochemical behavior of the biomacromolecule. In practice, successful design depends on aligning payload class, micelle architecture, loading mechanism, and preparation method instead of assuming that the same self-assembly logic used for conventional drug-loaded micelles will automatically translate to proteins and peptides.
Resources
Protein and Peptide-Based Micelles as Drug Delivery Carriers
Polymer micelles used for protein and peptide delivery are self-assembled or structurally engineered colloidal systems in which the carrier is expected not only to accommodate a biologically active macromolecule, but also to moderate the stresses that normally destabilize it. Unlike conventional micelles developed mainly for poorly soluble small molecules, these systems often rely on a more complex balance among amphiphilicity, electrostatic interaction, shell hydration, and interfacial protection. The decisive question is not whether a protein or peptide can be associated with a micelle at all, but whether that association remains useful through preparation, storage, administration, and release.
Definition of Protein- and Peptide-Loaded Polymer Micelles
Protein- and peptide-loaded polymer micelles are colloidal assemblies in which amphiphilic polymers create a core-shell structure that either encapsulates, complexes, associates with, or conjugates a proteinaceous payload. Depending on the system, the payload may reside in a hydrophobic core, interact at the core-shell interface, bind electrostatically to a charged domain, or remain associated through affinity-driven or covalent design. This broad definition is important because proteins and peptides do not all enter micelles through one universal mechanism. Delivery performance is therefore determined as much by the mode of association as by the presence of a micellar carrier itself.
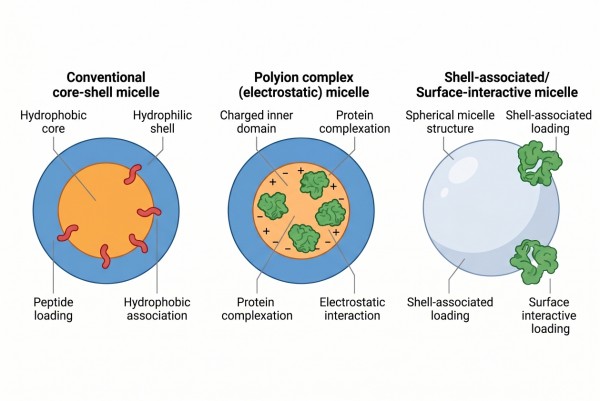 Fig. 1. Protein and peptide loading modes in polymer micelles (BOC Sciences Authorized).
Fig. 1. Protein and peptide loading modes in polymer micelles (BOC Sciences Authorized).
How They Differ from Conventional Drug-Loaded Micelles?
Conventional polymer micelles are often optimized around hydrophobic-core partitioning, where the main task is to increase apparent solubility and maintain nanoscale dispersion. Protein and peptide delivery changes that logic because many biomacromolecules are amphiphilic only in a partial or context-dependent sense, and some are strongly affected by interface composition, ionic environment, and chain mobility at the micelle surface. As a result, a formulation that works well for a lipophilic small molecule may fail for a peptide due to weak retention or may damage a protein through destabilizing interfacial contact. Protein/peptide micelles therefore demand a more protective and less assumption-driven design approach.
Why Protein and Peptide Delivery Requires a Different Design Logic?
The main difference lies in the fact that proteins and peptides are not only payloads but also dynamic structures whose function depends on maintaining a useful conformational state. In many cases, the carrier must reduce denaturation risk, limit aggregation, shield from enzymatic attack, and prevent uncontrolled loss during handling or release. That means the micelle cannot be judged solely by size and encapsulation efficiency. It must also be evaluated by how gently it forms, how it presents interfaces to the biomolecule, how well it preserves loading without excessive rigidity, and whether release conditions remain compatible with biological integrity after the carrier begins to reorganize.
Why Protein and Peptide Delivery Requires Specialized Micelle Design?
The main challenge in protein- and peptide-loaded micelles is that the carrier must solve multiple problems at once: it has to create a defined colloidal structure, maintain sufficient loading, protect the cargo from destabilizing environments, and still allow meaningful release. These requirements can conflict with one another. Stronger micelle cohesion may improve retention but hinder release or expose the biomolecule to harsher preparation conditions, while a more dynamic micelle may release efficiently but fail to preserve structure during storage or dilution. Specialized design is therefore necessary because proteins and peptides respond not only to chemistry, but to the full process history of the system.
Structural Fragility and Conformational Instability
Proteins and many peptides are conformationally sensitive because their useful state depends on noncovalent interactions that can be disrupted by solvent exchange, temperature change, local dehydration, or interaction with hydrophobic interfaces. Even short peptides may lose functional organization when their local microenvironment shifts abruptly. In micellar systems, this means the carrier must be designed to avoid forcing the biomolecule into a destabilizing contact state. A shell that is too poorly hydrated, a core that is too hydrophobic, or a preparation route that exposes the payload to organic solvent for too long can all generate structural changes that reduce activity before delivery is even evaluated.
Enzymatic Degradation and Environmental Sensitivity
Many proteins and peptides are vulnerable to proteolysis, hydrolysis, pH drift, ionic changes, and oxidation, which means the micelle may need to function as a temporary protective microenvironment rather than only as a carrier. For peptides in particular, modest improvements in shielding can already enhance apparent stability, while for larger proteins the carrier may also need to reduce direct access of degradative species to sensitive domains. This is one reason surface hydration and shell density matter so much in proteinaceous micelles: the shell is not merely a colloidal stabilizer, but often the first barrier controlling environmental exposure around the biomolecule.
Interfacial Stress During Preparation and Release
Micelles are interface-rich systems, and interfaces can be beneficial or harmful depending on the payload. A protein or peptide that accumulates at the core-shell boundary or at an air-liquid or solvent-liquid interface during preparation may undergo conformational stress, partial unfolding, or self-association. Release can create a second interfacial event, especially if the biomolecule passes through a changing shell environment or if the micelle reorganizes under dilution. For this reason, route selection and release testing are not secondary tasks in protein/peptide micelle development. They are central to determining whether the carrier protects the biomolecule or exposes it to repeated destabilizing transitions.
Protein and Peptide Types for Polymer Micelles
Polymer micelles can be used with a wide range of proteinaceous materials, but not all proteins and peptides interact with micellar systems in the same way. Some act as structural biomaterials or assembly partners, some behave as functional cargos with defined biological activity, and others are short peptides whose amphiphilicity or charge distribution makes them more naturally compatible with micellar interfaces. The most useful classification is therefore not by biological origin alone, but by how the molecular architecture of the payload influences loading, stability, and release within the micelle.
| Protein | Main Structural Features | Why Suitable for Polymer Micelles | Main Formulation Concern |
|---|
| Gelatin | Partially denatured collagen-derived protein with flexible chains, high hydration capacity, and abundant functional groups | Useful for forming hydrated and biocompatible micellar environments, improving interfacial compatibility, and supporting mild association with sensitive peptide or protein cargos | High sensitivity to temperature, moisture conditions, and structural heterogeneity during processing |
| Collagen | Fibrous structural protein with ordered triple-helical organization and strong intermolecular assembly tendency | Relevant when bioinspired structural compatibility and matrix-like interaction are desired in polymer-protein micellar systems | Conformational instability outside controlled conditions and limited tolerance to harsh preparation routes |
| Casein | Amphiphilic milk protein with natural colloidal behavior and strong self-association tendency in aqueous media | Particularly suitable for micellar or micelle-like carrier design because of its inherent colloidal assembly properties and hydrophobic interaction capacity | Composition variability and sensitivity to pH, ionic strength, and processing history |
| Silk | Protein material with tunable secondary structure, strong mechanical integrity, and controllable hydrophobic-hydrophilic balance | Useful for designing more structurally stable or bioinspired micellar systems with controllable association behavior | Structural transition during processing can alter loading behavior and release performance |
| Elastin | Elastic protein with responsive and repetitive sequence motifs that can influence hydration and self-assembly behavior | Attractive for responsive or temperature-sensitive micellar systems where dynamic structural change is desired | Sequence-dependent assembly behavior and sensitivity to formulation environment |
| Zein | Hydrophobic plant protein with strong self-aggregation tendency and limited water solubility | Useful when stronger hydrophobic interaction is needed for micelle formation or for association with lipophilic peptide-like cargos | Poor aqueous compatibility and risk of uncontrolled aggregation during formulation |
| Gliadin | Hydrophobic gluten-derived protein with amphiphilic domains and interfacial activity | Can support micelle-related delivery through interfacial association and hydrophobic domain interaction | Broad compositional heterogeneity and sensitivity to solvent conditions |
| Soy Proteins | Plant-derived protein mixture with amphiphilic character, colloidal activity, and multiple interactive domains | Useful in mixed micelle or protein-polymer assembly systems requiring accessible plant protein functionality | Batch variation, complex composition, and environment-dependent assembly behavior |
| Albumin | Globular soluble protein with defined tertiary structure, high aqueous compatibility, and multiple binding sites | Highly relevant for shell-associated loading, affinity-guided association, and mild aqueous micelle preparation strategies | Partial unfolding or loss of functional conformation at destabilizing interfaces |
| Keratin | Cysteine-rich structural protein with strong intermolecular crosslinking tendency and robust fibrous character | Useful in bioinspired micellar systems where structural resilience and functional group richness are beneficial | Limited solubility and difficult processing under mild formulation conditions |
Table 1. Comparison of representative proteins used in polymer micelle delivery.
Fibrous and Structural Proteins
Structural proteins such as gelatin, collagen, silk, elastin, and keratin are attractive in micellar delivery because they offer rich functionality, biopolymer-derived compatibility, and multiple opportunities for intermolecular association. Gelatin and collagen contribute hydration and biointerfacial compatibility, while silk and elastin-inspired systems can provide tunable assembly behavior and mechanically distinct domains. Keratin adds interesting cysteine-rich chemistry and structural robustness in some designs. These materials are especially relevant when the delivery platform benefits from protein-derived assembly behavior, but they also require careful control because their own higher-order organization can compete with or reshape micelle formation if the polymer environment is poorly matched.
Food and Plant Protein Materials
Casein, zein, gliadin, and soy proteins are often discussed in micellar and colloidal delivery because they combine amphiphilic features with accessible sourcing and well-studied self-assembly behavior. Casein is particularly noteworthy because its natural colloidal behavior already suggests compatibility with nanostructured carrier concepts. Zein and gliadin are useful when stronger hydrophobic association is needed, while soy proteins can offer broader interfacial behavior depending on processing state. These proteins are often relevant in formulation work not only as cargos but as structural contributors or co-assembling components, especially when the design seeks a protein-informed balance between hydrophobic interaction, dispersibility, and payload compatibility.
Globular and Functional Protein Cargos
Albumin represents an important example of a globular protein cargo or protein-based carrier component because it is soluble, structurally organized, and functionally sensitive to its local environment. More broadly, globular proteins require micelles that can reduce destabilizing contact while still allowing association strong enough for retention. These cargos often benefit from shell-mediated protection, affinity-guided loading, or carefully engineered interface chemistry rather than from simple hydrophobic sequestration alone. Their micellar compatibility therefore depends on whether the carrier can present a sufficiently mild environment to preserve useful conformation while still functioning as a nanoscale delivery system.
Short Peptides and Bioactive Peptide Therapeutics
Short peptides, antimicrobial peptides, signaling peptides, and other bioactive therapeutic peptides often show more variable micelle behavior than larger proteins because they can range from highly hydrophilic to strongly amphiphilic. Some peptides are compatible with core-associated loading due to partial hydrophobic character, while others interact more effectively with charged domains or shell interfaces. Their smaller size can simplify association, but it can also increase the risk of fast leakage if retention depends on weak noncovalent interaction alone. For peptide-focused micelles, the decisive variables are often sequence-dependent amphiphilicity, net charge, tendency toward self-association, and whether the therapeutic mechanism tolerates transient exposure to micellar interfaces.
Why Peptides and Proteins Behave Differently in Micellar Systems?
Proteins and peptides differ not simply in size, but in the way they respond to micellar microenvironments. Larger proteins often require broader conformational protection and may be more vulnerable to partial unfolding at interfaces, whereas shorter peptides may load more easily but escape more rapidly unless charge or affinity interactions strengthen retention. Some peptides can exploit amphiphilic sequence features to integrate more naturally into core-shell structures, while proteins more often require shell-protected association or gentler aqueous assembly. Recognizing this distinction prevents a common mistake in formulation: treating all biomacromolecular payloads as if they demand the same micelle architecture.
What Forms of Micelles Are Used for Protein and Peptide Delivery?
Not all micelles used for protein and peptide delivery share the same structural logic. Some are classical amphiphilic assemblies, some are organized in nonaqueous or inverse environments, some behave as unimolecular carriers with covalently fixed architecture, and others depend strongly on electrostatic complexation or mixed-chain co-assembly. Choosing the correct micelle form is often more important than choosing a nominally attractive polymer, because the form determines how the biomolecule is retained, how stable the system remains under dilution, and how much structural freedom the payload has during association and release.
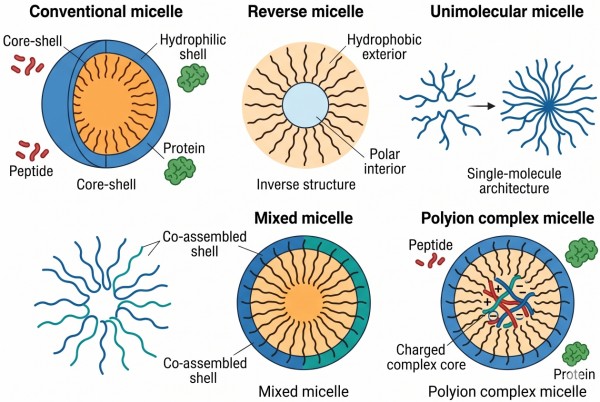 Fig. 2. Comparison of micelle forms for protein and peptide delivery (BOC Sciences Authorized).
Fig. 2. Comparison of micelle forms for protein and peptide delivery (BOC Sciences Authorized).
Conventional Micelles
Conventional micelles are formed when amphiphilic polymers self-assemble into a hydrophobic core and hydrophilic shell in a selective solvent, usually water. They are most useful when the protein or peptide can interact with the core, interface, or shell without severe destabilization. For proteins and peptides, conventional micelles often work best when the payload has at least some amphiphilic compatibility or when the shell provides sufficient protection against interfacial damage. Their main advantage is preparation simplicity and nanoscale dispersibility, but their dynamic nature can also lead to weak retention for biomacromolecules that do not strongly associate with the assembly.
Reverse Micelles
Reverse micelles invert the conventional arrangement by creating a relatively polar interior inside a surrounding hydrophobic medium. They can be useful when a hydrophilic biomolecule must be confined within a nanoscale interior under nonaqueous or low-water conditions during certain preparation routes or specialized delivery concepts. For proteins and peptides, reverse micelles are technically interesting because they offer a way to host hydrophilic macromolecules in environments that conventional aqueous micelles cannot provide. However, they also impose significant formulation challenges, including control of internal water pools, biomolecule stability under low-water processing, and transfer of the system into a final use state compatible with biological activity.
Unimolecular Micelles
Unimolecular micelles are built from single macromolecules whose architecture is preorganized to behave like a micelle without requiring dynamic multichain assembly. This can be advantageous for protein and peptide delivery because the carrier is less dependent on concentration-driven equilibrium and often more resistant to dilution-induced disassembly. Such systems are especially attractive when payload retention and structural persistence matter more than simple self-assembly convenience. Their main tradeoff is synthetic complexity: the carrier must be architecturally programmed in advance, and the resulting system often demands more sophisticated polymer design and characterization than conventional self-assembled micelles.
Mixed Micelles
Mixed micelles are formed from more than one amphiphilic component and are useful when one material alone cannot provide the necessary balance among loading, stability, and interface control. For protein and peptide delivery, mixed systems can combine a strongly assembling component with a more hydrophilic or more protective shell-forming component, or they can distribute functionality across multiple chains to reduce structural compromise in any single material. This architecture is particularly useful when a peptide needs stronger core affinity but the overall system still requires a hydrated shell, or when a protein benefits from softer surface presentation than a single amphiphile can provide.
Polyion Complex Micelles
Polyion complex micelles rely primarily on electrostatic interaction between oppositely charged segments rather than only on hydrophobic segregation. They are especially relevant for peptides and proteins that carry defined charge patterns or that interact more effectively through complexation than through hydrophobic partitioning. In these systems, the ionic complex often forms the internal association domain while a hydrophilic shell maintains colloidal stability. This architecture is highly valuable when biomacromolecule retention depends on charge matching, but it also requires careful tuning because excessive ionic strength or unfavorable pH conditions can destabilize the complex. For many protein and peptide formulations, polyion complex micelles provide a more chemically rational route than forcing the payload into a traditional hydrophobic core.
| Micelle Form | Structural Feature | Typical Loading Mechanism | Main Advantage | Main Limitation | Best-Fit Protein/Peptide Type |
|---|
| Conventional Micelles | Dynamic core-shell self-assembly | Core partitioning, interface association | Simple nanoscale dispersion | Weak retention for some biomacromolecules | Amphiphilic peptides, partially hydrophobic cargos |
| Reverse Micelles | Polar interior in hydrophobic medium | Internal confinement of hydrophilic payloads | Useful for specialized hydrophilic loading routes | Complex transfer to final formulation state | Hydrophilic proteins and peptides in specialized processing |
| Unimolecular Micelles | Architecturally fixed single-molecule carrier | Covalent, affinity, shell or domain-based loading | High dilution resistance | Synthetic complexity | Sensitive proteins requiring higher structural persistence |
| Mixed Micelles | Co-assembly of multiple amphiphiles | Combined core and shell tuning | Balanced stability and compatibility | More formulation variables | Peptides and proteins needing tailored interface properties |
| Polyion Complex Micelles | Charge-driven association with hydrophilic shell | Electrostatic complexation | Excellent for charged biomacromolecules | Sensitive to pH and ionic strength | Charged peptides and proteins |
Table 2. Major micelle architectures for protein and peptide delivery.
Need the right micelle architecture for protein or peptide delivery?
BOC Sciences provides custom polymer design, micelle formulation support, and route-specific development services for protein- and peptide-loaded micellar systems.
How Proteins and Peptides Are Incorporated into Polymer Micelles?
Loading mechanism is one of the most important variables in protein and peptide delivery because it determines not only how much biomolecule can be associated with the micelle, but also what stresses the biomolecule experiences during assembly, how strongly it is retained, and how it is likely to be released. A useful formulation begins by matching the payload's charge, amphiphilicity, size, and conformational sensitivity to the correct loading pathway. This prevents the common error of evaluating micelle performance only in terms of particle size while ignoring whether the biomolecule is actually associated in a stable and structurally acceptable state.
Hydrophobic Association and Core Partitioning
Hydrophobic association works best for peptides or partially hydrophobic protein domains that can tolerate interaction with a solvophobic micelle interior. This route is conceptually similar to loading poorly soluble small molecules, but in protein and peptide systems the mechanism is usually less straightforward because complete hydrophobic sequestration may destabilize the biomolecule or create only weak partial association. It is most useful for amphiphilic peptides, short bioactive sequences with hydrophobic residues, or protein fragments that can benefit from transient shielding in a core-like domain. Its main weakness is that biomacromolecules retained only by partial hydrophobic compatibility may leak rapidly when the surrounding medium changes.
Electrostatic Complexation and Polyion-Driven Assembly
Electrostatic complexation is especially powerful when the protein or peptide presents a defined charge pattern that can interact with an oppositely charged polymer segment. In these systems, loading is driven less by partitioning and more by charge-mediated association, often leading naturally to polyion complex micelles or mixed interfacial structures. This route is valuable because it avoids forcing hydrophilic biomacromolecules into a hydrophobic environment that may be structurally unfavorable. At the same time, charge-driven loading is highly sensitive to pH, ionic strength, and local charge density. A complex that looks stable in one buffer may weaken dramatically after dilution or salt exposure, so electrostatic loading must be evaluated under conditions that reflect actual formulation use.
Covalent Conjugation and Affinity-Guided Loading
Covalent conjugation and affinity-guided loading are useful when passive association is too weak or too unpredictable to support reliable retention. Covalent strategies anchor the payload to the polymer or to a linker that can later be cleaved, while affinity-based systems rely on more selective interactions such as ligand binding, host-guest recognition, or engineered complementary domains. These approaches can greatly improve control over loading state, but they also change what "micellar delivery" means because the payload is no longer simply entrapped. Instead, it becomes part of a more structured association network. This can be a major advantage when reproducibility matters, provided the conjugation or binding event does not interfere with biological function.
Shell Association and Surface-Interactive Loading
Some proteins and peptides are best associated with the shell or shell-adjacent region rather than with the core. This route can reduce exposure to a strongly hydrophobic microenvironment while still benefiting from nanoscale organization and shell-mediated shielding. Shell association is particularly relevant when the payload is hydrophilic, conformationally delicate, or structurally incompatible with full core incorporation. However, it can also increase the risk of fast exchange with the surrounding medium if the interaction is too weak. For this reason, shell-based loading works best when surface interaction is strengthened by charge balance, affinity features, or a shell environment specifically designed to maintain a hydrated but not overly labile association.
Preparation Methods for Protein- and Peptide-Loaded Polymer Micelles
Preparation method is especially important in protein and peptide micelle development because the process itself can determine whether the payload remains functional. Some routes are attractive for loading efficiency but expose the biomolecule to harsh solvent or interfacial conditions; others are milder but offer weaker association or less process flexibility. A successful route is therefore not the one that merely produces particles, but the one that preserves a useful balance among micelle formation, payload integrity, retention, and reproducibility. Method selection should start from protein or peptide sensitivity, not from laboratory habit.
Direct Dissolution
Direct dissolution is the mildest conceptual route when the polymer can assemble in the target aqueous medium and the protein or peptide does not require prior exposure to organic solvent for loading. This method is attractive because it minimizes processing steps and can reduce solvent-induced damage. It is often useful for shell-associated loading, charge-mediated interaction, or cases where the biomolecule can be introduced after micelle formation under controlled conditions. Its main limitation is that it provides less freedom for forcing difficult hydrophobic association, which means it may be insufficient when loading relies on strong core interaction rather than on interfacial or electrostatic compatibility.
Dialysis
Dialysis is widely used when the polymer requires an organic or mixed solvent before controlled transition into water. For protein and peptide delivery, the appeal of dialysis lies in its gradual solvent change, which can reduce abrupt aggregation and allow the assembly to organize more gently than direct antisolvent addition. This is useful when the carrier needs time to form around a sensitive payload or when mixed micelles or polyion structures benefit from slower equilibration. The method is not inherently gentle, however. If the biomolecule is exposed too long to partially unfavorable solvent conditions or if solvent exchange shifts ionic balance excessively, the same route can still cause structural compromise.
Oil-in-Water Emulsion Evaporation
Oil-in-water emulsion evaporation is more often associated with particle-forming systems, but it can also be relevant to micelle-oriented formulations when hydrophobic co-loading or phase-controlled assembly is needed. In this route, the main technical risk for protein and peptide payloads is interfacial stress. Emulsification creates large interfacial area, and proteins are especially prone to adsorption, unfolding, or aggregation at such boundaries. The method may still be useful for specific formulations where polymer organization depends on an emulsified intermediate, but it must be designed so that the biomolecule is either protected from the interface or introduced at a stage that avoids the most destabilizing conditions.
Cosolvent Evaporation
Cosolvent evaporation can be useful when the polymer and payload require a shared medium for initial association but full organic-solvent exposure must be limited. By reducing solvent gradually, the formulator can sometimes create a more controlled transition from dissolved state to micellar assembly. For protein and peptide systems, the effectiveness of this route depends on whether the cosolvent composition remains compatible with biomolecular stability during the critical association period. A small solvent fraction may assist assembly without severe damage, whereas a poorly chosen cosolvent or overly long evaporation step may compromise conformation before the final micelle is formed. Careful solvent screening is therefore essential.
Microphase Separation
Microphase separation routes are useful when local domain formation is needed to drive organized association between the polymer and the biomacromolecule. In practice, this may involve controlled phase behavior that encourages segregation into nanoscopic regions from which micelle-like structures emerge or are stabilized. The method is technically appealing because it can create defined internal organization without relying solely on conventional dilution-driven self-assembly. For protein and peptide delivery, however, the challenge lies in managing local concentration gradients. If the payload becomes trapped in an unfavorable microdomain during separation, the resulting formulation may exhibit good apparent particle formation but poor biological integrity or uneven loading state.
Thin-Film Hydration
Thin-film hydration is useful when the polymer or a hydrophobic co-component must first be distributed in a volatile solvent before reorganization in water. In protein and peptide systems, this route is most defensible when the biomolecule is added during hydration or after partial reconstitution rather than being forced through the full dry-film step unless it is known to tolerate that history. The method can support structured assembly and mixed-component systems, but it also introduces risks related to residual solvent, incomplete film redispersion, and high local polymer concentration during hydration. For delicate biomacromolecules, these factors can be as important as the polymer composition itself.
How to Characterize Protein- and Peptide-Loaded Micelles?
Characterization of protein- and peptide-loaded micelles must answer two different questions at the same time: what is the state of the carrier, and what is the state of the biomolecule inside or on that carrier? Measuring particle size alone is not sufficient, because a micelle can appear well formed while the protein has already unfolded or the peptide has largely leaked into solution. A useful characterization strategy therefore combines colloidal analysis, loading-state analysis, and biomolecular integrity analysis so that carrier performance is interpreted together with payload preservation.
Particle Size, Morphology, and Colloidal Stability
Particle size and morphology remain important because they reveal whether the micelle population is uniform, broadly aggregated, or structurally altered by loading. Dynamic light scattering can track hydrodynamic size and distribution, while microscopy helps determine whether the particles retain the expected spherical or near-spherical organization. For protein and peptide systems, these measurements are most informative when compared before and after loading and under relevant medium conditions. A formulation that shows acceptable size only in dilute buffer but loses stability in more realistic environments may not provide useful delivery performance regardless of its initial appearance.
Encapsulation Efficiency and Loading State
Loading should be characterized not only by how much protein or peptide is associated with the system, but by where and how that association occurs. A high apparent encapsulation efficiency can be misleading if the biomolecule is only loosely adsorbed at the outer shell or if a large fraction is associated in a rapidly exchangeable form. Distinguishing core-associated, shell-associated, and free fractions is therefore highly valuable. In practical terms, the loading state often predicts release behavior better than the loading number alone. This is especially true for peptides, which may appear efficiently loaded while still redistributing quickly under dilution or ionic change.
Structural Integrity of the Protein or Peptide
Structural integrity analysis is essential because many preparation routes and micellar interfaces can modify conformation without visibly disrupting the colloid. A protein may remain "loaded" but no longer retain its useful structural state, while a peptide may aggregate or reorganize in a way that changes bioactivity. Spectroscopic methods, electrophoretic evaluation, activity assays, or other structure-sensitive analytical tools should therefore be used wherever possible to determine whether the payload remains comparable to its intended functional form. For protein and peptide micelles, this is one of the most decisive differentiators between a technically successful nanocarrier and a merely well-dispersed particle suspension.
Release Behavior Under Relevant Conditions
Release testing should reflect the actual conditions the biomacromolecule is likely to encounter rather than relying only on simplified sink conditions. The purpose is not merely to show that release occurs, but to determine whether the release pathway preserves the value of the loaded state created during preparation. For proteins and peptides, this includes evaluating whether the released fraction remains structurally acceptable and whether release is governed by meaningful association mechanisms rather than by immediate passive loss. Relevant conditions may include ionic strength changes, buffer composition shifts, dilution, and other variables that affect both micelle stability and protein or peptide behavior after departure from the carrier.
Services
Advanced Polymer Micelle Synthesis and Formulation Services
BOC Sciences supports the development of protein- and peptide-loaded micellar systems through integrated capabilities in polymer design, route-specific formulation, and biomacromolecule-compatible process optimization. Our services cover custom amphiphilic polymer synthesis, architecture tuning for core-shell control, charged and functional segment design for complexation-based loading, and preparation planning for mild or staged self-assembly workflows. We help align polymer composition, payload interaction mode, and fabrication history so that the resulting micelles are not only colloidally well defined, but also better matched to the structural sensitivity and release requirements of protein and peptide cargos.

Custom Amphiphilic Polymer Design

Functional Segment & Interface Engineering
- Incorporation of charged, affinity-guided, or shell-modifying functional segments.
- Design of Polyethylene glycol-containing systems for hydration and interfacial shielding.
- Optimization of polymer architecture for conventional, mixed, or polyion-complex micelles.
- Reduction of interface-induced stress through tailored surface chemistry.

Micelle Preparation & Loading Strategy Development
- Preparation planning for direct dissolution, dialysis, film hydration, and staged assembly routes.
- Selection of loading strategies for hydrophobic association, charge complexation, or shell association.
- Optimization of process order to protect biomacromolecular integrity during micelle formation.
- Development of reproducible workflows for protein- and peptide-loaded micelle fabrication.

Analytical Characterization Support
- Comparative characterization of carrier structure and payload state in protein- and peptide-loaded micelles.
- Assessment of loading mode, colloidal stability, and route-dependent variability.
- Evaluation of release behavior together with biomolecular integrity retention.
- Documentation support for formulation consistency and process-transfer readiness.
Do You Need A Consultation?
Discuss your protein or peptide micelle project with BOC Sciences to identify the right balance among payload type, micelle form, loading mechanism, and preparation strategy.
Products
Unlock New Possibilities with Tailored and High-Performance Polymers
FAQs
Frequently Asked Questions
-
Can polymer micelles deliver both proteins and peptides?
Yes, but they do not behave the same way. Proteins are usually larger and more structurally sensitive, while peptides may show stronger amphiphilicity or easier association with the micellar core or interface.
-
Which proteins are commonly used in polymer micelle delivery?
Commonly discussed protein materials include gelatin, collagen, casein, silk, elastin, zein, gliadin, soy proteins, albumin, and keratin, each offering different loading and assembly behavior.
-
What is the difference between conventional micelles and polyion complex micelles?
Conventional micelles usually rely more on amphiphilic self-assembly and hydrophobic segregation, while polyion complex micelles are driven more strongly by electrostatic interactions between oppositely charged components.
-
Which micelle form is best for protein and peptide delivery?
There is no universal best option. The preferred micelle form depends on payload size, charge, hydrophobicity, structural fragility, and the required release behavior.
-
What preparation method is most suitable for protein-loaded polymer micelles?
Mild preparation routes such as direct dissolution or dialysis are often preferred when protein stability is critical, but the final choice depends on polymer solubility, payload sensitivity, and desired loading mechanism.

 Fig. 1. Protein and peptide loading modes in polymer micelles (BOC Sciences Authorized).
Fig. 1. Protein and peptide loading modes in polymer micelles (BOC Sciences Authorized). Fig. 2. Comparison of micelle forms for protein and peptide delivery (BOC Sciences Authorized).
Fig. 2. Comparison of micelle forms for protein and peptide delivery (BOC Sciences Authorized).